On the Effect of the Nature of Carbon Nanostructures on the Activity of Bifunctional Catalysts Based on Manganese Oxide Nanowires
Abstract
:1. Introduction
2. Results
2.1. Physical-Chemical Characterization
2.2. Effect of the Carbon Nanostructure on the Catalytic Activity towards the Oxygen Reduction and Evolution Reactions
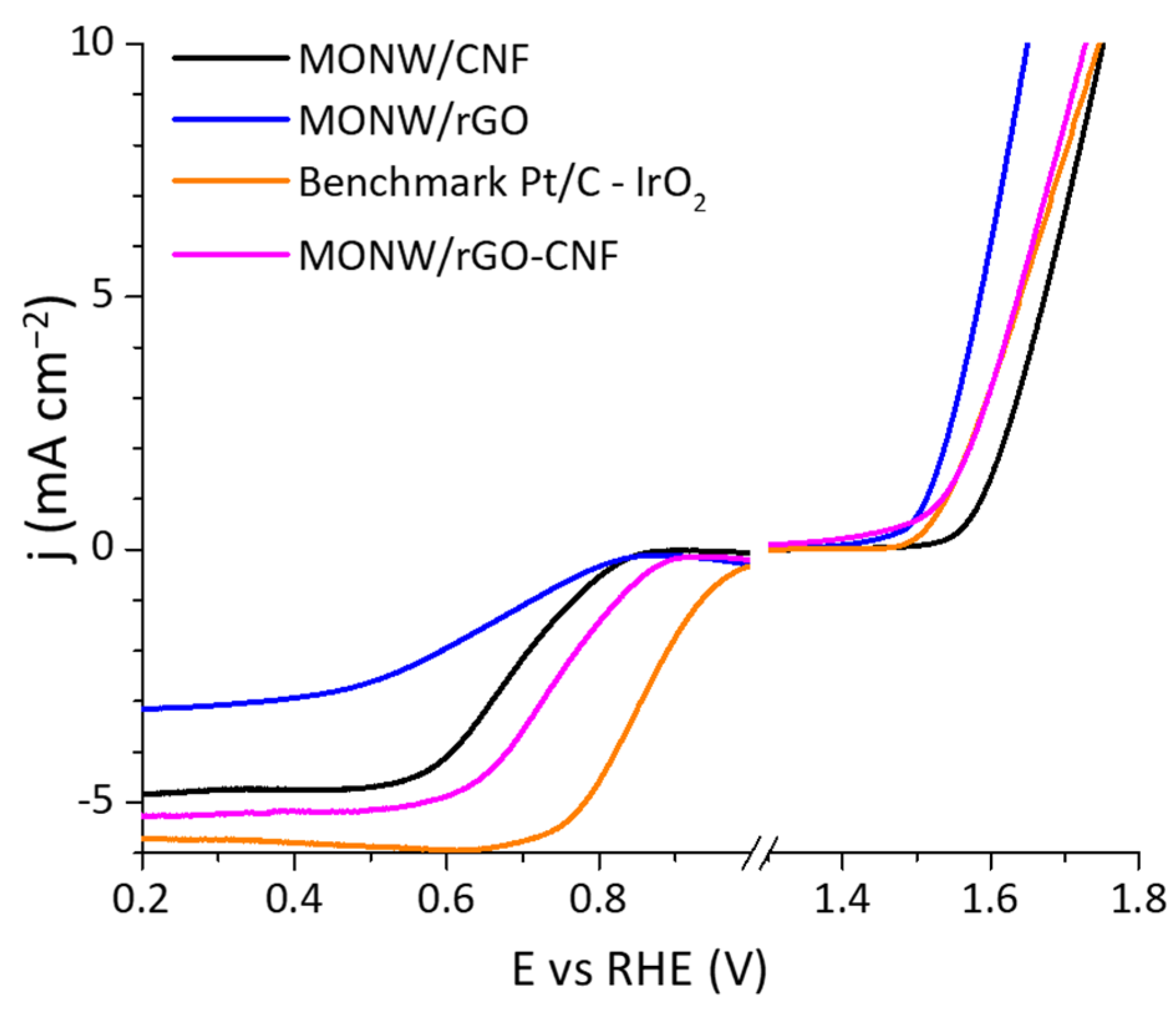
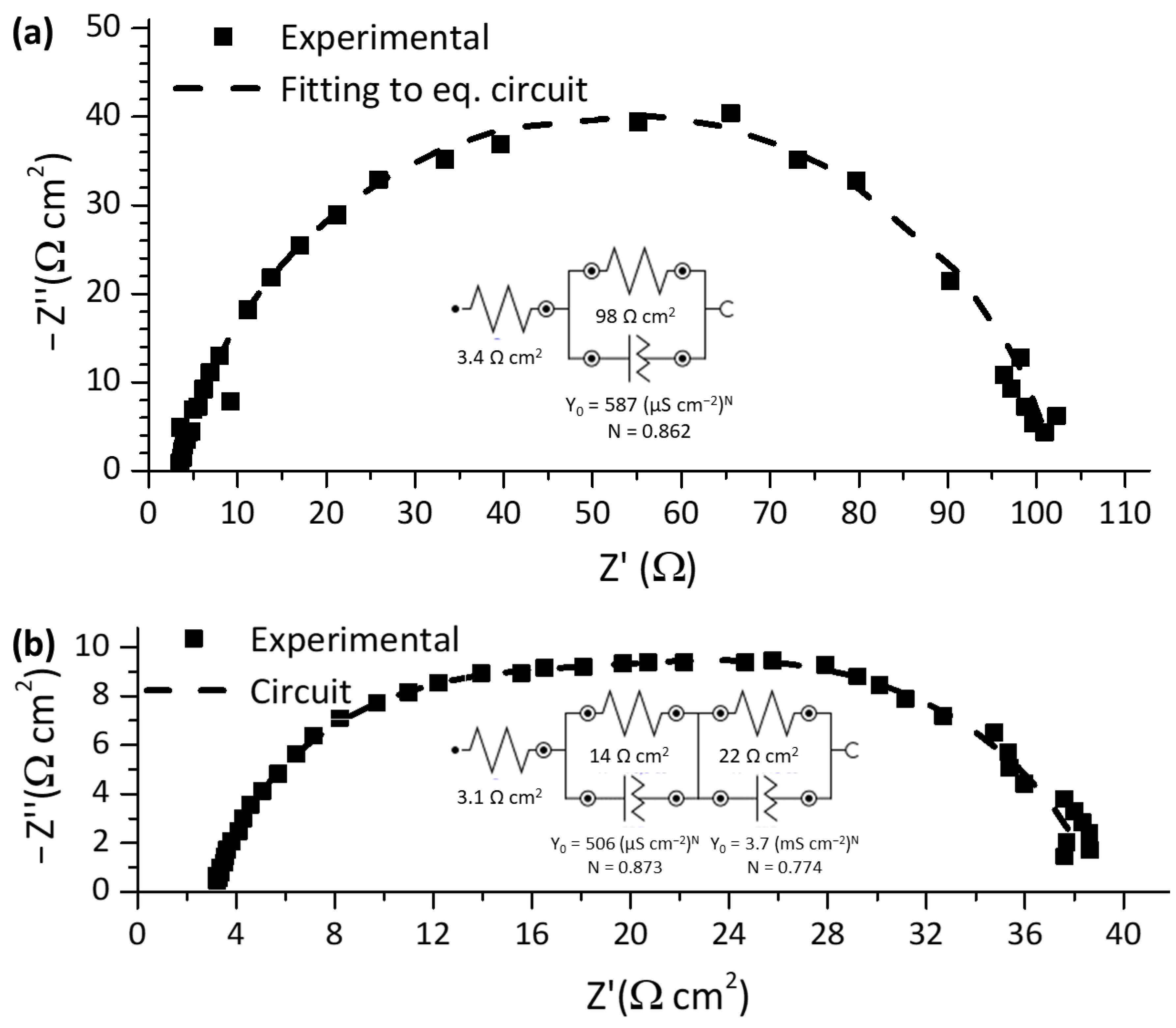
2.3. Catalytic Activity towards the Oxygen Reduction and Evolution Reactions for a Composite Material between Mn Oxide and Hybrid Carbon Nanostructure
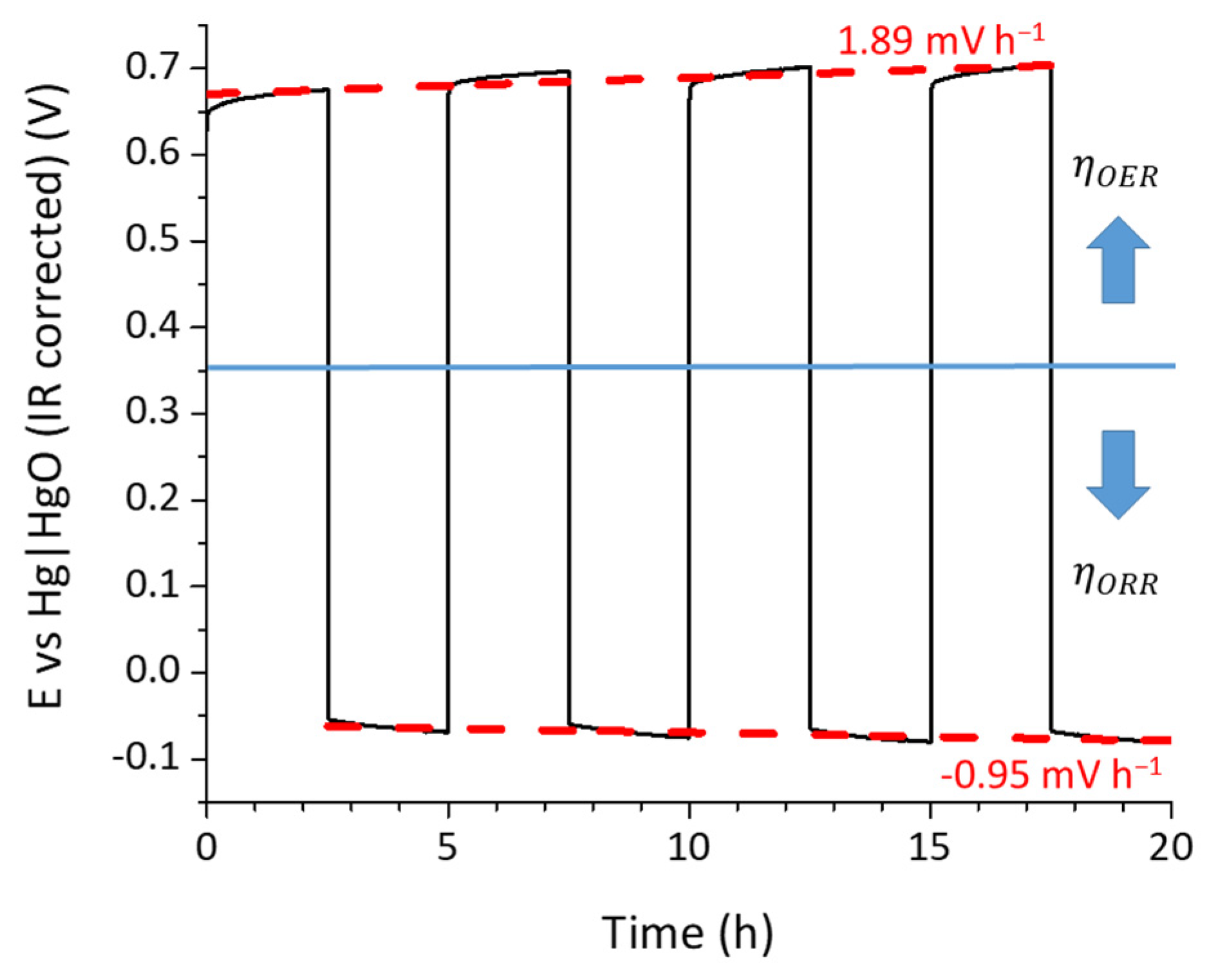
2.4. Stability Studies
3. Materials and Methods
3.1. Catalysts Synthesis
3.2. Physical-Chemical Characterization
3.3. Electrochemical Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- McKerracher, R.D.; Ponce de Leon, C.; Wills, R.G.A.; Shah, A.A.; Walsh, F.C. A Review of the Iron-Air Secondary Battery for Energy Storage. Chempluschem 2015, 80, 323–335. [Google Scholar] [CrossRef]
- Wang, Y.; Leung, D.Y.C.; Xuan, J.; Wang, H. A review on unitized regenerative fuel cell technologies, part B: Unitized regenerative alkaline fuel cell, solid oxide fuel cell, and microfluidic fuel cell. Renew. Sustain. Energy Rev. 2017, 75, 775–795. [Google Scholar] [CrossRef]
- Xie, X.; Du, L.; Yan, L.; Park, S.; Qiu, Y.; Sokolowski, J.; Wang, W.; Shao, Y. Oxygen Evolution Reaction in Alkaline Environment: Material Challenges and Solutions. Adv. Funct. Mater. 2022, 32, 2110036. [Google Scholar] [CrossRef]
- Shao, Y.; Jiang, Z.; Zhang, Q.; Guan, J. Progress in Nonmetal-Doped Graphene Electrocatalysts for the Oxygen Reduction Reaction. ChemSusChem 2019, 12, 2133–2146. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.J. An impending platinum crisis and its implications for the future of the automobile. Energy Policy 2009, 37, 1805–1808. [Google Scholar] [CrossRef]
- Minke, C.; Suermann, M.; Bensmann, B.; Hanke-Rauschenbach, R. Is iridium demand a potential bottleneck in the realization of large-scale PEM water electrolysis? Int. J. Hydrog. Energy 2021, 46, 23581–23590. [Google Scholar] [CrossRef]
- Alegre, C.; Stassi, A.; Modica, E.; Lo Vecchio, C.; Aricò, A.S.; Baglio, V. Investigation of the activity and stability of Pd-based catalysts towards the oxygen reduction (ORR) and evolution reactions (OER) in iron-air batteries. RSC Adv. 2015, 5, 25424–25427. [Google Scholar] [CrossRef]
- McKerracher, R.D.; Alegre, C.; Baglio, V.; Aricò, A.S.; Ponce De León, C.; Mornaghini, F.; Rodlert, M.; Walsh, F.C. A nanostructured bifunctional Pd/C gas-diffusion electrode for metal-air batteries. Electrochim. Acta 2015, 174, 508–515. [Google Scholar] [CrossRef]
