Application of Ba0.5Sr0.5FeO3−δ-NdMnO3−δ Composite Cathode in Proton-Conducting Solid Oxide Fuel Cells
Abstract
:1. Introduction
2. Results and Discussion
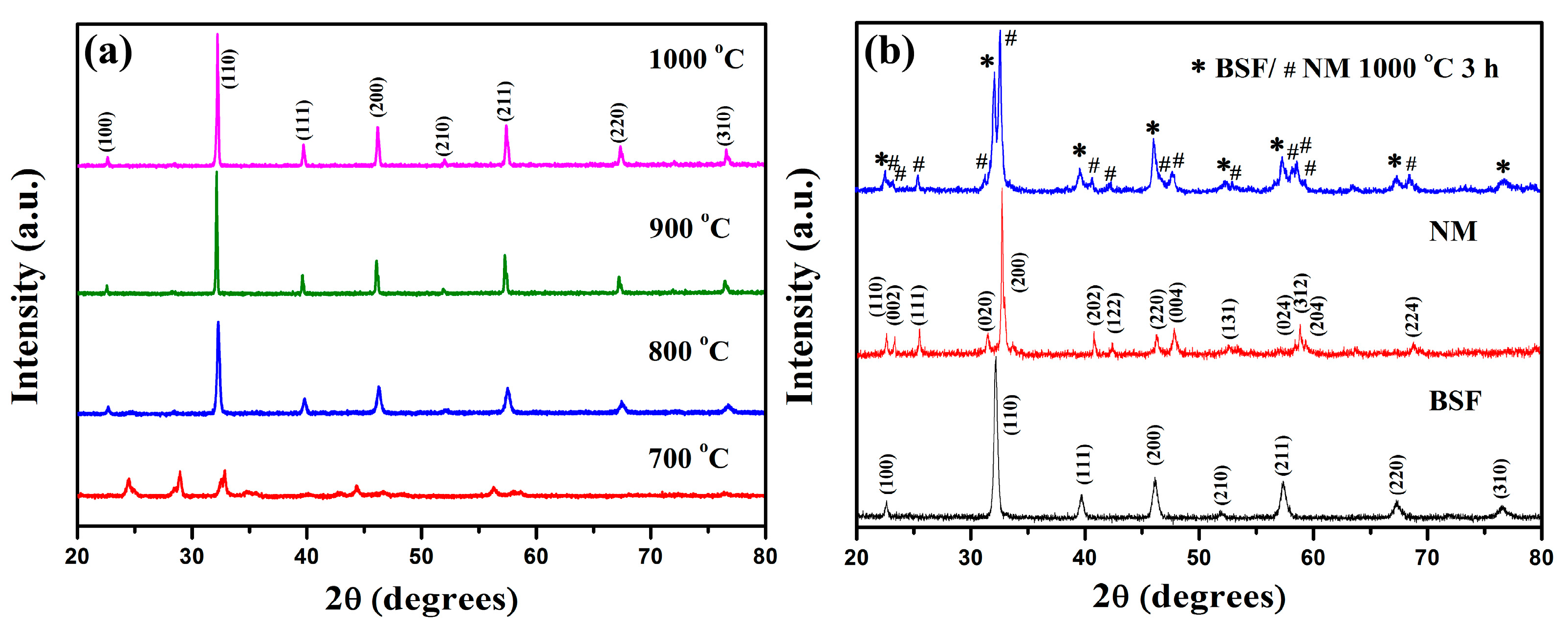
2.1. X-ray Diffraction Analysis and Average Diameter of the BSF Fiber
2.2. Scanning Electron Microscope Analysis
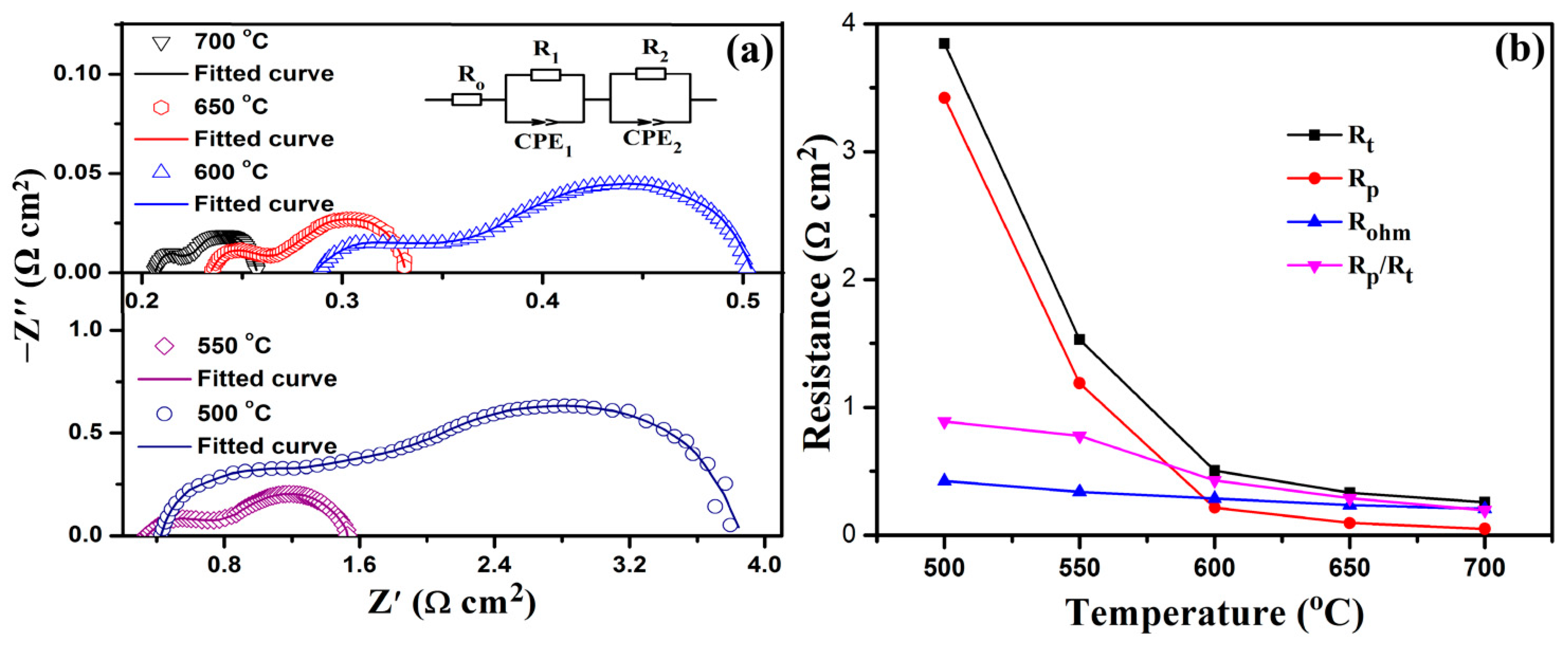
2.3. Electrochemical Performance Test and Analysis
3. Materials and Methods
3.1. Synthesis of Materials
3.2. Preparation of Single Cells
3.3. Characterizations and Electrochemical Properties
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, S.; Shen, J.; Zhu, Z.; Wang, Z.; Cao, Y.; Guan, X.; Wang, Y.; Wei, Z.; Chen, M. Further optimization of barium cerate properties via co-doping strategy for potential application as proton-conducting solid oxide fuel cell electrolyte. J. Power Sources 2018, 387, 24–32. [Google Scholar] [CrossRef]
- Duan, C.; Tong, J.; Shang, M.; Nikodemski, S.; Sanders, M.; Ricote, S.; Almansoori, A.; O’Hayre, R. Readily processed protonic ceramic fuel cells with high performance at low temperatures. Science 2015, 349, 1321–1326. [Google Scholar] [CrossRef]
- Noponena, M.; Torria, P.; Göösa, J.; Puranena, J.; Kaara, H.; Pylypkob, S.; Roostarb, M.; Õunpuub, E. Elcogen–Next Generation Solid Oxide Cell and Stack Technology. ECS Trans. 2019, 91, 91–97. [Google Scholar] [CrossRef]
- Dai, H.; Da’as, E.H.; Shafi, S.P.; Wang, H. Tailoring cathode composite boosts the performance of proton-conducting SOFCs fabricated by a one-step co-firing method. J. Eur. Ceram. Soc. 2018, 38, 2903–2908. [Google Scholar] [CrossRef]
- Han, D.; Shinoda, K.; Sato, S.; Majima, M.; Uda, T. Correlation between electroconductive and structural properties of proton conductive acceptor-doped barium zirconate. J. Mater. Chem. 2015, A3, 1243–1250. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Bi, L.; Zhao, X.S. Highly-conductive proton-conducting electrolyte membranes with a low sintering temperature for solid oxide fuel cells. J. Membr. Sci. 2018, 558, 17–25. [Google Scholar] [CrossRef]
