Carbon-Encapsulated Ni Nanoparticles Catalysts Derived from Ni-Hexamine Coordination Frameworks for Oxygen Reduction Reaction and Oxygen Evolution Reaction
Abstract
1. Introduction
2. Results and Discussion
2.1. Morphology and Structural Characterizations of the NiHMT
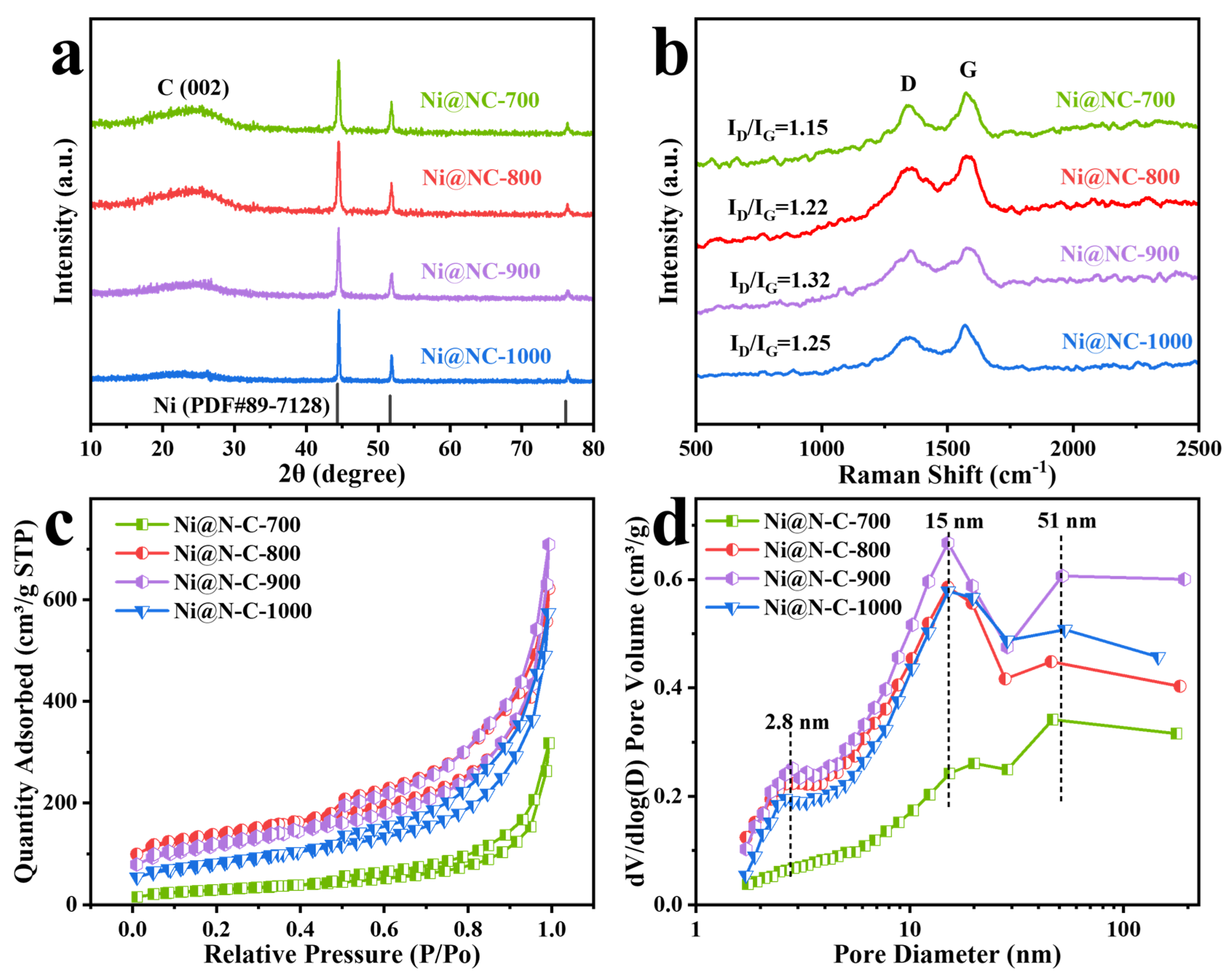
2.2. Morphology and Structural Characterizations of the Ni@NC-T
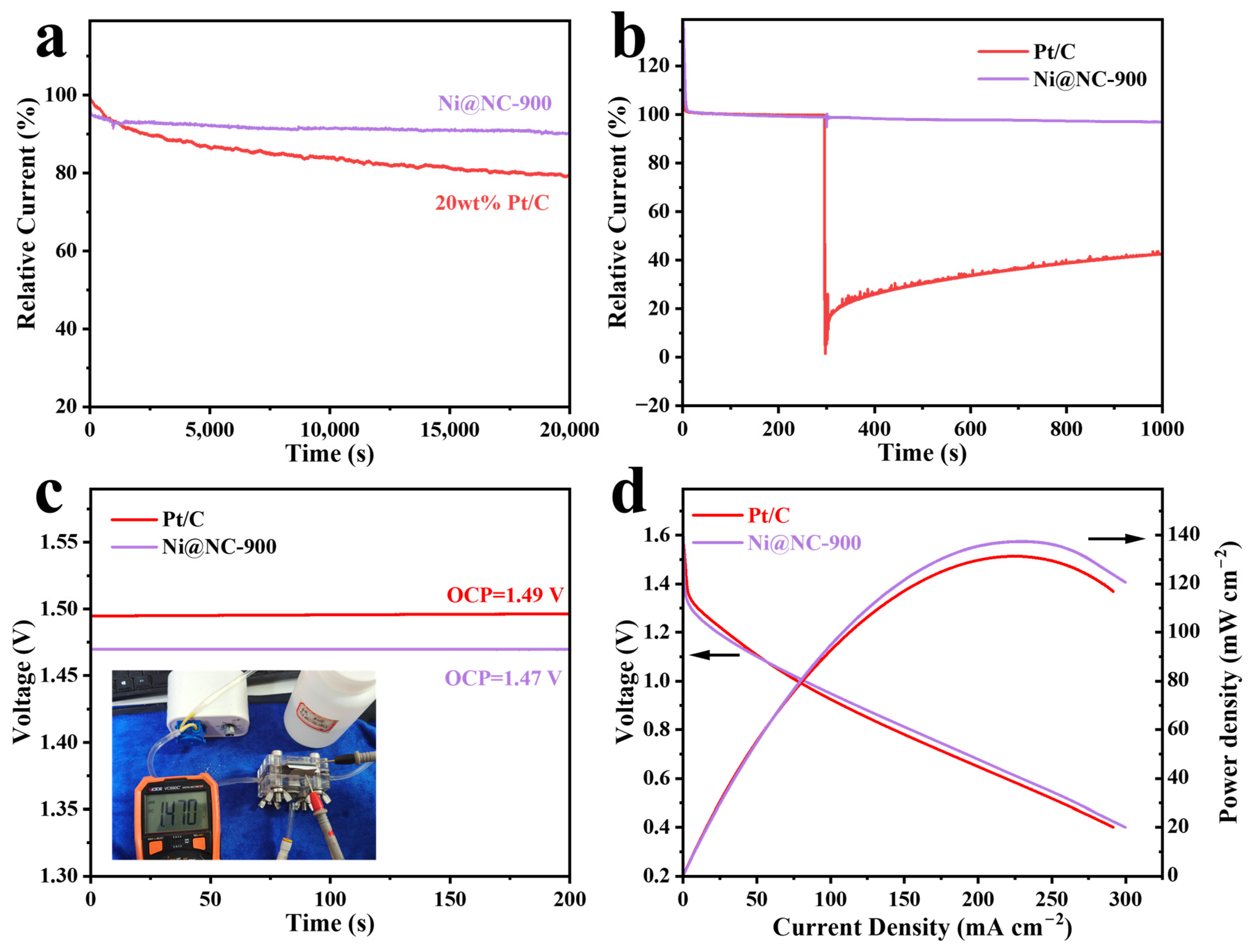
2.3. Electrochemical ORR/OER Performance and ZAB Testing
3. Experimental Section
3.1. Preparation of NiHMT
3.2. Preparation of Ni@NC-T
3.3. Materials Characterization
3.4. Electrochemical Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jaouen, F. Enabling Low-Cost and Sustainable Fuel Cells. Nat. Mater. 2022, 21, 733–735. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhao, Z.; Xia, Z.; Dai, L. A Metal-Free Bifunctional Electrocatalyst for Oxygen Reduction and Oxygen Evolution Reactions. Nat. Nanotechnol. 2015, 10, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Rebrov, E.V.; Gao, P.-Z. Molecular Catalysts for OER/ORR in Zn–Air Batteries. Catalysts 2023, 13, 1289. [Google Scholar] [CrossRef]
- Dai, L.; Xue, Y.; Qu, L.; Choi, H.-J.; Baek, J.-B. Metal-Free Catalysts for Oxygen Reduction Reaction. Chem. Rev. 2015, 115, 4823–4892. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, X.; Yang, Z.; Yan, S.; Zhang, C.; Liu, S. Space-Confined Synthesis of Lasagna-like N-Doped Graphene-Wrapped Copper–Cobalt Sulfides as Efficient and Durable Electrocatalysts for Oxygen Reduction and Oxygen Evolution Reactions. ACS Sustain. Chem. Eng. 2020, 8, 1004–1014. [Google Scholar] [CrossRef]
- Ma, Z.; Cano, Z.P.; Yu, A.; Chen, Z.; Jiang, G.; Fu, X.; Yang, L.; Wu, T.; Bai, Z.; Lu, J. Enhancing Oxygen Reduction Activity of Pt-Based Electrocatalysts: From Theoretical Mechanisms to Practical Methods. Angew. Chem. Int. Ed. 2020, 59, 18334–18348. [Google Scholar] [CrossRef]
- Wang, C.; Lan, F.; He, Z.; Xie, X.; Zhao, Y.; Hou, H.; Guo, L.; Murugadoss, V.; Liu, H.; Shao, Q.; et al. Iridium-Based Catalysts for Solid Polymer Electrolyte Electrocatalytic Water Splitting. ChemSusChem 2019, 12, 1576–1590. [Google Scholar] [CrossRef]
