1. Introduction
Protein phosphorylation increases the functional diversity of the proteome and is critical to the regulation of numerous cellular processes. About 13,000 human proteins have one or more “p-sites”—amino acid residues where reversible phosphorylation occurs [
1]. The presence or absence of a phosphoryl group at a p-site impacts protein structure, enzyme activity, interactions with other proteins, transport properties, and, ultimately, cell function and metabolism [
2]. Countering the action of phosphoryl attachment by protein kinase enzymes (phosphorylation), the protein phosphatases catalyze the hydrolysis that liberates a phosphate ion and restores the p-site residue (dephosphorylation). The hydroxyl groups of serine, threonine, and tyrosine are amenable to phosphoryl attachment via a phosphoester bond, and most p-sites are one of these three amino acids, with serine dominant, threonine second, and tyrosine a distant third [
3].
Our interest is the common mechanism by which the members of the phosphoprotein phosphatase (PPP) gene family (PPP1C, PPP2C, PPP3C/calcineurin, PPP4C, PPP5C, PPP6C, and PPPEF/PP7), a subcategory of the broader class of protein serine/threonine phosphatases, catalyze dephosphorylation at seryl and threonyl p-sites. The PPPs share a highly conserved catalytic core [
4] featuring a bimetal system (M
1/M
2) as the site of substrate binding and hydrolysis. In this study, we focused on serine/threonine protein phosphatase-5 (PP5) as representative of the PPP family. PP5 expression is known to affect cell proliferation [
5,
6] and stress-induced responses [
7,
8,
9], and its overexpression is implicated in human breast carcinoma [
10], non-small cell lung cancer [
11], and other cancers [
12].
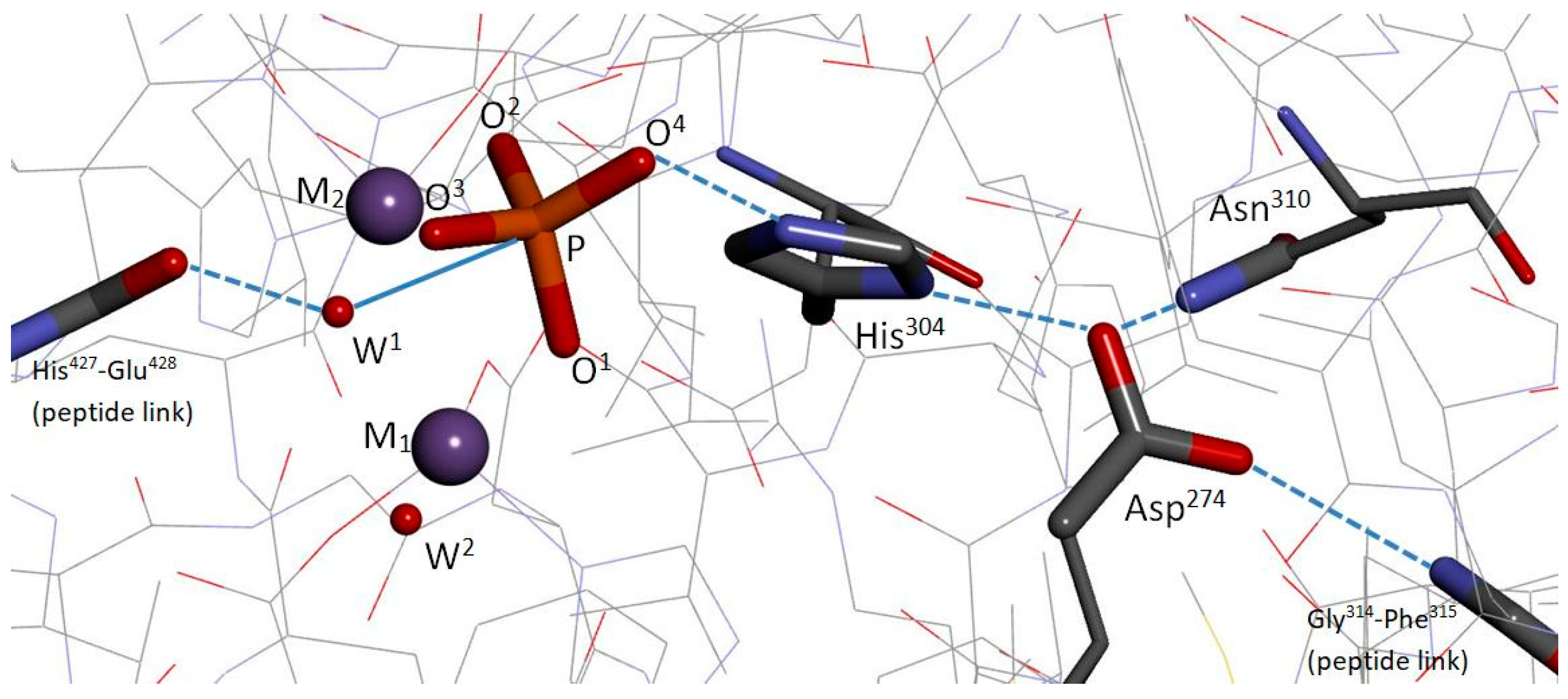
Co-crystal structures of PP5/phosphate [
13], PP1/tungstate [
14], and PP1/phosphate [
15], reveal common PPP catalytic-site features, including: A bimetal system held in place by the same amino-acid scaffold of six residues, a bridge hydroxide/water (W