- Alegre, C.; Modica, E.; Lo Vecchio, C.; Sebastián, D.; Lázaro, M.J.; Aricò, A.S.; Baglio, V. Carbon Nanofibers as Advanced Pd Catalyst Supports for the Air Electrode of Alkaline Metal-Air Batteries. Chempluschem 2015, 80, 1384–1388. [Google Scholar] [CrossRef]
- Zhao, Q.; Yan, Z.; Chen, C.; Chen, J. Spinels: Controlled Preparation, Oxygen Reduction/Evolution Reaction Application, and beyond. Chem. Rev. 2017, 117, 10121–10211. [Google Scholar] [CrossRef]
- Alegre, C.; Modica, E.; Aricò, A.S.; Baglio, V. Bifunctional oxygen electrode based on a perovskite/carbon composite for electrochemical devices. J. Electroanal. Chem. 2018, 808, 412–419. [Google Scholar] [CrossRef]
- Rashkova, V.; Kitova, S.; Konstantinov, I.; Vitanov, T. Vacuum evaporated thin films of mixed cobalt and nickel oxides as electrocatalyst for oxygen evolution and reduction. Electrochim. Acta 2002, 47, 1555–1560. [Google Scholar] [CrossRef]
- Indra, A.; Menezes, P.W.; Sahraie, N.R.; Bergmann, A.; Das, C.; Tallarida, M.; Schmeißer, D.; Strasser, P.; Driess, M. Unification of catalytic water oxidation and oxygen reduction reactions: Amorphous beat crystalline cobalt iron oxides. J. Am. Chem. Soc. 2014, 136, 17530–17536. [Google Scholar] [CrossRef]
- Hazarika, K.K.; Goswami, C.; Saikia, H.; Borah, B.J.; Bharali, P. Cubic Mn2O3 nanoparticles on carbon as bifunctional electrocatalyst for oxygen reduction and oxygen evolution reactions. Mol. Catal. 2018, 451, 153–160. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, D.; Li, S.; Zhang, L.; Zheng, G.; Guo, L. Layered copper manganese oxide for the efficient catalytic CO and VOCs oxidation. Chem. Eng. J. 2019, 357, 258–268. [Google Scholar] [CrossRef]
- Yang, S.; Wang, Z.; Cao, Z.; Mao, X.; Shi, M.; Li, Y.; Zhang, R.; Yin, Y. Facile synthesis of well dispersed spinel cobalt manganese oxides microsphere as efficient bi-functional electrocatalysts for oxygen reduction reaction and oxygen evolution reaction. J. Alloys Compd. 2017, 721, 482–491. [Google Scholar] [CrossRef]
- Gu, Y.; Yan, G.; Lian, Y.; Qi, P.; Mu, Q.; Zhang, C.; Deng, Z.; Peng, Y. MnIII-enriched α-MnO2 nanowires as efficient bifunctional oxygen catalysts for rechargeable Zn-air batteries. Energy Storage Mater. 2019, 23, 252–260. [Google Scholar] [CrossRef]
- Chatenet, M.; Pollet, B.G.; Dekel, D.R.; Dionigi, F.; Deseure, J.; Millet, P.; Braatz, R.D.; Bazant, M.Z.; Eikerling, M.; Staffell, I.; et al. Water electrolysis: From textbook knowledge to the latest scientific strategies and industrial developments. Chem. Soc. Rev. 2022, 51, 4583–4762. [Google Scholar] [CrossRef]
