β-Cyclodextrin-Silica Hybrid: A Spatially Controllable Anchoring Strategy for Cu(II)/Cu(I) Complex Immobilization
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation and Characterization of Silica-β-CD Derivatives
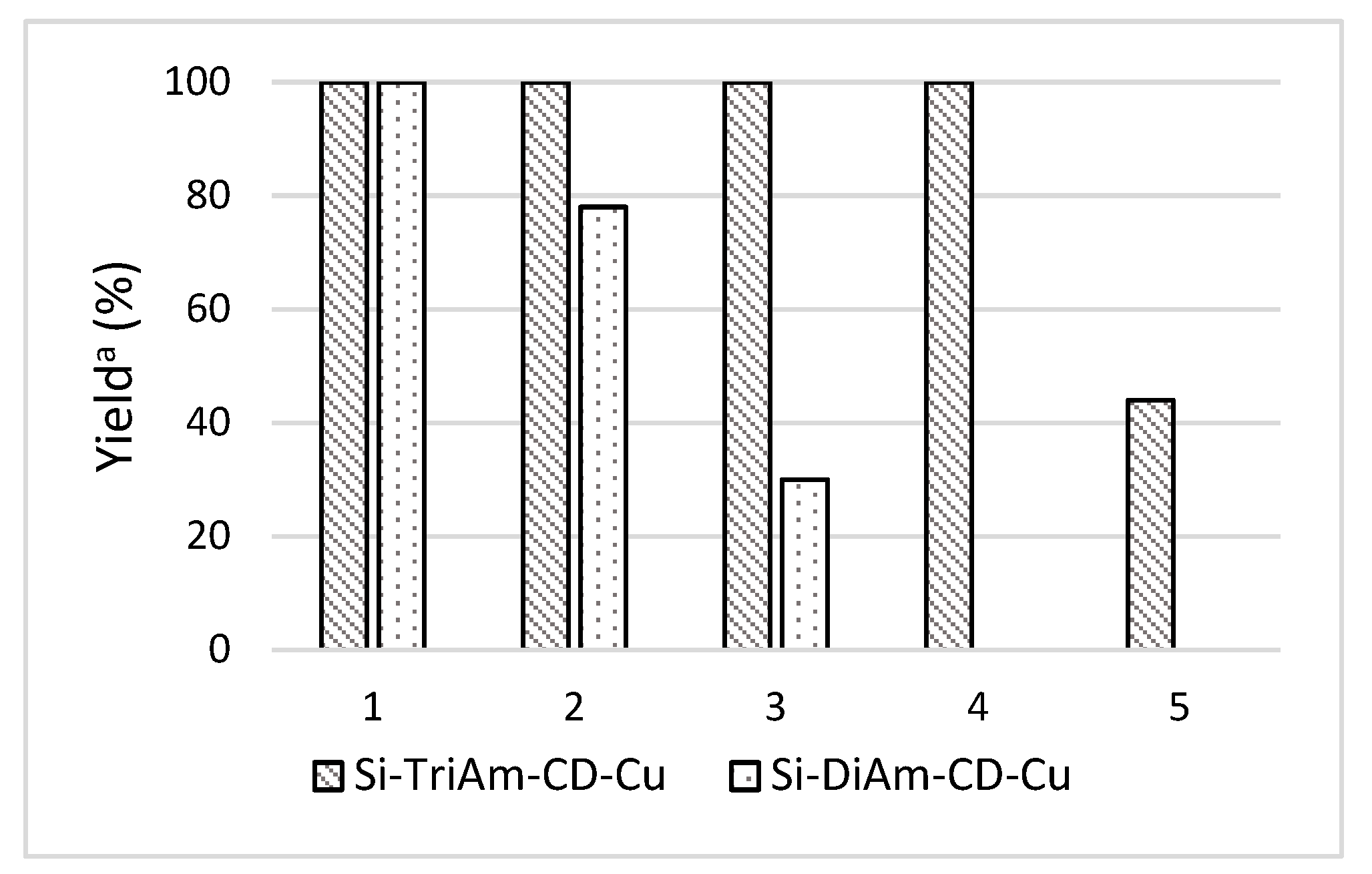
2.2. Catalytic Activity of Organic-Inorganic Silica-Supported β-CD-Cu(II)
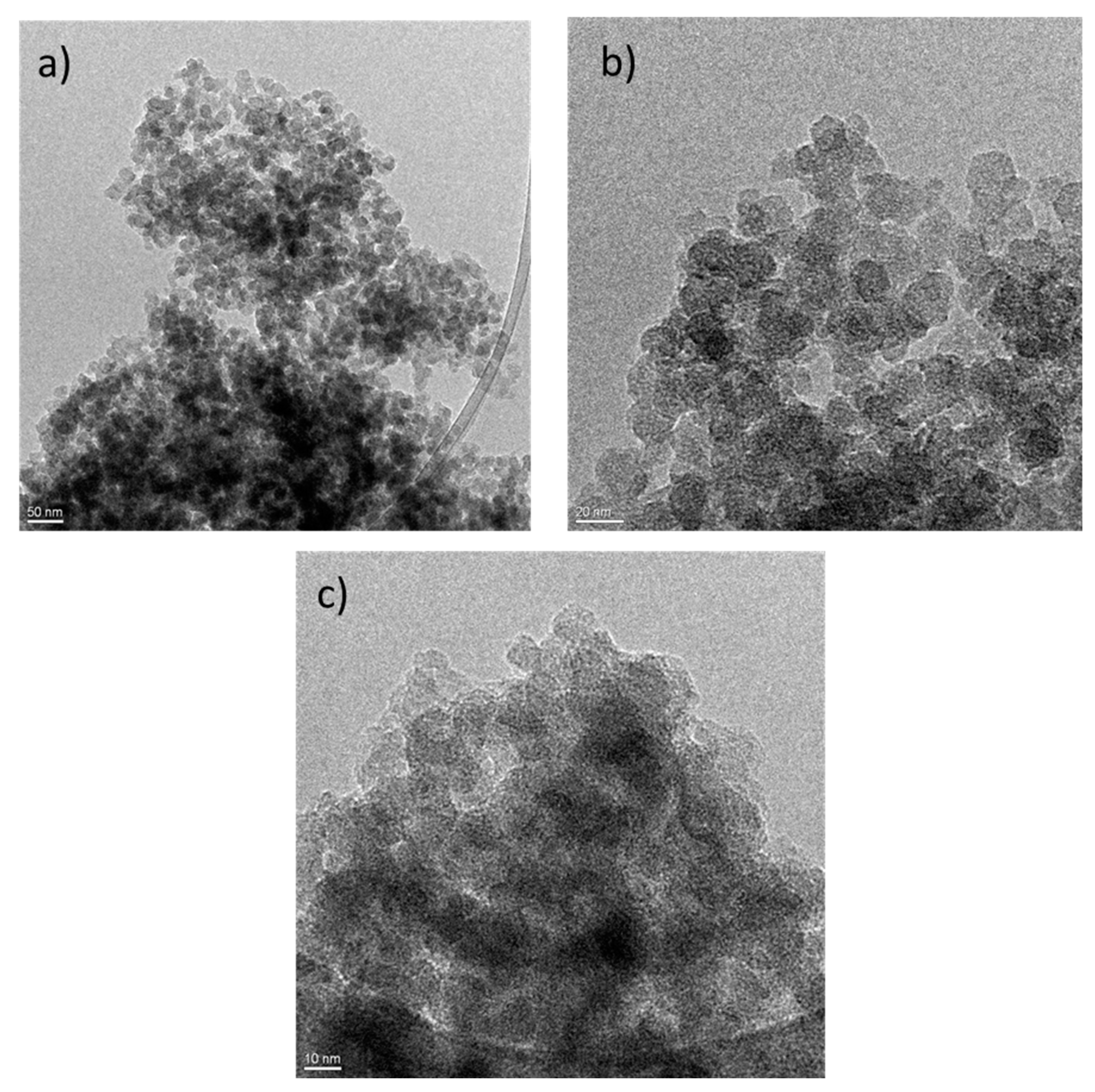
2.3. Characterization of Aged Si-TriAm-CD-Cu Catalyst
3. Materials and Methods
3.1. Materials
3.2. Catalyst Preparation
3.2.1. General Preparation of Alkoxy Silyl-Silica Derivatives (Si-Gly, Si-MonoAm, Si-TriAm)
3.2.2. General Preparation of Silica-β-CD Hybrid Systems (Si-Gly-CD, Si-MonoAm-CD, Si-TriAm-CD)
3.2.3. β-CD-Cu(II) Complexation
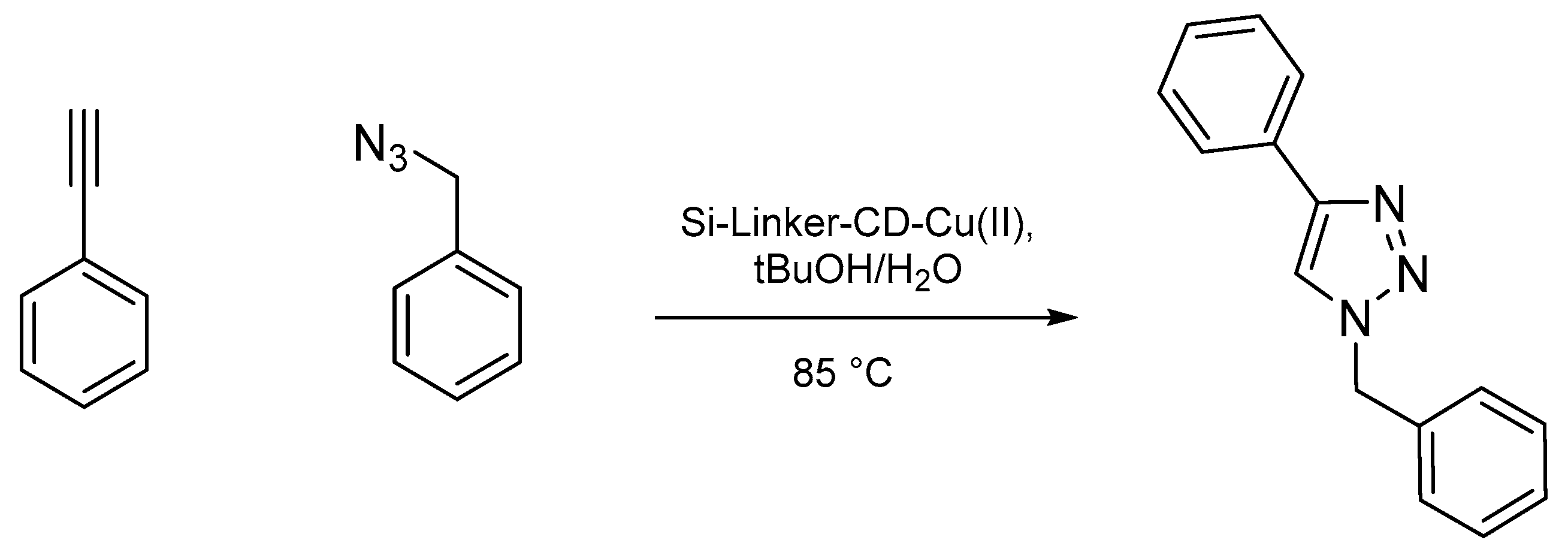
3.3. Click Chemistry Reaction
3.4. Catalysts Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chujo, Y. Organic-Inorganic hybrid materials. Curr. Opin. Solid State Mater. Sci. 1996, 1, 806–811. [Google Scholar] [CrossRef]
- Férey, G. Hybrid porous solids: Past, present, future. Chem. Soc. Rev. 2008, 37, 191–214. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Wilkes, G.L. Organic/Inorganic Hybrid Network Materials by the Sol-Gel Approach. Chem. Mater. 1996, 8, 1667–1681. [Google Scholar] [CrossRef]
- Mohammadi Ziarani, G.; Badiei, A.; Hajiabbas Tabar Amiri, P.; Lashgari, N. A Green Multicomponent One-pot Synthesis of 9, 10-Diaryl-7H-benzo [d,e] imidazo [2,1-a] isoquinolin-7-one Derivatives using Nanoporous Base Silica (SBA-Pr-NH2) as Catalyst. J. Nanostruct. 2019, 9, 1–7. [Google Scholar] [CrossRef]
- Suganya, K.S.U.; Govindaraju, K.; Vani, C.V.; Premanathan, M.; Kumar, V.K.G. In vitro biological evaluation of anti-diabetic activity of organic-inorganic hybrid gold nanoparticles. IET Nanobiotechnol. 2019, 13, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Cariati, E.; Lucenti, E.; Botta, C.; Giovanella, U.; Marinotto, D.; Righetto, S. Cu(I) hybrid inorganic–organic materials with intriguing stimuli responsive and optoelectronic properties. Coord. Chem. Rev. 2016, 306, 566–614. [Google Scholar] [CrossRef]
- Karthik, P.; Vinoth, R.; Selvam, P.; Balaraman, E.; Navaneethan, M.; Hayakawa, Y.; Neppolian, B. A visible-light active catechol–metal oxide carbonaceous polymeric material for enhanced photocatalytic activity. J. Mater. Chem. A 2017, 5, 384–396. [Google Scholar] [CrossRef]
- Xiao, Z.; Kerner, R.A.; Zhao, L.; Tran, N.L.; Lee, K.M.; Koh, T.-W.; Scholes, G.D.; Rand, B.P. Efficient perovskite light-emitting diodes featuring nanometre-sized crystallites. Nat. Photonics 2017, 11, 108. [Google Scholar] [CrossRef]