1), a metal-bound phosphate (or tungstate) positioned by the same four residues, and a cooperative histidine/aspartate tandem. As shown for PP5/phosphate,
Figure 1, the alignment of the phosphate with W
1 strongly implies a nucleophilic attack on the P center by W
1, with subsequent protonation of the exiting alkoxide at the O
4 site by H
+ transfer from N
ϵ of His
304. Furthermore, Swingle et al. argued that W
1 is a hydroxide ion, and that the exiting alkoxide oxygen is probably not metal bound [
13]. Of course, this inferred mechanistic scenario depends on a match between the phosphate ion’s binding mode seen in the PP5 co-crystal and the binding mode of the phosphoserine/phosphothreonine substrate, which is unknown. Notably, the co-crystals also reveal a second water/hydroxide (W
2) coordinated to M
1, a backbone carbonyl (PP5:His
427) positioned to direct W
1, and a backbone -NH group (PP5:Phe
315) anchoring the His/Asp tandem at the backend,
Figure 1. In PP5, Asp
274 is also anchored by the nonconserved Asn
310 (likewise in PP1 and PP7, threonine otherwise). Thus, these co-crystals, along with mutation studies [
16,
17] and analysis, established a plausible pathway for PPP dephosphorylation [
13,
14,
15]. The substrate’s anticipated mode of binding to M
1/M
2 and the W
1(OH
−) attack on the P center were later corroborated by density functional theory (DFT) calculations by Ribeiro et al. on a cluster model of PP5/phosphoserine [
18].
In their PP5 study [
18], Ribeiro et al. presented pathways for two different systems, the first of which they endorsed: (I) W
1(OH
−)/W
2(OH
−) system (one step, exothermic): Arg
275 loses a proton to the W
2(OH
−) pre-reaction. That is, W
2 is actually a water and Arg
275 is deprotonated during the reactant and TS stages of the pathway; the proton is returned to Arg
275 in the product state. The seryl alkoxide is protonated by direct H
+ transfer from His
304 as W
1 attacks. (II) W
1(OH
−)/W
2(H
2O) system (two steps, endothermic): The seryl alkoxide is protonated by Arg
275 in step 1, in which the W
1(OH
−) attack also occurs. In step 2, H
+ is transferred from His
304 to Arg
275 through the serine’s alcohol group.
