Abstract
This study explores the impact of scandium (Sc) as a promoter on the catalytic performance of 4Ni/MCM-41 catalysts for the partial oxidation of methane (POM). 4Ni+Sc/MCM-41 catalysts were synthesized with varying Sc loadings of 0, 0.2, 0.4, 0.6, and 0.8 wt.%. These catalysts were characterized using several techniques, including N2 physisorption, X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM), Raman spectroscopy, thermogravimetric analysis (TGA), and X-ray photoelectron spectroscopy (XPS). All catalysts exhibited a mesoporous structure characterized by narrow slit-shaped pores. Among them, the 4Ni+0.2Sc/MCM-41 catalyst showed the most consistent pore size distribution. The addition of Sc (scandium) facilitated the formation of strongly interacting nickel species, which enhanced the initial catalytic activity. However, a trade-off was observed between initial activity and long-term stability. The optimal Sc loading was determined to be 0.2 wt.%. This catalyst achieved the highest methane conversion rate of 63.9%, a hydrogen yield of 60%, and an H2/CO ratio of 2.7 while also demonstrating superior stability during extended operation. The 4Ni+0.2Sc/MCM-41 catalyst showed only a 7% weight loss in the thermogravimetric analysis (TGA), which shows that it will stay stable even after being used for a long time. The improved performance of the Sc-promoted catalysts is attributable to the increased availability of active sites, enhanced stability, and better dispersion of nickel. These efforts aim to create more sustainable and efficient methods for hydrogen production, minimizing the negative effects associated with traditional processes. By advancing these technologies, we can further support the transition to a cleaner energy future.
1. Introduction
The rising demand for energy and environmental concerns have driven the search for sustainable energy solutions. Hydrogen is a clean energy source with a higher heating value than gasoline and natural gas. During combustion, hydrogen does not produce carbon dioxide; the only byproduct is water (H2O). This makes hydrogen an environmentally appealing option for clean energy solutions [1,2,3]. Hydrogen is an essential green energy source for developing a sustainable society [4,5]. Hydrogen can be produced from methane, which is increasingly available due to processes like hydraulic fracturing [6,7]. However, thermal reforming, a common production method, has environmental drawbacks. Researchers are exploring innovative ways to reduce the environmental impact of and enhance hydrogen production [8]. Natural gas, which is composed mainly of about 80% methane, is an essential fuel source and plays a crucial role in chemical production. Methane is widely used both as a fuel and as a fundamental feedstock. It is key to producing a variety of chemical products, including hydrogen, various chemicals, liquid fuels, and electricity, through different methane conversion processes [9]. The steam reforming of methane is the leading industrial method for converting natural gas into hydrogen among the various processes that have been thoroughly investigated [10,11]. Additionally, other methods, such as partial oxidation [12,13,14,15], are gaining interest because they require less energy. Equation (1) displays the partial oxidation of a methane (POM) reaction.
POM is an effective method for methane conversion, providing high conversion efficiency, selectivity, and short residence times [16]. The POM directly converts methane into liquid oxygenates [17,18,19]. Thus, the POM route utilizes natural gas to generate syngas, mainly due to its exothermic nature and its ability to produce syngas with the necessary H2/CO ratio of 2, which is crucial for Fischer–Tropsch synthesis. Syngas, which is a mixture of hydrogen and carbon monoxide, is an essential intermediate in the manufacturing of hydrogen, ammonia, and value-added compounds like methanol and liquid fuels generated from Fischer–Tropsch [20,21].
The syngas ratio is often higher than 3 because of the existence of a water gas shift reaction (Equation (2)) [22].
Despite ongoing efforts, issues such as catalyst deactivation from metal sintering, the formation of hotspots, and coke deposition continue to persist in the POM. Noble metals like Pd, Ir, Pt, and Rh provide excellent activity and stability, but they are rare and expensive. The primary processes that form surface carbon are CO disproportionation (termed as the Boudouard reaction) and CH4 cracking.
To combat these challenges, researchers have investigated various catalysts, including transition and noble metals [23]. Despite their exceptional activity and stability, noble metals such as Pd, Ir, Pt, and Rh are costly and scarce [24,25]. Due to their stability and environmental friendliness, transition metals like Ni make good substitutes for this process.
However, nickel-based catalysts face challenges with poor activity, primarily due to coke formation and sintering [26,27]. Hence, the major challenge is to come up with Ni catalysts capable of resisting carbon formation and sintering. Research has demonstrated that interactions between metals and their supports can influence both the activity of catalysts and their longevity [28,29]. The type of support chosen is crucial in the context of POM [30,31]. The catalyst stability is controlled by the equilibrium between the rates of carbon gasification and methane decomposition [32]. In the literature, several supports, including molecular sieves, are considered [33]. The MCM-41 molecular sieve is a long-range ordered mesoporous material with uniform pore size [34]. The mesoporous material supports have uniform and customizable pore dimensions and a high specific surface area. It is currently used in various applications, including catalysis [35]. MCM-41 is a mesoporous silica material characterized by unidirectional channels arranged in a regular hexagonal pattern, with pore sizes ranging from 20 to 100 angstroms. Recent literature indicates various attempts to incorporate metals into the MCM-41 framework. This incorporation is typically carried out through methods such as wet impregnation or co-precipitation [36,37]. MCM-41 allows for the direct incorporation of transition metal ions, such as Ni, into the mesoporous framework by substituting Si atoms in their regular tetrahedral positions. The modified materials have shown remarkable catalytic activity for the partial oxidation of hydrocarbons under mild conditions [38]. The addition of trace amounts of promoters, which may not have any catalytic activity in and of themselves, is one of the most straightforward methods of altering the efficiency of catalysts [39]. Chien et al. investigated nickel/samarium-doped ceria (Ni/SDC) nanocatalysts, which showed optimal POM for syngas production with a moderate Ni–SDC interaction and a Sm/Ce ratio of ~1/9 to 2/8 [40]. Shiratori et al. highlighted the suitability of the Sc element and found that the conductivity of Sc scandia-stabilized zirconia (ScSZ) is approximately three times higher than that of yttria-stabilized zirconia (YSZ) [41]. Rajput et al. used Ni on scandia-ceria-zirconia supports to enhance catalytic efficiency and found that the stability of active sites, the stability of oxide vacancies, and the type of carbon deposits were significantly modified [42]. Arifin et al. investigated the characteristics of scandia-stabilized zirconia as a solid oxide fuel cell material. They found that scandia-stabilized zirconia possesses 1.5 to 3 times the conductivity of yttria-stabilized zirconia (YSZ) due to differences in their crystal structures. YSZ is stable up to 1200 °C, but doping with scandium oxide (Sc2O3) enhances its thermal stability to 1400 °C [43]. It is expected that the Sc promoter aids the active site, stabilizing the catalyst under high temperatures and enhancing oxygen mobility during the oxidation reaction in the POM process. In this study, scandium was introduced as a promoter for the first time in nickel catalysts supported on MCM-41 for the partial oxidation process. We will investigate how varying amounts of scandium affect hydrogen production. The results will be compared with characterization data obtained before and after the reaction using BET, XRD, TPR, TEM, TPD, and TGA techniques.
2. Results and Discussion
2.1. N2 Physisorption
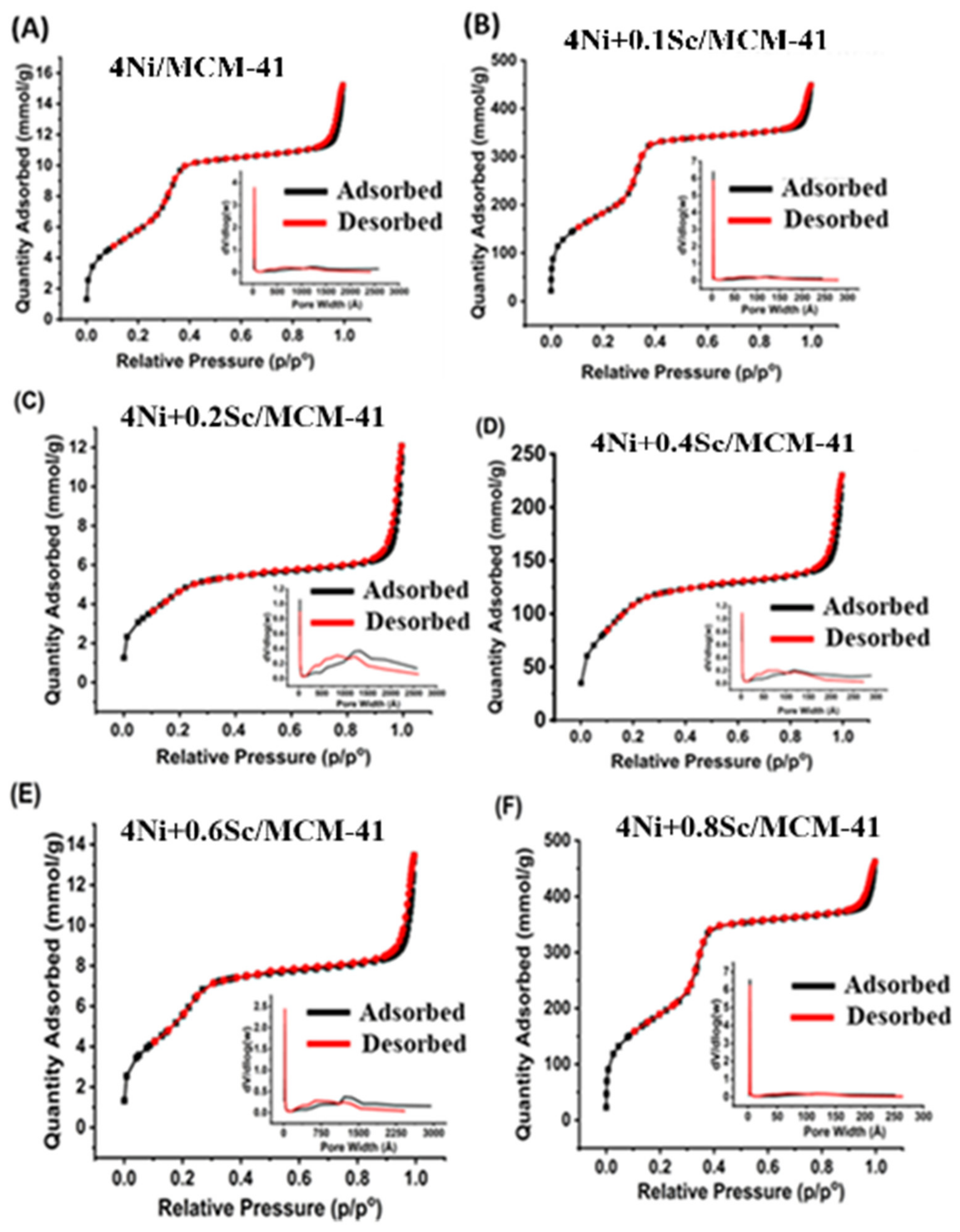
N2 physisorption was performed to determine surface area, pore volume, and pore size. As shown in Figure 1, the catalysts showed type IV isotherms, and capillary condensation occurred in the high P/P0 region [44]. Furthermore, the catalysts exhibited an H4 Hysteresis Loop associated with materials that have narrow slit-shaped pores [45]. The embedded distribution curves indicated a reasonably consistent pore size distribution in the mesopore region, with relatively narrow peaks around 25–100 Å. The 4Ni+0.2Sc/MCM-41 catalyst’s curve C showed the narrowest distribution, indicating a consistent distribution of pore sizes.
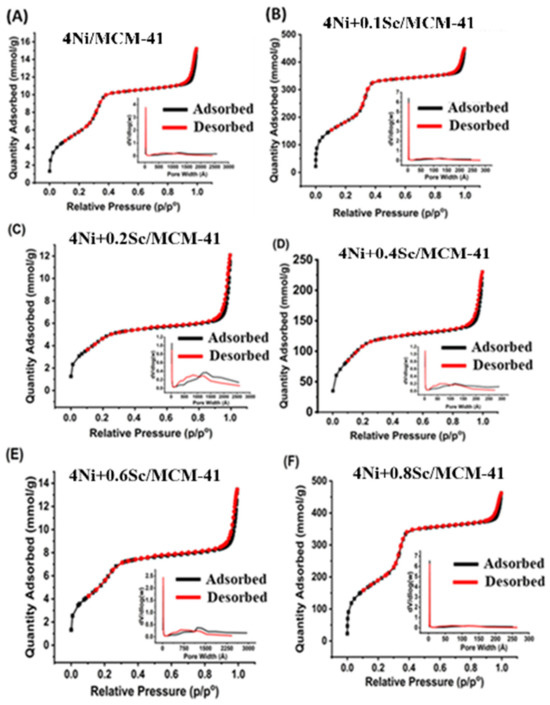
Figure 1.
