Current Challenges and Perspectives for the Catalytic Pyrolysis of Lignocellulosic Biomass to High-Value Products
Abstract
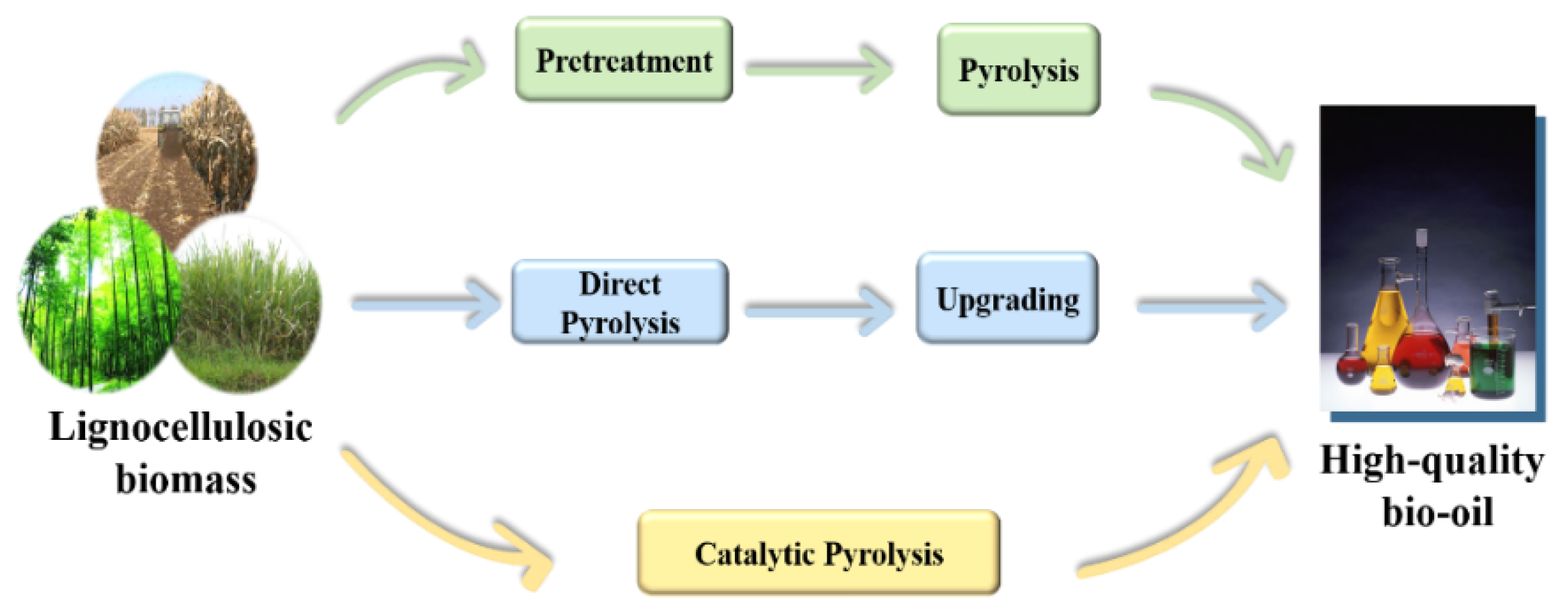
:1. Introduction
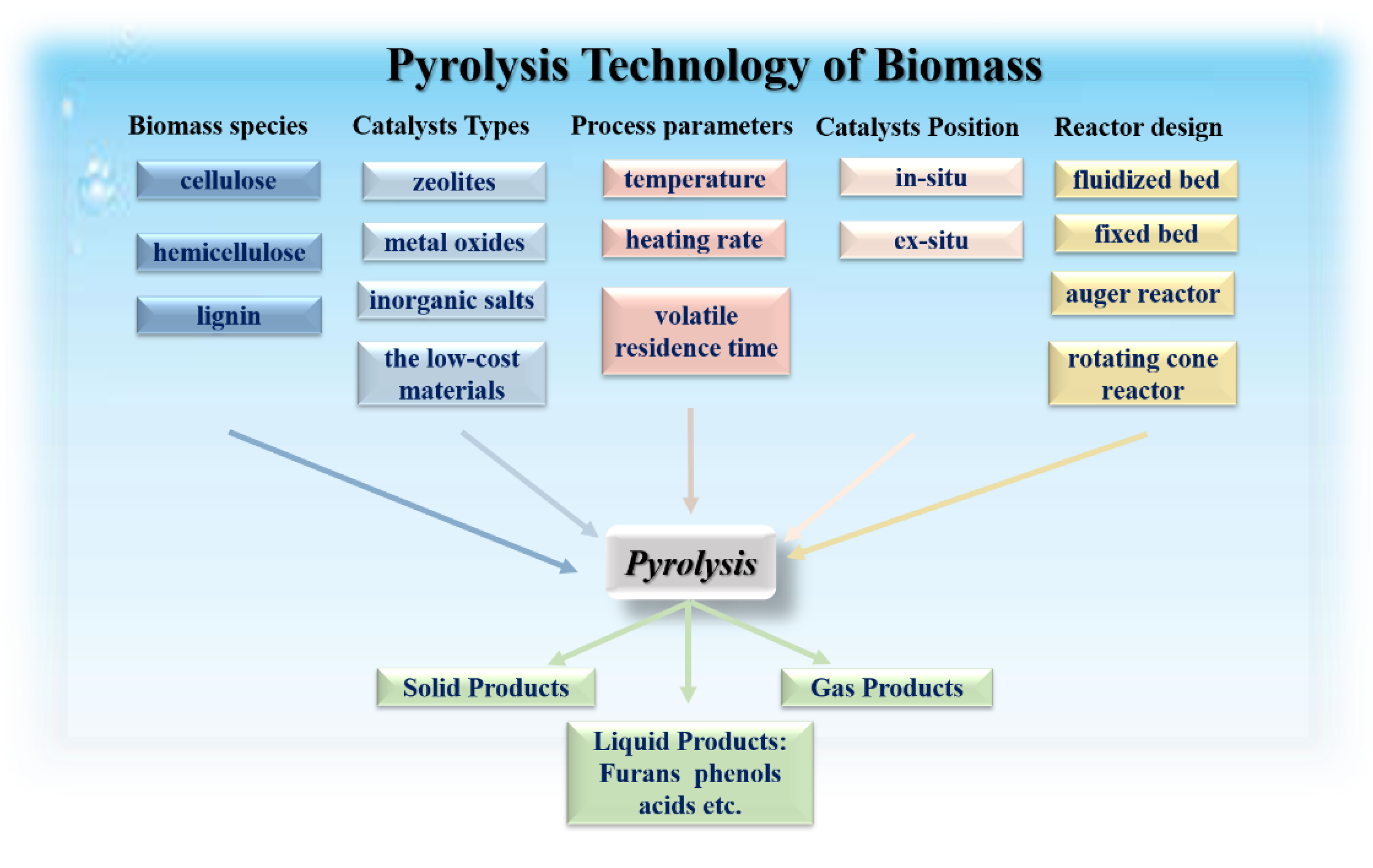
2. Factors Affecting the Catalytic Pyrolysis of Biomass
2.1. Lignocellulosic Biomass
2.1.1. Pyrolysis of Cellulose
2.1.2. Pyrolysis of Hemicellulose
2.1.3. Pyrolysis of Lignin
2.2. The Reactor Design and Process Control
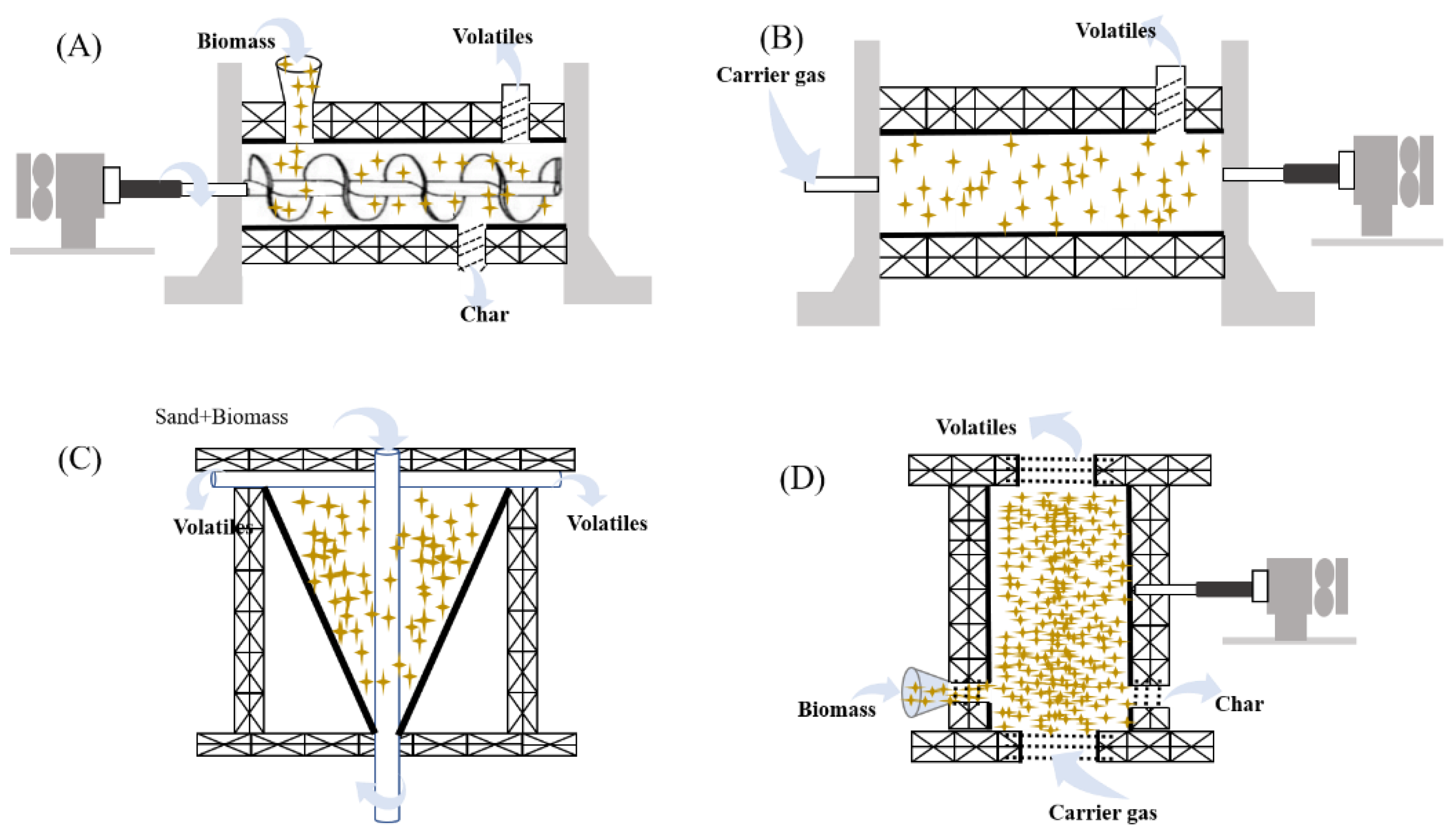
2.2.1. Diversity of Reactor
2.2.2. The Process Parameters
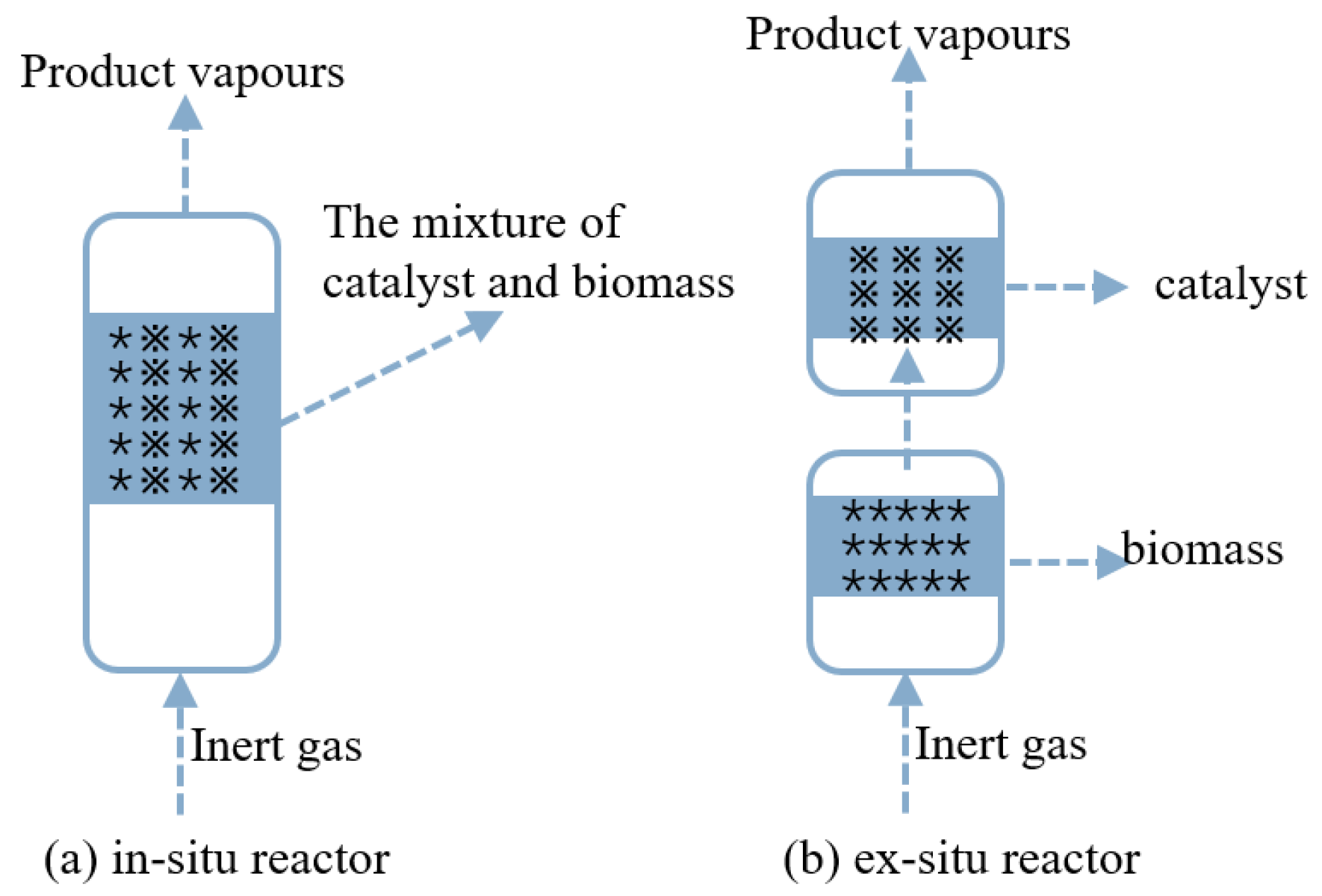
2.2.3. The Installation of Catalysts
2.3. The Types of Catalysts
2.3.1. Zeolites
2.3.2. Metal Oxide Catalysts
2.3.3. Soluble Inorganic Salts
2.3.4. Other Low-Cost Materials
3. Challenges and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Feng, Y.; Long, S.; Tang, X.; Sun, Y.; Luque, R.; Zeng, X.; Lin, L. Earth-abundant 3d-transition-metal catalysts for lignocellulosic biomass conversion. Chem. Soc. Rev. 2021, 50, 6042–6093. [Google Scholar] [CrossRef]
- Liang, J.; Shan, G.; Sun, Y. Catalytic fast pyrolysis of lignocellulosic biomass: Critical role of zeolite catalysts. Renew. Sustain. Energy Rev. 2021, 139, 110707. [Google Scholar] [CrossRef]
- Vikram, S.; Rosha, P.; Kumar, S. Recent Modeling Approaches to Biomass Pyrolysis: A Review. Energy Fuels 2021, 35, 7406–7433. [Google Scholar] [CrossRef]
- Zakzeski, J.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The Catalytic Valorization of Lignin for the Production of Renewable Chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef]
- Tian, X.; Wang, Y.; Zeng, Z.; Dai, L.; Xu, J.; Cobb, K.; Ke, L.; Zou, R.; Liu, Y.; Ruan, R. Research progress on the role of common metal catalysts in biomass pyrolysis: A state-of-the-art review. Green Chem. 2022, 24, 3922–3942. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Garg, R.; Anand, N.; Kumar, D. Pyrolysis of babool seeds (Acacia nilotica) in a fixed bed reactor and bio-oil characterization. Renew. Energy 2016, 96, 167–171. [Google Scholar] [CrossRef]
- Kollman, M.S.; Jiang, X.; Sun, R.; Zhang, X.; Li, W.; Chang, H.-M.; Jameel, H. Towards jet fuel from technical lignins: Feedstock-catalyst-product interactions revealed during catalytic hydrogenolysis. Chem. Eng. J. 2023, 451, 138464. [Google Scholar] [CrossRef]
- Hoang, A.T.; Ong, H.C.; Fattah, I.M.R.; Chong, C.T.; Cheng, C.K.; Sakthivel, R.; Ok, Y.S. Progress on the lignocellulosic biomass pyrolysis for biofuel production toward environmental sustainability. Fuel Process. Technol. 2021, 223, 106997. [Google Scholar] [CrossRef]
- Bhoi, P.R.; Ouedraogo, A.S.; Soloiu, V.; Quirino, R. Recent advances on catalysts for improving hydrocarbon compounds in bio-oil of biomass catalytic pyrolysis. Renew. Sustain. Energy Rev. 2020, 121, 109676. [Google Scholar] [CrossRef]
- Oh, Y.K.; Hwang, K.R.; Kim, C.; Kim, J.R.; Lee, J.S. Recent developments and key barriers to advanced biofuels: A short review. Bioresour. Technol. 2018, 257, 320–333. [Google Scholar] [CrossRef]
- Imran, A.; Bramer, E.A.; Seshan, K.; Brem, G. High quality bio-oil from catalytic flash pyrolysis of lignocellulosic biomass over alumina-supported sodium carbonate. Fuel Process. Technol. 2014, 127, 72–79. [Google Scholar] [CrossRef]
- Zhou, S.; Xue, Y.; Cai, J.; Cui, C.; Ni, Z.; Zhou, Z. An understanding for improved biomass pyrolysis: Toward a systematic comparison of different acid pretreatments. Chem. Eng. J. 2021, 411, 128513. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, M.; Jiang, E.; Wang, D.; Zhang, K.; Ren, Y.; Jiang, Y. Pyrolysis of Torrefied Biomass. Trends Biotechnol. 2018, 36, 1287–1298. [Google Scholar] [CrossRef] [Green Version]
- Chen, D.; Mei, J.; Li, H.; Li, Y.; Lu, M.; Ma, T.; Ma, Z. Combined pretreatment with torrefaction and washing using torrefaction liquid products to yield upgraded biomass and pyrolysis products. Bioresour. Technol. 2017, 228, 62–68. [Google Scholar] [CrossRef]
- Han, Y.; Gholizadeh, M.; Tran, C.-C.; Kaliaguine, S.; Li, C.-Z.; Olarte, M.; Garcia-Perez, M. Hydrotreatment of pyrolysis bio-oil: A review. Fuel Process. Technol. 2019, 195, 106140. [Google Scholar] [CrossRef]
- Tabassum, N.; Pothu, R.; Pattnaik, A.; Boddula, R.; Balla, P.; Gundeboyina, R.; Challa, P.; Rajesh, R.; Perugopu, V.; Mameda, N.; et al. Heterogeneous Catalysts for Conversion of Biodiesel-Waste Glycerol into High-Added-Value Chemicals. Catalysts 2022, 12, 767. [Google Scholar] [CrossRef]
- Wang, Y.; Akbarzadeh, A.; Chong, L.; Du, J.; Tahir, N.; Awasthi, M.K. Catalytic pyrolysis of lignocellulosic biomass for bio-oil production: A review. Chemosphere 2022, 297, 134181. [Google Scholar] [CrossRef]
- Qiu, B.; Tao, X.; Wang, J.; Liu, Y.; Li, S.; Chu, H. Research progress in the preparation of high-quality liquid fuels and chemicals by catalytic pyrolysis of biomass: A review. Energy Convers. Manag. 2022, 261, 115647. [Google Scholar] [CrossRef]
- Liu, R.; Sarker, M.; Rahman, M.M.; Li, C.; Chai, M.; Nishu; Cotillon, R.; Scott, N.R. Multi-scale complexities of solid acid catalysts in the catalytic fast pyrolysis of biomass for bio-oil production—A review. Prog. Energy Combust. Sci. 2020, 80, 100852. [Google Scholar] [CrossRef]
- Djakaria, K.; Tang, Z.; Shao, J.; Chen, X.; Xiao, H.; Smith, J.; Yang, H.; Chen, H. Improving the production of furfural from cellulose catalytic pyrolysis using WO3/γ-Al2O3 composite oxides. J. Anal. Appl. Pyrolysis 2022, 167, 105648. [Google Scholar] [CrossRef]
- Zhang, W.; Xia, H.; Deng, Y.; Zhang, Q.; Xin, C. Slow pyrolysis of waste navel orange peels with metal oxide catalysts to produce high-grade bio-oil. Green Process. Synth. 2022, 11, 218–228. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, L.; Li, Q.; Wang, Y.; Liu, Q.; Wei, T.; Dong, D.; Salavati, S.; Gholizadeh, M.; Hu, X. Catalytic pyrolysis of poplar wood over transition metal oxides: Correlation of catalytic behaviors with physiochemical properties of the oxides. Biomass Bioenergy 2019, 124, 125–141. [Google Scholar] [CrossRef]
- Wang, W.; Liu, Y.; Wang, Y.; Liu, L.; Hu, C. Effect of Ni(NO3)2 Pretreatment on the Pyrolysis of Organsolv Lignin Derived from Corncob Residue. Processes 2020, 9, 23. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Y.; Wang, W.; Liu, L.; Hu, C. Effects of MgCl2 Solution Pretreatment at Room Temperature on the Pyrolytic Behavior of Pubescens and the Properties of Bio-oil Obtained. Energy Fuels 2020, 34, 12665–12677. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, B.; Shujaa Aldeen, A.; Mwenya, S.; Cheng, H.; Xu, Z.; Zhang, H. Enhancing production of hydrocarbon-rich bio-oil from biomass via catalytic fast pyrolysis coupled with advanced oxidation process pretreatment. Bioresour. Technol. 2022, 359, 127450. [Google Scholar] [CrossRef]