- Yu, J.; He, Q.; Yang, G.; Zhou, W.; Shao, Z.; Ni, M. Recent Advances and Prospective in Ruthenium-Based Materials for Electrochemical Water Splitting. ACS Catal. 2019, 9, 9973–10011. [Google Scholar] [CrossRef]
- Tang, L.; Xu, Q.; Zhang, Y.; Chen, W.; Wu, M. MOF/PCP-Based Electrocatalysts for the Oxygen Reduction Reaction. Electrochem. Energy Rev. 2022, 5, 32–81. [Google Scholar] [CrossRef]
- Zasypkina, A.A.; Ivanova, N.A.; Spasov, D.D.; Mensharapov, R.M.; Sinyakov, M.V.; Grigoriev, S.A. Recent Advances in the Development of Nanocarbon-Based Electrocatalytic/Electrode Materials for Proton Exchange Membrane Fuel Cells: A Review. Catalysts 2024, 14, 303. [Google Scholar] [CrossRef]
- Zhang, W.; Li, J.; Wei, Z. Carbon-Based Catalysts of the Oxygen Reduction Reaction: Mechanistic Understanding and Porous Structures. Chin. J. Catal. 2023, 48, 15–31. [Google Scholar]
- Shi, W.; Lu, S.; Chao, M.; Zheng, X.; Gong, H.; Qian, Y.; Gao, F.; Guo, X.; Liu, Y.; Zhang, J.; et al. A Bottom-up Puzzle Strategy for P/B Co-Doped Carbon Nanosheets as Efficient Oxygen Reaction Electrocatalysts. Appl. Surf. Sci. 2024, 649, 159172. [Google Scholar]
- Lai, L.; Potts, J.R.; Zhan, D.; Wang, L.; Poh, C.K.; Tang, C.; Gong, H.; Shen, Z.; Lin, J.; Ruoff, R.S. Exploration of the Active Center Structure of Nitrogen-Doped Graphene-Based Catalysts for Oxygen Reduction Reaction. Energy Environ. Sci. 2012, 5, 7936–7942. [Google Scholar]
- Jeon, I.-Y.; Zhang, S.; Zhang, L.; Choi, H.-J.; Seo, J.-M.; Xia, Z.; Dai, L.; Baek, J.-B. Edge-Selectively Sulfurized Graphene Nanoplatelets as Efficient Metal-Free Electrocatalysts for Oxygen Reduction Reaction: The Electron Spin Effect. Adv. Mater. 2013, 25, 6138–6145. [Google Scholar]
- Yang, D.-S.; Bhattacharjya, D.; Inamdar, S.; Park, J.; Yu, J.-S. Phosphorus-Doped Ordered Mesoporous Carbons with Different Lengths as Efficient Metal-Free Electrocatalysts for Oxygen Reduction Reaction in Alkaline Media. J. Am. Chem. Soc. 2012, 134, 16127–16130. [Google Scholar]
- Qiao, X.; Deng, Y.; Cao, X.; Wu, J.; Guo, H.; Xiao, W.; Liao, S. Iron Carbide Nanoparticles Embedded in Edge-Rich, N and F Codoped Graphene/Carbon Nanotubes Hybrid for Oxygen Electrocatalysis. Catalysts 2022, 12, 1023. [Google Scholar] [CrossRef]
