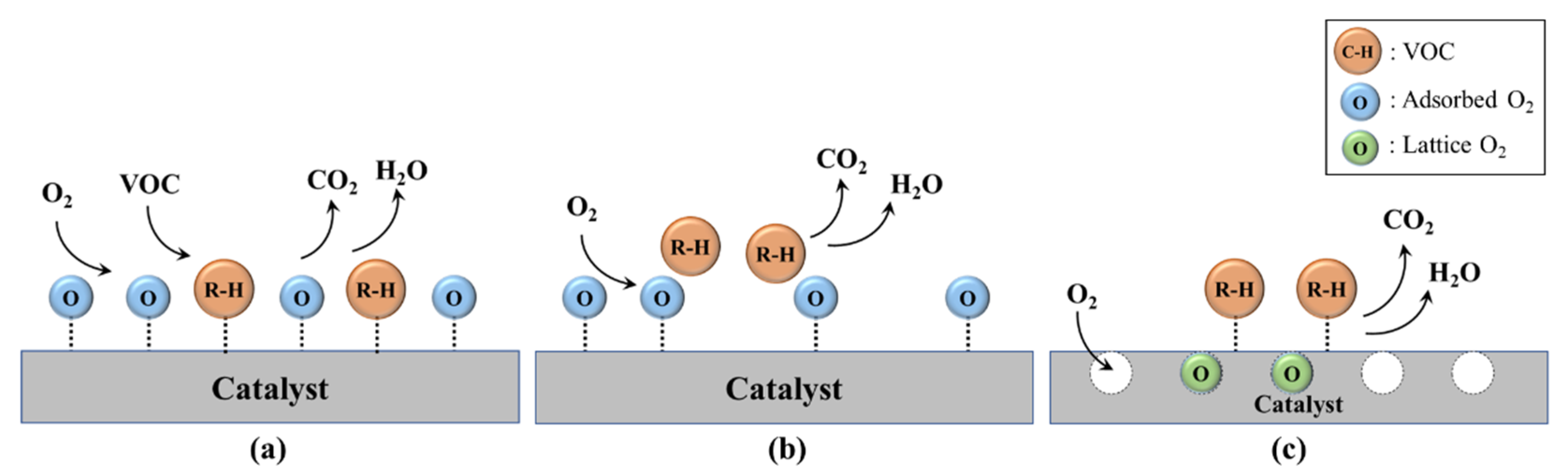
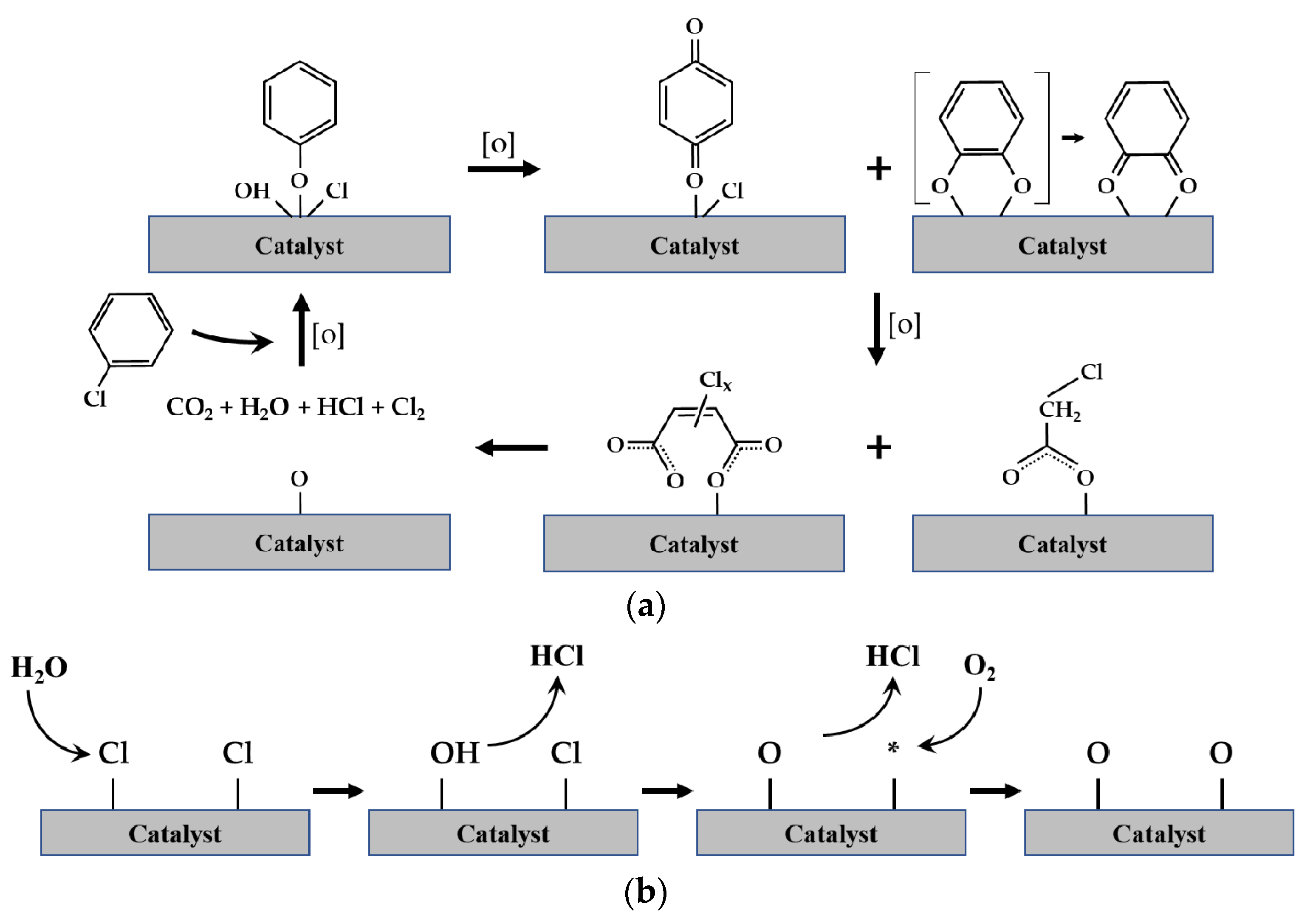
As a catalyst for VOC removal from industrial flue gas, noble-metal-based catalysts, such as Ag, Au, Pd, Pt, Rh, and Ru, which have high activity at low temperatures, are widely used. Ceramic or metallic materials are widely used as supports for noble-metal-based catalysts. These catalysts have different VOC combustion activities depending on the type of support and bonding type with the support. However, because noble metals are expensive, studies are actively being conducted to improve the economic feasibility, stability, and selectivity of noble-metal-based catalysts by improving the catalyst dispersion, particle size, specific surface area, and structure of the active material. Furthermore, a catalyst with both high performance and economic feasibility has been developed by mixing non-noble metal catalysts possessing relatively low activity with noble metal catalysts [
39].
3.1. Ag-Based Catalysts
VOC combustion generally occurs in the temperature range of 250–350 °C. However, it has been reported that Ag-based catalysts are less active in the oxidation of BTEX compounds in this temperature range, compared to other noble metal or transition metal catalysts. Baek et al. analyzed the characteristics of Ag catalysts during VOC combustion using Ag/HY-zeolite catalysts. As the Ag molecular content increased, the number of Ag nanoparticles increased, resulting in an increase in the catalytic activity [
40].
Although the optimal content of Ag molecules reported by researchers varied significantly, it has been reported that, when Ag content is increased, the number of active sites is limited owing to the increase in the catalyst crystal size and, consequently, the decrease in specific surface area and void fraction [
41].
However, according to Zhou et al., when Ag content was low, the VOC removal efficiency of the catalyst was reduced owing to the decreased content of the active phase [
42]. Moreover, the Cu-Mn-Ag catalyst was impregnated onto a cordite monolith support, and the combustion efficiency of toluene was evaluated. Consequently, the catalyst activity was the highest at 21.2 wt% Ag content, and at a relatively higher Ag content, the catalyst activity decreased because the active phase accumulated on the surface of the support.
Generally, oxygen vacancies are widely known to promote catalytic activity for VOC combustion [
43,
44,
45]. Ma et al. demonstrated that the binding of Ag species was related to the benzene combustion reaction. The binding of Ag species over CeO
2-Co
3O
4 induces electron transfer at the surface of the metal oxide, thus creating more active sites (Co
3+) and oxygen defect sites (Ce
3+) (
Figure 2). This promotes the adsorption of benzene molecules on the surface of the metal oxide catalyst and the reactivity of benzene combustion at low temperatures [
45].