- Su, J.; Miao, K.; Zhao, Y.; Li, T.; Zhao, Z.; Luo, G.; Wang, K. Production of gasoline components from biomass catalytic hydropyrolysis using zeolite-based bifunctional catalysts. Biomass Convers. Biorefin. 2022, 1–13. [Google Scholar] [CrossRef]
- Wu, L.; Xin, J.; Xia, D.; Sun, J.; Liang, J. Enhanced production of hydrocarbons from the catalytic pyrolysis of maize straw over hierarchical ZSM-11 zeolites. Appl. Catal. B 2022, 317, 121775. [Google Scholar] [CrossRef]
- Rahman, M.M.; Liu, R.; Cai, J. Catalytic fast pyrolysis of biomass over zeolites for high quality bio-oil—A review. Fuel Process. Technol. 2018, 180, 32–46. [Google Scholar] [CrossRef]
- Huang, X.; Ren, J.; Ran, J.-Y.; Qin, C.-L.; Yang, Z.-Q.; Cao, J.-P. Recent advances in pyrolysis of cellulose to value-added chemicals. Fuel Process. Technol. 2022, 229, 107175. [Google Scholar] [CrossRef]
- Vuppaladadiyam, A.K.; Vuppaladadiyam, S.S.V.; Sahoo, A.; Murugavelh, S.; Anthony, E.; Bashkar, T.; Zheng, Y.; Zhao, M.; Duan, H.; Zhao, Y.; et al. Bio-oil and biochar from the pyrolytic conversion of biomass: A current and future perspective on the trade-off between economic, environmental, and technical indicators. Sci. Total Environ. 2022, 857, 159155. [Google Scholar] [CrossRef]
- Kan, T.; Strezov, V.; Evans, T.; He, J.; Kumar, R.; Lu, Q. Catalytic pyrolysis of lignocellulosic biomass: A review of variations in process factors and system structure. Renew. Sustain. Energy Rev. 2020, 134, 110305. [Google Scholar] [CrossRef]
- Chen, X.; Che, Q.; Li, S.; Liu, Z.; Yang, H.; Chen, Y.; Wang, X.; Shao, J.; Chen, H. Recent developments in lignocellulosic biomass catalytic fast pyrolysis: Strategies for the optimization of bio-oil quality and yield. Fuel Process. Technol. 2019, 196, 106180. [Google Scholar] [CrossRef]
- Somerville, C.; Youngs, H.; Taylor, C.; Davis, S.C.; Long, S.P. Feedstocks for lignocellulosic biofuels. Science 2010, 329, 790–792. [Google Scholar] [CrossRef] [Green Version]
- Luby, I.H.; Miller, S.J.; Polasky, S. When and where to protect forests. Nature 2022, 609, 89–93. [Google Scholar] [CrossRef]
- Xu, S.; Wang, R.; Gasser, T.; Ciais, P.; Penuelas, J.; Balkanski, Y.; Boucher, O.; Janssens, I.A.; Sardans, J.; Clark, J.H.; et al. Delayed use of bioenergy crops might threaten climate and food security. Nature 2022, 609, 299–306. [Google Scholar] [CrossRef]
- Babu, B.V. Biomass pyrolysis: A state-of-the-art review. Biofuels Bioprod. Biorefin. 2008, 2, 393–414. [Google Scholar] [CrossRef]
- Pasangulapati, V.; Ramachandriya, K.D.; Kumar, A.; Wilkins, M.R.; Jones, C.L.; Huhnke, R.L. Effects of cellulose, hemicellulose and lignin on thermochemical conversion characteristics of the selected biomass. Bioresour. Technol. 2012, 114, 663–669. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, H.; Wang, X.; Chen, W.; Chen, H. Biomass Pyrolytic Polygeneration System: Adaptability for Different Feedstocks. Energy Fuels 2016, 30, 414–422. [Google Scholar] [CrossRef]
- Biswas, B.; Pandey, N.; Bisht, Y.; Singh, R.; Kumar, J.; Bhaskar, T. Pyrolysis of agricultural biomass residues: Comparative study of corn cob, wheat straw, rice straw and rice husk. Bioresour. Technol. 2017, 237, 57–63. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, W.; Wang, Y.; Liu, L.; Li, G.; Hu, C. Enhanced pyrolysis of lignocellulosic biomass by room-temperature dilute sulfuric acid pretreatment. J. Anal. Appl. Pyrolysis 2022, 166, 105588. [Google Scholar] [CrossRef]
- Mendes, F.L.; Ximenes, V.L.; de Almeida, M.B.B.; Azevedo, D.A.; Tessarolo, N.S.; de Rezende Pinho, A. Catalytic pyrolysis of sugarcane bagasse and pinewood in a pilot scale unit. J. Anal. Appl. Pyrolysis 2016, 122, 395–404. [Google Scholar] [CrossRef]
- Huang, W.; Gong, F.; Fan, M.; Zhai, Q.; Hong, C.; Li, Q. Production of light olefins by catalytic conversion of lignocellulosic biomass with HZSM-5 zeolite impregnated with 6wt.% lanthanum. Bioresour. Technol. 2012, 121, 248–255. [Google Scholar] [CrossRef]
- Fu, X.; Li, Q.; Hu, C. Identification and structural characterization of oligomers formed from the pyrolysis of biomass. J. Anal. Appl. Pyrolysis 2019, 144, 104696. [Google Scholar] [CrossRef]
- Ma, Y.; Bao, H.; Hu, X.; Wang, R.; Dong, W. Productions of phenolic rich bio-oil using waste chilli stem biomass by catalytic pyrolysis: Evaluation of reaction parameters on products distributions. J. Energy Inst. 2021, 97, 233–239. [Google Scholar] [CrossRef]
- Ozbay, N.; Yargic, A.S.; Yarbay Sahin, R.Z.; Yaman, E. Valorization of banana peel waste via in-situ catalytic pyrolysis using Al-Modified SBA-15. Renew. Energy 2019, 140, 633–646. [Google Scholar] [CrossRef]
- Zhao, J.; Yao, F.; Hu, C. Enhancing enzymatic hydrolysis efficiency of crop straws via tetrahydrofuran/water co-solvent pretreatment. Bioresour. Technol. 2022, 358, 127428. [Google Scholar] [CrossRef]
- Yao, F.; Shen, F.; Wan, X.; Hu, C. High yield and high concentration glucose production from corncob residues after tetrahydrofuran + H2O co-solvent pretreatment and followed by enzymatic hydrolysis. Renew. Sustain. Energy Rev. 2020, 132, 110107. [Google Scholar] [CrossRef]
- Pirbazari, S.M.; Norouzi, O.; Kohansal, K.; Tavasoli, A. Experimental studies on high-quality bio-oil production via pyrolysis of Azolla by the use of a three metallic/modified pyrochar catalyst. Bioresour. Technol. 2019, 291, 121802. [Google Scholar] [CrossRef]
- Dhepe, P.L.; Fukuoka, A. Cellulose Conversion under Heterogeneous Catalysis. ChemSusChem 2008, 1, 969–975. [Google Scholar] [CrossRef]
- Shen, D.; Jin, W.; Hu, J.; Xiao, R.; Luo, K. An overview on fast pyrolysis of the main constituents in lignocellulosic biomass to valued-added chemicals: Structures, pathways and interactions. Renew. Sustain. Energy Rev. 2015, 51, 761–774. [Google Scholar] [CrossRef]
- Girio, F.M.; Fonseca, C.; Carvalheiro, F.; Duarte, L.C.; Marques, S.; Bogel-Lukasik, R. Hemicelluloses for fuel ethanol: A review. Bioresour. Technol. 2010, 101, 4775–4800. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Wu, L.; Zheng, Z.; Dong, C.; Yang, Y. Lignin hydrolysis and phosphorylation mechanism during phosphoric acid-acetone pretreatment: A DFT study. Molecules 2014, 19, 21335–21349. [Google Scholar] [CrossRef] [PubMed]
- Neiva, D.M.; Rencoret, J.; Marques, G.; Gutierrez, A.; Gominho, J.; Pereira, H.; Del Rio, J.C. Lignin from Tree Barks: Chemical Structure and Valorization. ChemSusChem 2020, 13, 4537–4547. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Flores, F.G.; Dobado, J.A. Lignin as renewable raw material. ChemSusChem 2010, 3, 1227–1235. [Google Scholar] [CrossRef]
