Techno-Economic and Environmental Analysis for Direct Catalytic Conversion of CO2 to Methanol and Liquid/High-Calorie-SNG Fuels
Abstract
:1. Introduction
2. Results
2.1. Model Validation
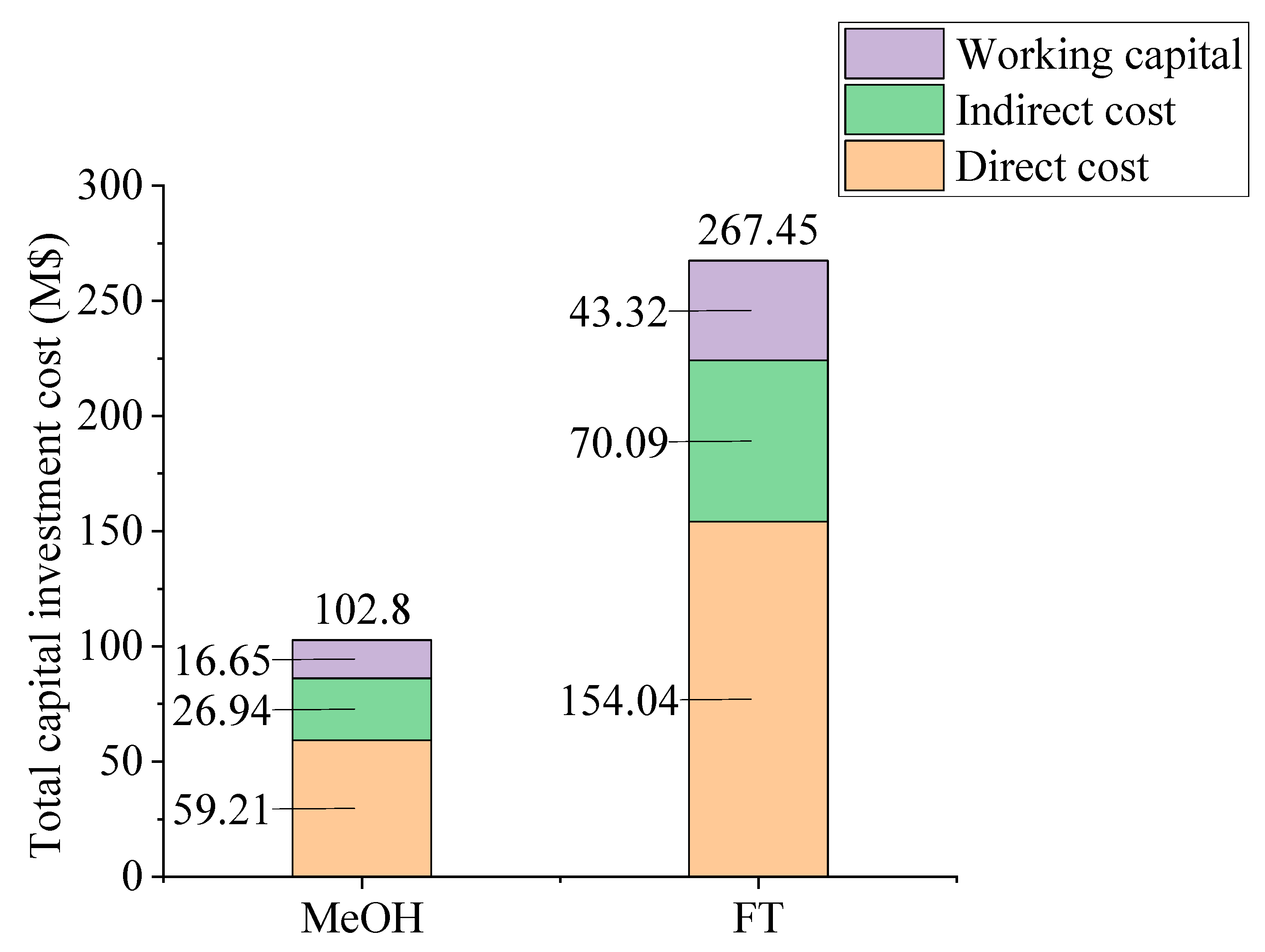
2.2. Economic Analysis Results
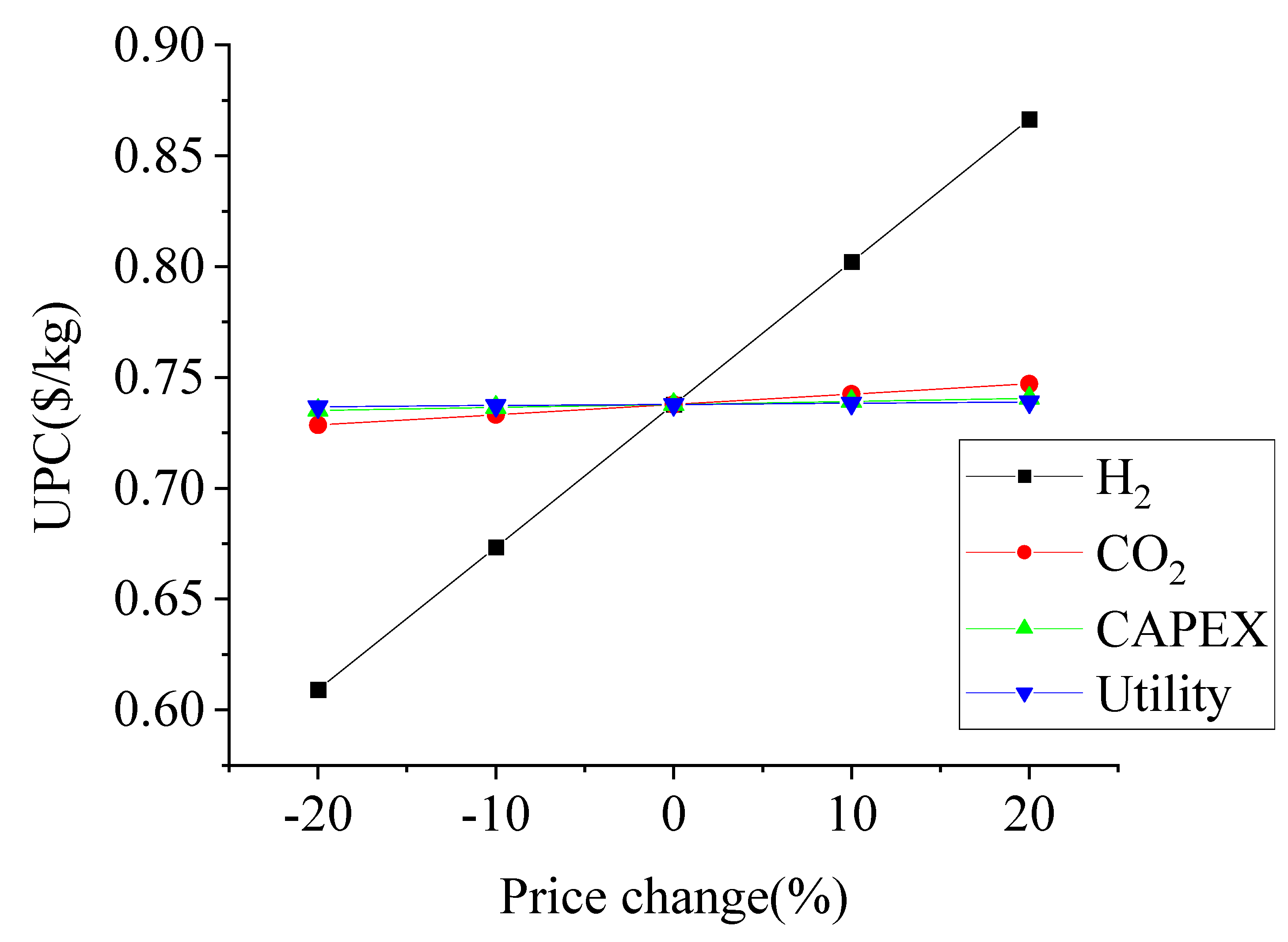
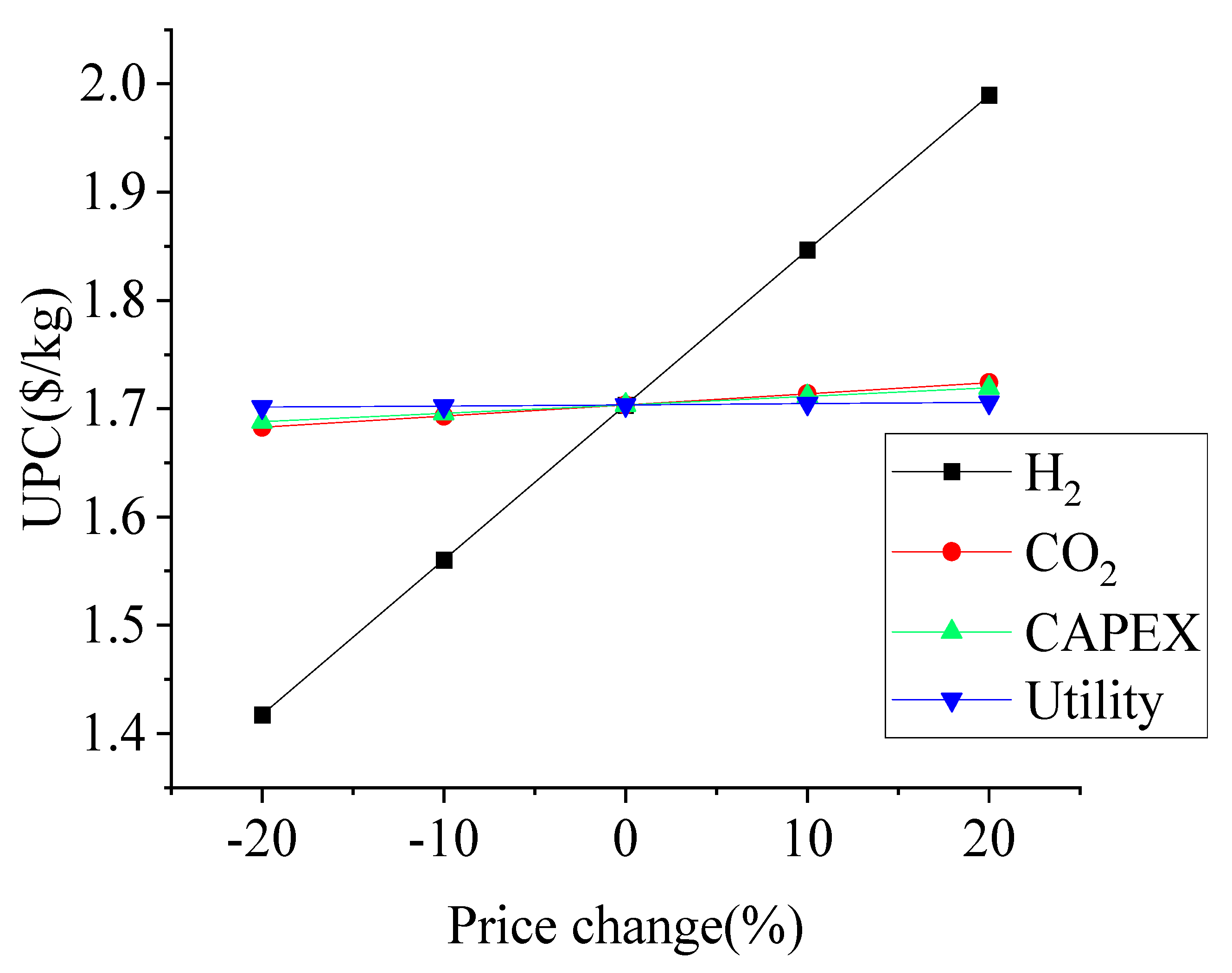
Sensitivity Analysis
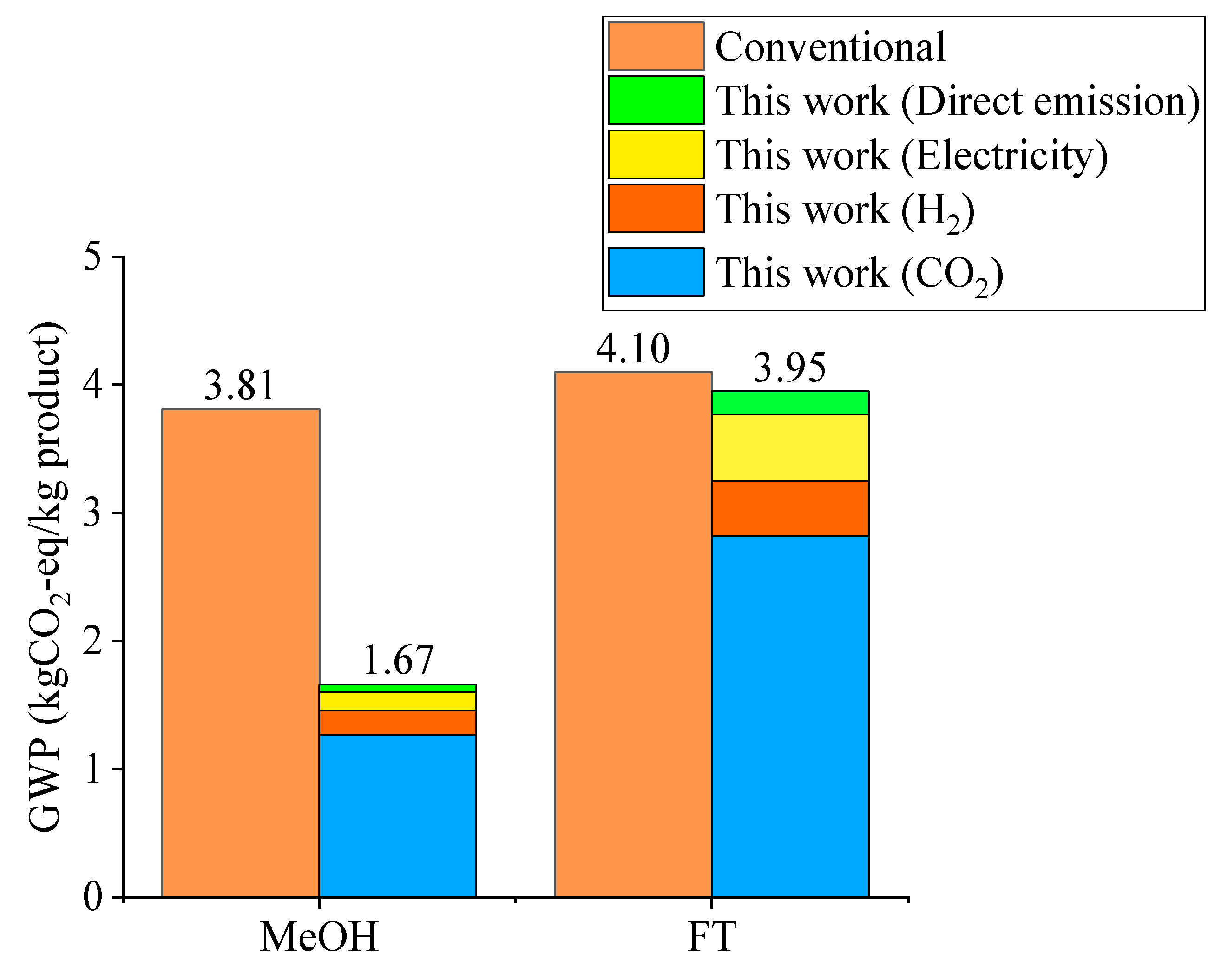
2.3. Environmental Assessment Results
2.4. Economic and Environmental Assessment Result: Comparison of Methanol Synthesis and Fischer–Tropsch Process Models
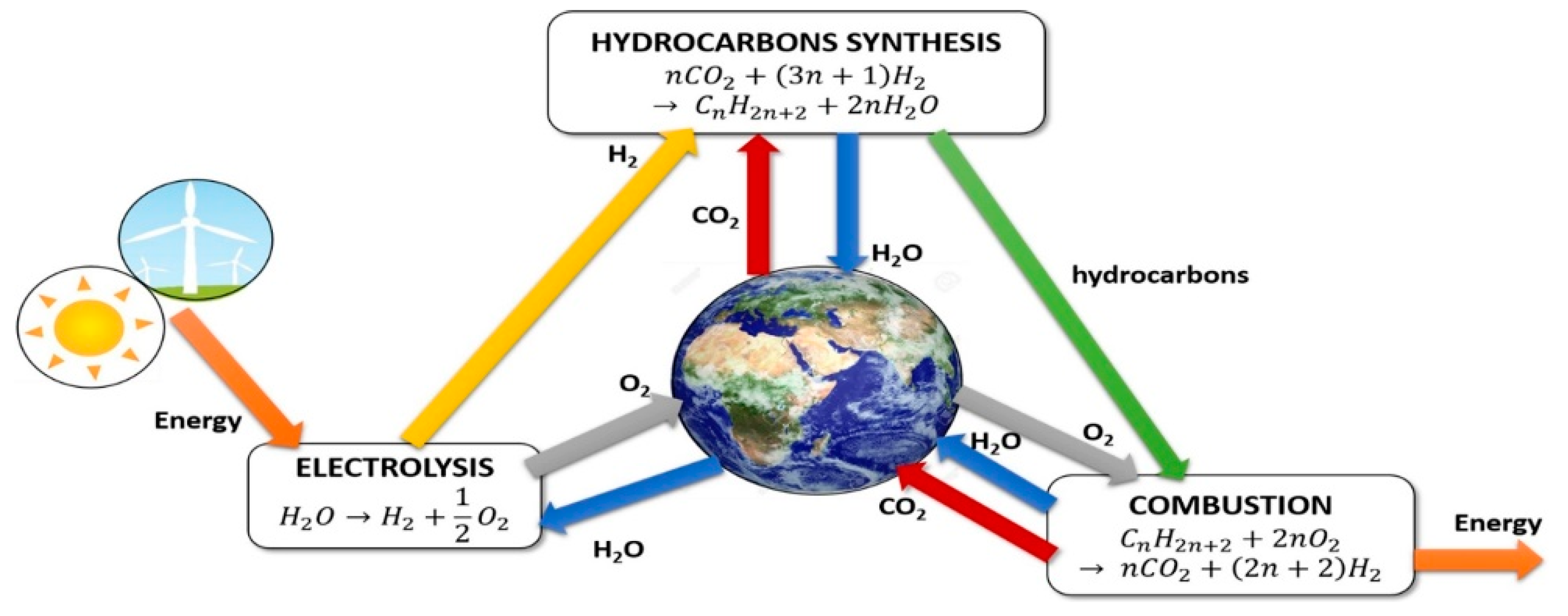
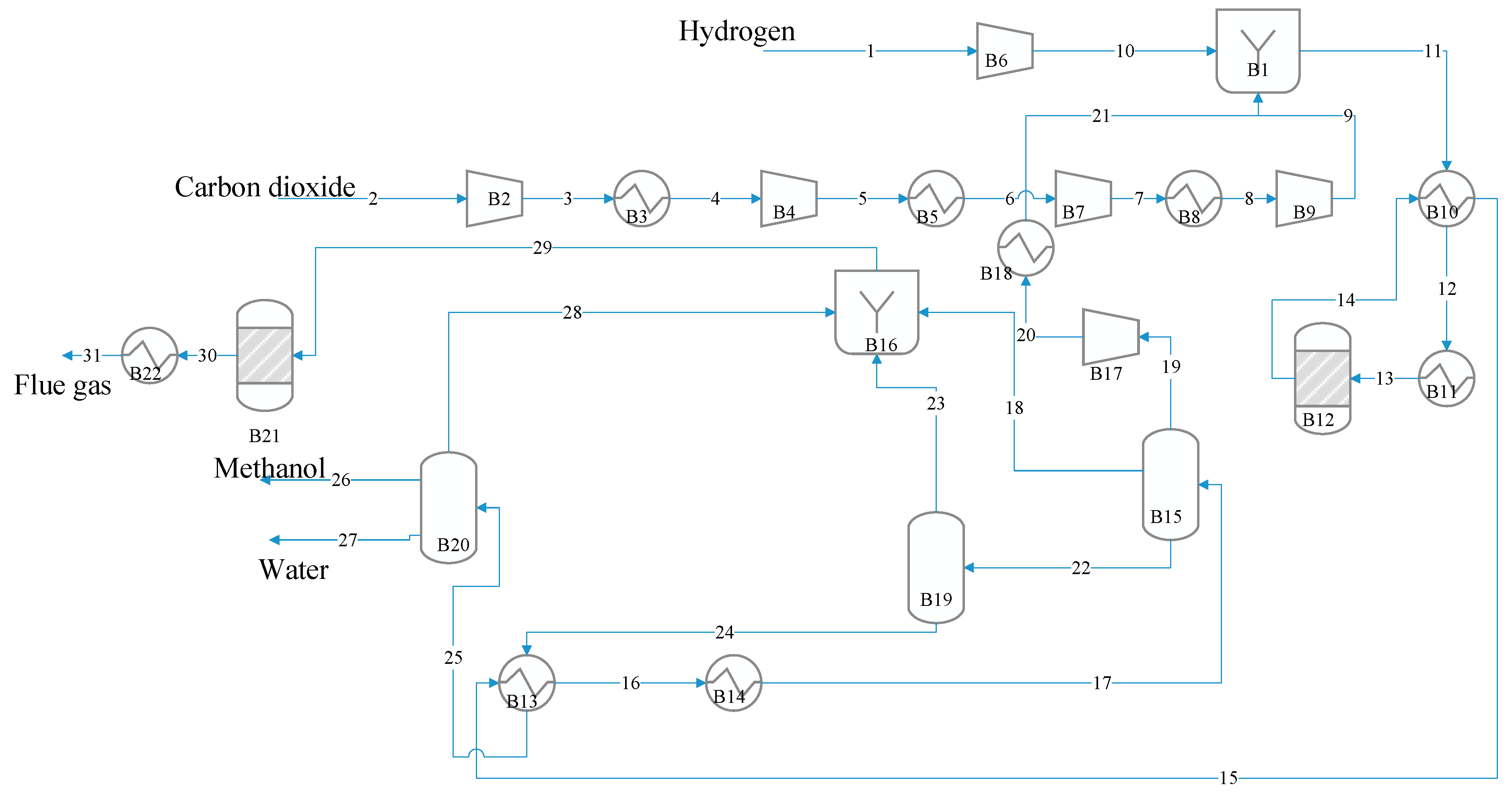
3. Process Description and Design
3.1. Assumptions
3.2. Methanol Synthesis
3.3. Fischer–Tropsch-Based Liquid/High-Calorie-SNG Fuel Synthesis
4. Process Assessment Methodology
4.1. Cost Estimation
4.2. Environmental Assessment
5. Conclusions and Recommendation