3.2. Au-Based Catalysts
Gold-based catalysts are known to exhibit high oxidation activity, as reported by many previous studies [
46,
47,
48,
49,
50]. For BTEX combustion, Au-based catalysts can react at temperatures higher than 190–400 °C, compared to Pd/Pt-based catalysts. Further, Au-based catalysts have the characteristic that they are less prone to deactivation caused by carbon coke formation inside the catalyst pores. Although Au-based catalysts have low economic efficiency compared to combustion efficiency due to the low oxygen changing rate, many studies are ongoing to address this problem by developing transition metal complexes to overcome thermal stabilities and enhance catalyst activities [
51].
In particular, for Au-based catalysts in VOC combustion, the preparation method is crucial because it is highly dependent on the size of the particles and the type of support [
52,
53,
54,
55]. The mechanism of the catalytic reaction of VOC oxidation may vary depending on the type of support material used. The oxidation of VOCs is affected by the size, distribution, shape, and oxidation state of Au particles, which are the metal active sites. Many reports emphasize the role of supports and describe various supports, such as metals [
23,
56], aluminum [
24,
47,
57,
58], titanium [
59,
60], and silica [
61], in Au-based catalysts. These studies optimized the shapes, sizes, contents, etc. of the Au catalyst, influencing the activity of the material in VOC oxidation, and described the correlation between catalyst and support in heterogeneous catalysts [
54,
62].
Table 2 lists the VOC oxidation characteristics of the Au catalysts according to the type of support material used.
Although some researchers have reported that the oxidation state of Au as a metal active site has an important effect on the activity of the VOC oxidation reaction, it was reported that the oxidation state is influenced by the particle size, shape, distribution, etc. of Au. Thus, research on increasing particle dispersion is in progress. Au-based catalysts are produced by various methods, such as precipitation and coprecipitation, to increase the dispersion of Au particles. It was reported that the deposition–precipitation method can produce particles in the range of 4 to 8 nm [
24,
63,
64].
According to Carabineiro et al., metals in Au-based catalysts play a role in improving the support reducibility and catalytic reactivity. Catalyst activity is enhanced by an increase in the oxygen exchange between the lattice and the support surface. The Au/Y
2O
3 catalyst with the Au
3+ oxidation state showed low catalytic activity. In contrast, for Au-based Fe
2O
3, NiO, and CuO catalysts, the toluene combustion activity increased with Au in the oxidation state of Au
+ and Au
0 [
23].
Li et al. studied the reaction characteristics of toluene oxidation using a LaCoO
3 supported on a crystallized three-dimensionally ordered microporous material (3DOM). The Au-based 3DOM LaCoO
3 catalyst, compared to the bulk LaCoO
3 catalyst, possessed several characteristics, such as uniform pore size, surface thickness, and high Au dispersion [
49]. In addition, it has been reported that the presence of oxygen species on the support surface promotes oxygen formation, which is advantageous for VOC oxidation.
3.3. Pd-Based Catalysts
Pd-based catalysts are used in a slightly higher temperature range of 200–300 °C compared to Pt-based catalysts for the VOC combustion reaction. The active phase of the Pd catalyst was Pd
0, and the oxidation state of Pd affected the catalyst activity. Therefore, it has been reported that VOC combustion activity is highly dependent on the Pd content in the catalyst [
65,
66]. Although the chemical state of the Pd species is an important factor influencing the catalytic activity, there is controversy about this effect, as shown in
Table 3.
According to Dégé et al., it was confirmed that the VOC oxidation activity was greatly affected by the Pd content and that the surface area, particle size, particle dispersion, and support also affect the activity of the catalyst [
66]. Moreover, the results indicated that the coke content generated at low temperatures differed depending on the acidity of the zeolite (that is, the change in the Si/Al ratio) used as the support. For the oxidation of o-xylene (C
8H
10), the higher the acidity of the zeolite, the higher the rate of coke formation. Furthermore, it was confirmed that an increase in Pd content also plays a role in suppressing coke formation at the same temperature. During toluene combustion, it was determined that the stability of the Pd-based catalyst was higher than that of the Pt-based catalyst (the activated carbon fiber support was impregnated with 3.86 wt% Pt or Pd), which is advantageous for the combustion of cyclic organic compounds [
67]. Bi et al. reported that a Pd-U-H catalyst formed nanoparticles and increased the surface Pd
0 and lattice oxygen content. As a result of this effect, the combustion ratio of toluene was maintained at a high level in the 6-cycle reaction experiment [
68].