- Di Tommaso, S.; Giannici, F.; Mossuto Marculescu, A.; Chiara, A.; Tealdi, C.; Martorana, A.; Labat, F.; Adamo, C. Theoretical insights into inorganic-organic intercalation products of the layered perovskite HLaNb2O7: Perspectives for hybrid proton conductors. Phys. Chem. Chem. Phys. 2019, 21, 16647–16657. [Google Scholar] [CrossRef]
- Dolbecq, A.; Dumas, E.; Mayer, C.R.; Mialane, P. Hybrid Organic−Inorganic Polyoxometalate Compounds: From Structural Diversity to Applications. Chem. Rev. 2010, 110, 6009–6048. [Google Scholar] [CrossRef]
- Lei, B.; Li, B.; Zhang, H.; Zhang, L.; Li, W. Synthesis, Characterization, and Oxygen Sensing Properties of Functionalized Mesoporous SBA-15 and MCM-41 with a Covalently Linked Ruthenium(II) Complex. J. Phys. Chem. C 2007, 111, 11291–11301. [Google Scholar] [CrossRef]
- Franville, A.-C.; Zambon, D.; Mahiou, R.; Troin, Y. Luminescence Behavior of Sol−Gel-Derived Hybrid Materials Resulting from Covalent Grafting of a Chromophore Unit to Different Organically Modified Alkoxysilanes. Chem. Mater. 2000, 12, 428–435. [Google Scholar] [CrossRef]
- Benzaqui, M.; Semino, R.; Carn, F.; Tavares, S.R.; Menguy, N.; Giménez-Marqués, M.; Bellido, E.; Horcajada, P.; Berthelot, T.; Kuzminova, A.I.; et al. Covalent and Selective Grafting of Polyethylene Glycol Brushes at the Surface of ZIF-8 for the Processing of Membranes for Pervaporation. ACS Sustain. Chem. Eng. 2019, 7, 6629–6639. [Google Scholar] [CrossRef]
- Georgakilas, V.; Otyepka, M.; Bourlinos, A.B.; Chandra, V.; Kim, N.; Kemp, K.C.; Hobza, P.; Zboril, R.; Kim, K.S. Functionalization of Graphene: Covalent and Non-Covalent Approaches, Derivatives and Applications. Chem. Rev. 2012, 112, 6156–6214. [Google Scholar] [CrossRef]
- Lashgari, N.; Badiei, A.; Mohammadi Ziarani, G. Modification of mesoporous silica SBA-15 with different organic molecules to gain chemical sensors: A review. Nanochem. Res. 2016, 1, 127–141. [Google Scholar] [CrossRef]
- Singh, S.; Kumar, R.; Setiabudi, H.D.; Nanda, S.; Vo, D.-V.N. Advanced synthesis strategies of mesoporous SBA-15 supported catalysts for catalytic reforming applications: A state-of-the-art review. Appl. Catal. A Gen. 2018, 559, 57–74. [Google Scholar] [CrossRef]
- Ndolomingo, M.J.; Meijboom, R. Noble and Base-Metal Nanoparticles Supported on Mesoporous Metal Oxides: Efficient Catalysts for the Selective Hydrogenation of Levulinic Acid to γ-Valerolactone. Catal. Lett. 2019, 149, 2807–2822. [Google Scholar] [CrossRef]