- Qu, L.; Liu, Y.; Baek, J.-B.; Dai, L. Nitrogen-Doped Graphene as Efficient Metal-Free Electrocatalyst for Oxygen Reduction in Fuel Cells. ACS Nano 2010, 4, 1321–1326. [Google Scholar]
- Gong, K.; Du, F.; Xia, Z.; Durstock, M.; Dai, L. Nitrogen-Doped Carbon Nanotube Arrays with High Electrocatalytic Activity for Oxygen Reduction. Science 2009, 323, 760–764. [Google Scholar]
- Wu, G.; Santandreu, A.; Kellogg, W.; Gupta, S.; Ogoke, O.; Zhang, H.; Wang, H.-L.; Dai, L. Carbon Nanocomposite Catalysts for Oxygen Reduction and Evolution Reactions: From Nitrogen Doping to Transition-Metal Addition. Nano Energy 2016, 29, 83–110. [Google Scholar]
- Lu, X.; Yang, P.; Wan, Y.; Zhang, H.; Xu, H.; Xiao, L.; Li, R.; Li, Y.; Zhang, J.; An, M. Active Site Engineering toward Atomically Dispersed M−N−C Catalysts for Oxygen Reduction Reaction. Coord. Chem. Rev. 2023, 495, 215400. [Google Scholar]
- Wang, D.; Yang, P.; Xu, H.; Ma, J.; Du, L.; Zhang, G.; Li, R.; Jiang, Z.; Li, Y.; Zhang, J.; et al. The Dual-Nitrogen-Source Strategy to Modulate a Bifunctional Hybrid Co/Co–N–C Catalyst in the Reversible Air Cathode for Zn-Air Batteries. J. Power Sources 2021, 485, 229339. [Google Scholar] [CrossRef]
- Zhang, L.; Qi, C.; Zhao, A.; Xu, G.; Xu, J.; Zhang, L.; Zhang, C.; Jia, D. N-Doped Porous Carbon-Encapsulated Fe Nanoparticles as Efficient Electrocatalysts for Oxygen Reduction Reaction. Appl. Surf. Sci. 2018, 445, 462–470. [Google Scholar] [CrossRef]
- Luo, Z.; Zhong, G.; Meng, Z.; Fu, X.; Liao, W.; Zheng, S.; Xu, Y.; Luo, S. MOF-Derived Two-Dimensional FeNx Catalysts Based on Hexamethylenetetramine for Efficient Oxygen Reduction Reactions. Colloids Surf. Physicochem. Eng. Asp. 2023, 671, 131631. [Google Scholar] [CrossRef]
- Oliveira, F.E.R.; Galiote, N.A.; Lima, F.H.B. Investigation of Earth-Abundant Oxygen Reduction Electrocatalysts for the Cathode of Passive Air-Breathing Direct Formate Fuel Cells. Catalysts 2018, 8, 320. [Google Scholar] [CrossRef]