Gil et al. examined the oxidation state of Pd according to the type of support (CeO
2, TiO
2, and Al
2O
3) through XPS analysis and evaluated the conversion rate of propene. Pd/Al
2O
3 and Pd/TiO
2 have a Pd
2+ phase, similar to bulk PdO, whereas Pd/CeO
2 has a Pd
4+ phase. These characteristics explain the poor catalytic activity of Pd/CeO
2 for propene combustion because the Pd
4+ species form strong bonds with the support [
69].
Weng et al. proposed that metallic Pd exhibited higher oxidation activity using the Pd/MgO-Al
2O
3 catalyst system. The oxidation state of the Pd-based catalyst was characterized by XPS analysis of fresh, hydrotreated, and spent catalysts after toluene reaction. After hydrogen treatment, the Pd
2+ state of the catalyst increased, indicating the formation of a Pd
2+/Pd
0 pair that aids in the toluene oxygen activity [
70].
Meanwhile, a portion of the metallic Pd is oxidized to Pd
2+ states, and the reaction proceeds in the form of reduction aided by hydrocarbons. For Pd
0, Pd
2+, or mixed Pd
2+/Pd
0 states, Pd
0 helps to improve the reaction rate by providing more adsorbable active sites for VOCs, and Pd
2+ plays a role in VOC combustion [
71,
72].
Table 3.
Catalytic oxidation of VOCs over Pd catalysts with different Pd states.
Table 3.
Catalytic oxidation of VOCs over Pd catalysts with different Pd states.
| Pd State | Catalyst | Used VOCs | Ref. |
|---|
| Pd0 | Pd/CeO2 | Propene | [69] |
| Pd/Al2O3 | Propene |
| Pd/MgO-Al2O3 | Toluene | [70] |
| Pd/Co3AlO | Toluene | [73] |
| Pd–CoAlO-Al | Toluene | [74] |
| Pd0, Pd2+ | Pd/HFAU(17) | o-xylene | [66] |
| Pd/Ti-SBA-15 | Benzene | [71] |
| Pd/ZSM-5/MCM-48 | Benzene | [72] |
| Pd-U-EG | Toluene | [68] |
| 0.5%Pd/SiO2 | Toluene | [75] |
| Pd0, Pd4+ | Pd/CeO2 | Propene | [69] |
As the surface area and structure of the catalyst affect the activity, several researchers have investigated the oxidation states of the catalyst with relation to the support. When activated carbon is used as a support, its physical and chemical properties affect the metal deposition, dispersion, and gas adsorption efficiency [
76].
Table 4 summarizes the activities of Pd-based catalysts based on different types of supports. According to Giraudon et al., the combustion performance of Pd/TiX showed superior chlorobenzene (C
6H
5Cl) conversion compared to Pd/ZrX. This is because the reducibility of TiO
2 was improved from Ti
4+ to Ti
3+ [
77]. Pérez-Cadenas et al. reported that the VOC combustion reactivity of a Pd-based catalyst is affected by the surface area of mesopores, since the activity of the mesoporous supports was higher than that of the microporous and monolithic supports [
78]. Mesoporous Pd/Co
3O
4 (3D) catalysts have relatively higher activity for o-xylene oxidation than the bulk counterpart Pd/Co
3O
4. This improvement might be due to the increased surface exposure of PdO on the mesoporous Co
3O
4 support [
79].
Nanoparticle catalysts have different physical and chemical properties compared to single metal catalysts; thus, there is a synergistic effect in the VOC combustion properties of Pd-based catalysts [
80]. Generally, alloy catalysts show a more flexible surface structure than single metal catalysts and induce affinity between the catalyst surface and the adsorbate, which contributes to high activity [
81].
Comparing the reaction mechanism of Pd-Ce/Al
2O
3 with that of Pd/Al
2O
3, the presence of Ce increased the active oxygen species and oxygen vacancy content on the catalyst surface. During the conversion of Ce
3+ to Ce
4+, electrons are donated to PdO to promote PdO to Pd, thereby generating lattice oxygens and promoting the reaction with adsorbed toluene. In addition, Ce can improve the catalyst activity by reducing the noble metal content in the catalyst [
14].
