Highly Dispersed Pd Species Supported on CeO2 Catalyst for Lean Methane Combustion: The Effect of the Occurrence State of Surface Pd Species on the Catalytic Activity
Abstract
:1. Introduction
2. Results and Discussion
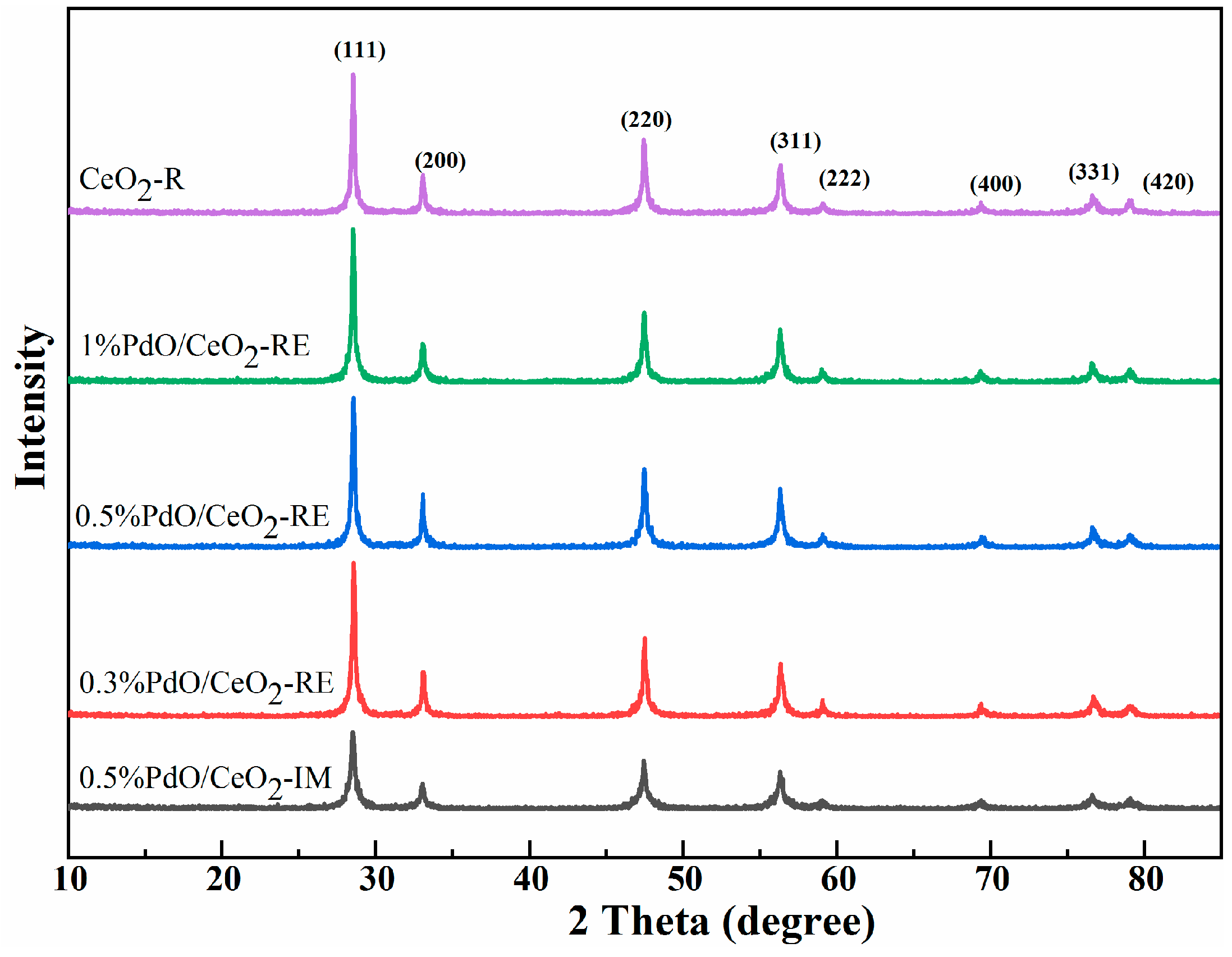
2.1. Structural Properties and Morphologies of Samples
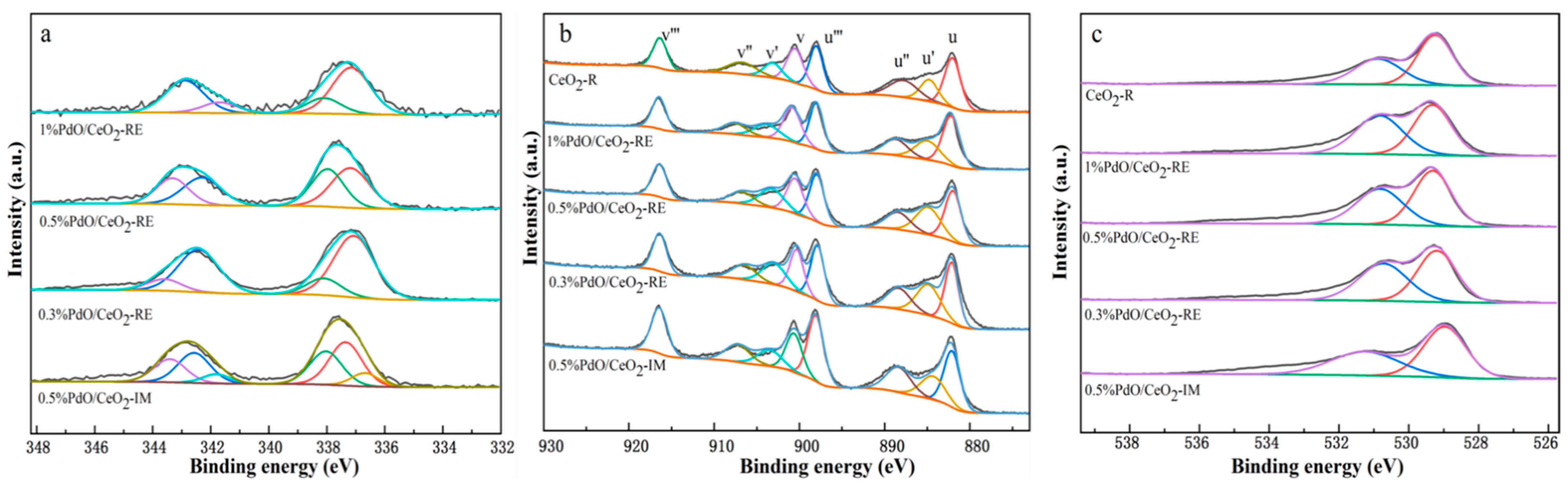
2.2. Chemical and Electronic States
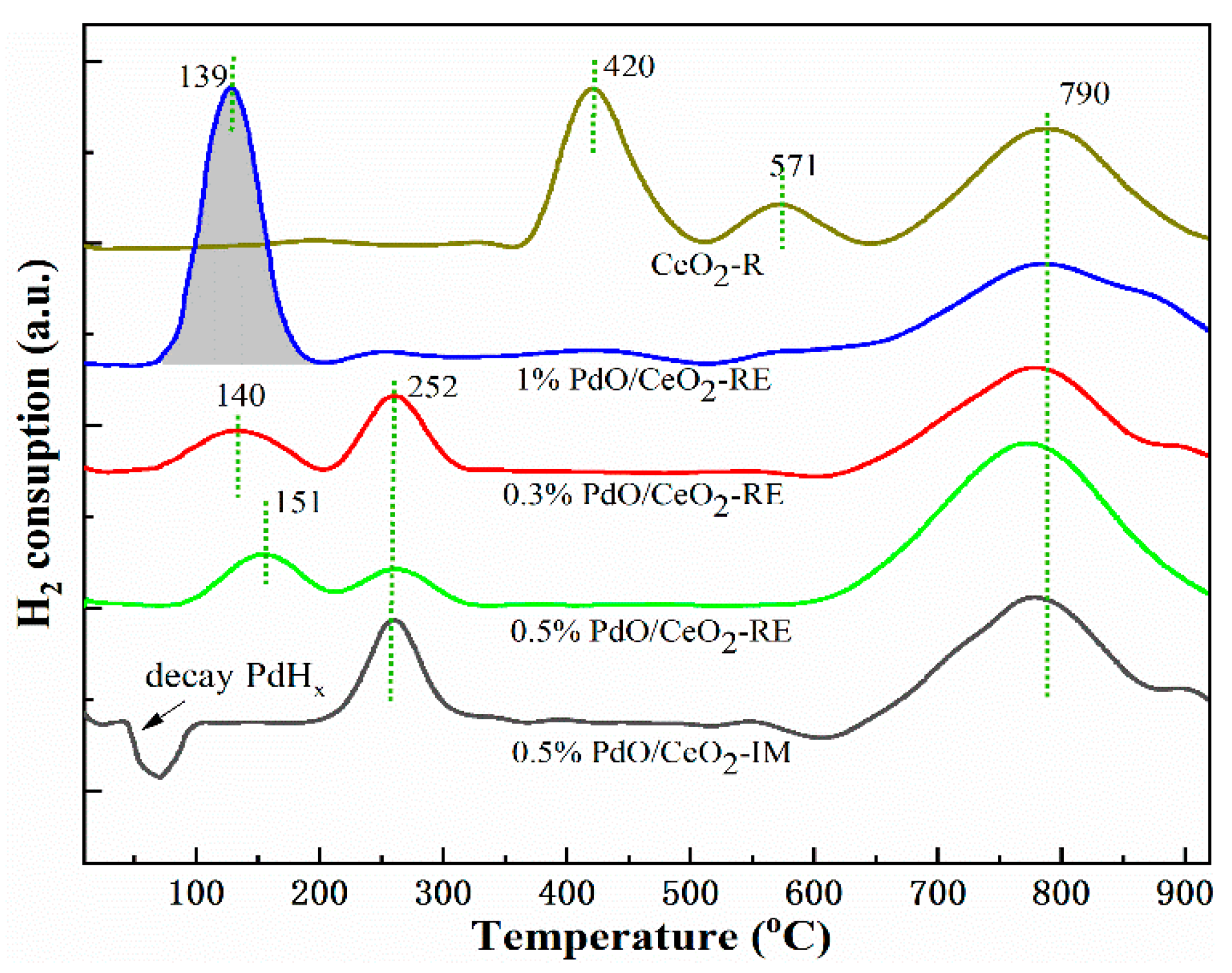
2.3. Redox Ability
2.4. Catalytic CH4 Oxidation
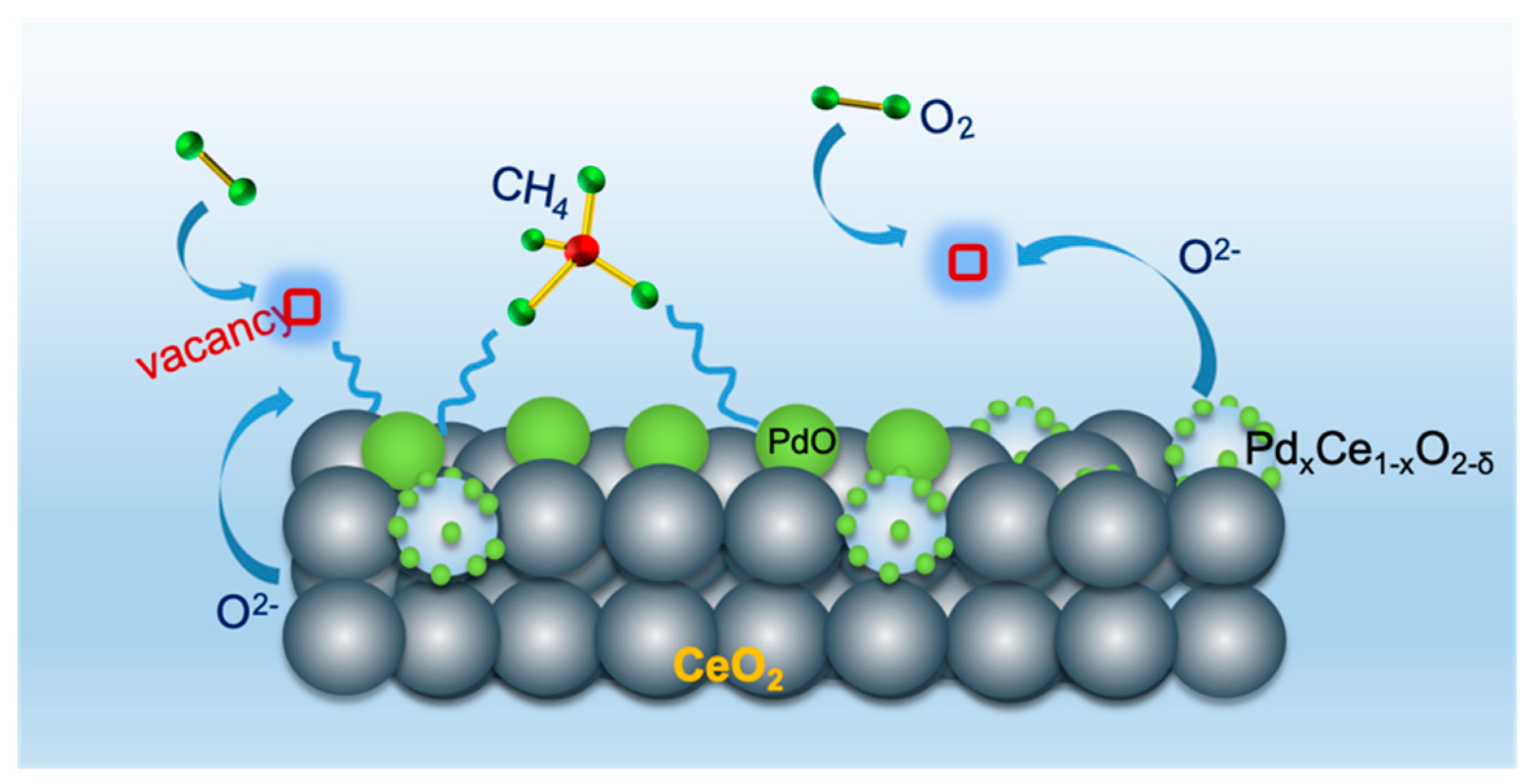
3. Discussion
4. Experimental Section
4.1. Catalyst Synthesis
4.1.1. Synthesis of Ceria Nanorods Support (CeO2-R)
4.1.2. Synthesis of Pd/CeO2 Catalyst
4.2. Catalyst Characterization
4.3. Evaluation of the Catalytic Performance
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Yang, H.; Xie, P.; Ni, L.; Flower, R.J. Underestimation of CH4 emission from freshwater lakes in China. Environ. Sci. Technol. 2011, 45, 4203–4204. [Google Scholar] [CrossRef]
- Choudhary, T.V.; Banerjee, S.; Choudhary, V.R. Catalysts for combustion of methane and lower alkanes. Appl. Catal. A Gen. 2002, 234, 1–23. [Google Scholar] [CrossRef]
- Liu, C.J.; Ye, J.; Jiang, J.; Pan, Y. Progresses in the preparation of coke resistant Ni-based catalyst for steam and CO2 reforming of methane. ChemCatChem 2011, 3, 529–541. [Google Scholar] [CrossRef]
- Murata, K.; Mahara, Y.; Ohyama, J.; Yamamoto, Y.; Arai, S.; Satsuma, A. The Metal–Support Interaction Concerning the Particle Size Effect of Pd/Al2O3 on Methane Combustion. Angew. Chem. Int. Ed. 2017, 56, 15993–15997. [Google Scholar] [CrossRef]