Both pathways (I) and (II) contain modeling artifacts. The Ribeiro et al. model system comprised 10 amino acid moieties + M
1/M
2 + W
1(OH
−)+ W
2 + phosphoserine. The amino acid moieties were terminated by frozen methyl groups. Asn
310 and the Gly
314-Phe
315 peptide linkage, which together bind Asp
274 (see
Figure 1), Asp
388, which binds Arg
400 (see
Figure 2), and Tyr
451, which abuts and interacts with Arg
275, were not represented. Consequently, the optimized positions of Asp
274, Arg
400, and Arg
275 diverged significantly from their native PP5/phosphate co-crystal positions. It should be added that a molecular dynamics (MD) study of PP5/phosphoserine by Wang and Yan [
19] concluded that W
1(OH
−)-based systems are stable, while W
1(H
2O)-based systems are not, in further support of the conclusion that W
1 is a hydroxide. Additionally, they found that the W
1(OH
−)/W
2(H
2O) system has greater substrate affinity versus the W
1(OH
−)/W
2(OH
−) system. However, Arg
275 and W
2 were apparently not modeled in the latter system, as would be prescribed for the reactant stage of pathway (I), with Arg
275 as a neutral species and W
2 as a water molecule.
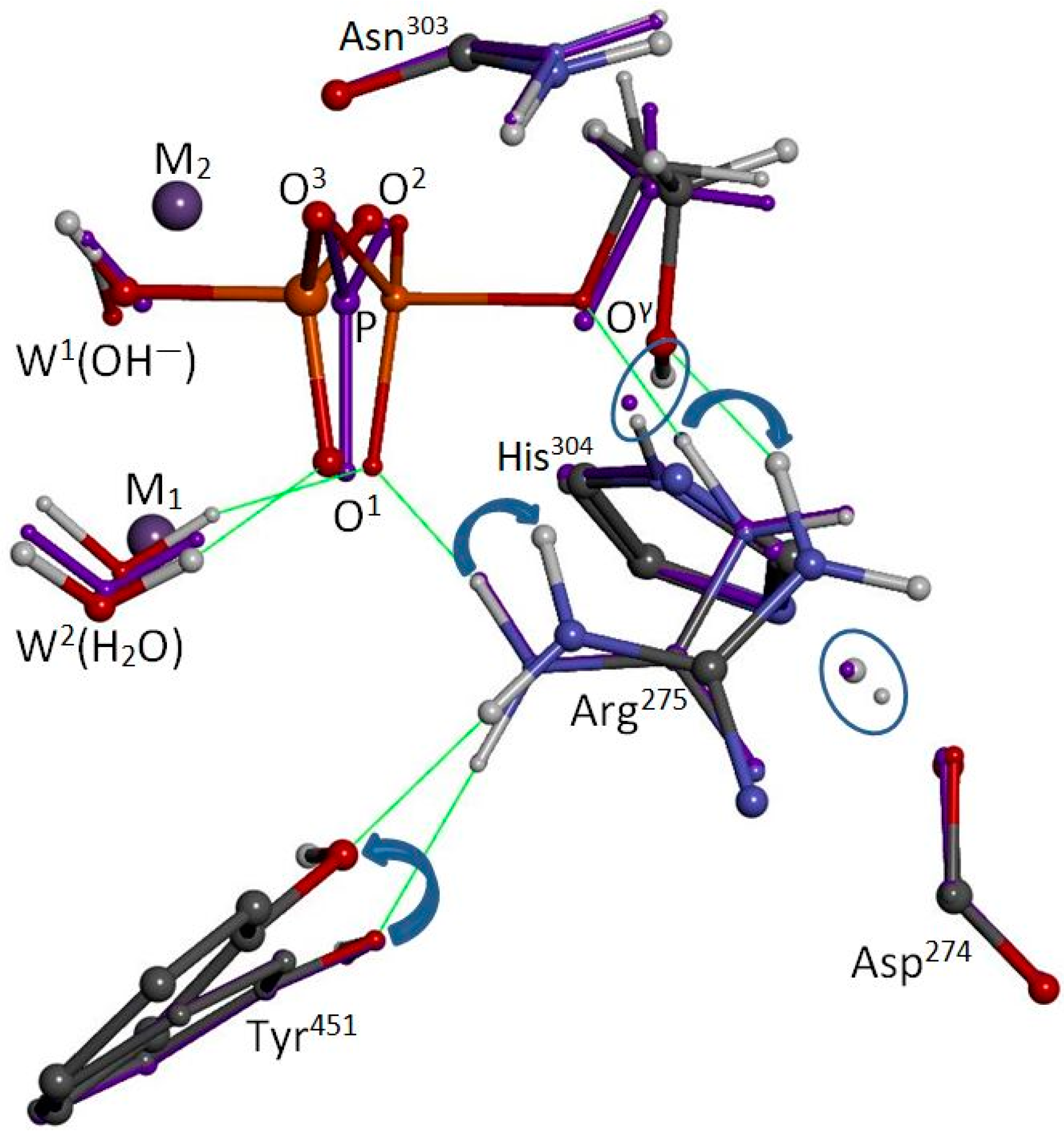
In the present work, we used a PP5 co-crystal (pdb entry 4ZX2 [
20]) as foundation for a 33-residue model of the PP5 catalytic site (
Figure 2). Our goal was to reexamine the W
1(OH
−)/W
2(H
2O) system, and our model was intended to include all relevant moieties that play a role in the reaction and/or serve as steric barriers to prevent nonnative reorientations during optimizations. Two types of calculations were performed: (1) Tests of the behavior of waters associated with the bimetal system and/or protonation states within the catalytic site and (2) tests of the W
1(OH
−)/W
2(H
2O) system for the reactant, transition state, and product stages of the reaction using methylphosphate dianion (CH
3OPO
32−) as a stand-in for phosphoserine. The dianionic species was used because kinetic isotope effect data indicated that PPPs act on the dianionic substrate [
21] and because the protonated species would have enhanced acidity when metal bound.
2. Results
The reactant, TS, and product states for the W
1(OH
−)/W
2(H
2O) system are presented in a truncated form in
Figure 3. According to both gas-phase and solvent-corrected energies, the reaction is exothermic (ΔH ≈ ΔE = −2.5 and −2.0 kcal/mol), with a low activation barrier (ΔH
‡ ≈ ΔE