- Chen, D.; Zhang, P.; Fang, Q.; Wan, S.; Li, H.; Yang, S.; Huang, C.; Dai, S. Coordination-supported organic polymers: Mesoporous inorganic–organic materials with preferred stability. Inorg. Chem. Front. 2018, 5, 2018–2022. [Google Scholar] [CrossRef]
- Azeez, A.; Polio, L.; Hanson, J.E.; Gorun, S.M. Photoreactive Superhydrophobic Organic–Inorganic Hybrid Materials Composed of Poly(vinylidene fluoride) and Titanium Dioxide-Supported Perfluorinated Phthalocyanines. ACS Appl. Polym. Mater. 2019, 1, 1514–1523. [Google Scholar] [CrossRef]
- Fedorov, P.P.; Luginina, A.A.; Kuznetsov, S.V.; Voronov, V.V.; Lyapin, A.A.; Ryabochkina, P.A.; Chernov, M.V.; Mayakova, M.N.; Pominova, D.V.; Uvarov, O.V.; et al. Preparation and properties of methylcellulose/nanocellulose/СаF2:Нo polymer-inorganic composite films for two-micron radiation visualizers. J. Fluor. Chem. 2017, 202, 9–18. [Google Scholar] [CrossRef]
- Ahadi, N.; Bodaghifard, M.A.; Mobinikhaledi, A. Cu (II)-β-cyclodextrin complex stabilized on magnetic nanoparticles: A retrievable hybrid promoter for green synthesis of spiropyrans. Appl. Organomet. Chem. 2019, 33, e4738. [Google Scholar] [CrossRef]
- Fallahi, M.; Ahmadi, E.; Mohamadnia, Z. Effect of inorganic oxide supports on the activity of chromium-based catalysts in ethylene trimerization. Appl. Organomet. Chem. 2019, 33, e4975. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Y.; Li, Q.; Zhang, G. Metal−organic framework composite membranes: Synthesis and separation applications. Chem. Eng. Sci. 2015, 135, 232–257. [Google Scholar] [CrossRef]
- Yin, P.T.; Shah, S.; Chhowalla, M.; Lee, K.-B. Design, Synthesis, and Characterization of Graphene–Nanoparticle Hybrid Materials for Bioapplications. Chem. Rev. 2015, 115, 2483–2531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, N.; Liu, X.; Huang, Y.; Wang, M.; Li, S.; Zong, M.; Liu, P. Novel nanocomposites of cobalt ferrite covalently-grafted on graphene by amide bond as superior electromagnetic wave absorber. J. Colloid Interface Sci. 2019, 540, 218–227. [Google Scholar] [CrossRef]
- de Sousa, M.E.; Fernández van Raap, M.B.; Rivas, P.C.; Mendoza Zélis, P.; Girardin, P.; Pasquevich, G.A.; Alessandrini, J.L.; Muraca, D.; Sánchez, F.H. Stability and Relaxation Mechanisms of Citric Acid Coated Magnetite Nanoparticles for Magnetic Hyperthermia. J. Phys. Chem. C 2013, 117, 5436–5445. [Google Scholar] [CrossRef] [Green Version]