- Son, I.H.; Lane, A.M.; Johnson, D.T. The study of the deactivation of water-pretreated Pt/γ-Al2O3 for low-temperature selective CO oxidation in hydrogen. J. Power Source 2003, 124, 415–419. [Google Scholar] [CrossRef]
- Rajasree, R.; Hoebink, J.H.B.J.; Schouten, J.C. Transient kinetics of carbon monoxide oxidation by oxygen over supported palladium/ceria/zirconia three-way catalysts in the absence and presence of water and carbon dioxide. J. Catal. 2004, 223, 36–43. [Google Scholar] [CrossRef]
- Singhania, A.; Krishnan, V.V.; Bhaskarwar, A.N.; Bhargava, B.; Parvatalu, D. Hydrogen-iodide decomposition over Pd–CeO2 nanocatalyst for hydrogen production in sulfur-iodine thermochemical cycle. Int. J. Hydrogen Energy 2018, 43, 3886–3891. [Google Scholar] [CrossRef] [Green Version]
- Gao, W.; Zhang, Z.; Li, J.; Ma, Y.; Qu, Y. Surface engineering on CeO2 nanorods by chemical redox etching and their enhanced catalytic activity for CO oxidation. Nanoscale 2015, 7, 11686–11691. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Sun, B.; Sun, J.; Hong, X.; Deng, Y.; Gao, F.; Dong, L. Solid state preparation of NiO-CeO2 catalyst for NO reduction. Catal. Today 2017, 281, 575–582. [Google Scholar] [CrossRef]
- Du, X.; Zhang, D.; Shi, L.; Gao, R.; Zhang, J. Morphology dependence of catalytic properties of Ni/CeO2 nanostructures for carbon dioxide reforming of methane. J. Phys. Chem. C 2012, 116, 10009–10016. [Google Scholar] [CrossRef]