‡ = +8.6 and +10.0 kcal/mol). Vibrational frequency analysis confirms the reactant and product structures as minima and assigns exactly one imaginary frequency (187
i cm
−1) to the TS. Free energy estimates under the rigid rotor/harmonic oscillator approximation afford ΔG = −7.2 and ΔG
‡ = +6.0 kcal/mol. The coordinates of the optimized structures and an animation of the imaginary mode are available in the
Supplementary Materials. W
2(H
2O) helps to stabilize the substrate via H-bonding with O
1, and this interaction is present in the TS and product states as well. The other expected stabilizing interactions for the phosphate group are also present in all three states, specifically, O
2 with Asn
303 and O
3 with Arg
400. Importantly, Arg
275 binds the substrate and TS in a bidentate fashion at O
1 and O
γ, just as it binds the phosphate ion in the co-crystal. In short, the reactant state is very similar to the geometry of the PP5/phosphate co-crystal, with no unusual reorientations of the five critical residues that were allowed to move during the optimization. The most significant difference between the co-crystal and the optimized reactant state is that the face of Arg
275 is turned about 49° to establish an H-bond with Tyr
451. We add that the reactant state’s coordination distance for W
2, r(W
2 − M
1) = 2.323 Å, is consistent with the X-ray crystal value of 2.295 Å and indicates that the assignment of W
2 as a water molecule is correct.
As in the previous report [
18], the TS is concerted; that is, W
1(OH
−) attacks the phosphorus center at the same time that the exiting seryl/threonyl alkoxide is protonated directly by the His
304/Asp
274 tandem. The primary atomic motion over the course of the reaction is the inversion of the P center along with the transfer of H
+ from His
304 to O
γ. As stated above, Arg
275 stabilizes the TS in a bidentate fashion through interactions with O
1 and O
γ. Post-TS, however, as the product alcohol recedes, Arg
275 turns to maintain the O
γ interaction, and the O
1 interaction is ended. Also post-TS, Tyr
451 rotates inward to maintain its H-bond with Arg
275. In the product state, Arg
275 is isolated from the HPO
42− product, which is ligated to M
1/M
2 through O
1, O
2, and now the bridge hydroxyl oxygen, formerly of the bridge hydroxide W
1(OH
−). As expected with the loss of formal negative charge, the bridge oxygen’s metal coordinations are longer in the product state than in the reactant state; meanwhile, the product alcohol lingers, engaged in an H-bonding trio along with Arg
275 and His
304.