- Jiang, J.; Li, Y.; Liu, J.; Huang, X.; Yuan, C.; Lou, X.W. Recent advances in metal oxide-based electrode architecture design for electrochemical energy storage. Adv. Mater. 2012, 24, 5166–5180. [Google Scholar] [CrossRef]
- Fu, G.; Yan, X.; Chen, Y.; Xu, L.; Sun, D.; Lee, J.M.; Tang, Y. Boosting Bifunctional Oxygen Electrocatalysis with 3D Graphene Aerogel-Supported Ni/MnO Particles. Adv. Mater. 2018, 30, 1704609. [Google Scholar] [CrossRef]
- Xu, N.; Zhang, Y.; Wang, M.; Fan, X.; Zhang, T.; Peng, L.; Zhou, X.D.; Qiao, J. High-performing rechargeable/flexible zinc-air batteries by coordinated hierarchical Bi-metallic electrocatalyst and heterostructure anion exchange membrane. Nano Energy 2019, 65, 104021. [Google Scholar] [CrossRef]
- Xiang, W.; Zhao, Y.; Jiang, Z.; Li, X.; Zhang, H.; Sun, Y.; Ning, Z.; Du, F.; Gao, P.; Qian, J.; et al. Palladium single atoms supported by interwoven carbon nanotube and manganese oxide nanowire networks for enhanced electrocatalysis. J. Mater. Chem. A 2018, 6, 23366–23377. [Google Scholar] [CrossRef]
- Poux, T.; Napolskiy, F.S.; Dintzer, T.; Kéranguéven, G.; Istomin, S.Y.; Tsirlina, G.A.; Antipov, E.V.; Savinova, E.R. Dual role of carbon in the catalytic layers of perovskite/carbon composites for the electrocatalytic oxygen reduction reaction. Catal. Today 2012, 189, 83–92. [Google Scholar] [CrossRef]
- Villanueva-Martínez, N.I.; Alegre, C.; Martínez-Visús, I.; Lázaro, M.J. Bifunctional oxygen electrocatalysts based on non-critical raw materials: Carbon nanostructures and iron-doped manganese oxide nanowires. Catal. Today 2023, 420, 114083. [Google Scholar] [CrossRef]
- He, M.; Jin, X.; Chen, F.; Chen, J.; Min, J.; Duan, H.; Kuang, X.; Li, J.; Wu, Z.; Li, J. Synergistic effect of Mn3+ and oxygen vacancy on the bifunctional oxygen electrocatalytic performance of MnOX/CNTs composites. J. Alloys Compd. 2023, 933, 167728. [Google Scholar] [CrossRef]
- Niu, Y.; Teng, X.; Gong, S.; Liu, X.; Xu, M.; Chen, Z. Boosting oxygen electrocatalysis for flexible zinc-air batteries by interfacing iron group metals and manganese oxide in porous carbon nanowires. Energy Storage Mater. 2021, 43, 42–52. [Google Scholar] [CrossRef]
- Cuong, T.V.; Pham, V.H.; Tran, Q.T.; Chung, J.S.; Shin, E.W.; Kim, J.S.; Kim, E.J. Optoelectronic properties of graphene thin films prepared by thermal reduction of graphene oxide. Mater. Lett. 2010, 64, 765–767. [Google Scholar] [CrossRef]
