2.1. Influence of Reaction Temperature on Char Gasification
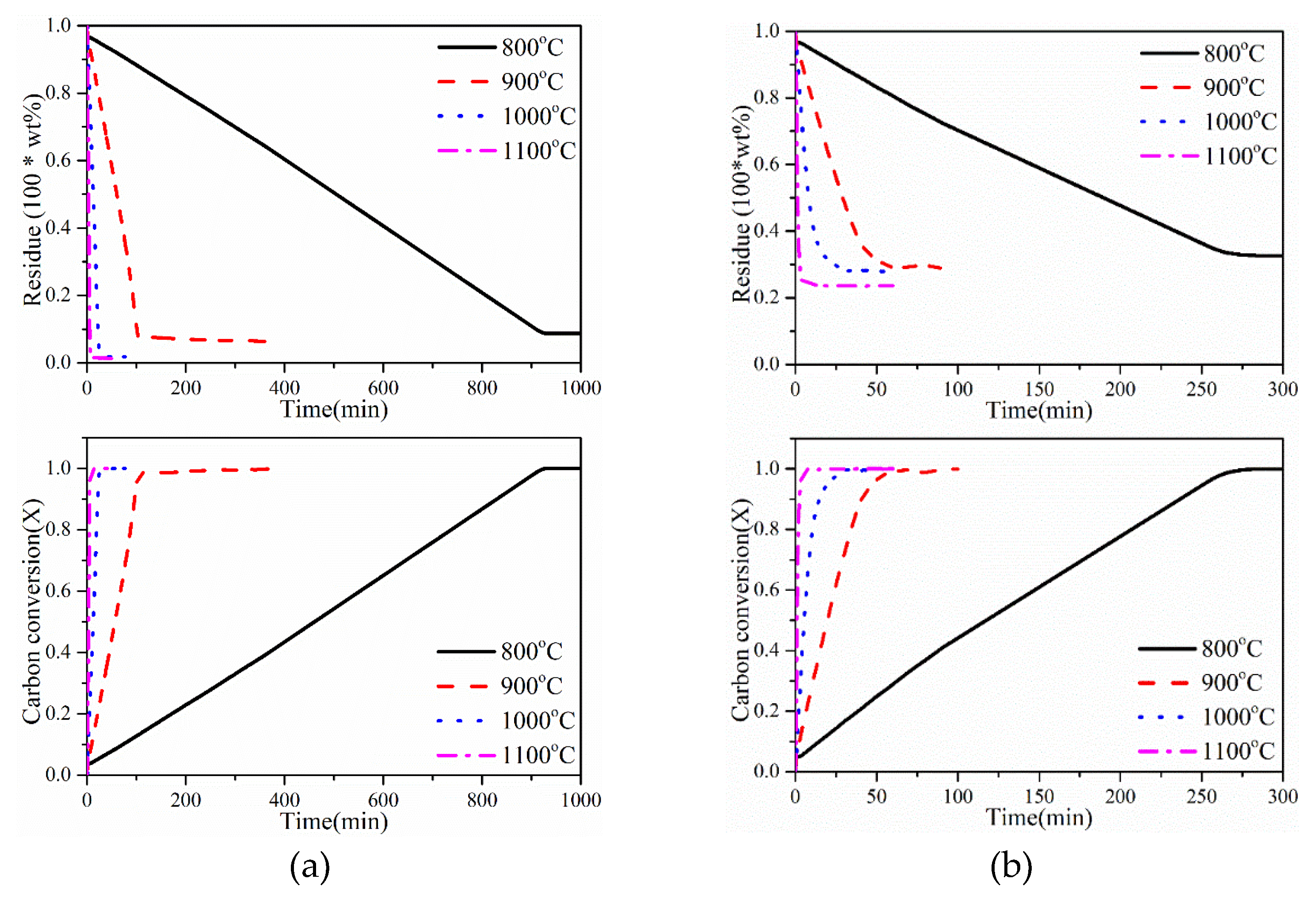
Figure 1 presents the influence of temperature on RAW coal and AW (acid-washing) coal char gasification. Two samples bear a resemblance that increasing temperature drastically shortens the reaction time and this result is foreseen because, in fact, Boudouard reaction is favored at high temperatures. In detail, it is clear that the reaction proceeds are very slow at low temperature (800 °C) and complete conversion is reached even more than 900 min and 260 min for AW and RAW samples. While with a 100 °C increase in temperature, completion of char reaction is achieved in approximately 100 min and 50 min respectively. These results demonstrate that the reaction temperature plays a dominant role in gasification kinetics.
Table 1 shows the change of gasification characteristic parameters as a function of reacting temperature. It can be found that these parameters are consistent with the changing trend of the curve in
Figure 1. With increasing temperature, the
time is getting shorter and a higher reactivity index is achieved, meaning a faster gasification rate. Meanwhile, the reactivity index of AW char increases by a large margin than RAW char. The reactivity index of AW char in 1000 °C is 37 times of that in 800 °C, but it is only 20 times for RAW char, which implies that the temperature has a deeper effect during the gasification process in terms of change in gasification rate rather than other factors.
It is worthy of notice that the difference of conversion degree from 1000 to 1100 °C are negligible compared to lower temperatures, which can be explained by the change of reaction control regime. It is considered that the process is under control of chemical reaction when the temperature is under 1000 °C, whereas the synergy of chemical reaction control and pore diffusion regime plays a part in 1100 °C. Some researchers [
11,
12,
13] have reached the same conclusion that 1000 °C is the critical temperature in the CO
2 gasification of coal char.