- The development of advanced methods to lower the price of renewable H2, such as improving the high electricity demand of electrolysis.
- The innovation of CO2 hydrogenation catalysts which are stable, selective, and capable of working at lower pressure to insure economic feasibility.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| CAPEX | Capital Expenditure |
| CO2-eq | Carbon dioxide equivalent |
| CEPCI | Chemical Engineering Plant Cost Index |
| FT | Fischer–Tropsch |
| GHSV | Gas hourly space velocity |
| GWP | Global warming potential |
| LCA | Life cycle assessment |
| MeOH | Methanol |
| M$ | Million USD |
| NPV | Net positive value |
| OPEX | Operating Expenditure |
| PSA | Pressure swing adsorption |
| PTG | Power to gas |
| PTL | Power to liquid |
| RWGS | Reversible water gas shift reaction |
| SNG | Synthetic natural gas |
| SRK | Redlich–Kwong–Soave |
| UPC | Unit production cost |
| yr | Year |
References
- Atsbha, T.A.; Yoon, T.; Seongho, P.; Lee, C.-J. A review on the catalytic conversion of CO2 using H2 for synthesis of CO, methanol, and hydrocarbons. J. CO2 Util. 2021, 44, 101413. [Google Scholar] [CrossRef]
- Michailos, S.; McCord, S.; Sick, V.; Stokes, G.; Styring, P. Dimethyl ether synthesis via captured CO2 hydrogenation within the power to liquids concept: A techno-economic assessment. Energy Convers. Manag. 2019, 184, 262–276. [Google Scholar] [CrossRef]
- Gonçalves, A.; Puna, J.F.; Guerra, L.F.; Rodrigues, J.C.; Gomes, J.F.; Santos, M.T.; Alves, D. Towards the development of syngas/biomethane electrolytic production, using liquefied biomass and heterogeneous catalyst. Energies 2019, 12, 3787. [Google Scholar] [CrossRef] [Green Version]
- Buttler, A.; Spliethoff, H. Current status of water electrolysis for energy storage, grid balancing and sector coupling via power-to-gas and power-to-liquids: A review. Renew. Sustain. Energy Rev. 2018, 82, 2440–2454. [Google Scholar] [CrossRef]