- Liu, T.H.; Gajewski, G.; Pao, C.W.; Chang, C.C. Structure, energy, and structural transformations of graphene grain boundaries from atomistic simulations. Carbon N. Y. 2011, 49, 2306–2317. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Veerapandian, M.; Yun, K.; Kim, S.J. The chemical and structural analysis of graphene oxide with different degrees of oxidation. Carbon N. Y. 2013, 53, 38–49. [Google Scholar] [CrossRef]
- Stobinski, L.; Lesiak, B.; Malolepszy, A.; Mazurkiewicz, M.; Mierzwa, B.; Zemek, J.; Jiricek, P.; Bieloshapka, I. Graphene oxide and reduced graphene oxide studied by the XRD, TEM and electron spectroscopy methods. J. Electron Spectros. Relat. Phenomena 2014, 195, 145–154. [Google Scholar] [CrossRef]
- Paulchamy, B.; Arthi, G.; Lignesh, B.D. A Simple Approach to Stepwise Synthesis of Graphene Oxide Nanomaterial. J. Nanomed. Nanotechnol. 2015, 6, 1000253. [Google Scholar] [CrossRef]
- Shen, J.; Hu, Y.; Shi, M.; Lu, X.; Qin, C.; Li, C.; Ye, M. Fast and facile preparation of graphene oxide and reduced graphene oxide nanoplatelets. Chem. Mater. 2009, 21, 3514–3520. [Google Scholar] [CrossRef]
- Saleem, H.; Haneef, M.; Abbasi, H.Y. Synthesis route of reduced graphene oxide via thermal reduction of chemically exfoliated graphene oxide. Mater. Chem. Phys. 2018, 204, 1–7. [Google Scholar] [CrossRef]
- Tientong, J.; Garcia, S.; Thurber, C.R.; Golden, T.D. Synthesis of nickel and nickel hydroxide nanopowders by simplified chemical reduction. J. Nanotechnol. 2014, 2014, 193162. [Google Scholar] [CrossRef]
- Hosseynizadeh Khezri, S.; Yazdani, A.; Khordad, R. Pure iron nanoparticles prepared by electric arc discharge method in ethylene glycol. EPJ Appl. Phys. 2012, 59, 30401. [Google Scholar] [CrossRef]
- Lu, X.; Qin, Z.; Zhang, Y.; Wang, X.; Li, F.; Ding, B.; Hu, Z. Study of the paramagnetic-antiferromagnetic transition and the γ → ε martensitic transformation in Fe-Mn alloys. J. Mater. Sci. 2000, 35, 5597–5603. [Google Scholar] [CrossRef]
- Mathur, A.; Halder, A. One-step synthesis of bifunctional iron-doped manganese oxide nanorods for rechargeable zinc-air batteries. Catal. Sci. Technol. 2019, 9, 1245–1254. [Google Scholar] [CrossRef]
- Shen, Y.; Zhu, Y.; Sunarso, J.; Guan, D.; Liu, B.; Liu, H.; Zhou, W.; Shao, Z. New Phosphorus-Doped Perovskite Oxide as an Oxygen Reduction Reaction Electrocatalyst in an Alkaline Solution. Chem. A Eur. J. 2018, 24, 6950–6957. [Google Scholar] [CrossRef]