2.2. Influence of Various Catalysts on Char Gasification
To better evaluate the catalytic efficacy of different metal chlorides in gasification, the TGA data versus time measured at the same final temperature are shown in
Figure 2. Results show that the catalysis of metal chlorides exhibits different behaviors. As gasified at 800 °C, the time needed to achieve complete conversion for AW coal char was approximately 900 min, however it is only 50 min and 130 min for AW-CaCl
2 and AW-KCl char, which implies that the AW gasification rate is 18 and 7 times slower than catalytic gasification rate respectively. The presence of CaCl
2 and KCl greatly enhances the gasification rate and sharply shortens reaction time. Extensive research [
4,
14,
15] has confirmed that AAEM as catalysts show outstanding performance in both pyrolysis and gasification. Contrarily, the additive of ZnCl
2 inhibits the catalytic gasification when the temperature is above 900 °C, contributing to lower gasification reactivity than AW sample. As for the use of NiCl
2, the weight loss curve is far akin to RAW coal char sample, which indicates that the NiCl
2 catalysts act as the same promoting function with inherent minerals in Raw sample. All results obtained above suggest that the gasification reactivity follows the anions order as: Ca > K > Ni > Zn. Such a trend is quite similar to those represented in the research for the catalytic gasification of char samples with CO
2 [
6,
16]. So the catalytic ability of metal chlorides can be divided into two groups, one group including CaCl
2, KCl, and NiCl
2 positively catalyzes the reaction, while another such as ZnCl
2 hinders the gasification process.
Concerning the mass of remaining residue after the gasification process, there are evident differences between AW samples and others. It is anticipated that the catalysts remain in different forms after reaction, while the inherent minerals only account for 0.52% in AW sample after acid-washing, thus almost complete conversion of AW coal char can be observed. It is of interest the residual amount of AW-CaCl
2 continues to be very high compared with other four samples. Matsukata at al. [
17] suggest that catalysts, such as calcium, were hard to vaporize and diffuse into the bulk, most of which kept constant on the char surface. There was no appreciable variation in Ca content at any carbon conversion level. In this case, CaCl
2 catalysts in the residue are left after the reaction. We note that the residuary amount of three samples with K, Zn, Ni catalysts after the reaction is fairly close, reveling that the metal chlorides can facilitate the gasification rate, but the final conversion is dependent on the intrinsic characteristics of char rather than these catalysts.
Moreover, to further elucidate the mechanism of catalysis,
Figure 3 demonstrates the reactivity data via carbon conversion at 1000 °C. It is obvious that the Ca and K exist in the coal chars largely enhance the char reactivity and change the reactivity profiles; this character is not noticeable with AW-Ni, -Zn chars. Du at al. [
18] points out that the catalysis species rather than active structures control the char reactivity with conversion, thus it is considered that the addition of metal chlorides alters reactivity profiles.
The reactivity of KCl-char is lower than that of CaCl
2-char at conversion ranges of 0.04 to 0.7, when the carbon conversion reaches a eigenvalue (
Xi = 0.7), the reactivity of KCl-char becomes the higher one. The shift in the X value suggests that CaCl
2 and KCl show different mechanisms during CO
2 gasification. Previous work has proved that the dispersion conditions [
19,
20] and the mobility of the catalysts [
21,
22] are significant properties for catalytic reactivities. Therefore, the good dispersion and the persistence of Ca species at high temperature both contribute to a sharp increase in char reactivity. While the saturation of Ca species in high conversion will promote sintering of Ca resulting in a poor dispersion of catalysts, corresponding to the remarkable decrease in reactivity, inducing lower reactivity than KCl-char. In contrast, the loss of KCl catalysts is the dominant factor for the deactivation process and the K metals are prone to vaporize and easily migrate into the bulk of carbon, increasing the reactivity and then reaching a maximum with the higher conversion. Consequently, the reactivity of KCl-char maintains at a relatively high level until the end of gasification.
2.4. Mechanism of Catalytic Gasification
The phase of metal chloride has changed due to the preparation of char by pyrolysis at 1000 °C, then the new phase may catalyze gasification process. In the previous study [
15,
23], the morphs of KCl, CaCl
2, and NiCl
2 are confirmed to become KCl, CaCO
3 and CaCl
2, Ni after pyrolysis respectively, resulting in the different mechanism of each sample. XRD measurements were carried out to find out the mechanism of each catalyst, and the spectra of three samples after gasification at 1000 °C is exhibited in
Figure 6. In the gasification reaction, the migration and transformation of K species largely occurred. It can be seen from
Figure 6a, most peaks of KCl disappeared in the sample after gasification, meaning the consumption and deactivation of the catalysts. While the other crystallites of K species were formed during the process, including C
nK and K
2CO
3. A reaction cycle based on the Electron Donor-Acceptor (EDA) complexes proposed by Wen at al. [
24] could account for this formation. As soon as the K metals are generated in the gasification process, some EDA complexes C
nK (n = 8, 16, 24) will be formed to catalyze the reactions with CO
2:
The peaks of CaCl
2, CaCO
3, and CaO are observed in the
Figure 6b, and the CaCl
2 first decomposes with water in coal as shown in Equation (7), then the resulting CaO tends to react with CO
2 to generate CaCO
3.
The mechanism of the sample can be explained by oxidation-reduction mechanism [
7], which involves the following reactions:
It is inferred that the phases of CaCl2 and CaCO3 both act as catalysts in the process. Therefore, the catalytic effects of CaCl2 is better than KCl with single mechanism, but the sintering of carbonates hinders the further catalysis in the later gasification process.
As for NiCl
2-char sample, spectra of Ni and NiO are found in the XRD results after the gasification. This may be described by the following reaction:
It is noteworthy that the lack of C atoms is unable to fully support the reduction reaction of NiO, conducing the coexisting of Ni/NiO. So, the Ni atoms and NiO synergistically work as catalysts in the reaction.