- Su, H.; Liu, Y.; Wang, D.; Wu, C.; Xia, C.; Gong, Q.; Song, B.; Ai, H. Amphiphilic starlike dextran wrapped superparamagnetic iron oxide nanoparticle clsuters as effective magnetic resonance imaging probes. Biomaterials 2013, 34, 1193–1203. [Google Scholar] [CrossRef]
- Mrowczynski, R.; Jedrzak, A.; Szutkowski, K.; Grzeskowiak, B.F.; Coy, E.; Markiewicz, R.; Jesionowski, T.; Jurga, S. Cyclodextrin-Based Magnetic Nanoparticles for Cancer Therapy. Nanomaterials (Basel) 2018, 8. [Google Scholar] [CrossRef] [Green Version]
- Rostamnia, S.; Doustkhah, E. Nanoporous silica-supported organocatalyst: A heterogeneous and green hybrid catalyst for organic transformations. RSC Adv. 2014, 4, 28238–28248. [Google Scholar] [CrossRef]
- Salvo, A.M.P.; Giacalone, F.; Gruttadauria, M. Advances in Organic and Organic-Inorganic Hybrid Polymeric Supports for Catalytic Applications. Molecules 2016, 21, 1288. [Google Scholar] [CrossRef] [Green Version]
- Ortuño, M.A.; López, N. Reaction mechanisms at the homogeneous–heterogeneous frontier: Insights from first-principles studies on ligand-decorated metal nanoparticles. Catal. Sci. Technol. 2019, 9, 5173–5185. [Google Scholar] [CrossRef]
- Karimi, B.; Zamani, A.; Clark, J.H. A Bipyridyl Palladium Complex Covalently Anchored onto Silica as an Effective and Recoverable Interphase Catalyst for the Aerobic Oxidation of Alcohols. Organometallics 2005, 24, 4695–4698. [Google Scholar] [CrossRef]
- Guo, H.; Gong, C.; Zeng, X.; Xu, H.; Zeng, Q.; Zhang, J.; Zhong, Z.; Xie, J. Isopolymolybdate-based inorganic–organic hybrid compounds constructed by multidentate N-donor ligands: Syntheses, structures and properties. Dalton Trans. 2019, 48, 5541–5550. [Google Scholar] [CrossRef]
- Goni, M.A.; Rosenberg, E.; Meregude, S.; Abbott, G. A methods study of immobilization of PONOP pincer transition metal complexes on silica polyamine composites (SPC). J. Organomet. Chem. 2016, 807, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Luca, O.R.; Crabtree, R.H. Redox-active ligands in catalysis. Chem. Soc. Rev. 2013, 42, 1440–1459. [Google Scholar] [CrossRef] [PubMed]
- Berben, L.A.; de Bruin, B.; Heyduk, A.F. Non-innocent ligands. Chem. Commun. 2015, 51, 1553–1554. [Google Scholar] [CrossRef] [Green Version]
- Skorjanc, T.; Benyettou, F.; Olsen, J.C.; Trabolsi, A. Design of Organic Macrocycle-Modified Iron Oxide Nanoparticles for Drug Delivery. Chemistry 2017, 23, 8333–8347. [Google Scholar] [CrossRef]