The N2 adsorption–desorption isotherms and pore size distribution of reduced samples of (A) 4Ni/MCM-41, (B) 4Ni+0.1Sc/MCM-41, (C) 4Ni+0.2Sc/MCM-41, (D) 4Ni+0.4Sc/MCM-41, (E) 4Ni+0.6Sc/MCM-41, and (F) 4Ni+0.8Sc/MCM-41.
2.2. TPR Analysis
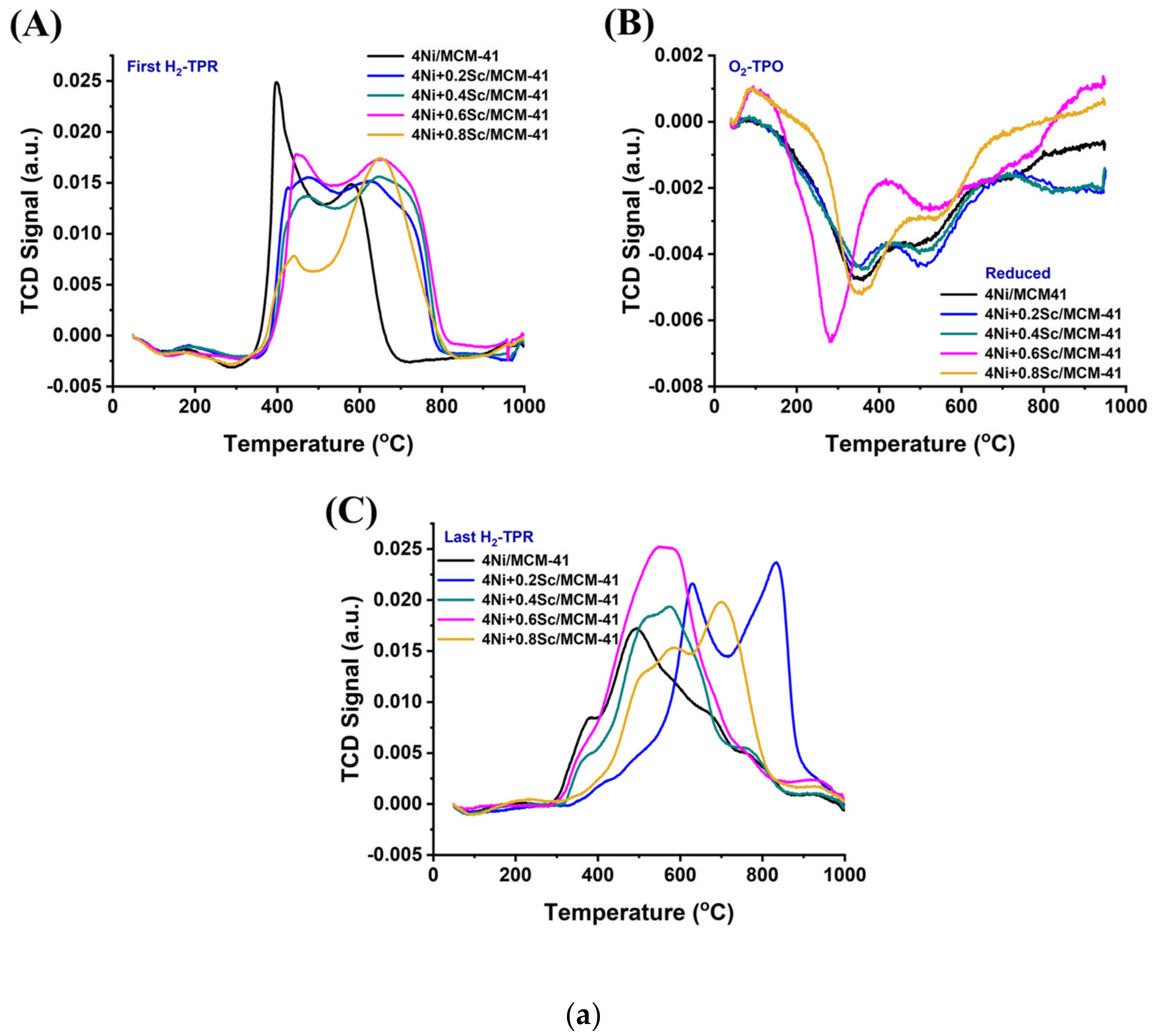
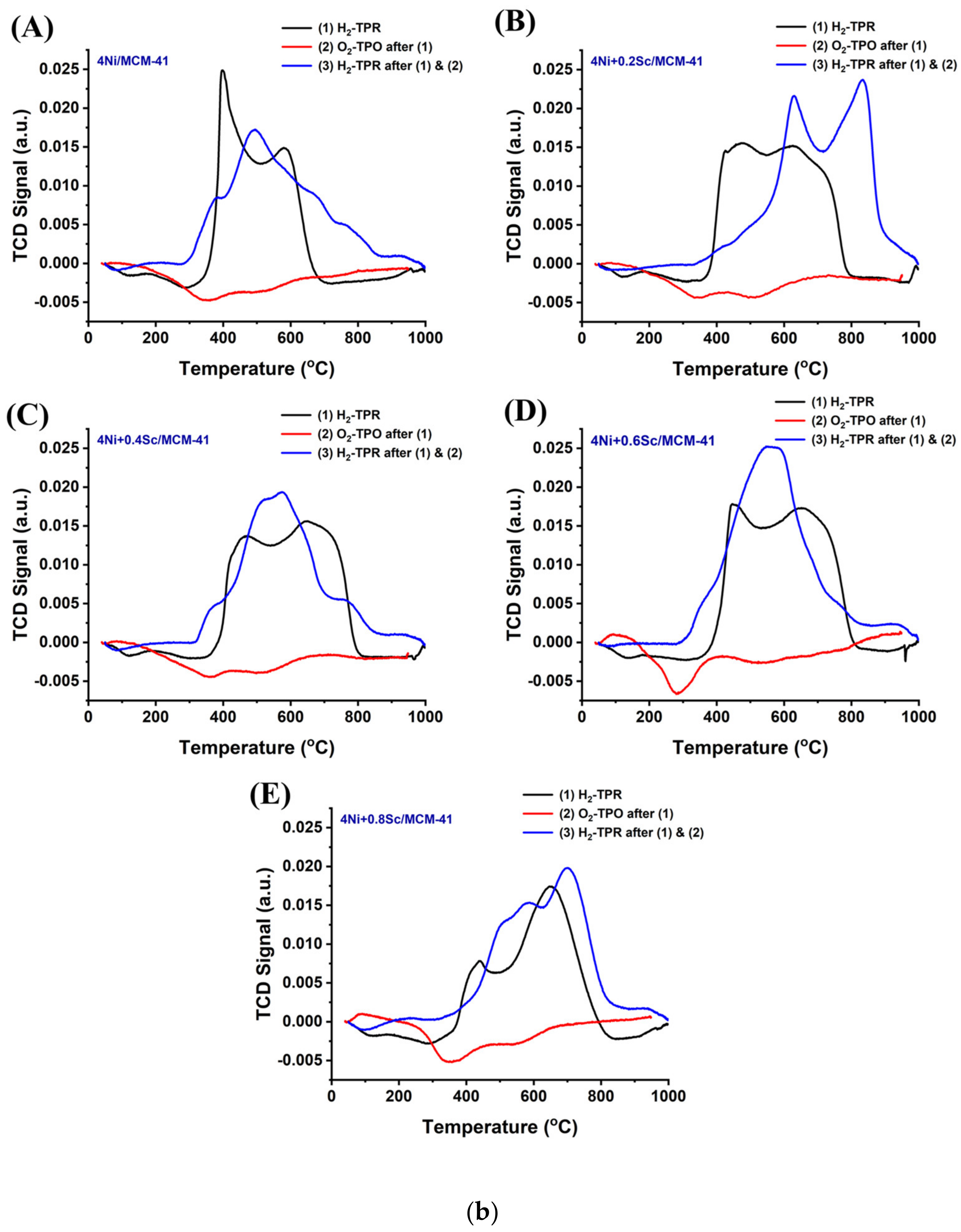
The profiles of the Ni-based supported MCM-14 catalysts after reduction are shown in Figure 2(aA). The presence of several metal oxide species with varying irreducibilities is indicated by the multiple reduction peaks observed in all the samples. As the Sc content increased to 0.6 wt.%, the peaks shifted to higher temperatures, and the low-temperature reduction peak became less pronounced. This indicates that upon the addition of Sr, the concentration of NiO, which was in a relatively weaker interaction with support was decreased, and the metal-supported interaction was raised. When the 4Ni/MCM-41 catalyst was modified with 0.6 wt.% Sc, it consumed the highest amount of H2 during H2-TPR (Table S1). This indicates the presence of the highest concentration of reducible species of interacted NiO species over the 4Ni+0.6Sr/MCM-41 catalyst. However, further Sr loading results saw a fall in H2 consumption, indicating the lowest concentration of active sites over 4Ni+0.8Sr/MCM-41 compared to the rest of the catalysts. During the POM reaction, the risk of oxidation of active site “Ni” by O2 was quite high, so the concentration of the actual active sites during the POM may vary. When O2-TPO was carried out in a sequence of H2-TPR, the metallic Ni was again oxidized. The extent of oxidation of metallic Ni can be quantified by carrying out sequential H2-TPR (mentioned as the last H2-TPR). This cyclic H2TPR-O2TPO-H2TPR experiment presents the exact amount of active site during the POM reaction. Table S1 indicates that in the last H2TPR, the hydrogen consumption by the catalyst was mostly increased (except the 4Ni+0.4Sr/MCM-41 catalyst). This shows that the catalyst’s consecutive exposure to O2 and H2 causes a higher number of active sites to increase significantly over the 4Ni+xSc/- (x = 0.2, 0.4, 0.6, and 0.8 wt.%) catalyst. Remarkably, 4Ni+0.2Sc/MCM-41 also used the most hydrogen in the most recent H2-TPR, and it similarly exhibited the greatest reduction profile change towards higher temperatures. That means that 4Ni+0.2Sc/MCM-41 carries the maximum number of active sites under a strong interaction with support. Thus, it can be concluded that the active site concentration was maximum over 4Ni+0.6Sc/MCM-41 initially, but during the POM (in the presence of H2 and O2), the maximum amount of stable active sites was generated over 4Ni+0.2Sc/MCM-41.
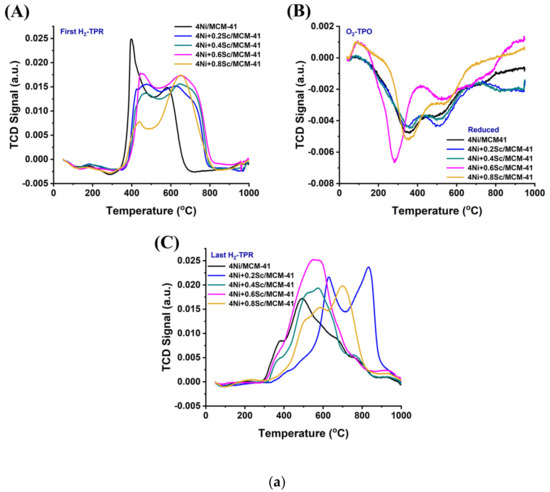
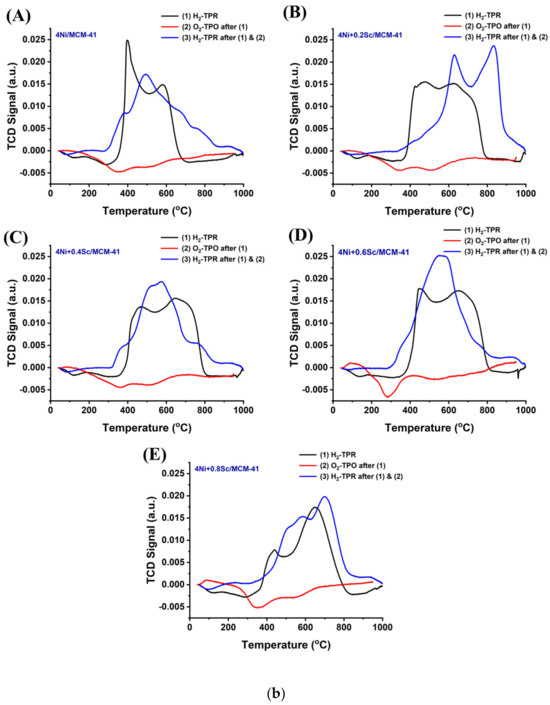
Figure 2.