- Briggs, D. Handbook of X-ray Photoelectron Spectroscopy C. D. Wanger, W.M. Riggs, L.E. Davis, J.F. Moulder and G. E.Muilenberg Perkin-Elmer Corp., Physical Electronics Division, Eden Prairie, Minnesota, USA, 1979. 190 pp. $195. Surf. Interface Anal. 1981, 3, v. [Google Scholar] [CrossRef]
- Santos, V.P.; Pereira, M.F.R.; Órfão, J.J.M.; Figueiredo, J.L. The role of lattice oxygen on the activity of manganese oxides towards the oxidation of volatile organic compounds. Appl. Catal. B Environ. 2010, 99, 353–363. [Google Scholar] [CrossRef]
- Kabalan, I.; Lebeau, B.; Fadlallah, M.B.; Toufaily, J.; Hamieh, T.; Bellat, J.P.; Daou, T.J. Hierarchical faujasite-type zeolite for molecular decontamination. J. Nanosci. Nanotechnol. 2016, 16, 9318–9322. [Google Scholar] [CrossRef]
- Waki, K.; Wong, R.A.; Oktaviano, H.S.; Fujio, T.; Nagai, T.; Kimoto, K.; Yamada, K. Non-nitrogen doped and non-metal oxygen reduction electrocatalysts based on carbon nanotubes: Mechanism and origin of ORR activity. Energy Environ. Sci. 2014, 7, 1950–1958. [Google Scholar] [CrossRef]
- Ruiz-Cornejo, J.C.; Vivo-Vilches, J.F.; Sebastián, D.; Martínez-Huerta, M.V.; Lázaro, M.J. Carbon nanofiber-supported tantalum oxides as durable catalyst for the oxygen evolution reaction in alkaline media. Renew. Energy 2021, 178, 307–317. [Google Scholar] [CrossRef]
- Ruiz-Cornejo, J.C.; Sebastián, D.; Pardo, J.I.; Martínez-Huerta, M.V.; Lázaro, M.J. Sulfur-doped carbon nanofibers as support for tantalum oxides bifunctional catalysts for the oxygen reduction and evolution reactions. J. Power Sources 2022, 546, 231988. [Google Scholar] [CrossRef]
- Chen, X.; Gao, P.; Liu, H.; Xu, J.; Zhang, B.; Zhang, Y.; Tang, Y.; Xiao, C. In situ growth of iron-nickel nitrides on carbon nanotubes with enhanced stability and activity for oxygen evolution reaction. Electrochim. Acta 2018, 267, 8–14. [Google Scholar] [CrossRef]
- Ahmed, M.S.; Begum, H.; Kim, Y.B. Iron nanoparticles implanted metal-organic-frameworks based Fe–N–C catalysts for high-performance oxygen reduction reaction. J. Power Sources 2020, 451, 227733. [Google Scholar] [CrossRef]
- Stoerzinger, K.A.; Risch, M.; Han, B.; Shao-Horn, Y. Recent Insights into Manganese Oxides in Catalyzing Oxygen Reduction Kinetics. ACS Catal. 2015, 5, 6021–6031. [Google Scholar] [CrossRef]
- Ramaswamy, N.; Mukerjee, S. Influence of inner- and outer-sphere electron transfer mechanisms during electrocatalysis of oxygen reduction in alkaline media. J. Phys. Chem. C 2011, 115, 18015–18026. [Google Scholar] [CrossRef]