- Huang, C.; Jiang, X.; Shen, X.; Hu, J.; Tang, W.; Wu, X.; Ragauskas, A.; Jameel, H.; Meng, X.; Yong, Q. Lignin-enzyme interaction: A roadblock for efficient enzymatic hydrolysis of lignocellulosics. Renew. Sustain. Energy Rev. 2022, 154, 111822. [Google Scholar] [CrossRef]
- Li, Y.; Liu, X.; Zhang, S.; Yao, Y.; Yao, X.; Xu, J.; Lu, X. Dissolving process of a cellulose bunch in ionic liquids: A molecular dynamics study. Phys. Chem. Chem. Phys. 2015, 17, 17894–17905. [Google Scholar] [CrossRef]
- Mazeau, K.; Heux, L. Molecular dynamics simulations of bulk native crystalline and amorphous structures of cellulose. J. Phys. Chem. B 2003, 107, 2394–2403. [Google Scholar] [CrossRef]
- Kim, D.-Y.; Nishiyama, Y.; Wada, M.; Kuga, S.; Okano, T. Thermal Decomposition of Cellulose Crystallites in Wood. Wood Res. Technol. Holzforshung 2001, 55, 521–524. [Google Scholar] [CrossRef]
- Trache, D.; Donnot, A.; Khimeche, K.; Benelmir, R.; Brosse, N. Physico-chemical properties and thermal stability of microcrystalline cellulose isolated from Alfa fibres. Carbohydr. Polym. 2014, 104, 223–230. [Google Scholar] [CrossRef]
- Wang, Z.; McDonald, A.G.; Westerhof, R.J.M.; Kersten, S.R.A.; Cuba-Torres, C.M.; Ha, S.; Pecha, B.; Garcia-Perez, M. Effect of cellulose crystallinity on the formation of a liquid intermediate and on product distribution during pyrolysis. J. Anal. Appl. Pyrolysis 2013, 100, 56–66. [Google Scholar] [CrossRef]
- Wang, Z.; Pecha, B.; Westerhof, R.J.M.; Kersten, S.R.A.; Li, C.-Z.; McDonald, A.G.; Garcia-Perez, M. Effect of Cellulose Crystallinity on Solid/Liquid Phase Reactions Responsible for the Formation of Carbonaceous Residues during Pyrolysis. Ind. Eng. Chem. Res. 2014, 53, 2940–2955. [Google Scholar] [CrossRef]
- Chen, L.; Liao, Y.; Guo, Z.; Cao, Y.; Ma, X. Products distribution and generation pathway of cellulose pyrolysis. J. Clean. Prod. 2019, 232, 1309–1320. [Google Scholar] [CrossRef]
- Wang, S.; Lin, H.; Zhang, L.; Dai, G.; Zhao, Y.; Wang, X.; Ru, B. Structural Characterization and Pyrolysis Behavior of Cellulose and Hemicellulose Isolated from Softwood Pinus armandii Franch. Energy Fuels 2016, 30, 5721–5728. [Google Scholar] [CrossRef]
- Zhou, X.; Nolte, M.W.; Mayes, H.B.; Shanks, B.H.; Broadbelt, L.J. Experimental and Mechanistic Modeling of Fast Pyrolysis of Neat Glucose-Based Carbohydrates. 1. Experiments and Development of a Detailed Mechanistic Model. Ind. Eng. Chem. Res. 2014, 53, 13274–13289. [Google Scholar] [CrossRef] [Green Version]
- Mettler, M.S.; Paulsen, A.D.; Vlachos, D.G.; Dauenhauer, P.J. The chain length effect in pyrolysis: Bridging the gap between glucose and cellulose. Green Chem. 2012, 14, 1284. [Google Scholar] [CrossRef]
- Patwardhan, P.R.; Satrio, J.A.; Brown, R.C.; Shanks, B.H. Product distribution from fast pyrolysis of glucose-based carbohydrates. J. Anal. Appl. Pyrolysis 2009, 86, 323–330. [Google Scholar] [CrossRef]
- Shen, D.K.; Gu, S. The mechanism for thermal decomposition of cellulose and its main products. Bioresour. Technol. 2009, 100, 6496–6504. [Google Scholar] [CrossRef]
- Ponder, G.R.; Richards, G.N.; Stevenson, T.T. Influence of linkage position and orientation in pyrolysis of polysaccharides: A study of several glucans. J. Anal. Appl. Pyrolysis 1992, 22, 217–229. [Google Scholar] [CrossRef]
- Paine, J.B.; Pithawalla, Y.B.; Naworal, J.D.; Thomas, C.E. Carbohydrate pyrolysis mechanisms from isotopic labeling. J. Anal. Appl. Pyrolysis 2007, 80, 297–311. [Google Scholar] [CrossRef]
- Maduskar, S.; Maliekkal, V.; Neurock, M.; Dauenhauer, P.J. On the Yield of Levoglucosan from Cellulose Pyrolysis. ACS Sustain. Chem. Eng. 2018, 6, 7017–7025. [Google Scholar] [CrossRef]
- Lindstrom, J.K.; Proano-Aviles, J.; Johnston, P.A.; Peterson, C.A.; Stansell, J.S.; Brown, R.C. Competing reactions limit levoglucosan yield during fast pyrolysis of cellulose. Green Chem. 2019, 21, 178–186. [Google Scholar] [CrossRef] [Green Version]
- Ding, K.; Zhong, Z.; Wang, J.; Zhang, B.; Fan, L.; Liu, S.; Wang, Y.; Liu, Y.; Zhong, D.; Chen, P.; et al. Improving hydrocarbon yield from catalytic fast co-pyrolysis of hemicellulose and plastic in the dual-catalyst bed of CaO and HZSM-5. Bioresour. Technol. 2018, 261, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Zheng, C. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 2007, 86, 1781–1788. [Google Scholar] [CrossRef]
- Aho, A.; Kumar, N.; Eranen, K.; Holmbom, B.; Hupa, M.; Salmi, T.; Murzin, D.Y. Pyrolysis of softwood carbohydrates in a fluidized bed reactor. Int. J. Mol. Sci. 2008, 9, 1665–1675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lv, G.; Wu, S. Analytical pyrolysis studies of corn stalk and its three main components by TG-MS and Py-GC/MS. J. Anal. Appl. Pyrolysis 2012, 97, 11–18. [Google Scholar] [CrossRef]
- Patwardhan, P.R.; Brown, R.C.; Shanks, B.H. Product distribution from the fast pyrolysis of hemicellulose. ChemSusChem 2011, 4, 636–643. [Google Scholar] [CrossRef]
- Zhou, X.; Li, W.; Mabon, R.; Broadbelt, L.J. A mechanistic model of fast pyrolysis of hemicellulose. Energy Environ. Sci. 2018, 11, 1240–1260. [Google Scholar] [CrossRef]
- Yang, X.; Zhao, Y.; Li, W.; Li, R.; Wu, Y. Unveiling the Pyrolysis Mechanisms of Hemicellulose: Experimental and Theoretical Studies. Energy Fuels 2019, 33, 4352–4360. [Google Scholar] [CrossRef]
- Vispute, T.P.; Zhang, H.; Sanna, A.; Xiao, R.; Huber, G.W. Renewable Chemical Commodity Feedstocks from Integrated Catalytic Processing of Pyrolysis Oils. Science 2010, 330, 1222–1227. [Google Scholar] [CrossRef]
- Jiang, Z.; Hu, C. Selective extraction and conversion of lignin in actual biomass to monophenols: A review. J. Energy Chem. 2016, 25, 947–956. [Google Scholar] [CrossRef]
- Chu, S.; Subrahmanyam, A.V.; Huber, G.W. The pyrolysis chemistry of a β-O-4 type oligomeric lignin model compound. Green Chem. 2013, 15, 125–136. [Google Scholar] [CrossRef]
- Dai, L.; Wang, Y.; Liu, Y.; He, C.; Ruan, R.; Yu, Z.; Jiang, L.; Zeng, Z.; Wu, Q. A review on selective production of value-added chemicals via catalytic pyrolysis of lignocellulosic biomass. Sci. Total Environ. 2020, 749, 142386. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Xiuwen, W.; Hu, J.; Liu, Q.; Shen, D.; Xiao, R. Thermal degradation of softwood lignin and hardwood lignin by TG-FTIR and Py-GC/MS. Polym. Degrad. Stab. 2014, 108, 133–138. [Google Scholar] [CrossRef]
- Jiang, G.; Nowakowski, D.J.; Bridgwater, A.V. Effect of the Temperature on the Composition of Lignin Pyrolysis Products. Energy Fuels 2010, 24, 4470–4475. [Google Scholar] [CrossRef]