- France, L.J.; Edwards, P.P.; Kuznetsov, V.L.; Almegren, H. The Indirect and Direct Conversion of CO2 into Higher Carbon Fuels; Elsevier: Amsterdam, The Netherlands, 2015; pp. 161–182. [Google Scholar]
- Panzone, C.; Philippe, R.; Chappaz, A.; Fongarland, P.; Bengaouer, A. Power-to-Liquid catalytic CO2 valorization into fuels and chemicals: Focus on the Fischer-Tropsch route. J. CO2 Util. 2020, 38, 314–347. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, C.; Gao, P.; Wang, H.; Li, X.; Zhong, L.; Wei, W.; Sun, Y. A review of the catalytic hydrogenation of carbon dioxide into value-added hydrocarbons. Catal. Sci. Technol. 2017, 7, 4580–4598. [Google Scholar] [CrossRef]
- Do, T.N.; Kim, J. Green C2–C4 hydrocarbon production through direct CO2 hydrogenation with renewable hydrogen: Process development and techno-economic analysis. Energy Convers. Manag. 2020, 214, 112866. [Google Scholar] [CrossRef]
- Pérez-Fortes, M.; Schöneberger, J.C.; Boulamanti, A.; Tzimas, E. Methanol synthesis using captured CO2 as raw material: Techno-economic and environmental assessment. Appl. Energy 2016, 161, 718–732. [Google Scholar] [CrossRef]
- Zhang, C.; Gao, R.; Jun, K.-W.; Kim, S.K.; Hwang, S.-M.; Park, H.-G.; Guan, G. Direct conversion of carbon dioxide to liquid fuels and synthetic natural gas using renewable power: Techno-economic analysis. J. CO2 Util. 2019, 34, 293–302. [Google Scholar] [CrossRef]
- Atsonios, K.; Panopoulos, K.D.; Kakaras, E. Investigation of technical and economic aspects for methanol production through CO2 hydrogenation. Int. J. Hydrogen Energy 2016, 41, 2202–2214. [Google Scholar] [CrossRef]
- Szima, S.; Cormos, C.-C. Improving methanol synthesis from carbon-free H2 and captured CO2: A techno-economic and environmental evaluation. J. CO2 Util. 2018, 24, 555–563. [Google Scholar] [CrossRef]
- Rivarolo, M.; Bellotti, D.; Magistri, L.; Massardo, A. Feasibility study of methanol production from different renewable sources and thermo-economic analysis. Int. J. Hydrogen Energy 2016, 41, 2105–2116. [Google Scholar] [CrossRef]