(a) (A) First H2-TPR of the catalysts; (B) O2-TPO of the catalysts; (C) last H2-TPR of the catalysts. (b) (A–E) H2-TPR-O2TPO-H2TPR cyclic experiment over the 4NixSc/MCM-41 (x = 0.1, 0.2, 0.4, 0.6, and 0.8 wt.%) catalysts: (A) 4Ni/MCM-41, (B) 4Ni+0.2Sc/MCM-41, (C) 4Ni+0.4Sc/MCM-41, (D) 4Ni+0.6Sc/MCM-41, and (E) 4Ni+0.8Sc/MCM-41.
2.3. XRD Profile
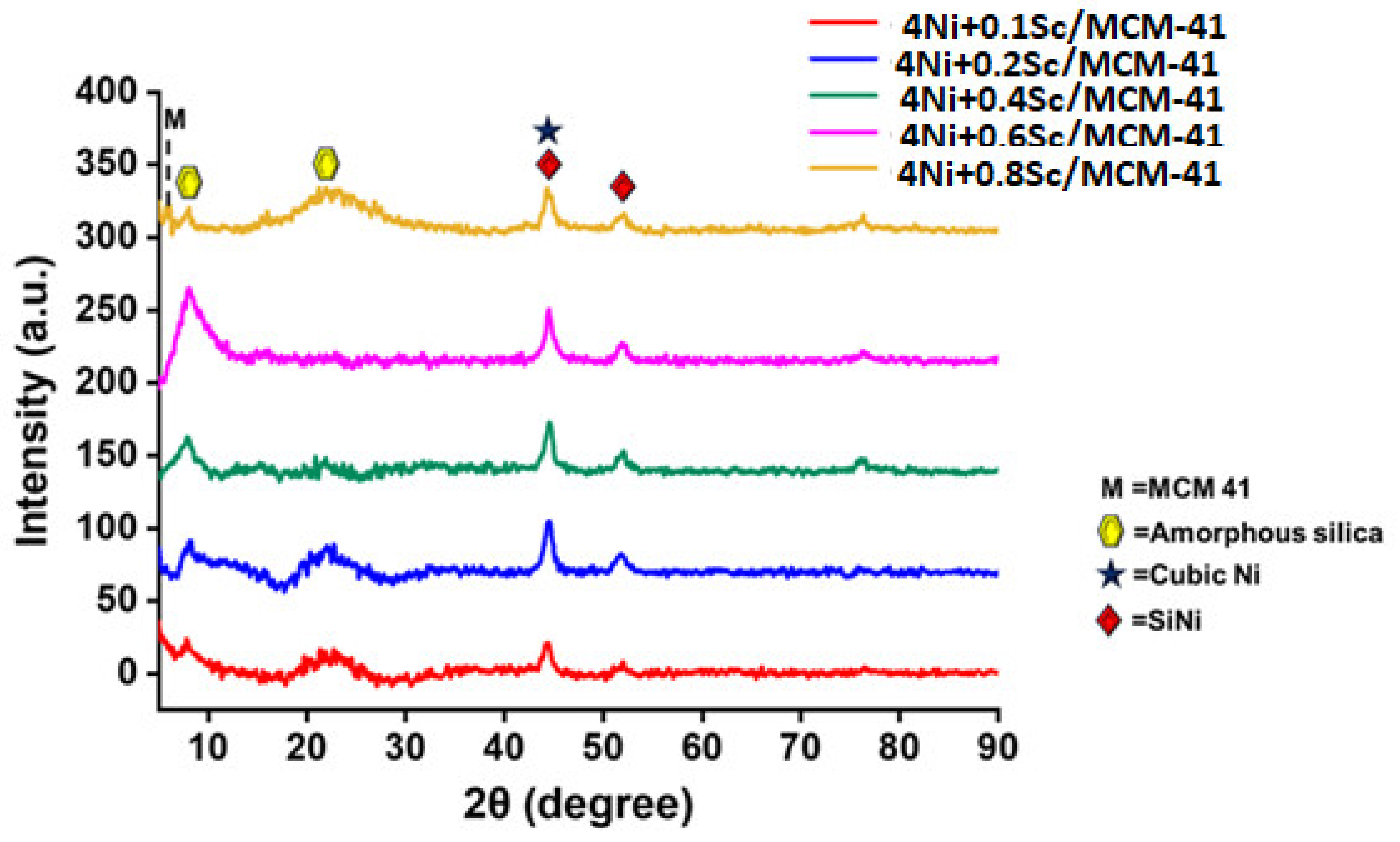
The X-ray diffraction (XRD) profiles used to identify the phase composition and crystal structures of the reduced catalysts are illustrated in Figure 3. The highly organized mesoporous structure of MCM-41 is characterized by peaks observed at low angles (about 2θ = 2.1°, 3.5°, and 5.3°) with reflections of (100), (110), and (200), respectively [46]. The presence of these peaks confirms that the MCM-41 structure was preserved across all samples. The XRD patterns show reflections at approximately 7.9° and 22.5° for the promoted catalysts, which are attributed to the amorphous silica present in the MCM-41 support, with Miller indices of (111) and (242) (ICDD = 96-900-8416) [47]. Additionally, peaks at 2θ = 44.5° and 51.9°, with Miller indices of (111) and (020) (ICDD = 96-152-2621), corresponded to SiNi. The significant peaks at higher angles, around 2θ = 44.4° and 51.7° with Miller indices of (111) and (200) (ICDD = 96-210-0656 and 96-900-8803), were due to the crystalline phases of nickel concerned with cubic crystal systems and Fm3m space groups [47]. Interestingly, upon increasing Sc loading from 0.1 wt.% to 0.2 wt.%, the crystalline peak intensity of Ni increased markedly (Figure S1). The peak intensity of the metallic Ni phase remained comparable up to 0.6 wt.% Sc incorporation in the 5Ni/MCM-41 catalyst. However, the crystalline peak intensity for the metallic Ni phase decreased upon further loading of Sc (>0.6 wt.%). This observation indicates that scandium incorporation up to 0.6 wt.% induces the growth of higher crystalline metallic Ni growth over the catalyst surface. The Sc promoter was highly dispersed within the MCM-41 matrix, with no distinct peaks observed for it. As the Sc loading increased, the peak intensities generally decreased. The peaks also tended to become slightly broader with increased Sc loading.
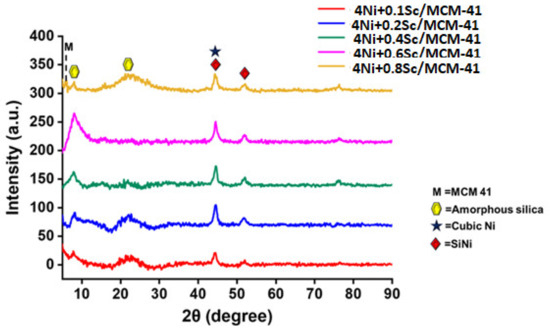
Figure 3.
XRD profiles of reduced 4NixSc/MCM-41 (x = 0.1, 0.2, 0.4, 0.6, 0.8 wt.%) catalysts.
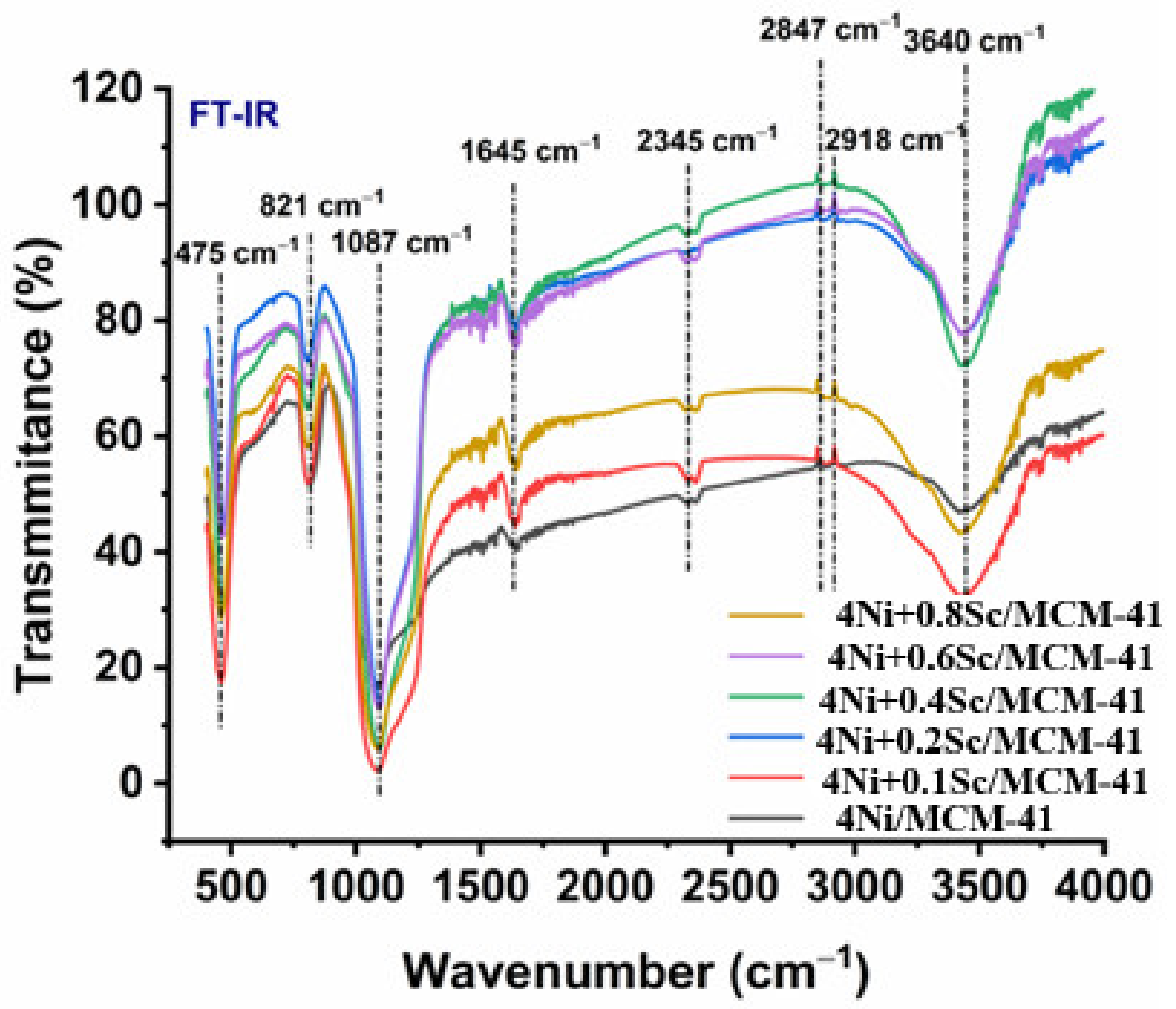
2.4. FTIR Spectroscopy
FTIR spectroscopy is a potent method for locating molecular structures and functional groups in materials. The stretching vibrations of O-H bonds connected to adsorbed water molecules on the catalyst surface were in charge of the broadband in Figure 4 at 3400–3500 cm−1. As the scandium level rises, this band’s strength fell, suggesting a potential drop in surface hydroxyl groups. The alkyl groups’ C-H stretching vibrations are represented by the band at 2918 cm−1, which was most likely the result of adsorbed species on the catalyst surface. The stretching vibrations of the C-N group were responsible for the band at 2345 cm−1, indicating the presence of nitrile groups, which could be from adsorbed species. The 1645 cm−1 band describes the bending vibrations of adsorbed water molecules. As the scandium content rose, its intensity likewise fell, which is in line with the O-H stretching band observation. The asymmetric and symmetric stretching vibrations of Si-O-Si bonds in the MCM-41 support were associated with the bands at 1087 cm−1 and 821 cm−1. These bands’ existence confirms the MCM-41 structure’s stability. The presence of the MCM-41 support was further supported by the band at 475 cm−1, which was attributed to the bending vibrations of the Si-O-Si bonds. The surface characteristics of the Ni/MCM-41 catalysts changed with the addition of Sc, as indicated by the FTIR spectra. As the concentration of Sc increased, the intensity of the O-H and H-O-H bending bands decreased, suggesting a reduction in the number of surface hydroxyl groups. This decrease may have been due to Sc interacting with the hydroxyl groups on the surface, leading to partial dihydroxylation. Additionally, the presence of C-H and C-N bands indicated the existence of adsorbed species on the catalyst surface.