- Poux, T.; Bonnefont, A.; Ryabova, A.; Kéranguéven, G.; Tsirlina, G.A.; Savinova, E.R. Electrocatalysis of hydrogen peroxide reactions on perovskite oxides: Experiment versus kinetic modeling. Phys. Chem. Chem. Phys. 2014, 16, 13595–13600. [Google Scholar] [CrossRef]
- Guan, D.; Shi, C.; Xu, H.; Gu, Y.; Zhong, J.; Sha, Y.; Hu, Z.; Ni, M.; Shao, Z. Simultaneously mastering operando strain and reconstruction effects via phase-segregation strategy for enhanced oxygen-evolving electrocatalysis. J. Energy Chem. 2023, 82, 572–580. [Google Scholar] [CrossRef]
- Shinagawa, T.; Garcia-Esparza, A.T.; Takanabe, K. Insight on Tafel slopes from a microkinetic analysis of aqueous electrocatalysis for energy conversion. Sci. Rep. 2015, 5, 13801. [Google Scholar] [CrossRef] [PubMed]
- Roche, I.; Chaînet, E.; Chatenet, M.; Vondrák, J. Carbon-supported manganese oxide nanoparticles as electrocatalysts for the Oxygen Reduction Reaction (ORR) in alkaline medium: Physical characterizations and ORR mechanism. J. Phys. Chem. C 2007, 111, 1434–1443. [Google Scholar] [CrossRef]
- Park, S.; Jiang, T.; Zheng, K.; Lin, Z.; Meng, Y.; Wang, M.; Chen, J.; Chen, W. Ultrafast Thermal Synthesis of Non-Noble Metal-Based Electrocatalysts for Overall Water Splitting. ACS Appl. Energy Mater. 2023, 6, 5787–5796. [Google Scholar] [CrossRef]
- Kapałka, A.; Fóti, G.; Comninellis, C. Determination of the Tafel slope for oxygen evolution on boron-doped diamond electrodes. Electrochem. Commun. 2008, 10, 607–610. [Google Scholar] [CrossRef]
- Sebastián, D.; Alegre, C.; Gálvez, M.E.; Moliner, R.; Lázaro, M.J.; Aricò, A.S.; Baglio, V. Towards new generation fuel cell electrocatalysts based on xerogel-nanofiber carbon composites. J. Mater. Chem. A 2014, 2, 13713–13722. [Google Scholar] [CrossRef]
- Xiao, X.; Zhang, W.; Zhao, H.; Li, L.; Deng, P.; Wu, Y.; Luo, S.; Chen, B. Ultrathin amorphous MnO2 modified prawn shells-derived porous carbon towards robust oxygen electrocatalyst for rechargeable Zn-air battery. Ceram. Int. 2022, 48, 6506–6511. [Google Scholar] [CrossRef]
- Meng, Y.; Song, W.; Huang, H.; Ren, Z.; Chen, S.Y.; Suib, S.L. Structure-property relationship of bifunctional MnO2 nanostructures: Highly efficient, ultra-stable electrochemical water oxidation and oxygen reduction reaction catalysts identified in alkaline media. J. Am. Chem. Soc. 2014, 136, 11452–11464. [Google Scholar] [CrossRef]
- Wahab, A.; Iqbal, N.; Noor, T.; Ashraf, S.; Raza, M.A.; Ahmad, A.; Khan, U.A. Thermally reduced mesoporous manganese MOF @reduced graphene oxide nanocomposite as bifunctional electrocatalyst for oxygen reduction and evolution. RSC Adv. 2020, 10, 27728–27742. [Google Scholar] [CrossRef]