- Carrettin, S.; Blanco, M.C.; Corma, A.; Hashmi, A.S.K. Heterogeneous gold-catalysed synthesis of phenols. Adv. Synth. Catal. 2006, 348, 1283–1288. [Google Scholar] [CrossRef]
- Aneggi, E.; Boaro, M.; De Leitenburg, C.; Dolcetti, G.; Trovarelli, A. Insights into the dynamics of oxygen storage/release phenomena in model ceria-zirconia catalysts as inferred from transient studies using H2, CO and soot as reductants. Catal. Today 2006, 112, 94–98. [Google Scholar] [CrossRef]
- Avgouropoulos, G.; Ioannides, T.; Papadopoulou, C.; Batista, J.; Hocevar, S.; Matralis, H.K. A comparative study of Pt/γ-Al2O3, Au/α-Fe2O3 and CuO-CeO2 catalysts for the selective oxidation of carbon monoxide in excess hydrogen. Catal. Today 2002, 75, 157–167. [Google Scholar] [CrossRef]
- Jeong, M.; Nunotani, N.; Moriyama, N.; Imanaka, N. Effect of introducing Fe2O3 into CeO2-ZrO2 on oxygen release properties and catalytic methane combustion over PdO/CeO2-ZrO2-Fe2O3/γ-Al2O3 catalysts. Catal. Sci. Technol. 2017, 7, 1986–1990. [Google Scholar] [CrossRef]
- Kappis, K.; Papadopoulos, C.; Papavasiliou, J.; Vakros, J.; Georgiou, Y.; Deligiannakis, Y.; Avgouropoulos, G. Tuning the catalytic properties of copper-promoted nanoceria via a hydrothermal method. Catalysts 2019, 9, 138. [Google Scholar] [CrossRef] [Green Version]
- Nolan, M.; Parker, S.C.; Watson, G.W. CeO2 catalysed conversion of CO, NO2 and NO from first principles energetics. Phys. Chem. Chem. Phys. 2006, 8, 216–218. [Google Scholar] [CrossRef] [Green Version]