- Velasco, M.I.; Krapacher, C.R.; de Rossi, R.H.; Rossi, L.I. Structure characterization of the non-crystalline complexes of copper salts with native cyclodextrins. Dalton Trans. 2016, 45, 10696–10707. [Google Scholar] [CrossRef] [Green Version]
- Kurokawa, G.; Sekii, M.; Ishida, T.; Nogami, T. Short Communication: Crystal Structure of a Molecular Complex from Native β-Cyclodextrin and Copper(II) Chloride. Supramol. Chem. 2004, 16, 381–384. [Google Scholar] [CrossRef]
- Rossi, L.I.; Kinen, C.O.; de Rossi, R.H. Important role of native β-cyclodextrin in the stabilization of transition metal salts. Comptes Rendus Chim. 2017, 20, 1053–1061. [Google Scholar] [CrossRef]
- Han, B.-H.; Antonietti, M. One-step synthesis of copper nanoparticles containing mesoporous silica by nanocasting of binuclear copper(ii) complexes with cyclodextrins. J. Mater. Chem. 2003, 13, 1793–1796. [Google Scholar] [CrossRef]
- Bahadorikhalili, S.; Ma’mani, L.; Mahdavi, H.; Shafiee, A. Copper supported β-cyclodextrin functionalized PEGylated mesoporous silica nanoparticle -graphene oxide hybrid: An efficient and recyclable nano-catalyst for straightforward synthesis of 2-arylbenzimidazoles and 1,2,3-triazoles. Microporous Mesoporous Mater. 2018, 262, 207–216. [Google Scholar] [CrossRef]
- Ye, R.-P.; Lin, L.; Liu, C.-Q.; Chen, C.-C.; Yao, Y.-G. One-Pot Synthesis of Cyclodextrin-Doped Cu-SiO2 Catalysts for Efficient Hydrogenation of Dimethyl Oxalate to Ethylene Glycol. ChemCatChem 2017, 9, 4587–4597. [Google Scholar] [CrossRef]
- Fedorova, A.A.; Morozov, I.V.; Kotovshchikov, Y.N.; Romanovsky, B.V.; Sirotin, S.V.; Knyazeva, E.E.; Lermontov, A.S.; Shaporev, A.S. Preparation and characterization of copper- and iron-containing mesoporous silica using β-cyclodextrin as a structure-directing agent. Mendeleev Commun. 2011, 21, 171–172. [Google Scholar] [CrossRef]
- Han, B.-H.; Polarz, S.; Antonietti, M. Cyclodextrin-based Porous Silica Materials as in Situ Chemical “Nanoreactors” for the Preparation of Variable Metal−Silica Hybrids. Chem. Mater. 2001, 13, 3915–3919. [Google Scholar] [CrossRef] [Green Version]
- Khalafi-Nezhad, A.; Panahi, F. Size-Controlled Synthesis of Palladium Nanoparticles on a Silica–Cyclodextrin Substrate: A Novel Palladium Catalyst System for the Heck Reaction in Water. ACS Sustain. Chem. Eng. 2014, 2, 1177–1186. [Google Scholar] [CrossRef]
- Martina, K.; Baricco, F.; Caporaso, M.; Berlier, G.; Cravotto, G. Cyclodextrin-Grafted Silica-Supported Pd Nanoparticles: An Efficient and Versatile Catalyst for Ligand-Free C−C Coupling and Hydrogenation. ChemCatChem 2016, 8, 1176–1184. [Google Scholar] [CrossRef]