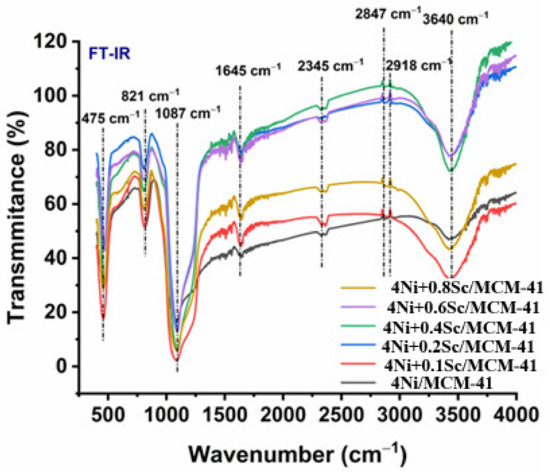
Figure 4.
Infrared spectra of fresh catalysts calcined at 550 °C for 4Ni/MCM-41, 4NixSc/MCM-41 (x = 0.1, 0.2, 0.4, 0.6, and 0.8 wt.%).
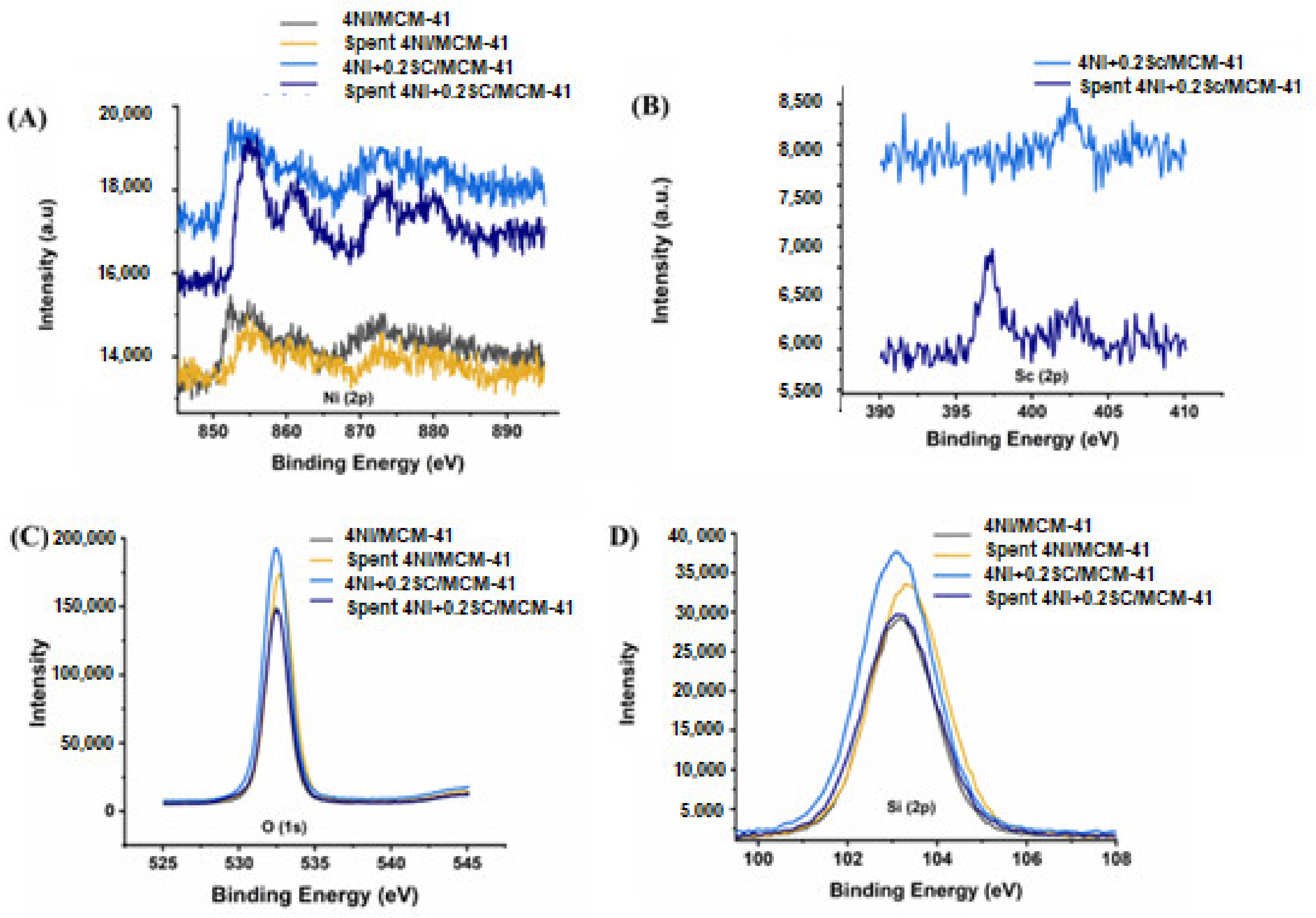
2.5. XPS Analysis
The XPS spectra confirmed the presence of Ni, Sc, O, and Si in the catalysts. Figure 5 exhibits the XPS for the 4Ni/MCM-41 and 4Ni+0.25Sr/MCM-41 catalysts. Over the reduced catalysts (4Ni/MCM-41 and 4Ni+0.2Sr/MCM-41), the metallic Ni state was confirmed by Ni (2p3/2) at 852.4 eV [48]. The Ni+2 oxidation state was verified by Ni (2p3/2) spectra at about 855 eV, along with a satellite peak at 861 eV [49,50] and Ni (2p1/2) spectra at 873 eV [51]. It is interesting to note that after the addition of 0.2 wt.% Sc, the XPS spectra for metallic Ni and Ni+2 were intensified, which indicated active site enrichment upon the loading of the Sc promoter. XRD also verified the enrichment of active sites (metallic Ni). Sc (2p3/2) XPS spectra verified the Sc+3 oxidation state (at 402.8 eV) in a reduced 4Ni+0.2Sr/MCM-41 catalyst [52]. However, in the spent 4Ni+0.2Sr/MCM-41 catalyst, the reduced state of Sc was also evident at a lower binding energy (~397 eV) [53]. This indicates the loss of oxygen from Sc2O3 during the oxidation reaction. The O (1s) spectra and Si (2p3/2) spectra of all catalysts were about 532.5 eV and 103 eV, respectively. Both are characteristic peaks of SiO2 [54,55]. The intensity of both O (1s) and Si (2p3/2) peaks were found to be maximum in the case of the Sc-promoted reduced 4Ni/MCM-41 catalyst. This indicates the surface enrichment of oxides by SiO2. Overall, the XPS spectra of reduced catalysts showed the presence of both metallic Ni and NiO (due to the partial reduction of NiO under hydrogen during reductive pretreatment), Sc2O3, and SiO2. The surface oxide enrichment, as well as the active site enrichment upon the addition of Sc were also confirmed by XPS.
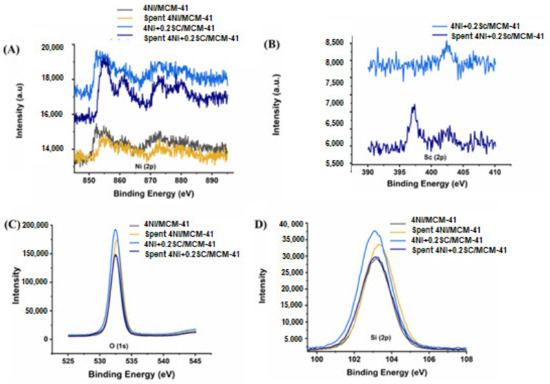
Figure 5.
XPS analysis of the non-promoted 4Ni/MCM-41 and best-promoted 4Ni+0.2Sc/MCM-41 catalysts. (A) Ni (2p) spectra, (B) Sc (2p) spectra, (C) O (1s) spectra, (D) Si (2p).