- Yuan, J.-M.; Li, H.; Xiao, L.-P.; Wang, T.-P.; Ren, W.-F.; Lu, Q.; Sun, R.-C. Valorization of lignin into phenolic compounds via fast pyrolysis: Impact of lignin structure. Fuel 2022, 319, 123758. [Google Scholar] [CrossRef]
- Wang, S.; Dai, G.; Yang, H.; Luo, Z. Lignocellulosic biomass pyrolysis mechanism: A state-of-the-art review. Prog. Energy Combust. Sci. 2017, 62, 33–86. [Google Scholar]
- Kawamoto, H.; Horigoshi, S.; Saka, S. Effects of side-chain hydroxyl groups on pyrolytic β-ether cleavage of phenolic lignin model dimer. J. Wood Sci. 2007, 53, 268–271. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Wang, D.; Sun, L. The pyrolysis of lignin: Pathway and interaction studies. Fuel 2021, 290, 120078. [Google Scholar] [CrossRef]
- Lv, X.; Li, Q.; Jiang, Z.; Wang, Y.; Li, J.; Hu, C. Structure characterization and pyrolysis behavior of organosolv lignin isolated from corncob residue. J. Anal. Appl. Pyrolysis 2018, 136, 115–124. [Google Scholar] [CrossRef]
- Mu, W.; Ben, H.; Ragauskas, A.; Deng, Y. Lignin Pyrolysis Components and Upgrading—Technology Review. Bioenergy Res. 2013, 6, 1183–1204. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, W.; Ma, H.; Wu, S. Relationship between the formation of oligomers and monophenols and lignin structure during pyrolysis process. Fuel 2020, 276, 118048. [Google Scholar] [CrossRef]
- Perkins, G.; Bhaskar, T.; Konarova, M. Process development status of fast pyrolysis technologies for the manufacture of renewable transport fuels from biomass. Renew. Sustain. Energy Rev. 2018, 90, 292–315. [Google Scholar] [CrossRef]
- Kabir, G.; Hameed, B.H. Recent progress on catalytic pyrolysis of lignocellulosic biomass to high-grade bio-oil and bio-chemicals. Renew. Sustain. Energy Rev. 2017, 70, 945–967. [Google Scholar] [CrossRef]
- Zhang, R.; Li, L.; Tong, D.; Hu, C. Microwave-enhanced pyrolysis of natural algae from water blooms. Bioresour. Technol. 2016, 212, 311–317. [Google Scholar] [CrossRef]
- Caudle, B.; Gorensek, M.B.; Chen, C.-C. A Novel Approach to Modeling Biomass Pyrolysis in a Fluidized Bed Reactor. ACS Sustain. Chem. Eng. 2020, 8, 14605–14615. [Google Scholar] [CrossRef]
- Vamvuka, D. Bio-oil, solid and gaseous biofuels from biomass pyrolysis processes—An overview. Int. J. Energy Res. 2011, 35, 835–862. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, J.; Shanks, B.H.; Brown, R.C. The deleterious effect of inorganic salts on hydrocarbon yields from catalytic pyrolysis of lignocellulosic biomass and its mitigation. Appl. Energy 2015, 148, 115–120. [Google Scholar] [CrossRef]
- Persson, H.; Yang, W. Catalytic pyrolysis of demineralized lignocellulosic biomass. Fuel 2019, 252, 200–209. [Google Scholar] [CrossRef]
- Rahman, M.M.; Chai, M.; Nishu; Sarker, M.; Liu, R. Catalytic pyrolysis of pinewood over ZSM-5 and CaO for aromatic hydrocarbon: Analytical Py-GC/MS study. J. Energy Inst. 2020, 93, 425–435. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, J.; Li, D.; Liu, C.; Lu, Y.; Lin, X.; Zheng, Z. Highly efficient catalytic pyrolysis of biomass vapors upgraded into jet fuel range hydrocarbon-rich bio-oil over a bimetallic Pt–Ni/γ-Al2O3 catalyst. Int. J. Hydrogen Energy 2021, 46, 27922–27940. [Google Scholar] [CrossRef]
- Li, Y.; Yellezuome, D.; Chai, M.; Li, C.; Liu, R. Catalytic pyrolysis of biomass over Fe-modified hierarchical ZSM-5: Insights into mono-aromatics selectivity and pyrolysis behavior using Py-GC/MS and TG-FTIR. J. Energy Inst. 2021, 99, 218–228. [Google Scholar] [CrossRef]
- Du, S.; Sun, Y.; Gamliel, D.P.; Valla, J.A.; Bollas, G.M. Catalytic pyrolysis of miscanthus × giganteus in a spouted bed reactor. Bioresour. Technol. 2014, 169, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Karanjkar, P.U.; Coolman, R.J.; Huber, G.W.; Blatnik, M.T.; Almalkie, S.; de Bruyn Kops, S.M.; Mountziaris, T.J.; Conner, W.C. Production of aromatics by catalytic fast pyrolysis of cellulose in a bubbling fluidized bed reactor. AIChE J. 2014, 60, 1320–1335. [Google Scholar] [CrossRef]
- Jae, J.; Coolman, R.; Mountziaris, T.J.; Huber, G.W. Catalytic fast pyrolysis of lignocellulosic biomass in a process development unit with continual catalyst addition and removal. Chem. Eng. Sci. 2014, 108, 33–46. [Google Scholar] [CrossRef]
- Iliopoulou, E.F.; Stefanidis, S.; Kalogiannis, K.; Psarras, A.C.; Delimitis, A.; Triantafyllidis, K.S.; Lappas, A.A. Pilot-scale validation of Co-ZSM-5 catalyst performance in the catalytic upgrading of biomass pyrolysis vapours. Green Chem. 2014, 16, 662–674. [Google Scholar] [CrossRef]
- Wagenaar, B.M.; Prins, W.; van Swaaij, W.P.M. Pyrolysis of biomass in the rotating cone reactor: Modelling and experimental justification. Chem. Eng. Sci. 1994, 49, 5109–5126. [Google Scholar] [CrossRef] [Green Version]
- Hu, G.; Gong, X.; Huang, H.; Fan, H.; Wang, Z. Experimental studies on flow and pyrolysis of coal with solid heat carrier in a modified rotating cone reactor. Chem. Eng. Process. 2008, 47, 1777–1785. [Google Scholar]
- Campuzano, F.; Brown, R.C.; Martínez, J.D. Auger reactors for pyrolysis of biomass and wastes. Renew. Sustain. Energy Rev. 2019, 102, 372–409. [Google Scholar] [CrossRef]
- Yildiz, G.; Pronk, M.; Djokic, M.; van Geem, K.M.; Ronsse, F.; van Duren, R.; Prins, W. Validation of a new set-up for continuous catalytic fast pyrolysis of biomass coupled with vapour phase upgrading. J. Anal. Appl. Pyrolysis 2013, 103, 343–351. [Google Scholar] [CrossRef]
- Isahak, W.N.R.W.; Hisham, M.W.M.; Yarmo, M.A.; Yun Hin, T.-Y. A review on bio-oil production from biomass by using pyrolysis method. Renew. Sustain. Energy Rev. 2012, 16, 5910–5923. [Google Scholar] [CrossRef]
- Jung, S.-H.; Kang, B.-S.; Kim, J.-S. Production of bio-oil from rice straw and bamboo sawdust under various reaction conditions in a fast pyrolysis plant equipped with a fluidized bed and a char separation system. J. Anal. Appl. Pyrolysis 2008, 82, 240–247. [Google Scholar] [CrossRef]
- Azargohar, R.; Jacobson, K.L.; Powell, E.E.; Dalai, A.K. Evaluation of properties of fast pyrolysis products obtained, from Canadian waste biomass. J. Anal. Appl. Pyrolysis 2013, 104, 330–340. [Google Scholar] [CrossRef]
- Akhtar, J.; Saidina Amin, N. A review on operating parameters for optimum liquid oil yield in biomass pyrolysis. Renew. Sustain. Energy Rev. 2012, 16, 5101–5109. [Google Scholar] [CrossRef]
- Onay, Ö.; Beis, S.H.; Koçkar, Ö.M. Fast pyrolysis of rape seed in a well-swept fixed-bed reactor. J. Anal. Appl. Pyrolysis 2001, 58–59, 995–1007. [Google Scholar] [CrossRef]
- Salehi, E.; Abedi, J.; Harding, T. Bio-oil from Sawdust: Pyrolysis of Sawdust in a Fixed-Bed System. Energy Fuels 2009, 23, 3767–3772. [Google Scholar] [CrossRef]
