1. Introduction
Sulforaphane (SFN) is an isothiocyanate widely recognized as a powerful anti-cancer and health-promoting compound [
1]. It occurs in cruciferous vegetables, mainly in broccoli. SFN formation takes place through the enzymatic hydrolysis of glucoraphanin, the main glucosinolate (GSL) found in broccoli, catalyzed by myrosinase (β-thioglucosidase glucohydrolase, EC 3.2.1.147). The conversion occurs through a two-step mechanism: hydrolysis of glucoraphanin by the action of myrosinase with the formation of an unstable intermediate (thiohydroxamate-O-sulfonate) and the release of an equimolar amount of glucose, followed by the spontaneous conversion of thiohydroxamate-O-sulfonate into sulforaphane or sulforaphane nitrile, depending on the chemical conditions. Acid pH together with the action of epithiospecifier protein (ESP) favor formation of sulforaphane nitrile, which is an undesirable product due to its potential toxicity. On the other hand, neutral pH favors the formation of sulforaphane.
Figure 1 depicts a scheme of glucoraphanin conversion into sulforaphane mediated by myrosinase. The hydrolysis begins with the nucleophilic attack of Glu or Asp on the anomeric carbon of glucoraphanin with the release of an aglucone (thyohydroxamate-O-sulfonate), followed by Lossen rearrangement and the release of glucose [
2]. The hydrolysis final product is sulforaphane at neutral pH, and sulforaphane nitrile at acid pH together with the action of epithiospecifier protein [
3].
The myrosinase-glucosinolate system constitutes a defense mechanism of the vegetal against biotic and abiotic stress. In the intact plant, myrosinase is compartmentalized in specialized myrosin cells, and GSL are ubiquitously distributed. As a result, the content of isothiocyanates in the intact vegetable tissue is relatively low. In order to increase the content of isothiocyanates, the plant tissue must be disrupted to allow that the enzyme gets in contact with GSL and the reaction to occur. This disruption can be achieved during mastication or by domestic or technological processing of the vegetable.
Several studies have sought to increase SFN content in the edible parts of broccoli by manipulating the post-harvest processing conditions, aiming at maximizing the functional properties of this vegetable. Pérez et al. [
4] reported the blanching temperature and time that maximized SFN content in broccoli florets, obtaining a 4-fold increase with respect to fresh broccoli. Mahn and Pérez [
5] optimized the incubation conditions, achieving an 8-fold increase in SFN content. Westphal et al. [
6] reported that high-pressure treatment of broccoli sprouts significantly increased SFN content. Tabart et al. [
7] investigated the effect of microwave processing of broccoli, reporting 4-fold increase of SFN content. Direct consumption of broccoli processed in such ways sometimes is not possible because of its unpleasant organoleptic characteristics and high perishability. Then, dehydration was proposed as a process to obtain a stable SFN-rich food ingredient that can be incorporated into further elaborated foods [
8]. A drawback arises when incorporating this broccoli-based ingredient into elaborated foods. SFN is thermo labile, it starts degrading above 40 °C [
9], and then any thermal process impairs SFN content in the final product. This limits the industrial application of the SFN-rich ingredient. An option to exploit the beneficial effects of sulforaphane and to give access to this compound to the consumers, is to deliver it as a food supplement or nutraceutical, avoiding the need of thermal processing. One way to obtain SFN from a natural source is to conduct glucoraphanin hydrolysis outside the vegetable, by extracting GSL from the vegetal tissues and adding exogenous myrosinase to perform the hydrolysis. In this way, SFN could be delivered directly to the consumer and not as part of a food matrix. In this sense, minimizing myrosinase activity during the glucoraphanin extraction process is desirable with the aim of obtaining the maximum conversion in SFN.
Ultrasound (US) processing is an emerging non-thermal technology recently adopted in the food industry with different purposes, such as tissue disruption and enzyme inactivation. It is considered a “green technology” that can assist traditional food processes [
10]. Ultrasound corresponds to acoustic waves with frequencies beyond the human audible range. The effect of US on food matrixes respond to the cavitation phenomenon produced by the formation and collision of bubbles accompanied by high temperature and pressure [
11]. This affects tissue structure by disrupting cell wall and membranes [
12] and by reducing enzyme activity due to the modification of the structure by breaking hydrogen interactions and van der Waals forces [
11]. Enzyme inactivation by US takes place at a higher rate than by thermal processing, thus reducing the processing time and temperature necessaries to achieve the desired denaturation [
13], and making US attractive to the industry.