- Kumar, K.; Dubau, L.; Jaouen, F.; Maillard, F. Review on the Degradation Mechanisms of Metal-N-C Catalysts for the Oxygen Reduction Reaction in Acid Electrolyte: Current Understanding and Mitigation Approaches. Chem. Rev. 2023, 123, 9265–9326. [Google Scholar] [CrossRef]
- Tang, T.; Ding, L.; Jiang, Z.; Hu, J.-S.; Wan, L.-J. Advanced Transition Metal/Nitrogen/Carbon-Based Electrocatalysts for Fuel Cell Applications. Sci. China Chem. 2020, 63, 1517–1542. [Google Scholar] [CrossRef]
- Niu, W.-J.; Sun, Q.-Q.; Wang, Y.-P.; Gu, B.-N.; Liu, M.-J.; He, J.-Z.; Chen, J.-L.; Chung, C.-C.; Liu, W.-W.; Chueh, Y.-L. Design of Co Nanoparticles-Encapsulated by Boron and Nitrogen Co-Doped Carbon Nanosheets as Highly Efficient Electrocatalyst for Oxygen Reduction Reaction. Adv. Mater. Interfaces 2021, 8, 2101454. [Google Scholar] [CrossRef]
- Jang, J.-H.; Anto Jeffery, A.; Min, J.; Jung, N.; Jong Yoo, S. Emerging Carbon Shell-Encapsulated Metal Nanocatalysts for Fuel Cells and Water Electrolysis. Nanoscale 2021, 13, 15116–15141. [Google Scholar] [CrossRef]
- Sharma, M.; Jang, J.-H.; Shin, D.Y.; Kwon, J.A.; Lim, D.-H.; Choi, D.; Sung, H.; Jang, J.; Lee, S.-Y.; Lee, K.Y.; et al. Work Function-Tailored Graphene via Transition Metal Encapsulation as a Highly Active and Durable Catalyst for the Oxygen Reduction Reaction. Energy Environ. Sci. 2019, 12, 2200–2211. [Google Scholar] [CrossRef]
- Choi, C.H.; Park, S.H.; Woo, S.I. N-Doped Carbon Prepared by Pyrolysis of Dicyandiamide with Various MeCl2·xH2O (Me = Co, Fe, and Ni) Composites: Effect of Type and Amount of Metal Seed on Oxygen Reduction Reactions. Appl. Catal. B Environ. 2012, 119–120, 123–131. [Google Scholar] [CrossRef]
- Ahuja, I.S.; Singh, R.; Yadava, C.L. Structural Information on Cobalt(II), Nickel(II), Copper(II), Zinc(II), Silver(I) and Cadmium(II) Nitrate Complexes with Hexamethylenetetramine from Their Magnetic Moments, Electronic and Infrared Spectra. J. Mol. Struct. 1980, 68, 333–339. [Google Scholar]
- Liu, S.; Zhou, J.; Song, H. Tailoring Highly N-Doped Carbon Materials from Hexamine-Based MOFs: Superior Performance and New Insight into the Roles of N Configurations in Na-Ion Storage. Small 2018, 14, 1703548. [Google Scholar] [CrossRef] [PubMed]