- Zhang, C.; Jun, K.-W.; Gao, R.; Kwak, G.; Park, H.-G. Carbon dioxide utilization in a gas-to-methanol process combined with CO2/Steam-mixed reforming: Techno-economic analysis. Fuel 2017, 190, 303–311. [Google Scholar] [CrossRef]
- Asif, M.; Gao, X.; Lv, H.; Xi, X.; Dong, P. Catalytic hydrogenation of CO2 from 600 MW supercritical coal power plant to produce methanol: A techno-economic analysis. Int. J. Hydrogen Energy 2018, 43, 2726–2741. [Google Scholar] [CrossRef]
- Alsayegh, S.; Johnson, J.; Ohs, B.; Wessling, M. Methanol production via direct carbon dioxide hydrogenation using hydrogen from photocatalytic water splitting: Process development and techno-economic analysis. J. Clean. Prod. 2019, 208, 1446–1458. [Google Scholar] [CrossRef]
- Smejkal, Q.; Rodemerck, U.; Wagner, E.; Baerns, M. Economic Assessment of the Hydrogenation of CO2 to Liquid Fuels and Petrochemical Feedstock. Chem. Ing. Tech. 2014, 86, 679–686. [Google Scholar] [CrossRef]
- Kiss, A.A.; Pragt, J.; Vos, H.; Bargeman, G.; de Groot, M. Novel efficient process for methanol synthesis by CO2 hydrogenation. Chem. Eng. J. 2016, 284, 260–269. [Google Scholar] [CrossRef] [Green Version]
- Numpilai, T.; Witoon, T.; Chanlek, N.; Limphirat, W.; Bonura, G.; Chareonpanich, M.; Limtrakul, J. Structure–activity relationships of Fe-Co/K-Al2O3 catalysts calcined at different temperatures for CO2 hydrogenation to light olefins. Appl. Catal. A: Gen. 2017, 547, 219–229. [Google Scholar] [CrossRef]
- Wernet, G.; Bauer, C.; Steubing, B.; Reinhard, J.; Moreno-Ruiz, E.; Weidema, B.P. The ecoinvent database version 3 (part I): Overview and methodology. Int. J. Life Cycle Assess. 2016, 21, 1218–1230. [Google Scholar] [CrossRef]
- Methanex Methanol Price Sheet. Methanex Corporation. 2021. Available online: www.methanex.com/our-business/pricing (accessed on 23 March 2021).