The use of US processing in broccoli is poorly documented so far. Briones-Labarca et al. [
14] found that US-assisted extraction produced a 5-fold increase in SFN recovery from Chilean papaya seeds in a methanol solution. Pongmalai et al. [
15] reported that US-assisted extraction increased extractability in methanol of glucoraphanin (1.8-fold) from steamed cabbage leaves in comparison with fresh cabbage. The authors used an ultrasound thermostatic bath, which does not ensure that ultrasound waves affected equally all the vegetal material. Aguilar-Camacho et al. [
16] studied the effect of ultrasound treatment on the content of secondary metabolites in broccoli florets, and found an 8-fold increase in glucoraphanin content. The authors did not consider SFN or myrosinase.
Currently there are no studies about the effect of ultrasound processing on SFN content and myrosinase activity in broccoli florets. The hypothesis of this work was that US-assisted blanching reduces myrosinase activity without significant SFN leakage to the blanching water. In this way, an extract from US-blanched broccoli will be enriched in glucoraphanin that can be converted into SFN by adding exogenous myrosinase. If the basal SFN content in the vegetable is not affected by US-blanching, then this amount can be added to the final extract. Accordingly, the aim of this work was to investigate the effect of ultrasound-assisted blanching on myrosinase inactivation and SFN content in broccoli florets.
2. Results
The effect of time and temperature in ultrasound-assisted blanching on myrosinase activity and SFN content was studied through a multilevel factorial design.
Table 1 shows the experimental conditions and the responses obtained in each experimental run. Statistically significant differences appeared on myrosinase activity and sulforaphane content. Myrosinase activity in fresh broccoli florets was 2450 ± 72 U, and in broccoli subjected to traditional blanching it was 1469 ± 17 U. In all runs, myrosinase activity was lower than the activity found in fresh broccoli. In runs 3, 4, 6, 9 and 14, myrosinase activity was lower than that found in traditionally blanched broccoli. The lowest activity corresponded to run 9 (508 ± 12 U). SFN content in fresh broccoli florets was 585.0 ± 9 µg g
−1 and in traditionally blanched broccoli it was 863 ± 11 µg g
−1. In runs 1, 2, 4, 7, 10, 11, 13 and 14, SFN content was significantly lower than that in the fresh vegetable. Runs 3, 6 and 9 showed SFN content significantly higher than in traditionally blanched broccoli. The lowest SFN content was obtained in run 13 (209 ± 21 µg/g), representing a 64% decrease in comparison with fresh broccoli. The highest SFN content was obtained in run 9, equal to 1185 ± 30 µg g
−1, 2-fold the SFN content in fresh broccoli.
The statistical analysis of the effect of each experimental factor on both responses is shown in
Figure 2 as Pareto charts and response surfaces. The experimental factors significantly affected myrosinase activity and SFN content. Blanching time (B) had a significant positive effect on myrosinase activity; the other experimental factors did not affect significantly this response, as shown in
Figure 2a. Temperature (A) and the interaction of temperature with itself (AA) had significant positive effect on SFN content, while blanching time had significant negative effect on this response (
Figure 2b).
Figure 2c shows the response surface for myrosinase activity. The minimum activity appears at 60 °C and 4 min of ultrasound-assisted blanching.
Figure 2d shows that there is no maximum, i.e., the processing conditions that maximize SFN content could not be detected.
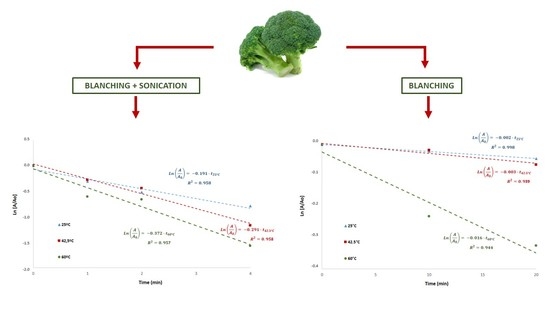
Myrosinase inactivation was studied considering a first-order kinetics. The experimental data were adjusted to a first-order inactivation model, as suggested in the literature [
13].
Figure 3 shows myrosinase inactivation kinetics during blanching at different temperatures with or without ultrasound processing. The determination coefficients (R
2) were higher than 0.94, confirming that this kinetic model represents adequately myrosinase inactivation. The kinetic constants obtained in US-assisted blanching (
Figure 3a) at the different temperatures were k
25 °C = 0.1910 min
−1, k
42.5 °C = 0.2913 min
−1 and k
60 °C = 0.3716 min
−1. The kinetic constants obtained in blanching without ultrasound (
Figure 3b) at the different temperatures were k
25 °C = 0.0022 min
−1, k
42.5 °C = 0.0031 min
−1 and k
60 °C = 0.0159 min
−1.