- Masa, J.; Xia, W.; Sinev, I.; Zhao, A.; Sun, Z.; Grützke, S.; Weide, P.; Muhler, M.; Schuhmann, W. MnxOy/NC and CoxOy/NC nanoparticles embedded in a nitrogen-doped carbon matrix for high-performance bifunctional oxygen electrodes. Angew. Chem. Int. Ed. 2014, 53, 8508–8512. [Google Scholar] [CrossRef]
- Gao, Y.; Zhao, H.; Chen, D.; Chen, C.; Ciucci, F. In situ synthesis of mesoporous manganese oxide/sulfur-doped graphitized carbon as a bifunctional catalyst for oxygen evolution/reduction reactions. Carbon N. Y. 2015, 94, 1028–1036. [Google Scholar] [CrossRef]
- Gebreslase, G.A.; Sebastián, D.; Martínez-Huerta, M.V.; Lázaro, M.J. Nitrogen-doped carbon decorated-Ni3Fe@Fe3O4 electrocatalyst with enhanced oxygen evolution reaction performance. J. Electroanal. Chem. 2022, 925, 116887. [Google Scholar] [CrossRef]
- Sebastián, D.; Suelves, I.; Moliner, R.; Lázaro, M.J. The effect of the functionalization of carbon nanofibers on their electronic conductivity. Carbon N. Y. 2010, 48, 4421–4431. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved synthesis of graphene oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef] [PubMed]

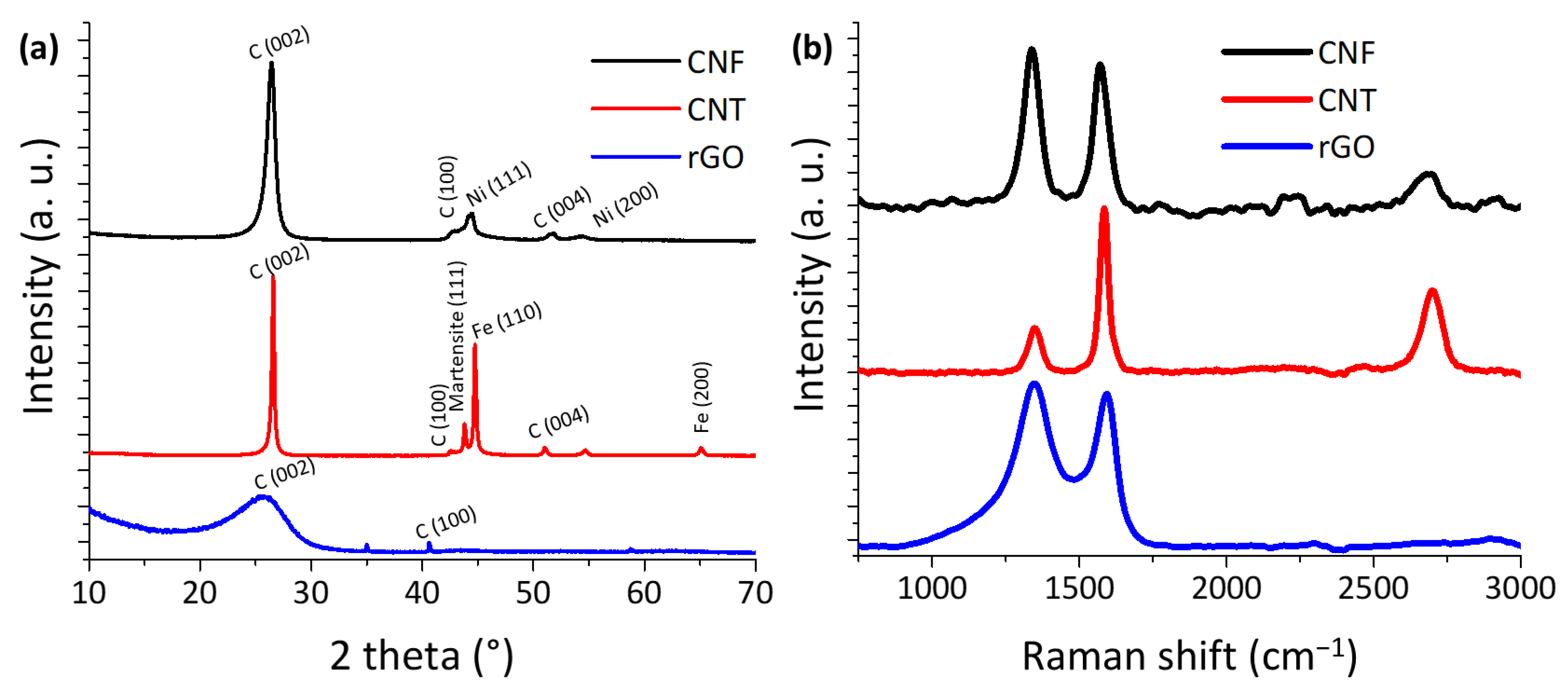
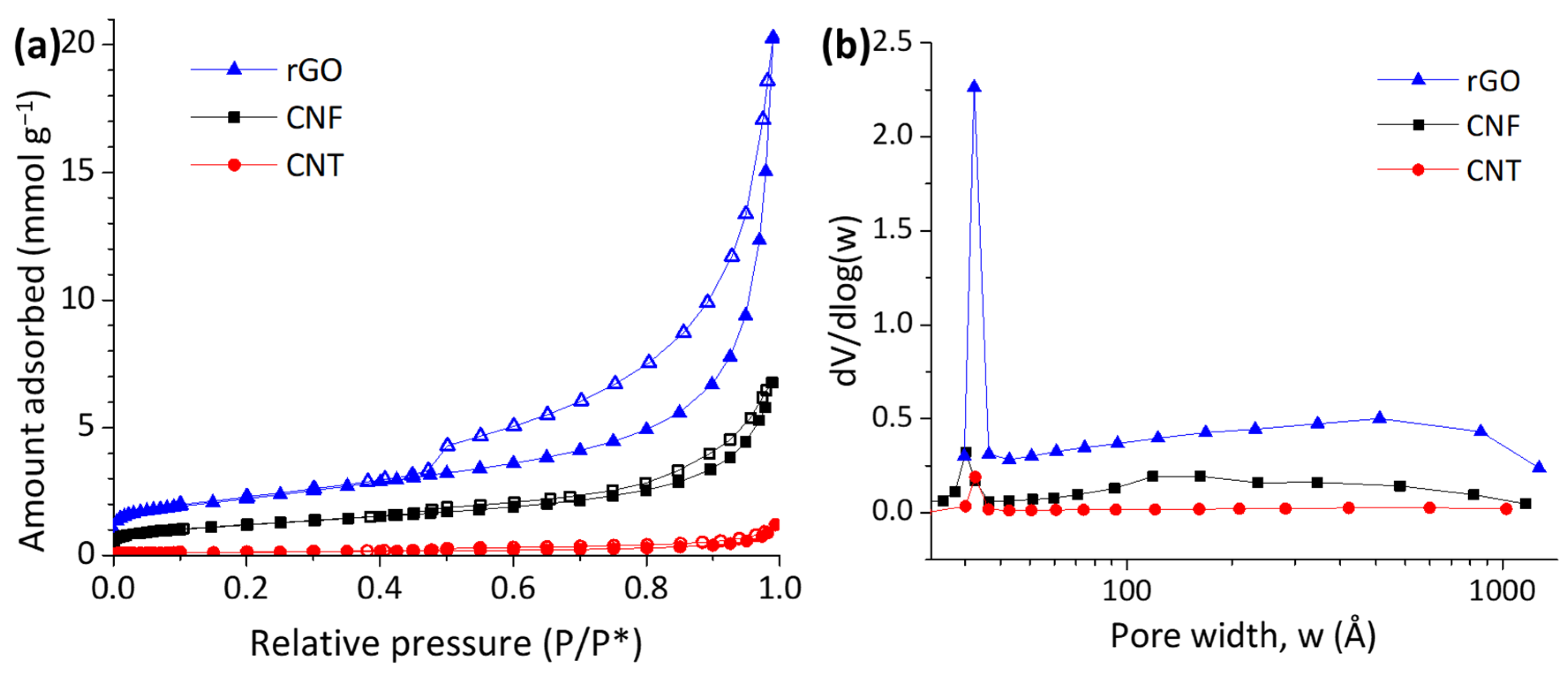

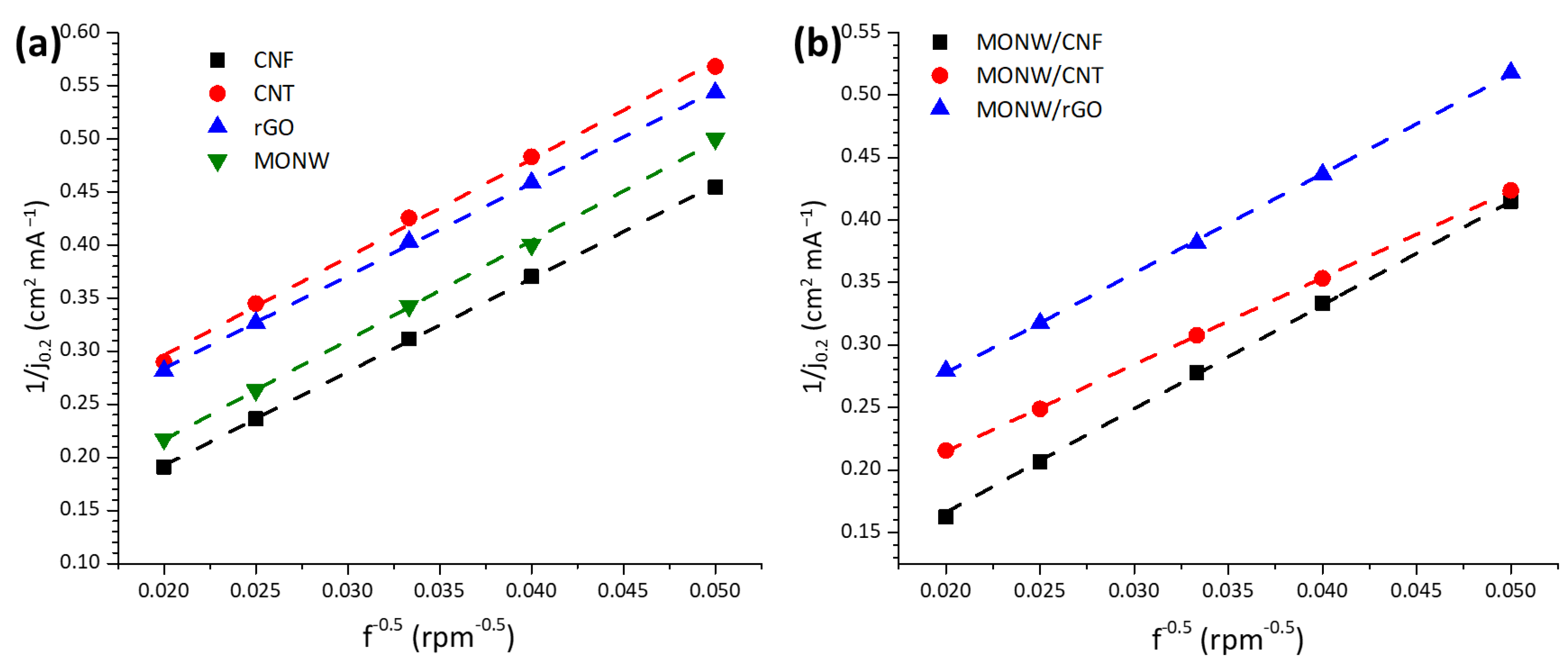

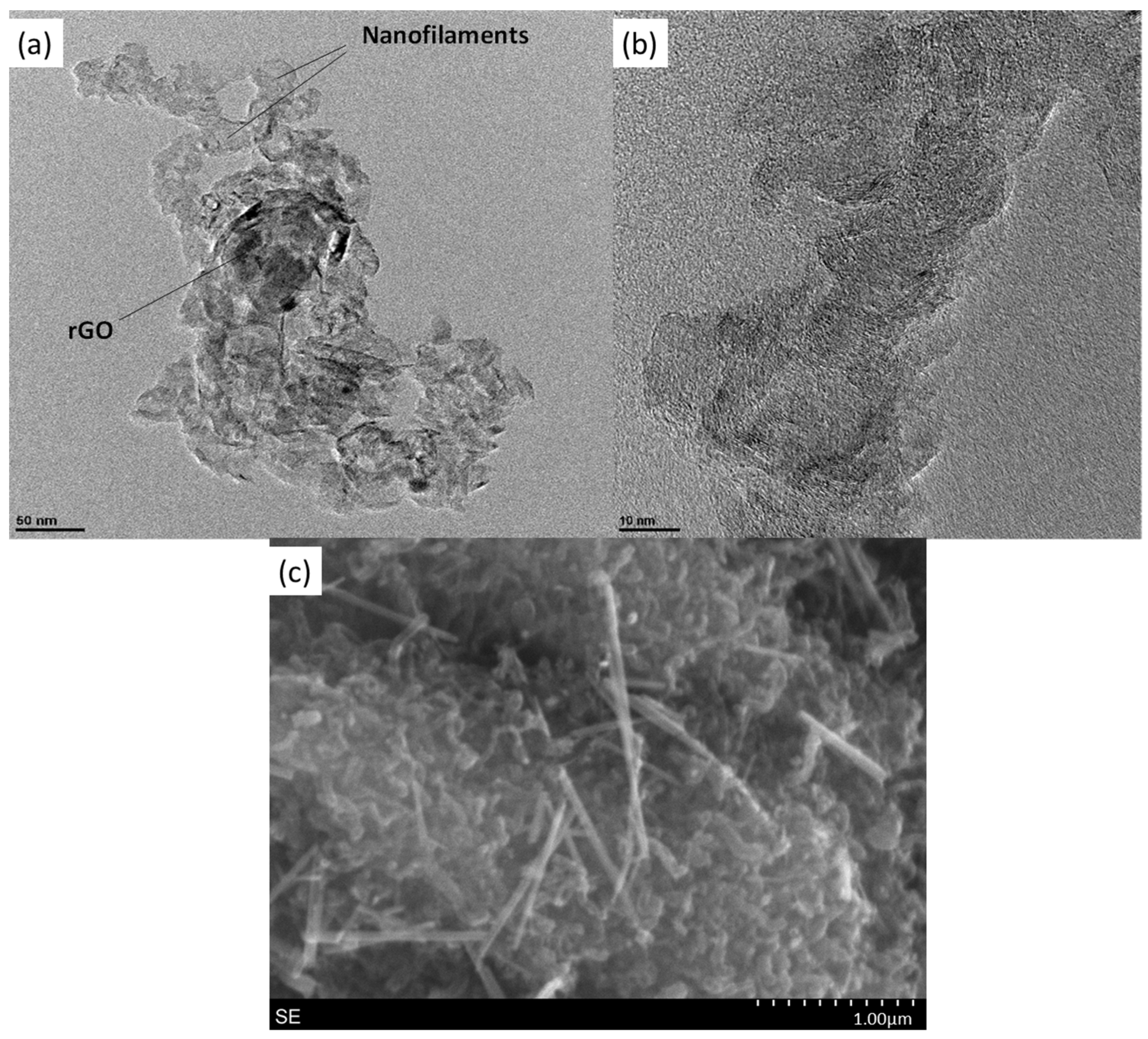
| Chemical Composition (wt%) | Textural Properties | |||||
|---|---|---|---|---|---|---|
| Material | Carbon | Oxygen | Iron * | Nickel * | SBET (m2 g−1) | Vpore (cm3 g−1) |
| CNF | 92.5 | - | - | 7.3 | 95 | 0.24 |
| CNT | 76.5 | - | 23.5 | - | 11 | 0.04 |
| rGO | 92.9 | 5.6 ** | - | - | 175 | 0.70 |
| Catalyst | Oxygen Reduction Reaction | Oxygen Evolution Reaction | Reversibility ORR/OER | ||||
|---|---|---|---|---|---|---|---|
| EORR,onset (V vs. RHE) | jd (mA cm−2) | E1/2 (V vs. RHE) | n | EOER,onset (V vs. RHE) | E10 (V vs. RHE) | ΔE * (V) | |
| CNF | 0.78 | −4.23 | 0.66 | 2.9 | 1.58 | 1.74 | 1.08 |
| CNT | 0.68 | −2.90 | 0.48 | 2.9 | 1.64 | 1.82 | 1.34 |
| rGO | 0.76 | −3.06 | 0.57 | 3.0 | 1.69 | 1.92 | 1.35 |
| MONW | 0.84 | −3.89 | 0.66 | 2.7 | 1.62 | 1.90 | 1.24 |
| MONW/CNF | 0.85 | −4.84 | 0.69 | 3.4 | 1.59 | 1.75 | 1.06 |
| MONW/CNT | 0.82 | −4.02 | 0.57 | 3.8 | 1.63 | 1.83 | 1.25 |
| MONW/rGO | 0.83 | −3.15 | 0.64 | 3.4 | 1.51 | 1.65 | 1.01 |
| Catalyst | Oxygen Reduction Reaction | Oxygen Evolution Reaction | ||
|---|---|---|---|---|
| j0 (mA cm−2) | Tafel Slope (mV dec−1) | j0 (mA cm−2) | Tafel Slope (mV dec−1) | |
| CNF | 1.8 × 10−10 | 76 | 1.7 × 10−4 | 87 |
| CNT | 6.8 × 10−11 | 88 | 8.5 × 10−6 | 38 |