- Jayaprabha, K.N.; Joy, P.A. Citrate modified β-cyclodextrin functionalized magnetite nanoparticles: A biocompatible platform for hydrophobic drug delivery. RSC Adv. 2015, 5, 22117–22125. [Google Scholar] [CrossRef]
- Bagabas, A.A.; Frasconi, M.; Iehl, J.; Hauser, B.; Farha, O.K.; Hupp, J.T.; Hartlieb, K.J.; Botros, Y.Y.; Stoddart, J.F. γ-Cyclodextrin Cuprate Sandwich-Type Complexes. Inorg. Chem. 2013, 52, 2854–2861. [Google Scholar] [CrossRef] [Green Version]
- Shin, J.-A.; Lim, Y.-G.; Lee, K.-H. Copper-Catalyzed Azide–Alkyne Cycloaddition Reaction in Water Using Cyclodextrin as a Phase Transfer Catalyst. J. Org. Chem. 2012, 77, 4117–4122. [Google Scholar] [CrossRef]
- Kaboudin, B.; Abedi, Y.; Yokomatsu, T. CuII–β-Cyclodextrin Complex as a Nanocatalyst for the Homo- and Cross-Coupling of Arylboronic Acids under Ligand- and Base-Free Conditions in Air: Chemoselective Cross-Coupling of Arylboronic Acids in Water. Eur. J. Org. Chem. 2011, 2011, 6656–6662. [Google Scholar] [CrossRef]
- Sadeghzadeh, S.M.; Zhiani, R.; Moradi, M. KCC-1 Supported Cu(II)-β-Cyclodextrin Complex as a Reusable Catalyst for the Synthesis of 3-Aryl-2-oxazolidinones from Carbon Dioxide, Epoxide, Anilines. ChemistrySelect 2018, 3, 3516–3522. [Google Scholar] [CrossRef]
- Gupta, D.; Mishra, A.; Kundu, S. Cu (II)-β-CD as Water-Loving Catalyst for One-Pot Synthesis of Triazoles and Biofuels Intermediate at Room Temperature without Any Other Additive. ChemistrySelect 2017, 2, 2997–3008. [Google Scholar] [CrossRef]
- Martina, K.; Calsolaro, F.; Zuliani, A.; Berlier, G.; Chavez-Rivas, F.; Moran, M.J.; Luque, R.; Cravotto, G. Sonochemically-Promoted Preparation of Silica-Anchored Cyclodextrin Derivatives for Efficient Copper Catalysis. Molecules 2019, 24, 2490. [Google Scholar] [CrossRef] [Green Version]
- Martina, K.; Baricco, F.; Berlier, G.; Caporaso, M.; Cravotto, G. Efficient Green Protocols for Preparation of Highly Functionalized β-Cyclodextrin-Grafted Silica. ACS Sustain. Chem. Eng. 2014, 2, 2595–2603. [Google Scholar] [CrossRef]
- Musso, G.E.; Bottinelli, E.; Celi, L.; Magnacca, G.; Berlier, G. Influence of surface functionalization on the hydrophilic character of mesoporous silica nanoparticles. Phys. Chem. Chem. Phys. 2015, 17, 13882–13894. [Google Scholar] [CrossRef]
- Negri, C.; Signorile, M.; Porcaro, N.G.; Borfecchia, E.; Berlier, G.; Janssens, T.V.W.; Bordiga, S. Dynamic Cu-II/Cu-I speciation in Cu-CHA catalysts by in situ Diffuse Reflectance UV-vis-NIR spectroscopy. Appl. Catal. A Gen. 2019, 578, 1–9. [Google Scholar] [CrossRef]