- Medvedev, D.A.; Lyagaeva, J.G.; Gorbova, E.V.; Demin, A.K.; Tsiakaras, P. Advanced materials for SOFC application: Strategies for the development of highly conductive and stable solid oxide proton electrolytes. Prog. Mater. Sci. 2016, 75, 38–79. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, L.; Li, Q.; Song, T.; Su, J.; Cai, B.; He, H. High performance In, Ta and Y-doped BaCeO3 electrolyte membrane for proton-conducting solid oxide fuel cells. Solid State Ion. 2018, 323, 25–31. [Google Scholar] [CrossRef]
- Blum, L.; Haart, L.G.J.; Malzbender, J.; Margaritis, N.; Menzler, N.H. Anode-Supported Solid Oxide Fuel Cell Achieves 70,000 Hours of Continuous Operation. Energy Technol. Gener. Convers. Storage Distrib. 2016, 4, 939–942. [Google Scholar]
- Shao, Z.; Zhou, W.; Zhu, Z. Advanced synthesis of materials for intermediate-temperature solid oxide fuel cells. Prog. Mater. Sci. 2012, 57, 804–874. [Google Scholar] [CrossRef]
- Xu, X.; Xia, C.; Huang, S.; Peng, D. YSZ thin films deposited by spin-coating for IT SOFCs. Ceram. Int. 2005, 31, 1061–1064. [Google Scholar] [CrossRef]
- Mushtaq, N.; Xia, C.; Dong, W.; Abbas, G.; Raza, R.; Ali, A.; Rauf, S.; Wang, B.; Kim, J.; Zhu, B. Perovskite SrFe1−xTixO3−δ (x ≤ 0.1) cathode for low temperature solid oxide fuel cell. Ceram. Int. 2018, 44, 10266–10272. [Google Scholar] [CrossRef] [Green Version]
- Tang, H.; Jin, Z.; Wu, Y.; Liu, W.; Bi, L. Cobalt-free nanofiber cathodes for proton conducting solid oxide fuel cells. Electrochem. Commun. 2019, 100, 108–112. [Google Scholar] [CrossRef]
- Huang, Y.; Ding, J.; Xia, Y.; Miao, L.; Li, K.; Zhang, Q.; Liu, W. Ba0.5Sr0.5Co0.8−xFe0.2NbxO3−δ (x ≤ 0.1) as cathode materials for intermediate temperature solid oxide fuel cells with an electron-blocking interlayer. Ceram. Int. 2020, 46, 10215–10223. [Google Scholar] [CrossRef]
- Huan, D.; Shi, N.; Zhang, L.; Tan, W.; Xie, Y.; Wang, W.; Xia, C.; Peng, R.; Lu, Y. New, Efficient, and Reliable Air Electrode Material for Proton-Conducting Reversible Solid Oxide Cells. ACS Appl. Mater. Interfaces 2018, 10, 1761–1770. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.C.; Shen, Y.; Wang, F.; Duan, Q. Nanofibrous La0.7Sr0.3FeO3−δ-SDC cobalt-free composite cathode for proton-conducting solid oxide fuel cell. Mater. Lett. 2023, 333, 133687. [Google Scholar] [CrossRef]