Wu et al. evaluated the toluene combustion characteristics of the Au-Pd/meso-Cr
2O
3 catalyst and reported that Au-Pd nanoparticles showed excellent toluene combustion activity, compared to single metal catalysts, owing to their strong interaction with Cr
2O
3 [
82]. Similarly, Chen et al. reported that, when a Pd-Pt-based nanoparticle catalyst was used for the benzene (C
6H
6) combustion reaction, it showed excellent conversion, which was due to the nano effect of the catalyst and the interaction between the active materials (Pd-Pt) [
83].
Table 4.
Comparison of Pd-based catalysts on various supports for VOC oxidation.
Table 4.
Comparison of Pd-based catalysts on various supports for VOC oxidation.
| Active Metal | Support Material | VOCs | Temperature (°C) | Conversion (%) | Ref. |
|---|
| Pd | UiO-66 | Toluene | 200 | 100 | [68] |
| Al2O3 | Toluene | 252 | 90 | [70] |
| MgO-Al2O3 | Toluene | 209 | 90 |
| Carbon | m-Xylene | 170 | 100 | [78] |
| Co3O4(3D) | o-Xylene | 249 | 90 | [79] |
| Pd-Ce | γ-Al2O3 | Toluene | 200 | 90 | [14] |
| Au-Pd | Cr2O3 | Toluene | 165 | 90 | [82] |
| Pd-Pt | Ce/γ-Al2O3 | Benzene | 190 | 95 | [83] |
| Pd-W | TiO2 | Propane | 375 | 100 | [84] |
3.4. Pt-Based Catalysts
Pt-based catalysts are known to be the most efficient element for the combustion of cyclic hydrocarbon compounds and exhibit activity for the combustion of BTEX compounds in the temperature range of 150–350 °C [
85,
86]. Because the Pt-based catalyst does not interact with the support, the physical/chemical properties are maintained after impregnation. The characteristics of the support affect the deposition and dispersion of the active element, which ultimately influences the durability and poisoning resistance of the catalyst [
87].
The dispersion degree of the Pt-based catalyst depends on the support, so it is important to improve this parameter to obtain high activity. Since the metal catalyst easily combines with the oxidized site of the support, the dispersibility of Pt can be improved by oxidizing the activated carbon support by acid treatment or air oxidation [
88]. To improve the deposition rate of the metal catalyst onto the support, a Pt catalyst was deposited on a porous support by dry impregnation rather than wet impregnation. In addition, the ion-exchange method was used to deposit metal catalysts onto the zeolite surface [
86].
Table 5 summarizes the oxidation characteristics of Pt-based catalysts using cerium oxide as a support (Pt/CeO
2). It has been reported that the Pt/CeO
2 catalyst has superior oxygen storage capacity, reducibility, and VOC combustion performance compared to the CeO
2 catalyst [
89]. In particular, when CeO
2-Al
2O
3 was used as a support, the Pt catalyst formed nanoparticles and exhibited high reducibility and activity in xylene and toluene oxidation. In addition, owing to the improvement in the dispersion degree of Pt particles, the high temperature (>300 °C) oxidation activity might be increased [
90]. The catalyst using Al
2O
3-CeO
2 as a support exhibited improved oxidation of dichloromethane (DCM, CH
2Cl
2) [
91]. The addition of Ce and Pt catalysts enhanced the selectivity of CO
2 and inhibits catalyst deactivation by reducing the formation of CH
3Cl, CH
2O, CO, etc.
The activity of the catalyst for VOC oxidation was greatly affected by the type and shape of the catalyst support. The surface oxygen vacancies, which play an important role in adsorbing gaseous oxygen and promoting combustion activity, depend on shape of catalyst. Peng et al. [
92] prepared catalysts by adding Pt catalysts to CeO
2 supports with various crystal sizes. A high-efficiency Pt/CeO
2 catalyst was optimized for toluene combustion by controlling the degree of exposure of the support surface and the role of CeO
2 was reported.
Recently, many studies have been reported on the combustion of chlorinated volatile organic compounds (CVOCs). As a result of evaluating the support of the Pt-based catalysts used for DOM combustion, the activity of Pt/Al
2O
3 was superior compared to that of Pt/TiO
2, Pt/CeO
2, and Pt/MgO. This is because when Al
2O
3 is used as a support, the particle size of Pt is reduced to less than 1.2 nm [
93].
Pt molecules serve to increase the number of weak acid sites and reduce the number of strong acid sites on the catalyst surface. This phenomenon strengthens the adsorption strength between the VOCs and the catalyst surface. Yang et al. confirmed that an insufficient active oxygen supply was improved by adding Pt to the Pd/CeO
2/γ-Al
2O
3 catalyst in the oxidation of various VOCs [
94].