- Zhou, K.; Wang, X.; Sun, X.; Peng, Q.; Li, Y. Enhanced catalytic activity of ceria nanorods from well-defined reactive crystal planes. J. Catal. 2005, 229, 206–212. [Google Scholar] [CrossRef]
- Hu, Z.; Liu, X.; Meng, D.; Guo, Y.; Guo, Y.; Lu, G. Effect of Ceria Crystal Plane on the Physicochemical and Catalytic Properties of Pd/Ceria for CO and Propane Oxidation. ACS Catal. 2016, 6, 2265–2279. [Google Scholar] [CrossRef]
- Ma, J.; Lou, Y.; Cai, Y.; Zhao, Z.; Wang, L.; Zhan, W.; Guo, Y.; Guo, Y. The relationship between the chemical state of Pd species and the catalytic activity for methane combustion on Pd/CeO2. Catal. Sci. Technol. 2018, 8, 2567–2577. [Google Scholar] [CrossRef]
- Xu, J.; Harmer, J.; Li, G.; Chapman, T.; Collier, P.; Longworth, S.; Tsang, S.C. Size dependent oxygen buffering capacity of ceria nanocrystals. Chem. Commun. 2010, 46, 1887–1889. [Google Scholar] [CrossRef] [PubMed]
- Priolkar, K.R.; Bera, P.; Sarode, P.R.; Hegde, M.S.; Emura, S.; Kumashiro, R.; Lalla, N.P. Formation of Ce1−xPdxO2−δ solid solution in combustion-synthesized Pd/CeO2 catalyst: XRD, XPS, and EXAFS investigation. Chem. Mater. 2002, 14, 2120–2128. [Google Scholar] [CrossRef]
- Meng, L.; Lin, J.J.; Pu, Z.Y.; Luo, L.F.; Jia, A.P.; Huang, W.X.; Luo, M.F.; Lu, J.Q. Identification of active sites for CO and CH4 oxidation over PdO/Ce1−xPdxO2−δ catalysts. Appl. Catal. B Environ. 2012, 119–120, 117–122. [Google Scholar] [CrossRef]
- Gulyaev, R.V.; Stadnichenko, A.I.; Slavinskaya, E.M.; Ivanova, A.S.; Koscheev, S.V.; Boronin, A.I. In situ preparation and investigation of Pd/CeO2 catalysts for the low-temperature oxidation of CO. Appl. Catal. A Gen. 2012, 439–440, 41–50. [Google Scholar] [CrossRef]