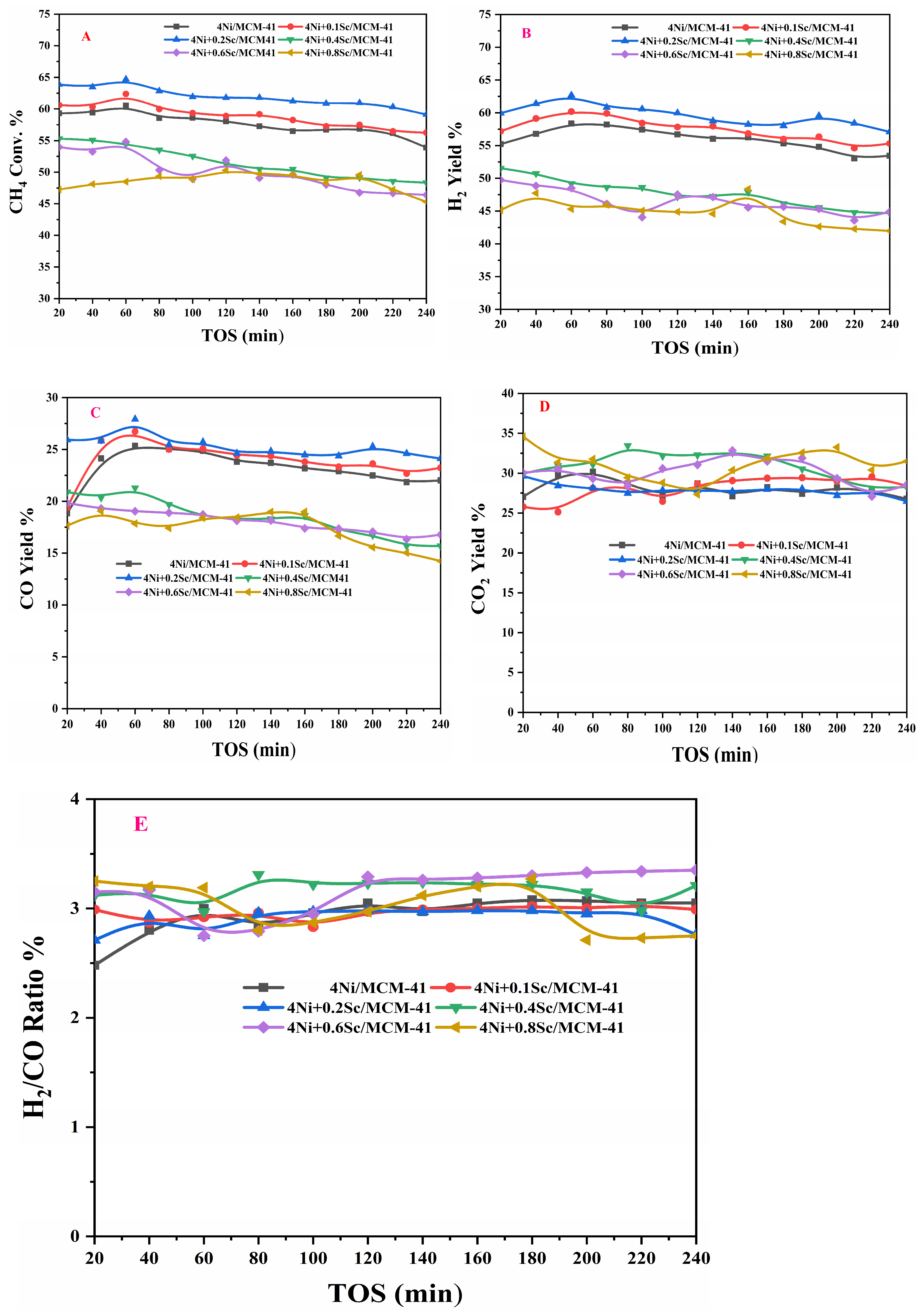
2.6. Catalytic Activity Results
The experimental setup for the POM is shown in Figure S2 of the catalytic activity Test S2 in the Supplementary Materials. The catalytic activities for the POM using both promoted and non-promoted catalysts at a reaction temperature of 650 °C over a time on stream (TOS) of 240 min are shown in Figure 6A. All catalysts exhibited a high initial conversion of methane, with the 4Ni+0.2Sc/MCM-41 catalyst demonstrating the highest conversion rate. However, all catalysts experienced deactivation over time, as indicated by the decreasing conversion rates. The non-promoted catalyst (4Ni/-) achieved an initial methane conversion of 59.9%, which decreased to 53.8% after 240 min of TOS. In contrast, adding a 0.1% Sc promoter affected the conversions positively; for example, it provided an initial methane conversion of 60.8% and a conversion of 56.4% at 240 min of TOS. This indicates that the Sc promoter enhanced the catalyst’s activity performance. When Sc was added, there were more active sites. When the concentration of Sc was further increased to 0.2%, the initial conversion rose to 63.9%, but after 240 min on stream (TOS), it decreased to 59.1%. Conversely, when the concentration of Sc exceeded 0.2%, the catalytic activity declined. This may have been due to excess Sc covering the sites on the Ni particles. Overall, the data suggests that achieving optimal performance in methane conversion over MCM-41-supported catalysts requires a careful balance of Ni and Sc loading. Figure 6B illustrates the hydrogen yield over TOS for various catalysts containing Ni supported on MCM-41, with different amounts of Sc. This figure indicates that the Sc promoter had a similar effect on both methane conversion and hydrogen yield in these catalysts. Figure 6C,D display the CO yield and CO2 yield, respectively, over TOS for the different catalysts. It is noted that the CO yield generally decreased over time for all catalysts. This decline is expected, as the active sites on the catalyst can become deactivated over time. The initial yield of CO was highest for the 4Ni+0.2Sc/MCM-41 catalyst, indicating that this catalyst is very effective for CO production. In contrast, the catalysts with higher Sc loading (4Ni+0.6Sc/MCM-41 and 4Ni+0.8Sc/MCM-41) exhibited lower initial CO yields. Figure 6E illustrates the H2/CO ratio as a function of TOS for the different catalysts. Generally, the H2/CO ratio decreased over time for all catalysts, suggesting a loss of selectivity for H2 production. The 4Ni+0.8Sc/MCM-41 catalyst started with the highest initial H2/CO ratio. Meanwhile, catalysts with lower Sc loading (4Ni/MCM-41, 4Ni+0.1Sc/MCM-41, and 4Ni+0.2Sc/MCM-41) demonstrated better stability in maintaining their H2/CO ratio over time, indicating a slower rate of deactivation compared to those with higher Sc loading. The catalysts with higher Sc loading (4Ni+0.4Sc/MCM-41, 4Ni+0.6Sc/MCM-41, and 4Ni+0.8Sc/MCM-41) exhibited a more rapid decrease in the H2/CO ratio, indicating faster deactivation. This rapid deactivation could be attributed to several factors, including the sintering of active sites and carbon deposition. Overall, the data suggest a trade-off between initial H2 selectivity and long-term stability. While catalysts with higher Sc loading yield higher initial H2/CO ratios, they deactivate more quickly. Conversely, catalysts with lower Sc loading show greater stability over time but lower initial H2 selectivity. The higher H2/CO ratios (over 2) can be attributed to the thermodynamically favorable reactions occurring within the same temperature range, which promote the reverse water–gas shift reaction (CO2 + H2 = CO + H2O). This reaction converts CO and H2O into H2 and CO2, further elevating the H2/CO ratio. As the catalysts deactivate over time, there can be changes in selectivity and product distribution, potentially impacting the H2/CO ratio. The varying concentrations of Sc indicate that the promoter can influence the H2/CO ratio. Higher Sc levels may lead to increased H2/CO ratios, possibly due to enhanced reverse water–gas shift activity. The current work is compared with the existing literature in Table S2 of the Supplementary Materials.
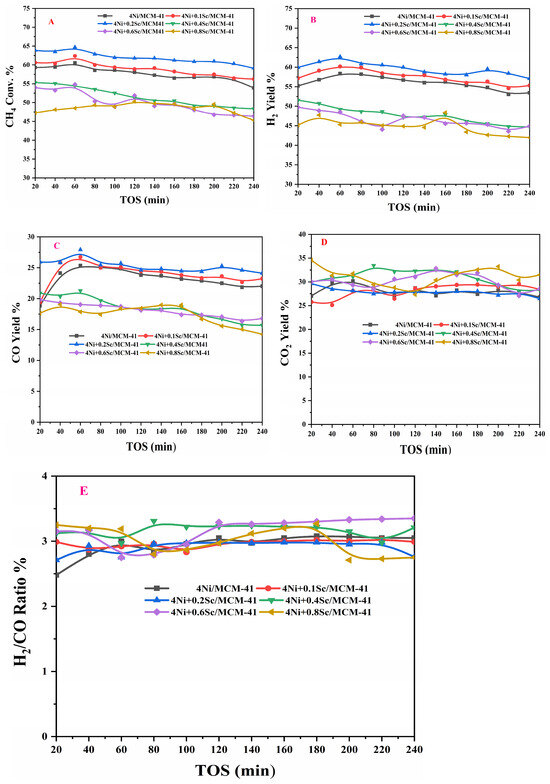
Figure 6.
Activity measurement: (A) CH4 conversion, (B) H2-yield, (C) CO-yield, (D) CO2-yield, and (E) H2/CO ratio.
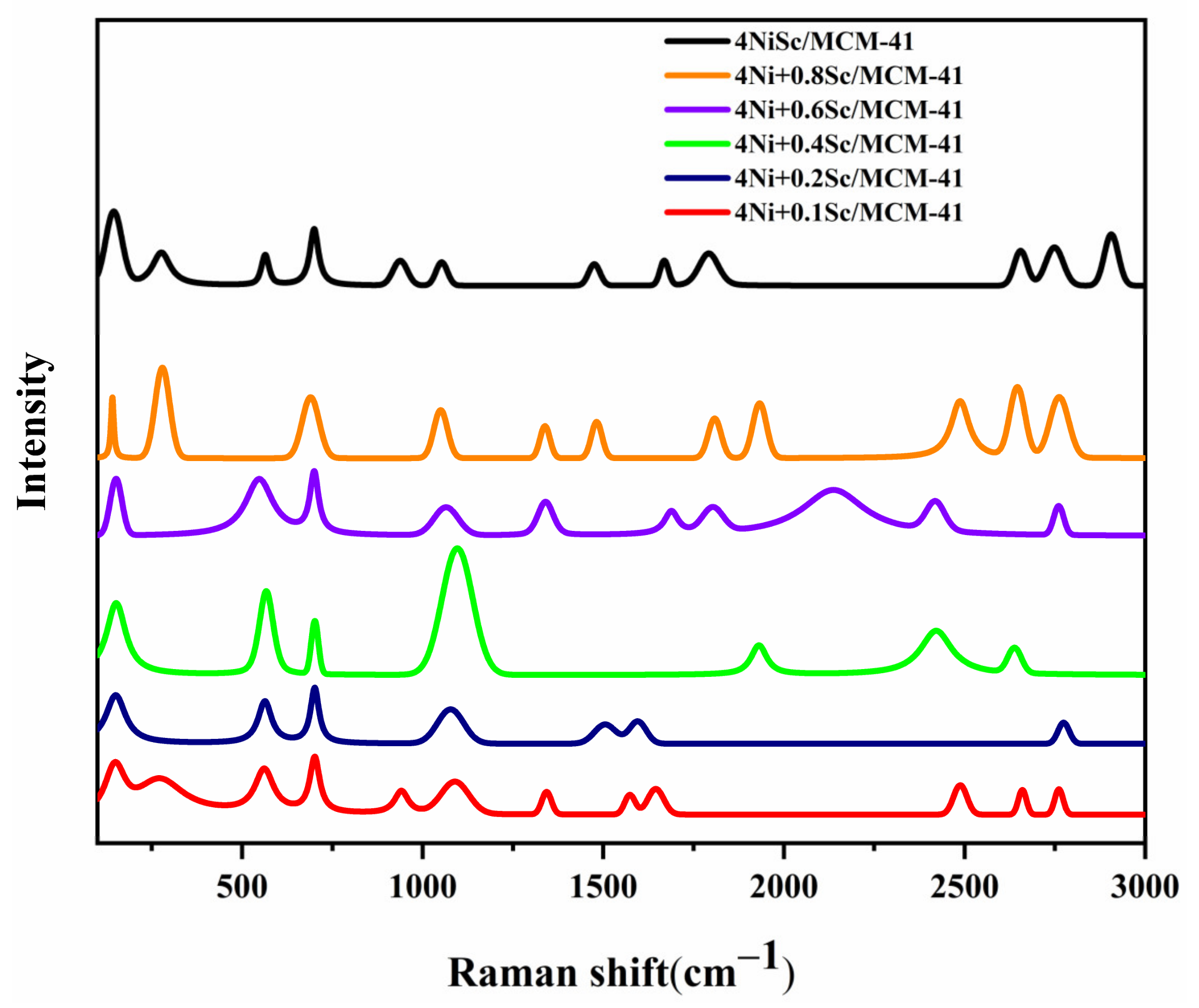
2.7. Raman Analysis
The Raman spectra provide valuable insights into the structural and chemical properties of the catalysts. Figure 7 displays the Raman spectra of the spent catalysts. The peaks in the lower wavenumber region (500–1000 cm−1) are associated with the vibrational modes of the MCM-41 support material. The intensity and shape of the peaks are influenced by the interaction between Ni and Sc species with the sharp peaks in the higher wavenumber region (1000–3000 cm−1) characteristic of the vibrational modes of metal–oxygen bonds, particularly Ni-O and Sc-O bonds. The presence and intensity of these peaks suggest the formation of Ni and Sc oxide species within the MCM-41 framework. The intensity of the peaks in the range of 1300–1600 cm−1 increased with the addition of Sc. These peaks are associated with the formation of Ni-O-Sc bonds. The peak at around 1580 cm−1 was attributed to the stretching vibrations of the Ni-O bonds. This peak shifted to lower wavenumbers with increasing Sc content, indicating a weakening of the Ni-O bond strength.
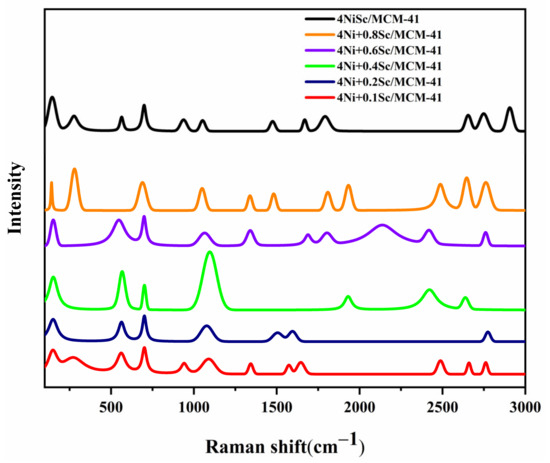
Figure 7.
Raman spectra analysis of the used catalysts.