- Ozbay, N.; Pütün, A.E.; Pütün, E. Bio-oil production from rapid pyrolysis of cottonseed cake: Product yields and compositions. Int. J. Energy Res. 2006, 30, 501–510. [Google Scholar] [CrossRef]
- Li, Q.; Fu, X.; Li, J.; Wang, Y.; Lv, X.; Hu, C. Effect of Heating Rate on Yields and Distribution of Oil Products from the Pyrolysis of Pubescen. Energy Technol. 2018, 6, 366–378. [Google Scholar] [CrossRef]
- Debdoubi, A.; El Amarti, A.; Colacio, E.; Blesa, M.J.; Hajjaj, L.H. The effect of heating rate on yields and compositions of oil products from esparto pyrolysis. Int. J. Energy Res. 2006, 30, 1243–1250. [Google Scholar] [CrossRef]
- Kan, T.; Strezov, V.; Evans, T.J. Lignocellulosic biomass pyrolysis: A review of product properties and effects of pyrolysis parameters. Renew. Sustain. Energy Rev. 2016, 57, 1126–1140. [Google Scholar] [CrossRef]
- Scott, D.S.; Majerski, P.; Piskorz, J.; Radlein, D. A second look at fast pyrolysis of biomass—The RTI process. J. Anal. Appl. Pyrolysis 1999, 51, 23–37. [Google Scholar] [CrossRef]
- Heo, H.S.; Park, H.J.; Dong, J.-I.; Park, S.H.; Kim, S.; Suh, D.J.; Suh, Y.-W.; Kim, S.-S.; Park, Y.-K. Fast pyrolysis of rice husk under different reaction conditions. J. Ind. Eng. Chem. 2010, 16, 27–31. [Google Scholar] [CrossRef]
- Qureshi, K.M.; Kay Lup, A.N.; Khan, S.; Abnisa, F.; Wan Daud, W.M.A. Optimization of palm shell pyrolysis parameters in helical screw fluidized bed reactor: Effect of particle size, pyrolysis time and vapor residence time. Cleaner Eng. Technol. 2021, 4, 100174. [Google Scholar] [CrossRef]
- Islam, M.R.; Parveen, M.; Haniu, H. Properties of sugarcane waste-derived bio-oils obtained by fixed-bed fire-tube heating pyrolysis. Bioresour. Technol. 2010, 101, 4162–4168. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, H.; Karim, A.M.; Sun, J.; Wang, Y. Catalytic fast pyrolysis of lignocellulosic biomass. Chem. Soc. Rev. 2014, 43, 7594–7623. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Hu, C.; Yang, Y.; Zhu, L.; Tong, D. Effect of the Interference Instant of Zeolite HY Catalyst on the Pyrolysis of Pubescens. Chin. J. Chem. Eng. 2010, 18, 351–354. [Google Scholar] [CrossRef]
- Hu, C.; Yang, Y.; Luo, J.; Pan, P.; Tong, D.; Li, G. Recent advances in the catalytic pyrolysis of biomass. Front. Chem. Sci. Eng. 2011, 5, 188–193. [Google Scholar] [CrossRef]
- Kumar, R.; Strezov, V.; Lovell, E.; Kan, T.; Weldekidan, H.; He, J.; Dastjerdi, B.; Scott, J. Bio-oil upgrading with catalytic pyrolysis of biomass using Copper/zeolite-Nickel/zeolite and Copper-Nickel/zeolite catalysts. Bioresour. Technol. 2019, 279, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Zhao, B.; Zhu, L.; Tong, D.; Hu, C. Catalytic pyrolysis of natural algae from water blooms over nickel phosphide for high quality bio-oil production. RSC Adv. 2013, 3, 10806–10816. [Google Scholar] [CrossRef]
- Veses, A.; Aznar, M.; López, J.M.; Callén, M.S.; Murillo, R.; García, T. Production of upgraded bio-oils by biomass catalytic pyrolysis in an auger reactor using low cost materials. Fuel 2015, 141, 17–22. [Google Scholar] [CrossRef]
- Wang, L.; Lei, H.; Bu, Q.; Ren, S.; Wei, Y.; Zhu, L.; Zhang, X.; Liu, Y.; Yadavalli, G.; Lee, J.; et al. Aromatic hydrocarbons production from ex situ catalysis of pyrolysis vapor over Zinc modified ZSM-5 in a packed-bed catalysis coupled with microwave pyrolysis reactor. Fuel 2014, 129, 78–85. [Google Scholar] [CrossRef]
- Yathavan, B.K.; Agblevor, F.A. Catalytic Pyrolysis of Pinyon–Juniper Using Red Mud and HZSM-5. Energy Fuels 2013, 27, 6858–6865. [Google Scholar] [CrossRef]
- Carlson, T.R.; Jae, J.; Lin, Y.-C.; Tompsett, G.A.; Huber, G.W. Catalytic fast pyrolysis of glucose with HZSM-5: The combined homogeneous and heterogeneous reactions. J. Catal. 2010, 270, 110–124. [Google Scholar] [CrossRef]
- Jae, J.; Tompsett, G.A.; Foster, A.J.; Hammond, K.D.; Auerbach, S.M.; Lobo, R.F.; Huber, G.W. Investigation into the shape selectivity of zeolite catalysts for biomass conversion. J. Catal. 2011, 279, 257–268. [Google Scholar] [CrossRef]
- Carlson, T.R.; Tompsett, G.A.; Conner, W.C.; Huber, G.W. Aromatic Production from Catalytic Fast Pyrolysis of Biomass-Derived Feedstocks. Top. Catal. 2009, 52, 241–252. [Google Scholar] [CrossRef] [Green Version]
- Yang, M.; Shao, J.; Yang, Z.; Yang, H.; Wang, X.; Wu, Z.; Chen, H. Conversion of lignin into light olefins and aromatics over Fe/ZSM-5 catalytic fast pyrolysis: Significance of Fe contents and temperature. J. Anal. Appl. Pyrolysis 2019, 137, 259–265. [Google Scholar] [CrossRef]
- Kurnia, I.; Karnjanakom, S.; Bayu, A.; Yoshida, A.; Rizkiana, J.; Prakoso, T.; Abudula, A.; Guan, G. In-situ catalytic upgrading of bio-oil derived from fast pyrolysis of lignin over high aluminum zeolites. Fuel Process. Technol. 2017, 167, 730–737. [Google Scholar] [CrossRef]
- Kelkar, S.; Saffron, C.M.; Andreassi, K.; Li, Z.; Murkute, A.; Miller, D.J.; Pinnavaia, T.J.; Kriegel, R.M. A survey of catalysts for aromatics from fast pyrolysis of biomass. Appl. Catal. B 2015, 174–175, 85–95. [Google Scholar] [CrossRef]
- Mukarakate, C.; Watson, M.J.; Ten Dam, J.; Baucherel, X.; Budhi, S.; Yung, M.M.; Ben, H.; Iisa, K.; Baldwin, R.M.; Nimlos, M.R. Upgrading biomass pyrolysis vapors over β-zeolites: Role of silica-to-alumina ratio. Green Chem. 2014, 16, 4891–4905. [Google Scholar] [CrossRef]
- Zhou, L.; Jia, Y.; Nguyen, T.-H.; Adesina, A.A.; Liu, Z. Hydropyrolysis characteristics and kinetics of potassium-impregnated pine wood. Fuel Process. Technol. 2013, 116, 149–157. [Google Scholar] [CrossRef] [Green Version]
- Patwardhan, P.R.; Satrio, J.A.; Brown, R.C.; Shanks, B.H. Influence of inorganic salts on the primary pyrolysis products of cellulose. Bioresour. Technol. 2010, 101, 4646–4655. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Strezov, V.; Zhou, X.; Kumar, R.; Kan, T. Pyrolysis of heavy metal contaminated biomass pre-treated with ferric salts: Product characterisation and heavy metal deportment. Bioresour. Technol. 2020, 313, 123641. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, P. Catalytic effects of copper(II) chloride and aluminum chloride on the pyrolytic behavior of cellulose. J. Anal. Appl. Pyrolysis 2012, 98, 86–97. [Google Scholar] [CrossRef]
- Lu, Q.; Wang, Z.; Dong, C.-Q.; Zhang, Z.-F.; Zhang, Y.; Yang, Y.-P.; Zhu, X.-F. Selective fast pyrolysis of biomass impregnated with ZnCl2: Furfural production together with acetic acid and activated carbon as by-products. J. Anal. Appl. Pyrolysis 2011, 91, 273–279. [Google Scholar] [CrossRef]
- Han, T.; Yang, W.; Jönsson, P.G. Pyrolysis and subsequent steam gasification of metal dry impregnated lignin for the production of H2-rich syngas and magnetic activated carbon. Chem. Eng. J. 2020, 394, 124902. [Google Scholar] [CrossRef]