3. Discussion
Crystal structure superposition shows that, in the modeling of Ribeiro et al. [
18], Arg
275 took a position that is partially occupied by Tyr
451 in the enzyme. This nonnative reorientation occurred because of the absence of Tyr
451 in their model, and it permitted the transfer of H
+ from Arg
275 to W
2(OH
−). In the enzyme, Tyr
451 blocks Arg
275 from contact with W
2 when a phosphate-bearing substrate is also ligated to the M
1/M
2 system. Also, the aforementioned reorientation of Asp
274 did not occur in our model because we included Asn
310 and the -NH peptide moiety needed to anchor Asp
274 (see
Figure 1); we froze Arg
400, but the presence of Asp
388 in our model (see
Figure 2) would have prevented an unrealistic reorientation for Arg
400 as well. Future improvements to our computational model would allow Arg
400 and the M
1/M
2 system to move because of their direct contacts with the substrate, TS, and product phosphate ion. Only subtle changes would be expected, and the states, as shown in
Figure 3, should remain qualitatively the same, but the reaction energetics might be impacted. Regarding movement of the M
1/M
2 system, an increase of about 0.12 Å in the M
1…M
2 separation distance did occur for pathways (I) and (II), upon going from reactant to the transition state(s) [
18].
Arg
275 is mobile and is known to take different positions depending upon the ligand present in the co-crystal [
9]. Additionally, we now see that Tyr
451 interacts with Arg
275 during the course of the reaction and that their motions are coupled. Tyr
451 moves to aid the disengagement of Arg
275 from the product phosphate, which, of course, must be achieved as part of displacing the product phosphate for site regeneration. After the TS, Tyr
451 follows Arg
275 while keeping contact with the face of His
244 and moves only a small distance. Generally, however, Tyr
451 is part of the flexible β12-β13 loop and is evidently somewhat mobile. Depending upon the ligand present, Tyr
451 is sometimes vertically farther away from His
244 and is not always engaged in H-bonding with Arg
275 in various crystal structures. Interestingly, PP2A:Arg
89 (counterpart of PP5′s Arg
275) can reorient to engage in an apparent salt-bridge interaction at the interface between the catalytic and regulatory domains of PP2A, at least in the presence of microcystin-LR (pdb entry 3FGA [
22]). In this orientation, PP2A:Arg
89 still contacts PP2A:Tyr
265 (counterpart of PP5′s Tyr
451). If a natural positioning of PP2A:Arg
89 is truly represented in this crystal structure, it seems reasonable to assume that PP2A does not function efficiently when in such a state.
Quantum-based modeling of some small phosphopeptides as PP5 substrates may be possible in the model we have developed here. Of particular interest, Oberoi et al. [
23] investigated the likely positions of the residues adjacent to the p-site serine of Cdc37 (pSer
13) by appending a mimic sequence (15 residues) to the catalytic domain of PP5. The resulting crystal structure (pdb entry 5HPE) indicates that the backbone –C=O and –NH groups of Cdc37:Val
12 interact with Arg
400, while the carboxylate of Cdc37:Asp
14 stacks parallel to the face of Arg
400. Also, the backbone –C=O of Cdc37:Asp
14 interacts with Arg
275. A substrate longer than a Val-pSer-Asp tripeptide, however, will probably require a larger model of the catalytic site to incorporate the additional contacting residues.
Our results are an improvement over both pathways presented in the earlier computational report by Ribeiro et al. [
18], as it is now practical computationally to enlarge the PP5 model system. In their report, they endorsed the W
1(OH
−)/W
2(OH
−) system and identified W
2 as a hydroxide ion: W
2(OH
−). The mechanism they presented, pathway (I), follows an initial H
+ extraction from Arg
275 by W
2(OH
−). The charge-zero Arg
275 binds the substrate and TS at O
1 but not O
γ, and its position overlaps with Tyr
451 (absent in the model) in order to interact with W
2. In short, Arg
275‘s mode of interaction with the substrate/TS in their model is not optimal for substrate/TS stabilization and does not match the expected bidentate binding seen in the PP5/phosphate complex. Moreover, we argue that an unprotonated arginine (pK
b ≈ 1.5) that is H-bonded to an electron-rich phosphate moiety would have a very low pK
b and a strong tendency to gain a proton from solvent. Of course, a protonated arginine would also be more effective in drawing the substrate’s electron density and, thus, promote hydroxide attack.