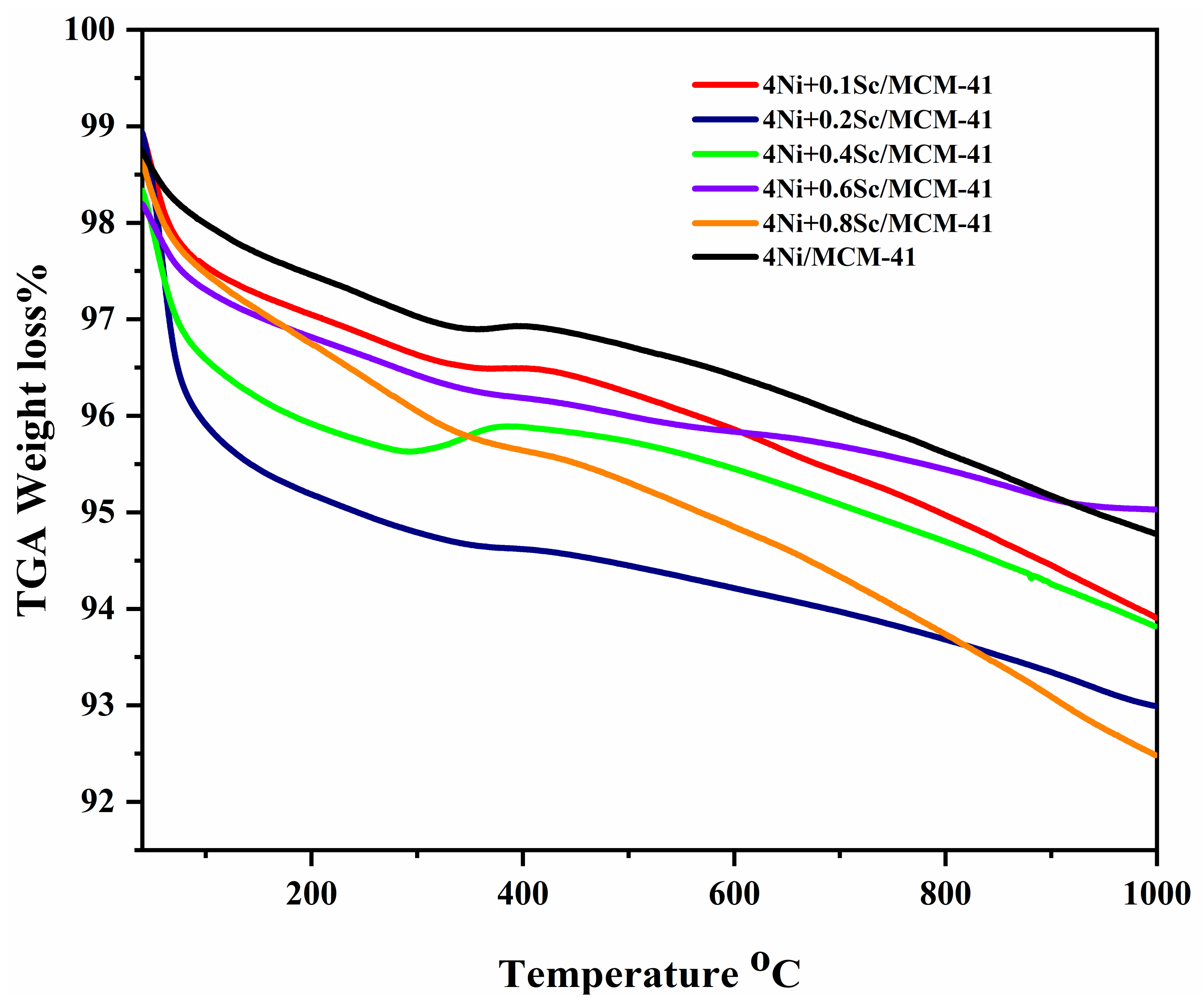
2.8. TGA Analysis
A thermogravimetric experiment measured the carbon deposited on spent catalysts in air (see Figure 8). All samples exhibited a gradual weight loss as the temperature increased. In the low-temperature range of 100–400 °C, the weight loss was due to water desorption and some volatile intermediate products. Between 400 °C and 1000 °C, the weight loss was attributed to the combustion of formed carbon. During this time, the weight of the spent catalysts increased, related to the oxidation of the metallic NiSc active component [56]. The addition of scandium seems to have had a slight effect on the weight loss profile. The curves for samples with higher Sc content (e.g., 0.4 Sc, 0.85 Sc) appeared to have had slightly steeper slopes in the initial temperature range. The weight loss occurred over a wide temperature range, from approximately 100 °C to 1000 °C. Although all samples experienced some weight loss at 1000 °C, they still retained a substantial amount of their initial weight. This indicates that the major weight loss events occurred at lower temperatures, and the remaining weight loss might have been due to more stable species [57].
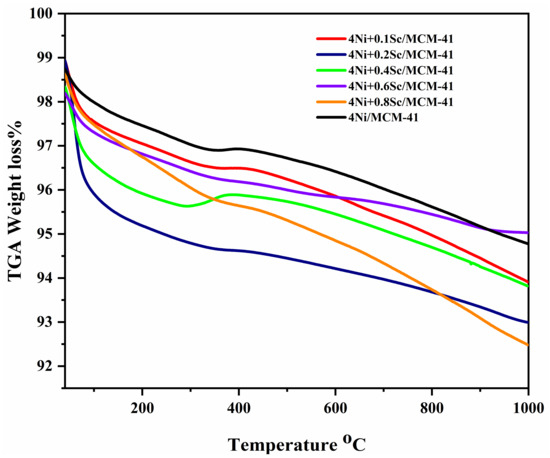
Figure 8.
Thermogravimetric analysis (TGA) of spent catalysts.
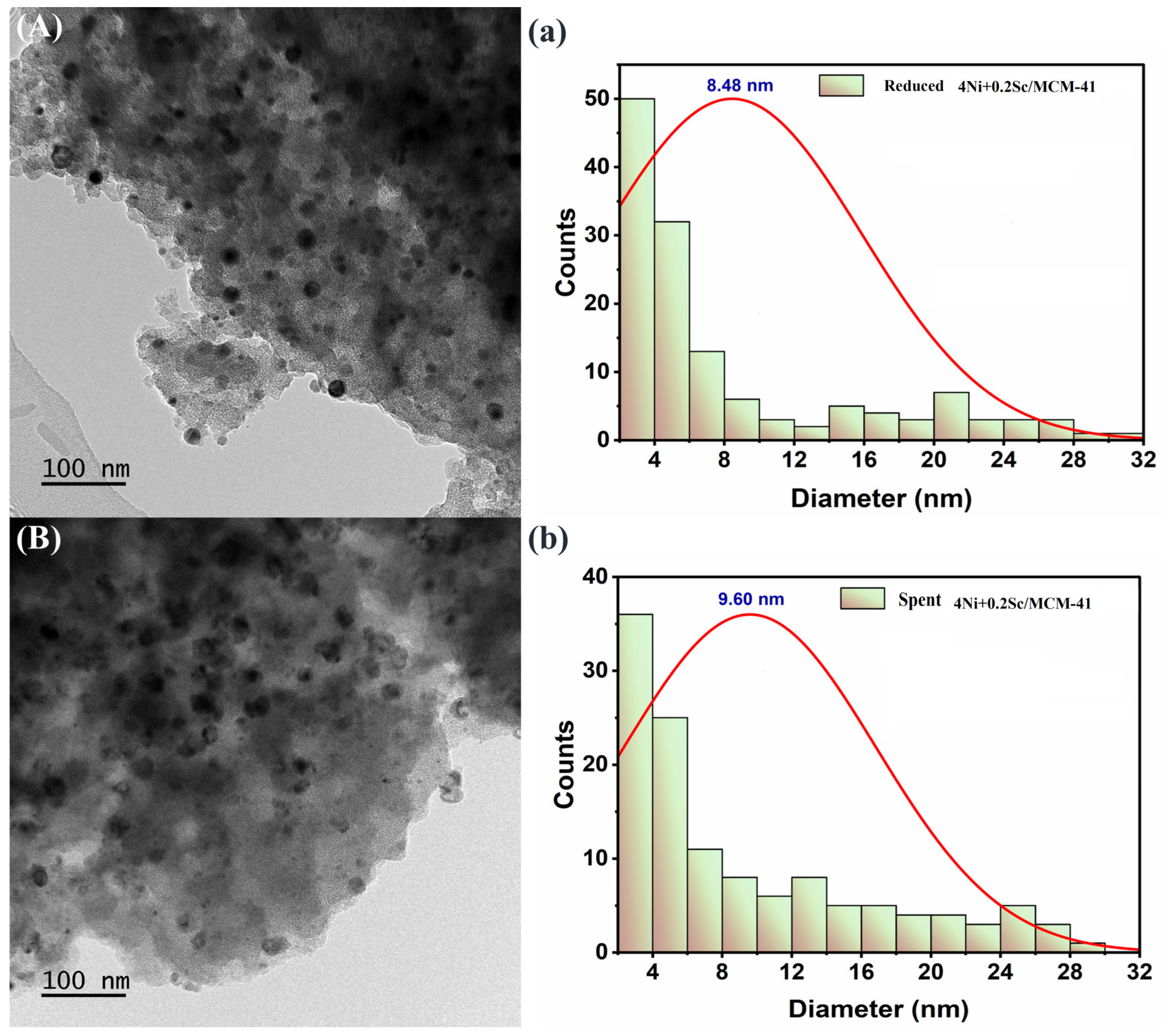
2.9. TEM Images
TEM is a potent method for nanoscale material imaging. It allows us to see individual particles and their size distribution. Figure 9 shows the best catalyst’s TEM, for both fresh and used samples. The fresh sample (Figure 9a) illustrates the porous structure of the MCM-41 support. The darker areas within the pores indicate the presence of nickel nanoparticles. These nanoparticles are relatively small and well dispersed throughout the support. The accompanying histogram reveals a narrow distribution of particle sizes, centered around 8.48 nm. This suggests that the nickel nanoparticles are quite uniform in size. In Figure 9b, the distribution of particles with a wider variety of sizes is less scattered in the spent sample. The plot shows a mean particle size of 9.60 nm, indicating a wider distribution. This implies that, in comparison to sample 5A, the particle size of sample 5B showed lower homogeneity. In general, the addition of scandium contributes to the improved dispersion and stability of the nickel nanoparticles.
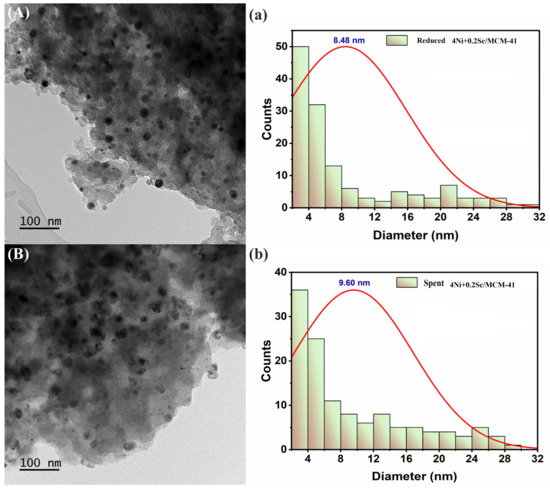
Figure 9.
(A,a) TEM images of fresh 4Ni+0.2Sc/MCM-41 catalyst. (a) Particle size distribution of fresh 4Ni+0.2Sc/MCM-41 catalyst. (B,b) TEM images of spent 4Ni+0.2Sc/MCM-41 catalyst. (b) Particle size distribution of spent 4Ni+0.2Sc/MCM-41 catalyst.
3. Materials and Method
3.1. Material
The materials used were Ni (NO3)2·6H2O (98%, Riedel-de Haen Ag, Seelze, Germany), Sc(NO3)2·4H2O (99.9%, Alfa Produkte, Ventron, France), and MCM-41.