- Zhong, G.; Zou, L.; Chi, X.; Meng, Z.; Chen, Z.; Li, T.; Huang, Y.; Fu, X.; Liao, W.; Zheng, S.; et al. Atomically Dispersed Mn–N Catalysts Derived from Mn-Hexamine Coordination Frameworks for Oxygen Reduction Reaction. Carbon Energy 2024, 6, e484. [Google Scholar]
- Kong, W.; Lu, C.; Zhang, W.; Pu, J.; Wang, Z. Homogeneous Core–Shell NiCo2S4 Nanostructures Supported on Nickel Foam for Supercapacitors. J. Mater. Chem. A 2015, 3, 12452–12460. [Google Scholar]
- Zhang, Z.; Sun, J.; Wang, F.; Dai, L. Efficient Oxygen Reduction Reaction (ORR) Catalysts Based on Single Iron Atoms Dispersed on a Hierarchically Structured Porous Carbon Framework. Angew. Chem. 2018, 130, 9176–9181. [Google Scholar] [CrossRef]
- Wu, X.; Yu, X.; Lin, Z.; Huang, J.; Cao, L.; Zhang, B.; Zhan, Y.; Meng, H.; Zhu, Y.; Zhang, Y. Nitrogen Doped Graphitic Carbon Ribbons from Cellulose as Non Noble Metal Catalyst for Oxygen Reduction Reaction. Int. J. Hydrogen Energy 2016, 41, 14111–14122. [Google Scholar] [CrossRef]
- Xing, T.; Li, L.H.; Hou, L.; Hu, X.; Zhou, S.; Peter, R.; Petravic, M.; Chen, Y. Disorder in Ball-Milled Graphite Revealed by Raman Spectroscopy. Carbon 2013, 57, 515–519. [Google Scholar]
- Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S.; Cançado, L.G.; Jorio, A.; Saito, R. Studying Disorder in Graphite-Based Systems by Raman Spectroscopy. Phys. Chem. Chem. Phys. 2007, 9, 1276–1290. [Google Scholar] [CrossRef]
- Zhong, H.; Luo, Y.; He, S.; Tang, P.; Li, D.; Alonso-Vante, N.; Feng, Y. Electrocatalytic Cobalt Nanoparticles Interacting with Nitrogen-Doped Carbon Nanotube in Situ Generated from a Metal–Organic Framework for the Oxygen Reduction Reaction. ACS Appl. Mater. Interfaces 2017, 9, 2541–2549. [Google Scholar]
- Wang, X.; Liu, T.; Li, H.; Han, C.; Su, P.; Ta, N.; Jiang, S.P.; Kong, B.; Liu, J.; Huang, Z. Balancing Mass Transfer and Active Sites to Improve Electrocatalytic Oxygen Reduction by B,N Codoped C Nanoreactors. Nano Lett. 2023, 23, 4699–4707. [Google Scholar]
- Fang, B.; Daniel, L.; Bonakdarpour, A.; Govindarajan, R.; Sharman, J.; Wilkinson, D.P. Dense Pt Nanowire Electrocatalyst for Improved Fuel Cell Performance Using a Graphitic Carbon Nitride-Decorated Hierarchical Nanocarbon Support. Small 2021, 17, 2102288. [Google Scholar]