3.5. Rh-Based Catalysts
It is known that Rh-based VOC combustion catalysts are far less explored than other novel metal-based catalysts. Recently, they have attracted attention because of their improved effects on the catalytic oxidation of CVOCs, aromatics, and alkanes.
Rh-based catalysts have high stability with the support material compared to other noble metal catalysts (Ru, Rh, Pd, and Pt) [
91,
95]. Noble metals have high thermal stability while forming an M–O–Ce bond with a CeO
2 support. Saburo et al. reported that the M-O–Ce bond was maintained above 800 °C for Ru-and Pd-based catalysts, while the M–O–Ce bond endured below 500 °C, and Ru was present as bulk RuO
2 below 500 °C. Therefore, it was concluded that the stability between noble metals and the CeO
2 support was high and followed the order of Rh > Pd > Pt > Ru [
96].
According to Pitkäaho et al. in
Table 6, the Rh-based catalytic activity sequence with different supports was Al
2O
3 > Al
2O
3-CeO
2 > Al
2O
3-TiO
2. When the same support was used, the Rh-based catalyst exhibited higher activity than other metal-based catalysts (Pd, V
2O
5, and Pt). For the combustion of CVOCs, selectivity to CO, CO
2, HCl, and high activity are required. The addition of Rh improved the HCl yield of the Al
2O
3 supported catalyst and had a HCl selectivity of 93%. For the Rh catalyst, the amount of CO formed differed depending on the support. It showed a high CO yield of 46% over the Al
2O
3 supported catalysts, 14% over Rh/Al
2O
3-TiO
2, and 1% over Rh/Al
2O
3-CeO
2 [
91].
In the reports on Rh-based catalysts, Rh/TiO
2 yielded the lowest Cl content of 0.61% after CVOC oxidation, which was consistent with the amount of polychlorinated by-products (dichlorobenzene, trichlorobenzene, tetra-chlorobenzene, pent-chlorobenzene, etc.) detected by XPS analyses. The maximum concentration of chlorobenzene combustion by-products, 1,3-dichlorobenzene and 1,4-dichlorobenzene, reached 23 ppm at 310 °C [
97].
We can now summarize the contents of verse 3, although the combustion characteristics of VOCs depend on the support; the characteristics of Pd, Pt, and Rh catalysts supported on TiO
2 were examined in the oxidation of chlorobenzene, as shown in
Figure 3 [
97]. For all three catalysts, the oxidation of chlorobenzene is initiated at 250 °C, and all of them exhibit a conversion rate of more than 95% at temperatures above 350 °C.: (1) T
90 (Pd/TiO
2) = 340 °C, (2) T
90 (Pt/TiO
2) = 337 °C, and (3) T
90 (Rh/TiO
2) = 339 °C. In particular, Pt/TiO
2 exhibited higher low-temperature catalytic activity than those of Pd/TiO
2 and Rh/TiO
2. According to the evaluation of the COx concentration, the CO yield was lower than 1%, which indicated that CO
2 selectivity was high. The generated trend for the by-products (PhCl
2, PhCl
3, PhCl
4, etc.) is shown in
Figure 3b. The maximum amounts of by-products are 65 ppm, 88 ppm, and 100 ppm at 360 °C, 340 °C, and 340 °C for Pt/TiO
2, Pd/TiO
2 and Rh/TiO
2, respectively. The Pt-based catalyst exhibited high VOC oxidation activity at temperatures ≤ 300 °C, and the use of the Rh-based catalyst was advantageous at temperatures ≥ 370 °C.
As mentioned above, most research on VOC oxidation over noble metals used Pt or Pd, which were manufactured using supports, such as TiO2, CeO2, Al2O3, and ZrO2. It has been reported that the acid–base properties of the support typically play an important role in catalyst performance. In particular, the Pd-based catalyst has been reported to have excellent durability in the catalytic oxidation of chlorinated VOCs. Meanwhile, some researchers have reported that a combination of Pt and Pd achieved higher VOC oxidation efficiency compared to that of single metal catalysts. In the oxidation reaction over noble metal catalysts, catalytic performance varies depending on the components of VOCs and supports. Therefore the main catalyst should be selected according to their application, such as the operation temperature, VOCs concentration, and components.