- Zhang, X.; Li, W.; Zhou, Z.; Chen, K.; Wu, M.; Yuan, L. High dispersed Pd supported on CeO2 (1 0 0) for CO oxidation at low temperature. Mol. Catal. 2021, 508, 111580. [Google Scholar] [CrossRef]
- Peng, N.; Zhou, J.; Chen, S.; Luo, X.; Chen, Y.; Gong, M. Synthesis of neodymium modified CeO2-ZrO2-Al2O3 support materials and their application in Pd-only three-way catalysts. J. Rare Earths 2012, 30, 342–349. [Google Scholar] [CrossRef]
- Polster, C.S.; Zhang, R.; Cyb, M.T.; Miller, J.T.; Baertsch, C.D. Selectivity loss of Pt/CeO2 PROX catalysts at low CO concentrations: Mechanism and active site study. J. Catal. 2010, 273, 50–58. [Google Scholar] [CrossRef]
- Lei, Y.; Li, W.; Liu, Q.; Lin, Q.; Zheng, X.; Huang, Q.; Guan, S.; Wang, X.; Wang, C.; Li, F. Typical crystal face effects of different morphology ceria on the activity of Pd/CeO2 catalysts for lean methane combustion. Fuel 2018, 233, 10–20. [Google Scholar] [CrossRef]
- Tan, H.; Wang, J.; Yu, S.; Zhou, K. Support Morphology-Dependent Catalytic Activity of Pd/CeO2 for Formaldehyde Oxidation. Environ. Sci. Technol. 2015, 49, 8675–8682. [Google Scholar] [CrossRef] [PubMed]
- Peng, R.; Sun, X.; Li, S.; Chen, L.; Fu, M.; Wu, J.; Ye, D. Shape effect of Pt/CeO2 catalysts on the catalytic oxidation of toluene. Chem. Eng. J. 2016, 306, 1234–1246. [Google Scholar] [CrossRef]