3.2. Catalyst Preparation
The MCM-41 was synthesized using silica sol, tetramethyl ammonium hydroxide solution, and trimethyl stearyl ammonium bromide through the hydrothermal method. Eighteen grams of silica sol (containing 50 wt.% SiO2) was dispersed in forty grams of distilled water while stirring vigorously. Next, fourteen grams of tetramethyl ammonium hydroxide (TMAOH) solution (25 wt.% concentration) and eight grams of trimethyl stearyl ammonium bromide were added to the mixture. Stirring continued until a homogeneous slurry was formed. This slurry was heated for three days at 120 °C in a Teflon-lined autoclave. After heating, the autoclave was cooled with water, and the product was separated from the mother liquor using glass filtration. The residue was washed five times with water and then dried overnight at room temperature. Lastly, the catalyst was calcined at 550 °C for 10 h to burn off the template. Ni- and Sc-impregnated MCM-41 was prepared by incipient wetness co-impregnation by using Ni-nitrate solution (equivalent to 4 wt.% Ni), Sc-nitrate solution (equivalent to 0.1 to 0.8 wt.% Sc), and MCM-41. The respective precursor solution and MCM-41 were stirred at 80 °C and dried at 120 °C overnight. The dried product was calcined at 550 °C for 3 h. The catalysts were called 4Ni/- and 4NixSc/MCM-41 (x = 0.1. 0.2, 0.4, 0.6, and 0.8 wt.%). A detailed description of the catalyst characterization, its activity performance, and catalyst synthesis is provided in the Supplementary Materials as Figures S1, S2, and S3, respectively. The measured composition was within 10% of the desired composition for both Ni and Sc (as shown in the EDX elemental composition in Figure S3).
4. Conclusions
This study successfully demonstrated the potential of Sc-promoted Ni/MCM-41 catalysts for the efficient and stable partial oxidation of methane (POM). All the catalysts exhibited a mesoporous structure with narrow, slit-shaped pores, while the 4Ni+0.2Sc/MCM-41 catalyst showed the most consistent pore size distribution. The addition of Sc as a promoter significantly enhanced catalytic performance by improving the dispersion of Ni active sites, inhibiting sintering, and promoting the formation of strongly interacting Ni species, including Ni-O-Sc bonds. The optimal Sc loading of 0.2 wt.% provided the best balance between initial activity and long-term stability. The reduction of surface hydroxyl groups contributed to improved stability at high temperatures. While the addition of scandium (Sc) enhances initial catalytic activity and promotes the formation of active sites, it also affects long-term stability and product selectivity. Although higher Sc loadings improved the initial performance, they led to faster deactivation, indicating a trade-off between activity and stability. Additionally, Sc’s impact on the overall carbon deposition and combustion behavior of the spent catalysts was limited, but it did promote the enrichment of active sites on the catalyst surface. Future research should focus on refining the catalyst formulation and reaction conditions to achieve higher stability and performance. A deeper understanding of the mechanisms underlying the effects of the Sc promoter could provide valuable insights for designing more efficient catalysts for partial oxidation methane (POM) and related reactions.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal15040337/s1, Catalyst Characterization S1, Catalytic Activity Test S2; Catalyst Synthesis S3; Figure S1. XRD of reduced catalysts in selected Bragg’s angle range (showing metallic cubic Ni phase); Figure S2. Schematic of the experimental setup; Figure S3. EDX elemental profile; Table S1. H2-TPR-O2-TPO-H2-TPR Cyclic Experiments; Table S2. A comparison of the present work activity with the results cited in the previous literature. References [58,59,60,61,62,63,64,65,66] are cited in the Supplementary Materials.
Author Contributions
Methodology, conceptualization, writing—original draft preparation: A.A.I. and A.S.A.-F.; formal analysis and characterization: O.A.B., F.M.A.-A. and M.S.A.; project administration and editing: A.H.F. and A.E.A.; funding and visualization: A.S.B. All authors have read and agreed to the published version of the manuscript.
Funding
Researchers Supporting Project (number RSP2025R368), King Saud University, Riyadh, Saudi Arabia.
Data Availability Statement
Data is contained within the article and Supplementary Material.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kalinci, Y.; Hepbasli, A.; Dincer, I. Biomass-Based Hydrogen Production: A Review and Analysis. Int. J. Hydrogen Energy 2009, 34, 8799–8817. [Google Scholar] [CrossRef]
- Acar, C.; Dincer, I. Review and Evaluation of Hydrogen Production Options for Better Environment. J. Clean. Prod. 2019, 218, 835–849. [Google Scholar] [CrossRef]
- Fakeeha, A.H.; Vadodariya, D.M.; Alotibi, M.F.; Abu-Dahrieh, J.K.; Ibrahim, A.A.; Abasaeed, A.E.; Alarifi, N.; Kumar, R.; Al-Fatesh, A.S. Pd+Al2O3-Supported Ni-Co Bimetallic Catalyst for H2 Production through Dry Reforming of Methane: Effect of Carbon Deposition over Active Sites. Catalysts 2023, 13, 1374. [Google Scholar] [CrossRef]
- Yang, M.; Sun, Z.; Hu, Y.H. Novel WS2-Based 3D Electrode with Protecting Scaffold for Efficient and Stable Hydrogen Evolution. J. Phys. Chem. C 2019, 123, 12142–12148. [Google Scholar] [CrossRef]
- Sher, F.; Al-Shara, N.K.; Iqbal, S.Z.; Jahan, Z.; Chen, G.Z. Enhancing Hydrogen Production from Steam Electrolysis in Molten Hydroxides via Selection of Non-Precious Metal Electrodes. Int. J. Hydrogen Energy 2020, 45, 28260–28271. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, X.; Jha, A.N.; Rogers, H. Natural Gas from Shale Formation—The Evolution, Evidences and Challenges of Shale Gas Revolution in United States. Renew. Sustain. Energy Rev. 2014, 30, 1–28. [Google Scholar] [CrossRef]
- Themelis, N.J.; Ulloa, P.A. Methane Generation in Landfills. Renew. Energy 2007, 32, 1243–1257. [Google Scholar] [CrossRef]
- Afarideh, M.; Esfanjani, P.; Sarlak, F.; Valipour, M.S. A Review on Solar Methane Reforming Systems for Hydrogen Production. Int. J. Hydrogen Energy 2024, in press. [Google Scholar] [CrossRef]
- Ganguli, A.; Bhatt, V. Hydrogen Production Using Advanced Reactors by Steam Methane Reforming: A Review. Front. Therm. Eng. 2023, 3, 1–15. [Google Scholar] [CrossRef]
- Wang, S.; Nabavi, S.A.; Clough, P.T. A Review on Bi/Polymetallic Catalysts for Steam Methane Reforming. Int. J. Hydrogen Energy 2023, 48, 15879–15893. [Google Scholar] [CrossRef]
- Massarweh, O.; Al-khuzaei, M.; Al-Shafi, M.; Bicer, Y.; Abushaikha, A.S. Blue Hydrogen Production from Natural Gas Reservoirs: A Review of Application and Feasibility. J. CO2 Util. 2023, 70, 102438. [Google Scholar] [CrossRef]
- Khaleel, A.; Jobe, S.; Ahmed, M.; Al-Zuhair, S.; Tariq, S. Enhanced Selectivity of Syngas in Partial Oxidation of Methane: A New Route for Promising Ni-Alumina Catalysts Derived from Ni/γ-AlOOH with Modified Ni Dispersion. Int. J. Energy Res. 2020, 44, 12081–12099. [Google Scholar] [CrossRef]
- Ma, L.; Ding, C.; Wang, J.; Xu, H.; Zhang, K. Lanthanide Modified Pt/CeO2-Based Catalysts for Methane Partial Oxidation: Relationship between Catalytic Activity and Structure. Int. J. Hydrogen Energy 2023, 48, 19074–19086. [Google Scholar] [CrossRef]
- Gazi, M.J.; Khurana, D.; Kaishyop, J.; Khan, T.S.; Bhandari, S.; Bordoloi, A. Solution Combustion Derived Nanoalloys: Robust and Efficient Catalyst Systems for Partial Oxidation of Methane. Int. J. Hydrogen Energy 2024, 51, 562–579. [Google Scholar] [CrossRef]
- Al-anazi, A.; Bellahwel, O.; Kavitha, C.; Abu-dahrieh, J.; Ibrahim, A.A.; Santhosh, S.; Abasaeed, A.E.; Fakeeha, A.H.; Al-fatesh, A.S. Promoter Impact on 4Ni/SAPO-5 Catalyst for H2 Production via Methane Partial Oxidation. Catalysts 2024, 14, 316. [Google Scholar] [CrossRef]
- Zeng, H.; Wang, Y.; Gong, S.; Shi, Y.; Cai, N. Catalytically Enhanced Methane-Rich Combustion by Porous Media Reactor. Fuel 2019, 248, 65–75. [Google Scholar] [CrossRef]
- Shi, S.; Sun, Z.; Bao, C.; Gao, T.; Hu, Y.H. The Special Route toward Conversion of Methane to Methanol on a Fluffy Metal-Free Carbon Nitride Photocatalyst in the Presence of H2O2. Int. J. Energy Res. 2020, 44, 2740–2753. [Google Scholar] [CrossRef]
- Jin, Z.; Wang, L.; Zuidema, E.; Mondal, K.; Zhang, M.; Zhang, J.; Wang, C.; Meng, X.; Yang, H.; Mesters, C.; et al. Hydrophobic Zeolite Modification for In Situ Peroxide Formation in Methane Oxidation to Methanol. Science 2020, 367, 193–197. [Google Scholar] [CrossRef]
- Jung, S.; Lee, J.; Moon, D.H.; Kim, K.H.; Kwon, E.E. Upgrading Biogas into Syngas through Dry Reforming. Renew. Sustain. Energy Rev. 2021, 143, 110949. [Google Scholar] [CrossRef]
- Galadima, A.; Muraza, O. From Synthesis Gas Production to Methanol Synthesis and Potential Upgrade to Gasoline Range Hydrocarbons: A Review. J. Nat. Gas Sci. Eng. 2015, 25, 303–316. [Google Scholar] [CrossRef]
- Ibrahim, A.A.; Fakeeha, A.H.; Abasaeed, A.E.; Wazeer, I.; Bentalib, A.; Siva Kumar, N.; Abu-Dahrieh, J.K.; Al-Fatesh, A.S. The Effectiveness of Ni-Based Bimetallic Catalysts Supported by MgO-Modified Alumina in Dry Methane Reforming. Catalysts 2023, 13, 1420. [Google Scholar] [CrossRef]
- Angeli, S.D.; Turchetti, L.; Monteleone, G.; Lemonidou, A.A. Catalyst Development for Steam Reforming of Methane and Model Biogas at Low Temperature. Appl. Catal. B Environ. 2016, 181, 34–46. [Google Scholar] [CrossRef]
- Chien, A.C.; Ye, N.J. Effect of Preparation Method and Particle Size of Ni/SDC Catalyst on Methane Oxidation. Catal. Commun. 2021, 154, 106312. [Google Scholar] [CrossRef]
- Pakhare, D.; Spivey, J. A Review of Dry (CO2) Reforming of Methane over Noble Metal Catalysts. Chem. Soc. Rev. 2014, 43, 7813–7837. [Google Scholar]
- Niu, J.; Du, X.; Ran, J.; Wang, R. Dry (CO2) Reforming of Methane over Pt Catalysts Studied by DFT and Kinetic Modeling. Appl. Surf. Sci. 2016, 376, 79–90. [Google Scholar] [CrossRef]