- Sharifi, T.; Hu, G.; Jia, X.; Wågberg, T. Formation of Active Sites for Oxygen Reduction Reactions by Transformation of Nitrogen Functionalities in Nitrogen-Doped Carbon Nanotubes. ACS Nano 2012, 6, 8904–8912. [Google Scholar] [PubMed]
- Zhou, Q.; Yang, Y.; Ye, Q.; Xue, T.; Tu, M.; Liu, Y.; Li, H.; Yan, X.-H.; Zou, Z.-L.; Wang, B.-P.; et al. Graphitic-Nitrogen-Enriched Carbon Skeleton with Embedment of Fe3C for Superior Performance Air Cathode in Zinc-Air Battery. Mater. Today Energy 2023, 31, 101194. [Google Scholar]
- Wu, J.; Ma, L.; Yadav, R.M.; Yang, Y.; Zhang, X.; Vajtai, R.; Lou, J.; Ajayan, P.M. Nitrogen-Doped Graphene with Pyridinic Dominance as a Highly Active and Stable Electrocatalyst for Oxygen Reduction. ACS Appl. Mater. Interfaces 2015, 7, 14763–14769. [Google Scholar]
- Shen, S.Y.; Zhao, T.S.; Xu, J.B.; Li, Y.S. Synthesis of PdNi Catalysts for the Oxidation of Ethanol in Alkaline Direct Ethanol Fuel Cells. J. Power Sources 2010, 195, 1001–1006. [Google Scholar]
- Song, C.; Zhang, J. Electrocatalytic Oxygen Reduction Reaction. In PEM Fuel Cell Electrocatalysts and Catalyst Layers: Fundamentals and Applications; Zhang, J., Ed.; Springer: London, UK, 2008; pp. 89–134. [Google Scholar]
- Wu, Y.; Muthukrishnan, A.; Nagata, S.; Nabae, Y. Kinetic Understanding of the Reduction of Oxygen to Hydrogen Peroxide over an N-Doped Carbon Electrocatalyst. J. Phys. Chem. C 2019, 123, 4590–4596. [Google Scholar]
- Kirillov, A.M. Hexamethylenetetramine: An old new building block for design of coordination polymers. Coord. Chem. Rev. 2011, 255, 1603–1622. [Google Scholar]
- Reyes-Rodríguez, J.L.; Sathish-Kumar, K.; Solorza-Feria, O. Synthesis and Functionalization of Green Carbon as a Pt Catalyst Support for the Oxygen Reduction Reaction. Int. J. Hydrogen Energy 2015, 40, 17253–17263. [Google Scholar]
- Francioso, O.; Sanchez-Cortes, S.; Bonora, S.; Roldán, M.L.; Certini, G. Structural Characterization of Charcoal Size-Fractions from a Burnt Pinus Pinea Forest by FT-IR, Raman and Surface-Enhanced Raman Spectroscopies. J. Mol. Struct. 2011, 994, 155–162. [Google Scholar]