- Yang, L.; Zuo, C.; Wang, S.; Cheng, Z.; Liu, M. A novel composite cathode for low temperature SOFCs based on oxide proton conductors. Adv. Mater. 2008, 20, 3280–3283. [Google Scholar] [CrossRef]
- Sun, C.C.; Shen, Y.; Wang, F.; Duan, Q. Optimization of a Cobalt-free La0.7Sr0.3FeO3−δ-BaZr0.1Ce0.7Y0.2O3−δ composite cathode for proton-conducting solid oxide fuel cells. J. Alloys Compd. 2022, 923, 166447. [Google Scholar] [CrossRef]
- Sun, W.P.; Shi, Z.; Fang, S.; Yan, L.; Zhu, Z.; Liu, W. A high performance BaZr0.1Ce0.7Y0.2O3−δ-based solid oxide fuel cell with a cobalt-free Ba0.5Sr0.5FeO3−δ-Ce0.8Sm0.2O2−δ composite cathode. Int. J. Hydrog. Energy 2010, 35, 7925–7929. [Google Scholar] [CrossRef]
- Mary, T.A.; Evans, J.S.O.; Vogt, T.; Sleight, A.W. Negative thermal expansion from 0.3 to 1050 Kelvin in ZrW2O8. Science 1996, 272, 90–92. [Google Scholar] [CrossRef] [Green Version]
- Evans, J.S.O.; Hanson, J.C.; Sleight, A.W. Room-temperature superstructure of Zr2V2O7. Acta Crystallogr. 1998, B54, 705–713. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Yang, M.; Chao, M.; Guo, J.; Gao, Q.; Jiao, Y.; Tang, X.; Liang, E. Negative thermal expansion property of β-Cu2V2O7. Solid State Ion. 2019, 343, 115086. [Google Scholar] [CrossRef]
- Oka, K.; Azuma, M.; Chen, W.T.; Yusa, H.; Belik, A.A.; Takayama-Muromachi, E.; Mizumaki, M.; Ishimatsu, N.; Hiraoka, N.; Tsujimoto, M.; et al. Pressure-induced spin-state transition in BiCoO3. J. Am. Chem. Soc. 2010, 132, 9438–9443. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Jiao, Y.; Yang, M.; Chao, M.; Zhu, Y.; Chen, R.; Guo, J.; Gao, Q.; Liang, E. Anomalous thermal expansion properties in perovskite Sm0.85Zn0.15MnO3. Solid State Ion. 2020, 344, 115139. [Google Scholar] [CrossRef]
- Chao, M.; Li, Y.; Zhou, W.; Xiao, X.; Liu, Y.; Zhang, N.; Liang, E.; Li, Y.; Feng, D. Negative Thermal Expansion Material NdMnO3 and its Preparing Method. Chinese Patent CN104495942B, 25 January 2017. [Google Scholar]