- Wang, N.; Shen, K.; Yu, X.; Qian, W.; Chu, W. Preparation and Characterization of a Plasma Treated NiMgSBA-15 Catalyst for Methane Reforming with CO2 to Produce Syngas. Catal. Sci. Technol. 2013, 3, 2278–2287. [Google Scholar] [CrossRef]
- Liu, C.J.; Ye, J.; Jiang, J.; Pan, Y. Progresses in the Preparation of Coke Resistant Ni-Based Catalyst for Steam and CO2 Reforming of Methane. ChemCatChem 2011, 3, 529–541. [Google Scholar]
- Chien, A.C.; Liao, B.Y.; Chen, W.Y.; Chien, A.C. Studies of Exsolution and Catalytic Activity of Metal Nanocatalysts from Parent Perovskite. Catal. Sci. Technol. 2021, 11, 4570–4580. [Google Scholar] [CrossRef]
- Bradford, M.C.J.; Vannice, M.A. The Role of Metal-Support Interactions in CO2 Reforming of CH4. Catal. Today 1999, 50, 87–96. [Google Scholar] [CrossRef]
- Tang, M.; Liu, K.; Roddick, D.M.; Fan, M. Enhanced Lattice Oxygen Reactivity over Fe2O3/Al2O3 Redox Catalyst for Chemical-Looping Dry (CO2) Reforming of CH4: Synergistic La-Ce Effect. J. Catal. 2018, 368, 38–52. [Google Scholar] [CrossRef]
- Faria, E.C.; Neto, R.C.R.; Colman, R.C.; Noronha, F.B. Hydrogen Production through CO2 Reforming of Methane over Ni/CeZrO2/Al2O3 Catalysts. In Catalysis Today; Elsevier: Amsterdam, The Netherlands, 2014; pp. 138–144. [Google Scholar]
- Stagg-Williams, S.M.; Noronha, F.B.; Fendley, G.; Resasco, D.E. CO2 Reforming of CH4 over Pt/ZrO2 Catalysts Promoted with La and Ce Oxides. J. Catal. 2000, 194, 240–249. [Google Scholar] [CrossRef]
- Hasnan, N.S.N.; Timmiati, S.N.; Lim, K.L.; Yaakob, Z.; Kamaruddin, N.H.N.; Teh, L.P. Recent Developments in Methane Decomposition over Heterogeneous Catalysts: An Overview. Mater. Renew. Sustain. Energy 2020, 9, 8. [Google Scholar] [CrossRef]
- Sun, X.; Xu, D.; Dai, P.; Liu, X.; Tan, F.; Guo, Q. Efficient Degradation of Methyl Orange in Water via Both Radical and Non-Radical Pathways Using Fe-Co Bimetal-Doped MCM-41 as Peroxymonosulfate Activator. Chem. Eng. J. 2020, 402, 125881. [Google Scholar] [CrossRef]
- Kumar, N.; Leino, E.; Mäki-Arvela, P.; Aho, A.; Käldström, M.; Tuominen, M.; Laukkanen, P.; Eränen, K.; Mikkola, J.P.; Salmi, T.; et al. Synthesis and Characterization of Solid Base Mesoporous and Microporous Catalysts: Influence of the Support, Structure, and Type of Base Metal. Microporous Mesoporous Mater. 2012, 152, 71–77. [Google Scholar] [CrossRef]
- Dündar-Tekkaya, E.; Yürüm, Y. Effect of Loading Bimetallic Mixture of Ni and Pd on Hydrogen Storage Capacity of MCM-41. Int. J. Hydrogen Energy 2015, 40, 7636–7643. [Google Scholar] [CrossRef]
- Li, M.; Hui, K.N.; Hui, K.S.; Lee, S.K.; Cho, Y.R.; Lee, H.; Zhou, W.; Cho, S.; Chao, C.Y.H.; Li, Y. Influence of Modification Method and Transition Metal Type on the Physicochemical Properties of MCM-41 Catalysts and Their Performances in the Catalytic Ozonation of Toluene. Appl. Catal. B Environ. 2011, 107, 245–252. [Google Scholar] [CrossRef]
- Pârvulescu, V.; Anastasescu, C.; Su, B.L. Bimetallic Ru-(Cr, Ni, or Cu) and La-(Co or Mn) Incorporated MCM-41 Molecular Sieves as Catalysts for Oxidation of Aromatic Hydrocarbons. J. Mol. Catal. A Chem. 2004, 211, 143–148. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, L.; Cui, Y.; Xing, Y.; Su, W. Research on Nickel-Based Catalysts for Carbon Dioxide Methanation Combined with Literature Measurement. J. CO2 Util. 2022, 63, 102117. [Google Scholar] [CrossRef]
- Chien, A.C.; Ye, N.J.; Huang, C.W.; Tseng, I.H. Studies of Nickel/Samarium-Doped Ceria for Catalytic Partial Oxidation of Methane and Effect of Oxygen Vacancy. Catalysts 2021, 11, 731. [Google Scholar] [CrossRef]
- Shiratori, Y.; Teraoka, Y.; Sasaki, K. Ni1-x-YMgxAlyO-ScSZ Anodes for Solid Oxide Fuel Cells. Solid State Ion. 2006, 177, 1371–1380. [Google Scholar] [CrossRef]
- Rajput, Y.B.; Al-Fatesh, A.S.; Osman, A.I.; Bayazed, M.O.; Ibrahim, A.A.; Fakeeha, A.H.; Abasaeed, A.E.; Almubaddel, F.S.; Alothman, O.; Kumar, R. Enhancing Hydrogen Production via Dry Reforming of Methane: Optimization of Co and Ni on Scandia-Ceria-Zirconia Supports for Catalytic Efficiency and Economic Feasibility. Fuel 2024, 378, 132843. [Google Scholar] [CrossRef]
- Reza Loghman-Estarki, M.; Shoja Razavi, R.; Edris, H. Synthesis of Scandia, Yttria Stabilized Zirconia (SYSZ) Nanoparticles by New Wet Chemistry Method. Curr. Nanosci. 2012, 8, 767–775. [Google Scholar] [CrossRef]
- Al-Fatesh, A.S.; Atia, H.; Abu-Dahrieh, J.K.; Ibrahim, A.A.; Eckelt, R.; Armbruster, U.; Abasaeed, A.E.; Fakeeha, A.H. Hydrogen Production from CH4 Dry Reforming over Sc Promoted Ni / MCM-41. Int. J. Hydrogen Energy 2019, 44, 20770–20781. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, J.; Ding, J.; Liu, T.; Shi, G.; Li, X.; Dang, W.; Cheng, Y.; Guo, R. Pore Structure and Fractal Characteristics of Different Shale Lithofacies in the Dalong Formation in the Western Area of the Lower Yangtze Platform. Minerals 2020, 10, 72. [Google Scholar] [CrossRef]
- Tang, Z.; Zhang, Y.; Guo, Q. Catalytic Hydrocracking of Pyrolytic Lignin to Liquid Fuel in Supercritical Ethanol. Ind. Eng. Chem. Res. 2010, 49, 2040–2046. [Google Scholar] [CrossRef]
- Cho, Y.S.; Park, J.C.; Lee, B.; Kim, Y.; Yi, J. Preparation of Mesoporous Catalyst Supported on Silica with Finely Dispersed Ni Particles. Catal. Lett. 2002, 81, 89–96. [Google Scholar] [CrossRef]
- Schreifels, J.A.; Maybury, P.C.; Swartz, W.E. X-Ray Photoelectron Spectroscopy of Nickel Boride Catalysts: Correlation of Surface States with Reaction Products in the Hydrogenation of Acrylonitrile. J. Catal. 1980, 65, 195–206. [Google Scholar] [CrossRef]
- Lian, K.K.; Kirk, D.W.; Thorpe, S.J. Erratum: Investigation of a “Two-State” Tafel Phenomenon for the Oxygen Evolution Reaction on an Amorphous Ni-Co Alloy. Electrochem. Soc. 1995, 142, 4309. [Google Scholar] [CrossRef]
- Bianchi, C.L.; Cattania, M.G.; Villa, P. XPS Characterization of Ni and Mo Oxides before and after “in Situ” Treatments. Appl. Surf. Sci. 1993, 70, 211–216. [Google Scholar] [CrossRef]
- Mansour, A.N. Characterization of β -Ni (OH)2 by XPS. Surf. Sci. Spectra 1994, 3, 239–246. [Google Scholar] [CrossRef]
- Gimzewski, J.K.; Fabian, D.J.; Watson, L.M.; Affrossman, S. X-Ray Photoelectron Studies of Scandium and Its Hydride and Oxide. J. Phys. F Met. Phys. 1977, 7, L305. [Google Scholar] [CrossRef]
- Myers, C.E.; Franzen, H.F.; Anderegg, J.W. X-ray photoelectron spectra and bonding in transition-metal phosphides. Inorg. Chem. 1985, 24, 1822–1824. [Google Scholar] [CrossRef]
- Al-Fatesh, A.S.; Kasim, S.O.; Ibrahim, A.A.; Osman, A.I.; Abasaeed, A.E.; Atia, H.; Armbruster, U.; Frusteri, L.; Bin Jumah, A.; Mohammed Alanazi, Y.; et al. Greenhouse Gases Utilization via Catalytic Reforming with Sc Promoted Ni/SBA-15. Fuel 2022, 330, 125523. [Google Scholar] [CrossRef]
- Takigawa, Y.; Kurosawa, K.; Sasaki, W.; Yoshida, K.; Fujiwara, E.; Kato, Y. SiO Bond Breaking in SiO2 by Vacuum Ultraviolet Laser Radiation. J. Non-Cryst. Solids 1990, 116, 293–296. [Google Scholar] [CrossRef]
- Al-Fatesh, A.S.; Arafat, Y.; Kasim, S.O.; Ibrahim, A.A.; Abasaeed, A.E.; Fakeeha, A.H. In Situ Auto-Gasification of Coke Deposits over a Novel Ni-Ce/W-Zr Catalyst by Sequential Generation of Oxygen Vacancies for Remarkably Stable Syngas Production via CO2-Reforming of Methane. Appl. Catal. B Environ. 2021, 280, 119445. [Google Scholar] [CrossRef]
- Damaskinos, C.M.; Vasiliades, M.A.; Efstathiou, A.M. The Effect of Ti4+ Dopant in the 5 Wt% Ni/Ce1-XTixO2-Δ Catalyst on the Carbon Pathways of Dry Reforming of Methane Studied by Various Transient and Isotopic Techniques. Appl. Catal. A Gen. 2019, 579, 116–129. [Google Scholar] [CrossRef]
- Du, G.; Lim, S.; Yang, Y.; Wang, C.; Pfefferle, L.; Haller, G.L. Catalytic Performance of Vanadium Incorporated MCM-41 Catalysts for the Partial Oxidation of Methane to Formaldehyde. Appl. Catal. A Gen. 2006, 302, 48–61. [Google Scholar] [CrossRef]
- Ding, C.; Wang, J.; Li, Y.; Ma, Q.; Ma, L.; Guo, J.; Ma, Z.; Liu, P.; Zhang, K. The Role of Active Sites Location in Partial Oxidation of Methane to Syngas for MCM-41 Supported Ni Nanoparticles. Catalysts 2019, 9, 606. [Google Scholar] [CrossRef]
- Liu, R.; Yang, M.; Huang, C.; Weng, W.; Wan, H. Partial Oxidation of Methane to Syngas over Mesoporous Co-Al2O3 Catalysts. Chin. J. Catal. 2013, 34, 146–151. [Google Scholar] [CrossRef]
- Moral, A.; Reyero, I.; Llorca, J.; Bimbela, F.; Gandía, L.M. Partial Oxidation of Methane to Syngas Using Co/Mg and Co/Mg-Al Oxide Supported Catalysts. Catal. Today 2019, 333, 259–267. [Google Scholar] [CrossRef]
- Lucrédio, A.F.; Jerkiewicz, G.; Assaf, E.M. Cobalt Catalysts Promoted with Cerium and Lanthanum Applied to Partial Oxidation of Methane Reactions. Appl. Catal. B Environ. 2008, 84, 106–111. [Google Scholar] [CrossRef]
- Fakeeha, A.H.; Arafat, Y.; Ibrahim, A.A.; Shaikh, H.; Atia, H.; Abasaeed, A.E.; Armbruster, U.; Al-Fatesh, A.S. Highly Selective Syngas/H2 Production via Partial Oxidation of CH4 Using (Ni, Co and Ni-Co)/ZrO2-Al2O3 Catalysts: Influence of Calcination Temperature. Processes 2019, 7, 141. [Google Scholar] [CrossRef]
- Xia, W.S.; Hou, Y.H.; Chang, G.; Weng, W.Z.; Han, G.-B.; Wan, H.-L. Partial Oxidation of Methane into Syngas (H2 + CO) over Effective High-Dispersed Ni/SiO2 Catalysts Synthesized by a Sol–Gel Method. Int. J. Hydrogen Energy 2012, 37, 8343–8353. [Google Scholar] [CrossRef]
- Emamdoust, A.; La Parola, V.; Pantaleo, G.; Testa, M.L.; Farjami Shayesteh, S.; Venezia, A.M. Partial Oxidation of Methane over SiO2 Supported Ni and NiCe Catalysts. J. Energy Chem. 2020, 47, 1–9. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Tang, Q.; Guo, Q.; Zhang, Q.; Wan, H. MCM-41-Supported Iron Phosphate Catalyst for Partial Oxidation of Methane to Oxygenates with Oxygen and Nitrous Oxide. J. Catal. 2003, 217, 457–467. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).