- Bordiga, S.; Groppo, E.; Agostini, G.; van Bokhoven, J.A.; Lamberti, C. Reactivity of Surface Species in Heterogeneous Catalysts Probed by In Situ X-ray Absorption Techniques. Chem. Rev. 2013, 113, 1736–1850. [Google Scholar] [CrossRef] [Green Version]
- Solomon, E.I.; Heppner, D.E.; Johnston, E.M.; Ginsbach, J.W.; Cirera, J.; Qayyum, M.; Kieber-Emmons, M.T.; Kjaergaard, C.H.; Hadt, R.G.; Tian, L. Copper Active Sites in Biology. Chem. Rev. 2014, 114, 3659–3853. [Google Scholar] [CrossRef] [Green Version]
- Negri, C.; Borfecchia, E.; Cutini, M.; Lomachenko, K.A.; Janssens, T.V.W.; Berlier, G.; Bordiga, S. Evidence of Mixed-Ligand Complexes in Cu-CHA by Reaction of Cu Nitrates with NO/NH3 at Low Temperature. Chemcatchem 2019, 11, 3828–3838. [Google Scholar] [CrossRef]
- Kau, L.S.; Spirasolomon, D.J.; Pennerhahn, J.E.; Hodgson, K.O.; Solomon, E.I. X-Ray Absorption-Edge Determination of The Oxidation-State and Coordination-Number Of Copper—Application To The Type-3 Site In Rhus-Vernicifera Laccase And Its Reaction With Oxygen. J. Am. Chem. Soc. 1987, 109, 6433–6442. [Google Scholar] [CrossRef]
- Borfecchia, E.; Beato, P.; Svelle, S.; Olsbye, U.; Lamberti, C.; Bordiga, S. Cu-CHA—A model system for applied selective redox catalysis. Chem. Soc. Rev. 2018, 47, 8097–8133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dent, A.J.; Cibin, G.; Ramos, S.; Smith, A.D.; Scott, S.M.; Varandas, L.; Pearson, M.R.; Krumpa, N.A.; Jones, C.P.; Robbins, P.E. B18: A core XAS spectroscopy beamline for Diamond. In Proceedings of the 14th International Conference on X-Ray Absorption Fine Structure, Camerino (MC), Italy, 26–31 July 2009; DiCicco, A., Filipponi, A., Eds.; IOP Publishing: Bristol, UK, 2009; Volume 190. [Google Scholar]
- Ravel, B.; Newville, M. Athena, Artemis, Hephaestus: Data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 2005, 12, 537–541. [Google Scholar] [CrossRef] [PubMed] [Green Version]






| Entry | Product | Linker | Loading [w/w%] | Loading [μmol/g] |
|---|---|---|---|---|
| 1 | Si-DETA | -NH(CH2)2NH(CH2)2NH2 | 15.3 (a) | 1.5 × 103 (a) |
| 2 | Si-MonoAm | -(CH2)3NH2 | 9.19 (a) | 513 (a) |
| 3 | Si-DiAm | -(CH2)3NH(CH2)2NH2 | 12.1 (a) | 543 (a) |
| 4 | Si-TriAm | -(CH2)3NH(CH2)2NH(CH2)2NH2 | 10.36 (a) | 390 (a) |
| 5 | Si-Gly | -(CH2)3OCH2CHOCH2 | 6.13 (a) | 259 (a) |
| 6 | Si-NH-CD | -NH-βCD | 7.12 (b)–0.68 (c) | 62 (b)-6 (c) |
| 7 | Si-MonoAm-CD | -(CH2)3NH-βCD | 3.82 (b)–1.28 (c) | 34 (b)–11.3 (c) |