- Miao, L.; Hou, J.; Gong, Z.; Jin, Z.; Liu, W. A high-performance cobalt-free Ruddlesden-Popper phase cathode La1.2Sr0.8Ni0.6Fe0.4O4+δ for low temperature proton-conducting solid oxide fuel cells. Int. J. Hydrog. Energy 2019, 44, 7531–7537. [Google Scholar] [CrossRef]
- Lu, F.; Yang, M.; Shi, Y.; Wu, C.; Jia, X.; He, H.; Su, J.; Chao, M.; Cai, B. Application of a negative thermal expansion oxide in SOFC cathode. Ceram. Int. 2021, 47, 1095–1100. [Google Scholar] [CrossRef]
- Hanif, M.B.; Rauf, S.; Abadeen, Z.; Khan, K.; Tayyab, Z.; Qayyum, S.; Mosiałek, M.; Shao, Z.P.; Li, C.X.; Motola, M.; et al. Proton-conducting solid oxide electrolysis cells: Relationship of composition-structure-property, their challenges, and prospects. Matter 2023, 6, 1782–1830. [Google Scholar] [CrossRef]
- Li, Q.; Sun, L.P.; Zhao, H.; Wang, H.L.; Huo, L.H.; Rougier, A.; Fourcade, S.; Grenier, J.C. La1.6Sr0.4NiO4 one-dimensional nanofibers as cathode for solid oxide fuel cells. J. Power Sources 2014, 263, 125–129. [Google Scholar] [CrossRef]
- Fan, L.Q.; Xiong, Y.P.; Liu, L.B.; Wang, Y.W.; Kishimoto, H.; Yamaji, K.; Horita, T. Performance of Gd0.2Ce0.8O1.9 infiltrated La0.2Sr0.8TiO3 nanofiber scaffolds as anodes for solid oxide fuel cells. J. Power Sources 2014, 265, 125–131. [Google Scholar] [CrossRef]
- Aruna, S.T.; Balaji, L.S.; Senthil Kumar, S.; Shri Prakash, B. Electrospinning in solid oxide fuel cells—A review. Renew. Sustain. Energy Rev. 2017, 67, 673–682. [Google Scholar] [CrossRef]
- Zhi, M.J.; Mariani, N.; Gemmen, R.; Gerdes, K.; Wu, N.Q. Nanofiber scaffold for cathode of solid oxide fuel cell. Energy Environ. Sci. 2011, 4, 417–420. [Google Scholar] [CrossRef]
- Zhi, M.; Lee, S.; Miller, N.; Menzler, N.H.; Wu, N. An intermediate-temperature solid oxide fuel cell with electrospun nanofiber cathode. Energy Environ. Sci. 2012, 5, 7066–7071. [Google Scholar] [CrossRef] [Green Version]
- Choi, J.Y.; Kim, B.S.; Shin, D.W. Performance evaluation of Sm0.5Sr0.5CoO3−δ fibers with embedded Sm0.2Ce0.8O1.9 particles as a solid oxide fuel cell composite cathode. J. Eur. Ceram. Soc. 2013, 33, 2269–2273. [Google Scholar] [CrossRef]