- Gu, D.; Jia, C.J.; Bongard, H.; Spliethoff, B.; Weidenthaler, C.; Schmidt, W.; Schüth, F. Ordered mesoporous Cu-Ce-O catalysts for CO preferential oxidation in H2-rich gases: Influence of copper content and pretreatment conditions. Appl. Catal. B Environ. 2014, 152–153, 11–18. [Google Scholar] [CrossRef]
- Song, S.; Zhang, C.; Lou, Y.; Wu, Y.; Wang, L.; Guo, Y.; Zhan, W.; Guo, Y. Effects of water on CO catalytic oxidation over Pd/CeO2. J. Rare Earths 2020, 38, 891–898. [Google Scholar] [CrossRef]
- Burch, R.; Hayes, M.J. CH bond activation in hydrocarbon oxidation on solid catalysts. J. Mol. Catal. A Chem. 1995, 100, 13–33. [Google Scholar] [CrossRef]
- Trovarelli, A.; Llorca, J. Ceria Catalysts at Nanoscale: How Do Crystal Shapes Shape Catalysis? ACS Catal. 2017, 7, 4716–4735. [Google Scholar] [CrossRef]
- Monai, M.; Montini, T.; Gorte, R.J.; Fornasiero, P. Catalytic Oxidation of Methane: Pd and Beyond. Eur. J. Inorg. Chem. 2018, 2018, 2884–2893. [Google Scholar] [CrossRef]
- Wang, R.; Dangerfield, R. Seed-mediated synthesis of shape-controlled CeO2 nanocrystals. RSC Adv. 2014, 4, 3615–3620. [Google Scholar] [CrossRef]




| Samples | Pd Loading a (wt%) | Specific Surface Area b (m2/g) | Pd Dispersion c (%) | Grain Size d (nm) |
|---|---|---|---|---|
| CeO2-R | - | 52.8 | - | 12.0 |
| 0.3% PdO/CeO2-RE | 0.31 | 35.3 | 31.4 | 11.9 |
| 0.5% PdO/CeO2-RE | 0.47 | 30.2 | 33.2 | 12.0 |
| 1% PdO/CeO2-RE | 0.89 | 29.4 | 29.1 | 12.0 |
| 0.5% PdO/CeO2-IM | 0.48 | 27.9 | 30.2 | 12.1 |
| Sample | Surface Pd Species Content (%) | Ce3+/(Ce3++Ce4+) (%) | Oads/(Olat+Oads) (%) | Pd Atomic (%) | TOF a (×103 s−1) | Ea (kJ/mol) | ||
|---|---|---|---|---|---|---|---|---|