- Mikkelsen, M.; Jørgensen, M.; Krebs, F.C. The teraton challenge. A review of fixation and transformation of carbon dioxide. Energy Environ. Sci. 2010, 3, 43–81. [Google Scholar] [CrossRef]
- Venvik, H.J.; Yang, J. Catalysis in microstructured reactors: Short review on small-scale syngas production and further conversion into methanol, DME and Fischer-Tropsch products. Catal. Today 2017, 285, 135–146. [Google Scholar] [CrossRef]
- Li, W.; Wang, H.; Jiang, X.; Zhu, J.; Liu, Z.; Guo, X.; Song, C. A short review of recent advances in CO2 hydrogenation to hydrocarbons over heterogeneous catalysts. RSC Adv. 2018, 8, 7651–7669. [Google Scholar] [CrossRef] [Green Version]
- Mahmoudi, H.; Mahmoudi, M.; Doustdar, O.; Jahangiri, H.; Tsolakis, A.; Gu, S.; LechWyszynski, M. A review of Fischer Tropsch synthesis process, mechanism, surface chemistry and catalyst formulation. Biofuels Eng. 2017, 2, 11–31. [Google Scholar] [CrossRef]
- Xie, T.; Wang, J.; Ding, F.; Zhang, A.; Li, W.; Guo, X.; Song, C. CO2 hydrogenation to hydrocarbons over alumina-supported iron catalyst: Effect of support pore size. J. CO2 Util. 2017, 19, 202–208. [Google Scholar] [CrossRef]
- Alfa Aesar, Copper Based Methanol Synthesis Catalyst. 2014. Available online: http://www.alfa.com/en/catalog (accessed on 20 February 2021).
- Spath, P.L.; Mann, M.K. Life Cycle Assessment of Renewable Hydrogen Production via Wind/Electrolysis. Nrel 2004, 2004, 1–13. [Google Scholar]



| CO2 Conversion (%) | Methanol Yield (%) | |||
|---|---|---|---|---|
| Temperature (°C) | [18] | This Work | [18] | This Work |
| 210 | 12.44 | 12.38 | 11.52 | 11.47 |
| 230 | 19.57 | 19.50 | 15.97 | 15.92 |
| 250 | 23.98 | 23.98 | 15.30 | 15.30 |
| 270 | 24.04 | 24.05 | 10.58 | 10.59 |
| Parameters | FT Reactor Stream Outlet (This Work) | FT Reactor Stream Outlet [19] |
|---|---|---|
| Temperature | 320 °C | 320 °C |
| Pressure | 20 bar | 20 bar |
| Yield | 12.2% (liquid fuel) | 12.2% (liquid fuel) |
| 32.2% (high-calorie SNG) | 32.2% (high-calorie SNG) |
| Scheme | 1 | 2 | 3 | 4 | 5 |
| % Reduction of H2 Cost | 65 | 70 | 75 | 80 | 85 |
| Reduced H2 Cost ($/kg) | 1.24 | 1.06 | 0.88 | 0.71 | 0.53 |
| Parameter | Value |
|---|---|
| Project lifetime | 20 years |
| Operating hours per period (year) | 8000 |
| Number of weeks per period (year) | 52 |
| Income tax rate | 35% |
| Discount rate | 8% |
| Depreciation | Straight line |
| Depreciation period | 20 |
| Location | South Korea |
| CO2 cost | $0.035/kg [7] |
| H2 cost | $3.53/kg [7] |
| Cooling water cost | $0.028/ton [2] |
| Electricity cost | $0.063/kWh [9] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atsbha, T.A.; Yoon, T.; Yoo, B.-H.; Lee, C.-J. Techno-Economic and Environmental Analysis for Direct Catalytic Conversion of CO2 to Methanol and Liquid/High-Calorie-SNG Fuels. Catalysts 2021, 11, 687. https://doi.org/10.3390/catal11060687
Atsbha TA, Yoon T, Yoo B-H, Lee C-J. Techno-Economic and Environmental Analysis for Direct Catalytic Conversion of CO2 to Methanol and Liquid/High-Calorie-SNG Fuels. Catalysts. 2021; 11(6):687. https://doi.org/10.3390/catal11060687
Chicago/Turabian StyleAtsbha, Tesfalem Aregawi, Taeksang Yoon, Byung-Hoon Yoo, and Chul-Jin Lee. 2021. "Techno-Economic and Environmental Analysis for Direct Catalytic Conversion of CO2 to Methanol and Liquid/High-Calorie-SNG Fuels" Catalysts 11, no. 6: 687. https://doi.org/10.3390/catal11060687