- Eibner, S.; Broust, F.; Blin, J.; Julbe, A. Catalytic effect of metal nitrate salts during pyrolysis of impregnated biomass. J. Anal. Appl. Pyrolysis 2015, 113, 143–152. [Google Scholar] [CrossRef] [Green Version]
- Shimada, N.; Kawamoto, H.; Saka, S. Different action of alkali/alkaline earth metal chlorides on cellulose pyrolysis. J. Anal. Appl. Pyrolysis 2008, 81, 80–87. [Google Scholar] [CrossRef] [Green Version]
- Khelfa, A.; Bensakhria, A.; Weber, J.V. Investigations into the pyrolytic behaviour of birch wood and its main components: Primary degradation mechanisms, additivity and metallic salt effects. J. Anal. Appl. Pyrolysis 2013, 101, 111–121. [Google Scholar] [CrossRef]
- Carvalho, W.S.; Cunha, I.F.; Pereira, M.S.; Ataíde, C.H. Thermal decomposition profile and product selectivity of analytical pyrolysis of sweet sorghum bagasse: Effect of addition of inorganic salts. Ind. Crops Prod. 2015, 74, 372–380. [Google Scholar] [CrossRef]
- Han, T.; Ding, S.; Yang, W.; Jönsson, P. Catalytic pyrolysis of lignin using low-cost materials with different acidities and textural properties as catalysts. Chem. Eng. J. 2019, 373, 846–856. [Google Scholar] [CrossRef]
- Gupta, J.; Papadikis, K.; Kozhevnikov, I.V.; Konysheva, E.Y. Exploring the potential of red mud and beechwood co-processing for the upgrading of fast pyrolysis vapours. J. Anal. Appl. Pyrolysis 2017, 128, 35–43. [Google Scholar] [CrossRef]
- Wang, S.; Li, Z.; Bai, X.; Yi, W.; Fu, P. Catalytic pyrolysis of lignin with red mud derived hierarchical porous catalyst for alkyl-phenols and hydrocarbons production. J. Anal. Appl. Pyrolysis 2018, 136, 8–17. [Google Scholar] [CrossRef]
- Elfadly, A.M.; Zeid, I.F.; Yehia, F.Z.; Abouelela, M.M.; Rabie, A.M. Production of aromatic hydrocarbons from catalytic pyrolysis of lignin over acid-activated bentonite clay. Fuel Process. Technol. 2017, 163, 1–7. [Google Scholar] [CrossRef]
- Kar, Y. Catalytic cracking of pyrolytic oil by using bentonite clay for green liquid hydrocarbon fuels production. Biomass Bioenergy 2018, 119, 473–479. [Google Scholar] [CrossRef]
- Rabie, A.M.; Mohammed, E.A.; Negm, N.A. Feasibility of modified bentonite as acidic heterogeneous catalyst in low temperature catalytic cracking process of biofuel production from nonedible vegetable oils. J. Mol. Liq. 2018, 254, 260–266. [Google Scholar] [CrossRef]
- Li, J.; Liu, Z.; Tian, Y.; Zhu, Y.; Qin, S.; Qiao, Y. Catalytic conversion of gaseous tars using land, coastal and marine biomass-derived char catalysts in a bench-scale downstream combined fixed bed system. Bioresour. Technol. 2020, 304, 122735. [Google Scholar] [CrossRef]
- Yao, D.; Hu, Q.; Wang, D.; Yang, H.; Wu, C.; Wang, X.; Chen, H. Hydrogen production from biomass gasification using biochar as a catalyst/support. Bioresour. Technol. 2016, 216, 159–164. [Google Scholar] [CrossRef]
- Shen, Y.; Yoshikawa, K. Tar Conversion and Vapor Upgrading via in Situ Catalysis Using Silica-Based Nickel Nanoparticles Embedded in Rice Husk Char for Biomass Pyrolysis/Gasification. Ind. Eng. Chem. Res. 2014, 53, 10929–10942. [Google Scholar] [CrossRef]
- Ren, S.; Lei, H.; Wang, L.; Bu, Q.; Chen, S.; Wu, J. Hydrocarbon and hydrogen-rich syngas production by biomass catalytic pyrolysis and bio-oil upgrading over biochar catalysts. RSC Adv. 2014, 4, 10731–10737. [Google Scholar] [CrossRef]
- Yan, Q.; Wan, C.; Liu, J.; Gao, J.; Yu, F.; Zhang, J.; Cai, Z. Iron nanoparticles in situ encapsulated in biochar-based carbon as an effective catalyst for the conversion of biomass-derived syngas to liquid hydrocarbons. Green Chem. 2013, 15, 1631. [Google Scholar] [CrossRef]
- Kastner, J.R.; Mani, S.; Juneja, A. Catalytic decomposition of tar using iron supported biochar. Fuel Process. Technol. 2015, 130, 31–37. [Google Scholar] [CrossRef] [Green Version]
- Čejka, J.; Wichterlová, B.; Bednářová, S. Alkylation of toluene with ethene over H-ZSM-5 zeolites. Appl. Catal. A 1991, 79, 215–226. [Google Scholar]
- Kubů, M.; Millini, R.; Žilková, N. 10-ring Zeolites: Synthesis, characterization and catalytic applications. Catal. Today 2019, 324, 3–14. [Google Scholar] [CrossRef]
- Alonso, D.M.; Bond, J.Q.; Dumesic, J.A. Catalytic conversion of biomass to biofuels. Green Chem. 2010, 12, 1493. [Google Scholar] [CrossRef]
- Pinar, A.B.; Gómez-Hortigüela, L.; McCusker, L.B.; Pérez-Pariente, J. Controlling the Aluminum Distribution in the Zeolite Ferrierite via the Organic Structure Directing Agent. Chem. Mater. 2013, 25, 3654–3661. [Google Scholar] [CrossRef]
- Holm, M.S.; Svelle, S.; Joensen, F.; Beato, P.; Christensen, C.H.; Bordiga, S.; Bjørgen, M. Assessing the acid properties of desilicated ZSM-5 by FTIR using CO and 2,4,6-trimethylpyridine (collidine) as molecular probes. Appl. Catal. A 2009, 356, 23–30. [Google Scholar] [CrossRef]
- Tan, S.; Zhang, Z.; Sun, J.; Wang, Q. Recent progress of catalytic pyrolysis of biomass by HZSM-5. Chin. J. Catal. 2013, 34, 641–650. [Google Scholar] [CrossRef]
- Ma, Z.; Troussard, E.; van Bokhoven, J.A. Controlling the selectivity to chemicals from lignin via catalytic fast pyrolysis. Appl. Catal. A 2012, 423–424, 130–136. [Google Scholar] [CrossRef]
- Li, X.; Su, L.; Wang, Y.; Yu, Y.; Wang, C.; Li, X.; Wang, Z. Catalytic fast pyrolysis of Kraft lignin with HZSM-5 zeolite for producing aromatic hydrocarbons. Front. Environ. Sci. Eng. 2012, 6, 295–303. [Google Scholar] [CrossRef]
- Stephanidis, S.; Nitsos, C.; Kalogiannis, K.; Iliopoulou, E.F.; Lappas, A.A.; Triantafyllidis, K.S. Catalytic upgrading of lignocellulosic biomass pyrolysis vapours: Effect of hydrothermal pre-treatment of biomass. Catal. Today 2011, 167, 37–45. [Google Scholar] [CrossRef]
- Dai, L.; Wang, Y.; Liu, Y.; Ruan, R.; Duan, D.; Zhao, Y.; Yu, Z.; Jiang, L. Catalytic fast pyrolysis of torrefied corn cob to aromatic hydrocarbons over Ni-modified hierarchical ZSM-5 catalyst. Bioresour. Technol. 2019, 272, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.Y.; Hu, C.; Li, G.; Guo, L.H.; Yang, Y.; Luo, J.; Miao, X.; Du, Y. Catalytic pyrolysis of several kinds of bamboos over zeolite NaY. Green Chem. 2006, 8, 183–190. [Google Scholar] [CrossRef]
- Qi, W.Y.; Yang, Y.; Hu, C. Catalytic Performance of Ultrastable Y Type Zeolite and Mg-Modified Ultrastable Y Type Zeolite on the Pyrolysis of Neosinocalamus affinis. Asian J. Chem. 2013, 25, 4005–4008. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, F.; Yang, X.; Huang, Y.; Liu, C.; Zheng, Z.; Gu, J. Study on aromatics production via the catalytic pyrolysis vapor upgrading of biomass using metal-loaded modified H-ZSM-5. J. Anal. Appl. Pyrolysis 2017, 126, 169–179. [Google Scholar] [CrossRef]