| Pd2+ | PdxCe1−xO2−δ | Pd0 | ||||||
| CeO2-R | − | − | − | 0.16 | 0.41 | − | − | − |
| 0.3% PdO/CeO2-RE | 55.28 | 44.72 | − | 0.25 | 0.49 | 0.12 | 22.3 | 72.7 |
| 0.5% PdO/CeO2-RE | 72.66 | 27.34 | − | 0.24 | 0.47 | 0.36 | 25.5 | 61.6 |
| 1% PdO/CeO2-RE | 81.34 | 18.66 | − | 0.18 | 0.43 | 1.34 | 5.0 | 93.1 |
| 0.5% PdO/CeO2-IM | 49.33 | 37.91 | 12.76 | 0.19 | 0.45 | 0.30 | 10.5 | 74.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Hu, C.; Bian, L. Highly Dispersed Pd Species Supported on CeO2 Catalyst for Lean Methane Combustion: The Effect of the Occurrence State of Surface Pd Species on the Catalytic Activity. Catalysts 2021, 11, 772. https://doi.org/10.3390/catal11070772
Liu Y, Hu C, Bian L. Highly Dispersed Pd Species Supported on CeO2 Catalyst for Lean Methane Combustion: The Effect of the Occurrence State of Surface Pd Species on the Catalytic Activity. Catalysts. 2021; 11(7):772. https://doi.org/10.3390/catal11070772
Chicago/Turabian StyleLiu, Yanxiong, Changhua Hu, and Longchun Bian. 2021. "Highly Dispersed Pd Species Supported on CeO2 Catalyst for Lean Methane Combustion: The Effect of the Occurrence State of Surface Pd Species on the Catalytic Activity" Catalysts 11, no. 7: 772. https://doi.org/10.3390/catal11070772
APA StyleLiu, Y., Hu, C., & Bian, L. (2021). Highly Dispersed Pd Species Supported on CeO2 Catalyst for Lean Methane Combustion: The Effect of the Occurrence State of Surface Pd Species on the Catalytic Activity. Catalysts, 11(7), 772. https://doi.org/10.3390/catal11070772