| rGO | 3.5 × 10−10 | 86 | 4.3 × 10−4 | 131 |
| MONW | 4.9 × 10−10 | 136 | 1.0 × 10−5 | 73 |
| MONW/CNF | 9.1 × 10−9 | 53 | 1.4 × 10−6 | 60 |
| MONW/CNT | 2.0 × 10−8 | 101 | 3.3 × 10−6 | 70 |
| MONW/rGO | 4.8 × 10−8 | 109 | 1.0 × 10−5 | 57 |
| Catalyst | Oxygen Reduction Reaction | Oxygen Evolution Reaction | Reversibility ORR/OER | Ref. | ||||
|---|---|---|---|---|---|---|---|---|
| EORR, onset † (V vs. RHE) | jd @ 1600 rpm (mA cm−2) | E1/2 (V vs. RHE) | n | EOER, onset † (V vs. RHE) | E10 (V vs. RHE) | ΔE (V) | ||
| rGO-CNF | 0.79 | −3.80 | 0.69 | 2.9 | 1.58 | 1.76 | 1.07 | This work |
| MONW/rGO-CNF | 0.90 | −5.26 | 0.74 | 3.7 | 1.54 | 1.73 | 0.99 | This work |
| Pt/C–IrO2 | 1.02 | −5.72 | 0.85 | 4 | 1.53 | 1.75 | - | This work |
| MnOx/CNTs | 0.85 * | −4.90 | 0.77 * | 3.6 | 1.58 * | 0.91 * | 0.91 * | [25] |
| MnO2-C(ultrathin amorphous) | 0.97 | −5.81 | 0.81 | 4 | 1.45 | 1.59 | 0.78 | [56] |
| α-MnO2(cryptomelane)/Vulcan | 0.88 * | −4.1 | 0.79 * | 4 | 1.58 * | 1.73 * | 0.94 * | [57] |
| Mn-MOF @rGO | 1.09 * | −2.03 | 0.98 * | - | 1.75 * | 1.84 * | 0.86 * | [58] |
| MnxOy/N-carbon | 0.85 | −5.0 | 0.77 | - | 1.61 | 1.68 | 0.91 | [59] |
| MnOx/S-carbon | 0.94 | −4.5 | 0.81 | 3.7 | 1.33 * | 1.62 * | 0.81 * | [60] |
| Catalyst | Oxygen Reduction Reaction | Oxygen Evolution Reaction | ||
|---|---|---|---|---|
| j0 (mA cm−2) | Tafel Slope (mV dec−1) | j0 (mA cm−2) | Tafel Slope (mV dec−1) | |
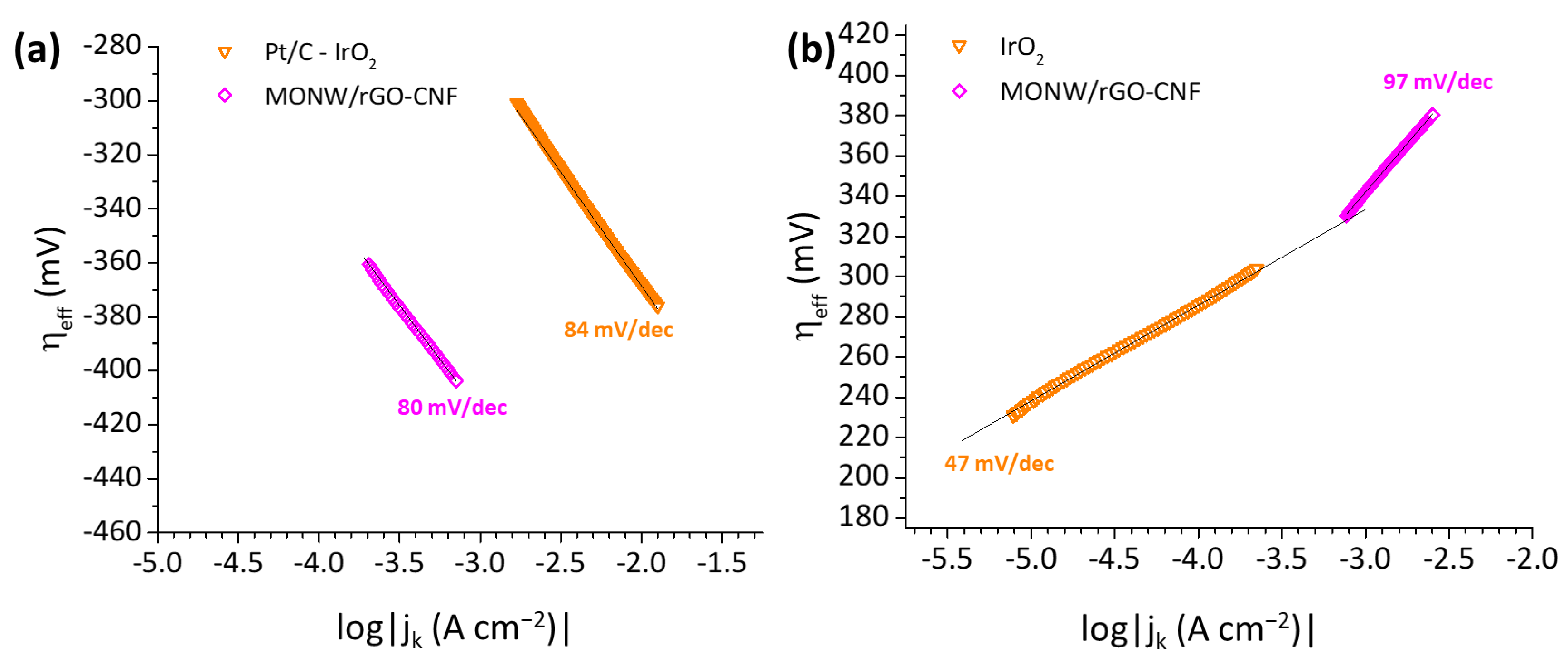
| rGO-CNF | 2.6 × 10−12 | 42 | 4.6 × 10−4 | 98 |
| MONW/rGO-CNF | 6.3 × 10−6 | 80 | 2.9 × 10−6 | 97 |
| Pt/C–IrO2 | 4.2 × 10−4 | 84 | 1.0 × 10−7 | 47 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villanueva-Martínez, N.I.; Alegre, C.; Sebastián, D.; Orozco, N.; Lázaro, M.J. On the Effect of the Nature of Carbon Nanostructures on the Activity of Bifunctional Catalysts Based on Manganese Oxide Nanowires. Catalysts 2023, 13, 1240. https://doi.org/10.3390/catal13091240
Villanueva-Martínez NI, Alegre C, Sebastián D, Orozco N, Lázaro MJ. On the Effect of the Nature of Carbon Nanostructures on the Activity of Bifunctional Catalysts Based on Manganese Oxide Nanowires. Catalysts. 2023; 13(9):1240. https://doi.org/10.3390/catal13091240
Chicago/Turabian StyleVillanueva-Martínez, Nicolás Ignacio, Cinthia Alegre, David Sebastián, Nataly Orozco, and María Jesús Lázaro. 2023. "On the Effect of the Nature of Carbon Nanostructures on the Activity of Bifunctional Catalysts Based on Manganese Oxide Nanowires" Catalysts 13, no. 9: 1240. https://doi.org/10.3390/catal13091240