- Maisano, S.; Urbani, F.; Mondello, N.; Chiodo, V. Catalytic pyrolysis of Mediterranean sea plant for bio-oil production. Int. J. Hydrogen Energy 2017, 42, 28082–28092. [Google Scholar] [CrossRef]
- Shahsavari, S.; Sadrameli, S.M. Production of renewable aromatics and heterocycles by catalytic pyrolysis of biomass resources using rhenium and tin promoted ZSM-5 zeolite catalysts. Process Saf. Environ. Prot. 2020, 141, 305–320. [Google Scholar] [CrossRef]
- Hao, Q.; Wang, C.; Lu, D.; Wang, Y.; Li, D.; Li, G. Production of hydrogen-rich gas from plant biomass by catalytic pyrolysis at low temperature. Int. J. Hydrogen Energy 2010, 35, 8884–8890. [Google Scholar]
- Chen, X.; Chen, Y.; Yang, H.; Wang, X.; Che, Q.; Chen, W.; Chen, H. Catalytic fast pyrolysis of biomass: Selective deoxygenation to balance the quality and yield of bio-oil. Bioresour. Technol. 2019, 273, 153–158. [Google Scholar] [CrossRef]
- Pothu, R.; Challa, P.; Rajesh, R.; Boddula, R.; Balaga, R.; Balla, P.; Perugopu, V.; Radwan, A.B.; Abdullah, A.M.; Al-Qahtani, N. Vapour-Phase Selective Hydrogenation of gamma-Valerolactone to 2-Methyltetrahydrofuran Biofuel over Silica-Supported Copper Catalysts. Nanomaterials 2022, 12, 3414. [Google Scholar] [CrossRef]
- Pothu, R.; Mameda, N.; Mitta, H.; Boddula, R.; Gundeboyina, R.; Perugopu, V.; Radwan, A.B.; Abdullah, A.M.; Al-Qahtani, N. High Dispersion of Platinum Nanoparticles over Functionalized Zirconia for Effective Transformation of Levulinic Acid to Alkyl Levulinate Biofuel Additives in the Vapor Phase. J. Compos. Sci. 2022, 6, 300. [Google Scholar] [CrossRef]
- Wang, W.; Liu, Y.; Wang, Y.; Liu, L.; Hu, C. Effect of nickel salts on the production of biochar derived from alkali lignin: Properties and applications. Bioresour. Technol. 2021, 341, 125876. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Liu, X. Applications of red mud as an environmental remediation material: A review. J. Hazard. Mater. 2021, 408, 124420. [Google Scholar] [CrossRef] [PubMed]
- Kaya, K.; Kurtoğlu, S.F.; Uzun, A.; Soyer-Uzun, S. Consequences of Simple Acid-Pretreatments on Geopolymerization and Thermal Stability of Red Mud-Based Geopolymers. Ind. Eng. Chem. Res. 2018, 57, 7156–7168. [Google Scholar] [CrossRef]
- Cornelius, T. Variation of Physico-Chemical and Textural Properties of Laboratory Prepared Red Mud Through Acid and Thermal Activations. Adv. Mater. 2017, 6, 11. [Google Scholar] [CrossRef]
- Bendou, S.; Amrani, M. Effect of Hydrochloric Acid on the Structural of Sodic-Bentonite Clay. J. Miner. Mater. Charact. Eng. 2014, 2, 404–413. [Google Scholar] [CrossRef]
- Ma, J.; Sun, S.; Chen, K. Facile and scalable synthesis of magnetite/carbon adsorbents by recycling discarded fruit peels and their potential usage in water treatment. Bioresour. Technol. 2017, 233, 110–115. [Google Scholar] [CrossRef]
- Li, Y.; Xing, B.; Ding, Y.; Han, X.; Wang, S. A critical review of the production and advanced utilization of biochar via selective pyrolysis of lignocellulosic biomass. Bioresour. Technol. 2020, 312, 123614. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, modification and environmental application of biochar: A review. J. Cleaner Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Leng, L.; Xu, S.; Liu, R.; Yu, T.; Zhuo, X.; Leng, S.; Xiong, Q.; Huang, H. Nitrogen containing functional groups of biochar: An overview. Bioresour. Technol. 2020, 298, 122286. [Google Scholar] [CrossRef]
- Luo, H.; Bao, L.; Wang, H.; Kong, L.; Sun, Y. Microwave-assisted in-situ elimination of primary tars over biochar: Low temperature behaviours and mechanistic insights. Bioresour. Technol. 2018, 267, 333–340. [Google Scholar] [CrossRef]
- Wu, Y.; Sun, Y.; Liang, K.; Yang, Z.; Tu, R.; Fan, X.; Cheng, S.; Yu, H.; Jiang, E.; Xu, X. Enhancing Hydrodeoxygenation of Bio-oil via Bimetallic Ni-V Catalysts Modified by Cross-Surface Migrated-Carbon from Biochar. ACS Appl. Mater. Interfaces 2021, 13, 21482–21498. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Li, J.; Shen, D.; He, L.; Tong, D.; Hu, C. Catalytic pyrolysis of Pubescens to phenols over Ni/C catalyst. Sci. China–Chem. 2010, 53, 1487–1491. [Google Scholar] [CrossRef]


| Type of Biomass | Cellulose/(wt.%) | Hemicellulose/(wt.%) | Lignin/(wt.%) | Ref. |
|---|---|---|---|---|
| Willow | 53.0 | 7.5 | 19.3 | [41] |
| Cypress | 52.9 | 7.3 | 23.9 | [41] |
| Pinewood | 39.0 | 27.5 | 26.9 | [42] |
| Sawdust | 41.9 | 19.3 | 29.6 | [43] |
| Apple branch | 35.6 | 17.7 | 32.5 | [44] |
| Mulberry wood | 44.5 | 15.1 | 22.2 | [44] |
| Sugarcane bagasse | 36.3 | 20.7 | 22.9 | [42] |
| Sugarcane bagasse | 43.8 | 26.2 | 21.8 | [43] |
| Chili stem | 27.1 | 44.2 | 18.4 | [45] |
| Banana peel | 40.2 | 10.5 | 24.3 | [46] |
| Wheat straw | 59.7 | 5.9 | 20.4 | [44] |
| Wheat straw | 40.3 | 20.7 | 20.6 | [47] |
| Sorghum straw | 36.4 | 26.4 | 18.6 | [47] |
| Corn stover | 42.9 | 21.3 | 20.5 | [47] |
| Corncob residues | 62.7 | 3.6 | 25.2 | [48] |
| Corn straw | 45.9 | 19.8 | 16.6 | [44] |
| Rice straw | 35.3 | 17.1 | 24.6 | [47] |
| Rice husk | 44.1 | 21.9 | 25.7 | [43] |
| Pubescens | 39.8 | 14.3 | 20.6 | [41] |
| Azolla.f | 29.0 | 16.5 | 14.7 | [49] |
| Cellulose | Hemicellulose | Lignin | |
|---|---|---|---|
| Temperature for decomposition | 240–350 °C | 200–250 °C | 280–500 °C |
| Structure features | amorphous and crystalline | unstable polysaccharide | disordered polymer rich in phenols |
| Characteristic that impacted pyrolysis | crystallinity index and degree of polymerization | - | three structure units (G/H/S) and abundance of C-C and C-O-C linkage |
| Reaction mechanisms | free radical mechanism, ionic mechanism and concerted mechanism | similar with the mechanism of cellulose, except for the unstable intermediate from xylosyl cation | free radical mechanism |
| Main products | anhydrosugars, furans and humins | furan or pyran ring derivatives, anhydro sugars and acids | phenol, guaiacol, syringol and their derivatives |
| Advantages | Disadvantages | |
|---|---|---|
| Micro py-GC/MS | beneficial for pyrolysis mechanism investigation | difficulties in the calculation of macrolevel indexes of the process performance |
| Fluidized bed | ease in design and operation, high heat transfer rates, scale-up possibility and excellent temperature control | small biomass particles needed |
| Rotating cone | ease in solid mixing and up-scaling, no carrier gas needed, small investment cost | large amount of energy needed for bed heat transfer small biomass particles needed |
| Auger reactors | ease in design and operation, effective temperature control, efficient heat transfer and well mixing of biomass and catalyst | comparatively higher residence and poor mixing at the radical direction in large scale applications |