- Hanif, M.B.; Motola, M.; Qayyum, S.; Rauf, S.; Khalid, A.; Li, C.J.; Li, C.X. Recent advancements, doping strategies and the future perspective of perovskite-based solid oxide fuel cells for energy conversion. Chem. Eng. J. 2022, 428, 132603. [Google Scholar] [CrossRef]
- Sacanell, J.; Leyva, A.G.; Bellino, M.G.; Lamas, D.G. Nanotubes of rare earth cobalt oxides for cathodes of intermediate-temperature solid oxide fuel cells. J. Power Sources 2010, 195, 1786–1792. [Google Scholar] [CrossRef]
- Zhao, E.Q.; Ma, C.; Yang, W.; Xiong, Y.P.; Li, J.Q.; Sun, C.W. Electrospinning La0.8Sr0.2Co0.2Fe0.8O3−δ tubes impregnated with Ce0.8Gd0.2O1.9 nanoparticles for an intermediate temperature solid oxide fuel cell cathode. Int. J. Hydrog. Energy 2013, 38, 6821–6829. [Google Scholar] [CrossRef]
- Tahir, N.N.M.; Baharuddin, N.A.; Samat, A.A.; Osman, N.; Somalu, M.R. A review on cathode materials for conventional and proton-conducting solid oxide fuel cells. J. Alloys Compd. 2022, 894, 162458. [Google Scholar] [CrossRef]
- Hagen, A.; Barfod, R.; Hendriksen, P.V.; Liu, Y.L.; Ramousse, S. Degradation of Anode Supported SOFCs as a Function of Temperature and Current Load. J. Electrochem. Soc. 2006, 153, A1165–A1171. [Google Scholar] [CrossRef]
- Kim, J.W.; Virkar, A.; Fung, K.Z.; Mehta, K.; Singhal, S. Polarization Effects in Intermediate Temperature, Anode-Supported Solid Oxide Fuel Cells. J. Electrochem. Soc. 1999, 146, 69–78. [Google Scholar] [CrossRef]
- Cai, B.; Song, T.F.; Su, J.R.; He, H.; Liu, Y. Comparison of (Pr, Ba, Sr)FeO3−δ-SDC composite cathodes in proton-conducting solid oxide fuel cells. Solid State Ion. 2020, 353, 115379. [Google Scholar] [CrossRef]
- Ding, H.; Lin, B.; Liu, X.; Meng, G. High performance protonic ceramic membrane fuel cells (PCMFCs) with Ba0.5Sr0.5Zn0.2Fe0.8O3−δ perovskite cathode. Electrochem. Commun. 2008, 10, 1388–1391. [Google Scholar] [CrossRef]
- Zhao, L.; He, B.; Ling, Y.; Xun, Z.; Peng, R.; Meng, G.; Liu, X. Cobalt-free oxide Ba0.5Sr0.5Fe0.8Cu0.2O3−δ for proton-conducting solid oxide fuel cell cathode. Int. J. Hydrog. Energy 2010, 35, 3769–3774. [Google Scholar] [CrossRef]