The pathway offered in [
18] for the W
1(OH
−)/W
2(H
2O) system is endothermic with a high activation energy (>25 kcal/mol), and the assignment option of W
2(H
2O) was consequently discarded [
18]. The mechanism presented for this system in [
18], pathway (II), involves two steps, with an initial proton transfer to the alkoxide coming from Arg
275, not from His
304. Instead, in this study of the W
1(OH
−)/W
2(H
2O) system, we have presented a concerted transition state (TS) in which W
1(OH
−) attacks the phosphate center at the same time that the exiting seryl/threonyl alkoxide is protonated directly by the His
304/Asp
274 tandem. Arg
275, proximal to M
1, stabilizes the substrate and TS in a bidentate fashion, and post-TS movement of the adjacent Tyr
451 decouples Arg
275 from the product phosphate ion. The reaction is exothermic (ΔH = −2.0 kcal/mol), as expected, and occurs in one step with a low activation barrier (ΔH
‡ = +10.0 kcal/mol).
4. Materials and Methods
First, our model was built from pdb entry 4ZX2 [
20] (PP5 catalytic domain, 1.23 Å res.) by removal of the inhibitor (a modified norcantharidin) and by selection of proximal residues of the catalytic site. Second, we applied aldehyde (–CHO) and neutral amine (–NH
2) terminations where we had broken the enzyme’s peptide backbone. Third, hydrogen atoms were added, including one for protonation of His
304. After placement of the methylphosphate dianion, the bridge hydroxide (W
1), and a water of hydration (W
2), the added species and all hydrogens were coarsely optimized using the PM7 semi-empirical method as implemented in Gaussian16 [
24]. Finally, we selected the atoms of the “high-level” region of our hybrid computational system, as indicated in
Figure 2, and carried out ONIOM(UB3LYP/6-31G(d):UPM7) [
25,
26,
27,
28,
29,
30] partial optimizations. (Of course, depending upon the nature of each computational test, we inserted other waters and removed the hydroxide, the His
304 proton, or substrate, as appropriate.) The system, as shown in
Figure 2, has zero total charge. During optimizations, the following residues were allowed to move: His
304, Asp
274, Arg
275, Tyr
451, and Asn
303, as were any added species. The first two residues are the His/Asp tandem. The second two, Arg
275 and Tyr
451, are known to freely move to accommodate ligand binding and were adjusted to approximate their PP5/phosphate co-crystal positions. The last, Asn
303, coordinates M
2 and the substrate’s phosphoryl group at O
2. All calculations were carried out using the Gaussian16 suite of programs [
24].
The Mn
2+/Mn
2+ system is an antiferromagnetic singlet state (5 “up”/5 “down”). The proper “high-level” B3LYP electronic state was attained by first constructing an appropriate spin-unrestricted guess wavefunction, as in earlier work [
18,
31]. The “low-level” PM7 wavefunctions could not be manipulated in the same manner to get antiferromagnetic states in the current implementation of Gaussian16; instead, we converged to stable spin-unrestricted open-shell singlets (1 “up”/1 “down”) generated from closed-shell wavefunctions (STABLE = opt). PM7 convergence was aided by using the quadratic convergence option (SCF = yqc). The transition state search was initiated by first carrying out a partial optimization with the inversion dihedral frozen in a nearly planar conformation (GEOM = addgic). Optimizations of the reactant, TS, and product systems were completed using INT = grid = superfine. Optimizations were judged complete when the RMS force value met the default target, as satisfying the full set of default convergence criteria was not practical for a system of this size. Analytic frequencies were computed to confirm the curvature of the energy surface at the two stable structures and the transition state. Single-point calculations using the default continuum solvation model in Gaussian16 (SCRF = solvent = water, sas) were used to estimate solvent-corrected energies.