The activation energy (Ea) for myrosinase inactivation with and without ultrasound processing was calculated considering that the constants depend on temperature, and they can be expressed by the Arrhenius equation. In ultrasound-assisted blanching, the Ea for myrosinase inactivation was 15.8 kJ mol−1; in blanching without ultrasound, Ea was 57.3 kJ mol−1.
3. Discussion
Myrosinase activity in broccoli florets decreased after all treatments with respect to myrosinase activity in the fresh vegetable (2450 ± 72 U). This may obey to the inactivation of myrosinase by ultrasound-assisted blanching. The SFN content in broccoli florets before treatment was 585 ± 8 µg g
−1. In runs 1, 2, 4, 7, 10, 11, 13 and 14, SFN content was significantly lower than that in the fresh vegetable, being the lowest content equal to 208.6 ± 20.9 µg g
−1. This represents a 65% decrease (run 13). The highest SFN content was obtained in run 9, being equal to 1185 ± 30 µg g
−1. This represents a 2-fold increase with respect to the fresh vegetable. This value is lower than those reported in literature [
4], showing a 4-fold increase in SFN content after blanching at 57 °C for 13 min without using ultrasound. The lower SFN values obtained in the present work may be due to myrosinase inactivation that prevents the hydrolysis of glucoraphanin to yield SFN. SFN concentration in the blanching water was negligible (data not shown), probably because of the highly hydrophobic nature of this compound. Therefore, the modest increase in SFN content cannot be explained by lixiviation into the blanching water. Additionally, glucoraphanin, a water-soluble compound, was not detected in the blanching water, suggesting that US processing did not affect significantly the structure integrity of the vegetal tissue.
Response surface analyses suggest that the minimum activity appears at 60 °C and 4 min of ultrasound-assisted blanching. SFN content cannot be maximized by manipulating the experimental factors considered in this work since the response surface showed no maximum. However, higher temperature and shorter time produced a higher SFN content in ultrasound-assisted blanched broccoli. US-assisted blanching at 60 °C and 4 min resulted in the highest SFN content (see run 9 in
Table 1). This behavior relates to the low leakage of SFN into the blanching water because of the low solubility of this hydrophobic compound, resulting in SFN accumulation in the vegetable tissue, in addition to the apparently slight tissue damage produced by ultrasound processing. SFN formation probably occurred during the temperature stabilization period at the beginning of blanching, before the significant inactivation of myrosinase.
Ultrasound processing resulted in significantly higher inactivation kinetic constants, being two orders of magnitude higher that the constants obtained in blanching without ultrasound processing (
Figure 3). Accordingly, ultrasound processing significantly increases the inactivation rate of broccoli myrosinase. The kinetic constants obtained in blanching without ultrasound (k
42.5 °C = 0.0031 min
−1 and k
60 °C = 0.0159 min
−1) agree with the constants reported elsewhere [
17] for thermal inactivation of myrosinase in rehydrated lyophilized broccoli: k
40 °C = 0.0033 min
−1 and k
60 °C = 0.0079 min
−1 at 40 and 60 °C, respectively.
In ultrasound-assisted blanching, the Ea for myrosinase inactivation was 15.8 kJ mol
−1; in traditional blanching, Ea was 57.3 kJ mol
−1. The latter value agrees with Olivero et al. [
17], who reported an Ea in the same order of magnitude (44.5 ± 10.6 kJ mol
−1). Accordingly, ultrasound processing reduces the activation energy required for myrosinase inactivation. This could represent significant energy savings in an industrial process, constituting a more efficient blanching process in comparison with traditional blanching.
Myrosinase inactivation may be useful to preserve glucoraphanin in the vegetable tissue if the objective is extracting the precursor of SFN, glucoraphanin, to conduct the hydrolysis exogenously under controlled conditions. This will prevent the formation of undesirable compounds such as nitriles and isothionitriles which compete with SFN formation, thus maximizing the conversion of glucoraphanin in SFN. Auspiciously, the minimum myrosinase activity agreed with the highest SFN content, and then ultrasound-assisted blanching at 60 °C for 4 min could constitute a step in an SFN production process. This process should include a size reduction operation, ultrasound-assisted blanching in the optimal conditions, and extraction of glucoraphanin to finally conduct the hydrolysis externally. These findings open the opportunity to valorize broccoli byproducts such as stalks, secondary inflorescences and leaves, all of them currently considered as discards.