| Fixed bed | easy design and operation | poor heat transfer, difficulties in a continuous operation and char removal |
| Type of Pyrolysis | Operating Conditions | The Main Pyrolytic Products | ||||
|---|---|---|---|---|---|---|
| Residence Time | Temperature/°C | Heating Rate | Coke/% | Bio-Oil/% | Gas/% | |
| Slow pyrolysis | 5–30 min | 400–600 | <50 °C/min | <35 | <30 | <40 |
| Fast pyrolysis | <5 s | 400–600 | ~1000 °C/s | <25 | <75 | <20 |
| Flash pyrolysis | <0.1 s | 650–900 | ~1000 °C/s | <20 | <20 | <70 |
| Feedstocks | Reactor | Catalyst | Pyrolysis Mode | Ref. | |
|---|---|---|---|---|---|
| Zeolites | Pinewood sawdust | Py-GC/MS | ZSM-5 | in-situ | [100] |
| Pinewood, rice straw and wheat straw | Conical spouted bed reactor | ZSM-5 | in-situ | [103] | |
| Cellulose | Bubbling fluidized bed reactor | ZSM-5 | in-situ | [104] | |
| Sugarcane bagasse and pinewood | Circulating fluidized bed | ZSM-5 | in-situ | [42] | |
| Maize straw | Fixed-bed reactor | ZSM-11 | in-situ | [28] | |
| Beechwood | Bench-scale fixed-bed tubular reactor | Co/ZSM-5 | ex-situ | [106] | |
| Rice husk, sawdust, sugarcane bagasse, cellulose, hemicellulose and lignin | Flowing fixed bed | La/HZSM-5 | in-situ | [43] | |
| Douglas fir sawdust | A packed-bed catalysis closely coupled with microwave pyrolysis | Zn/ZSM-5 | in-situ | [131] | |
| Pubescens | Fixed-bed reactor | HY | ex-situ | [126] | |
| Pine sawdust | Micro-pyrolyzer | Mo/HZSM-5, Ga/HZSM-5, W/HZSM-5 | ex-situ | [27] | |
| Hemicellulose and plastic | Py-GC/MS | HZSM-5 | ex-situ | [73] | |
| Pinyon−juniper | Fluidized bed reactor | HZSM-5 | in-situ | [132] | |
| Glucose | Py-GC/MS | HZSM-5 | in-situ | [133] | |
| Banana peel | Fixed-bed reactor | Al/SBA-15 | in-situ | [46] | |
| Glucose | Py-GC/MS | Small pore ZK-5, SAPO-34, medium pore Ferrierite, ZSM-23, MCM-22, SSZ-20, ZSM-11, ZSM-5, IM-5, TNU-9, and large pore SSZ-55, Beta zeolite, Y zeolite | in-situ | [134] | |
| Poplar sawdust | Py-GC/MS | Fe-modified hierarchical ZSM-5 | in-situ | [102] | |
| Glucose, xylitol, cellobiose and cellulose | Model 2000 pyroprobe analytical pyrolizer | ZSM-5, silicalite, beta, Y-zeolite and silica–alumina | in-situ | [135] | |
| Lignin | Fixed-bed reactor | Fe/ZSM-5 | ex-situ | [136] | |
| Lignin | A quartz fixed-bed reactor | H-Ferrierite, H-Mordenite, H-ZSM-5, H-Beta and H-USY | in-situ | [137] | |
| Poplar | Py-GC/MS | HZSM-5 (SAR 23, 30, 55, 80 and 280), SO42− ZrO2/MCM-41 | in-situ | [138] | |
| Yellow pine | Py-GC/MS | β-zeolite | in-situ | [139] | |
| Metal oxides | Waste navel orange peels | Fixed-bed reactor | Cu2O, CaO, V2O5, Fe2O3, and ZnO | in-situ | [22] |
| Poplar wood | Fixed-bed reactor | CoO, Cr2O3, CuO, Fe2O3, Mn2O3, NiO, TiO2, V2O5, and CeO2 | ex-situ | [23] | |
| Hemicellulose and plastic | Py-GC/MS | CaO | ex-situ | [73] | |
| Pinewood sawdust | Py-GC/MS | CaO | in-situ | [100] | |
| Pine sawdust | Py-GC/MS | Pt-Ni/γ-Al2O3 | [101] | ||
| Chili stem | Fixed-bed reactor | Ni–Ca/SiO2 | in-situ | [45] | |
| Cellulose | Py-GC/MS | WO3/γ-Al2O3 | ex-situ | [21] | |
| Wood fibers | Fixed-bed reactor | Na2CO3/γ-Al2O3 | ex-situ | [12] | |
| Soluble inorganic salts | Pine wood | TGA | CH3COOK | in-situ | [140] |
| Cellulose | Py-GC/MS | NaCl, KCl, MgCl2, CaCl2, Ca(OH)2, Ca(NO3)2, CaCO3 and CaHPO4 | in-situ | [141] | |
| Pubescens | Fixed-bed reactor | MgCl2 | in-situ | [25] | |
| Lignin | Fixed-bed reactor | Ni(NO3)2 | in-situ | [24] | |
| Avicennia marina biomass | Fixed-bed horizontal furnace | FeCl3 and Fe(NO3)3 | in-situ | [142] | |
| Cellulose | A horizontal oven with infrared heating system and dynamic cooling system | CuCl2 and AlCl3 | in-situ | [143] | |
| Corncob, fir wood, bagasse and rice husk | Lab-scale fixed bed | ZnCl2 | in-situ | [144] | |
| Kraft lignin | A horizontal furnace | FeSO4 | in-situ | [145] | |
| Eucalyptus | fixed-bed reactor | Ce, Mn, Fe, Co, Ni, Cu and Zn (nitrates) | in-situ | [146] | |
| Cellulose | Fixed-bed reactor | NaCl, KCl, MgCl2 and CaCl2 | in-situ | [147] | |
| Birch wood | Py-GC/MS | MgCl2, NiCl2 | in-situ | [148] | |
| Sweet sorghum | Py-GC/MS | ZnCl2 and MgCl2 | in-situ | [149] | |
| Low-cost materials | Pine woodchips | Auger reactor | Sepiolite, bentonite, attapulgite and red mud | in-situ | [130] |
| Kraft lignin | Modified fixed bed | Ilmenite (FeTiO3), bentonite (Al-Si-OH), activated carbon (AC) and red mud (RM), | in-situ | [150] | |
| Pinyon−juniper | Fluidized bed reactor | Red mud | in-situ | [132] | |
| Beechwood | Py-GC/MS | Red mud | in-situ | [151] | |
| Corn cob lignin | Vertical fixed-bed microreactor | Red mud | ex-situ | [152] | |
| Alkaline lignin | Down-flow fixed-bed quartz reactor | Acid-activated bentonite clay | in-situ | [153] | |
| Almond shell | Oil cracking | Bentonite clay | - | [154] | |
| Castor oil and jatropha oil | Oil cracking | Acidic bentonite clay | - | [155] | |
| Azolla.f wastes | Fixed-bed reactor | Mg-Ni-Mo/modified pyro-char | ex-situ | [49] | |
| Pyrolytic tar | A bench-scale combined fixed-bed reactor | Biochar of corn stalks, reed and Sargassum horneri | ex-situ | [156] | |
| Wheat straw | Two-stage fixed-bed reactor | Ni/char (char from wheat straw, rice husk and cotton stalk) | ex-situ | [157] | |
| Pyrolytic tar | A downdraft fixed-bed pyrolysis-reforming facility | Nano Ni/rice husk char | in-situ | [158] | |
| Douglas fir sawdust | Batch microwave oven | Corn stover biochar | in-situ | [159] | |
| Bio-syngas | Fixed-bed reactor | Fe0/biochar (from pine) | in-situ | [160] | |
| Toluene | Continuous flow packed bed reactor system | Fe/biochar (from pine) | in-situ | [161] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Gu, Y.; Zhou, C.; Hu, C. Current Challenges and Perspectives for the Catalytic Pyrolysis of Lignocellulosic Biomass to High-Value Products. Catalysts 2022, 12, 1524. https://doi.org/10.3390/catal12121524
Wang W, Gu Y, Zhou C, Hu C. Current Challenges and Perspectives for the Catalytic Pyrolysis of Lignocellulosic Biomass to High-Value Products. Catalysts. 2022; 12(12):1524. https://doi.org/10.3390/catal12121524
Chicago/Turabian StyleWang, Wenli, Yaxin Gu, Chengfen Zhou, and Changwei Hu. 2022. "Current Challenges and Perspectives for the Catalytic Pyrolysis of Lignocellulosic Biomass to High-Value Products" Catalysts 12, no. 12: 1524. https://doi.org/10.3390/catal12121524
APA StyleWang, W., Gu, Y., Zhou, C., & Hu, C. (2022). Current Challenges and Perspectives for the Catalytic Pyrolysis of Lignocellulosic Biomass to High-Value Products. Catalysts, 12(12), 1524. https://doi.org/10.3390/catal12121524