| Cathode | Electrolyte | T (°C) | Rp (Ω cm2) | Pm (mW cm−2) | Ref. |
|---|---|---|---|---|---|
| Ba0.5Sr0.5FeO3−δ-SDC powder | BZCY | 750 | 0.030 | 792 | [19] |
| Ba0.5Sr0.5FeO3−δ powder | BZCY | 750 | 0.120 | 661 | [27] |
| Ba0.5Sr0.5FeO3−δ-NdMnO3−δ powder | BZCY | 750 | 0.054 | 941 | [27] |
| Ba0.5Sr0.5FeO3−δ-SDC powder | BZCY | 750 | 0.135 | 657 | [41] |
| Pr0.5Ba0.25Sr0.25FeO3−δ-SDC powder | BZCY | 750 | 0.092 | 811 | [41] |
| Ba0.5Sr0.5Zn0.2Fe0.8O3−δ powder | BZCY | 700 | 0.080 | 486 | [42] |
| Ba0.5Sr0.5Fe0.8Cu0.2O3−δ-BZCY powder | BZCY | 700 | 0.160 | 430 | [43] |
| BSF nanofiber-NM powder | BZCY | 700 | 0.050 | 1170 | [this work] |
| T (°C) | Ro (Ω cm2) | R1 (Ω cm2) | R2 (Ω cm2) | Rp (Ω cm2) | Rt (Ω cm2) |
|---|---|---|---|---|---|
| 500 | 0.425 ± 0.014 | 1.072 ± 0.063 | 2.349 ± 0.155 | 3.421 | 3.846 |
| 550 | 0.339 ± 0.019 | 0.360 ± 0.049 | 0.829 ± 0.073 | 1.189 | 1.529 |
| 600 | 0.287 ± 0.017 | 0.047 ± 0.005 | 0.169 ± 0.015 | 0.216 | 0.504 |
| 650 | 0.235 ± 0.011 | 0.028 ± 0.002 | 0.068 ± 0.007 | 0.096 | 0.331 |
| 700 | 0.207 ± 0.008 | 0.013 ± 0.002 | 0.037 ± 0.003 | 0.050 | 0.257 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, C.; Shen, Y.; Wang, F.; Duan, Q.; Jin, F. Application of Ba0.5Sr0.5FeO3−δ-NdMnO3−δ Composite Cathode in Proton-Conducting Solid Oxide Fuel Cells. Catalysts 2023, 13, 1054. https://doi.org/10.3390/catal13071054
Sun C, Shen Y, Wang F, Duan Q, Jin F. Application of Ba0.5Sr0.5FeO3−δ-NdMnO3−δ Composite Cathode in Proton-Conducting Solid Oxide Fuel Cells. Catalysts. 2023; 13(7):1054. https://doi.org/10.3390/catal13071054
Chicago/Turabian StyleSun, Cuicui, Yu Shen, Fang Wang, Qian Duan, and Fangjun Jin. 2023. "Application of Ba0.5Sr0.5FeO3−δ-NdMnO3−δ Composite Cathode in Proton-Conducting Solid Oxide Fuel Cells" Catalysts 13, no. 7: 1054. https://doi.org/10.3390/catal13071054
APA StyleSun, C., Shen, Y., Wang, F., Duan, Q., & Jin, F. (2023). Application of Ba0.5Sr0.5FeO3−δ-NdMnO3−δ Composite Cathode in Proton-Conducting Solid Oxide Fuel Cells. Catalysts, 13(7), 1054. https://doi.org/10.3390/catal13071054