| 8 | Si-DiAm-CD | -(CH2)3NH(CH2)2NH-βCD | 7.66 (b)–2.09 (c) | 67 (b)-18.4 (c) |
| 9 | Si-TriAm-CD | -(CH2)3NH(CH2)2NH(CH2)2NH-βCD | 7.7 (b)–3.60 (c) | 68 (b)–32 (c) |
| 10 | Si-Gly-CD | -(CH2)3OCH2CHOHCH2-βCD | 6.08 (b)–1.04 (c) | 54 (b)–9.2 (c) |
| Entry | Catalyst | Cu mol.% | Yield [%] (a) |
|---|---|---|---|
| 1 | Si-NHCD-Cu | 4 | 5 (b) |
| 2 | Si-DiAm-CD-Cu | 4 | >99 (b) |
| 3 | Si-DiAm-CD-Cu | 2 | 65 (b) |
| 4 | Si-Gly-CD-Cu | 4 | 45 |
| 5 | Si-MonoAm-CD-Cu | 4 | 4 |
| 6 | Si-TriAm-CD-Cu | 4 | >99 |
| 7 | Si-TriAm-CD-Cu | 2 | >99 |
| 8 | Si-TriAm-CD-Cu | 1 | 85 |
| Entry | Alkyne | Azide | Product | Yield [%] (a) |
|---|---|---|---|---|
| 1 | 
| 
| 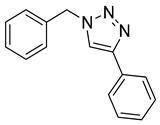
| 99 |
| 2 | 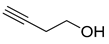
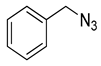
| 
| 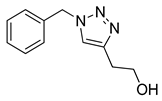
| 99 (b) |
| 3 | 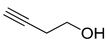
| 
| 
| 99 (b) |
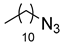
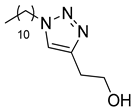
| 4 | 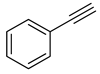
| 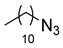
| 
| 99 (b) |
| 5 | 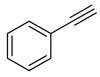
| 
| 
| 99 (c) |
| 6 | 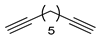
| 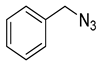
| 
| 99 (d) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calsolaro, F.; Martina, K.; Borfecchia, E.; Chávez-Rivas, F.; Cravotto, G.; Berlier, G. β-Cyclodextrin-Silica Hybrid: A Spatially Controllable Anchoring Strategy for Cu(II)/Cu(I) Complex Immobilization. Catalysts 2020, 10, 1118. https://doi.org/10.3390/catal10101118
Calsolaro F, Martina K, Borfecchia E, Chávez-Rivas F, Cravotto G, Berlier G. β-Cyclodextrin-Silica Hybrid: A Spatially Controllable Anchoring Strategy for Cu(II)/Cu(I) Complex Immobilization. Catalysts. 2020; 10(10):1118. https://doi.org/10.3390/catal10101118
Chicago/Turabian StyleCalsolaro, Federica, Katia Martina, Elisa Borfecchia, Fernando Chávez-Rivas, Giancarlo Cravotto, and Gloria Berlier. 2020. "β-Cyclodextrin-Silica Hybrid: A Spatially Controllable Anchoring Strategy for Cu(II)/Cu(I) Complex Immobilization" Catalysts 10, no. 10: 1118. https://doi.org/10.3390/catal10101118
APA StyleCalsolaro, F., Martina, K., Borfecchia, E., Chávez-Rivas, F., Cravotto, G., & Berlier, G. (2020). β-Cyclodextrin-Silica Hybrid: A Spatially Controllable Anchoring Strategy for Cu(II)/Cu(I) Complex Immobilization. Catalysts, 10(10), 1118. https://doi.org/10.3390/catal10101118