- Wang, R.; Jia, J.; Li, H.; Li, X.; Wang, H.; Chang, Y.; Kang, J.; Lei, Z. Nitrogen-Doped Carbon Coated Palygorskite as an Efficient Electrocatalyst Support for Oxygen Reduction Reaction. Electrochim. Acta 2011, 56, 4526–4531. [Google Scholar]
- Li, T.; Li, M.; Zhang, M.; Li, X.; Liu, K.; Zhang, M.; Liu, X.; Sun, D.; Xu, L.; Zhang, Y.; et al. Immobilization of Fe3N Nanoparticles within N-Doped Carbon Nanosheet Frameworks as a High-Efficiency Electrocatalyst for Oxygen Reduction Reaction in Zn-Air Batteries. Carbon 2019, 153, 364–371. [Google Scholar]
- Yang, T.; Chen, Y.; Liu, Y.; Liu, X.; Gao, S. Self-Sacrificial Template Synthesis of Fe, N Co-Doped Porous Carbon as Efficient Oxygen Reduction Electrocatalysts towards Zn-Air Battery Application. Chin. Chem. Lett. 2022, 33, 2171–2177. [Google Scholar]
- Deng, Y.; Tian, X.; Chi, B.; Wang, Q.; Ni, W.; Gao, Y.; Liu, Z.; Luo, J.; Lin, C.; Ling, L.; et al. Hierarchically Open-Porous Carbon Networks Enriched with Exclusive Fe–Nx Active Sites as Efficient Oxygen Reduction Catalysts towards Acidic H2–O2 PEM Fuel Cell and Alkaline Zn–Air Battery. Chem. Eng. J. 2020, 390, 124479. [Google Scholar]
- Liu, S.; Wang, Z.; Zhou, S.; Yu, F.; Yu, M.; Chiang, C.-Y.; Zhou, W.; Zhao, J.; Qiu, J. Metal–Organic-Framework-Derived Hybrid Carbon Nanocages as a Bifunctional Electrocatalyst for Oxygen Reduction and Evolution. Adv. Mater. 2017, 29, 1700874. [Google Scholar]
- Wang, Q.; Lei, Y.; Chen, Z.; Wu, N.; Wang, Y.; Wang, B.; Wang, Y. Fe/Fe3 C@C Nanoparticles Encapsulated in N-Doped Graphene–CNTs Framework as an Efficient Bifunctional Oxygen Electrocatalyst for Robust Rechargeable Zn–Air Batteries. J. Mater. Chem. A 2018, 6, 516–526. [Google Scholar]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, H.; Huang, J.; Zhong, G.; Xu, S.; Chen, H.; Fu, X.; Kang, S.; Tu, J.; Tuo, Y.; Liao, W.; et al. Carbon-Encapsulated Ni Nanoparticles Catalysts Derived from Ni-Hexamine Coordination Frameworks for Oxygen Reduction Reaction and Oxygen Evolution Reaction. Catalysts 2025, 15, 338. https://doi.org/10.3390/catal15040338
Huang H, Huang J, Zhong G, Xu S, Chen H, Fu X, Kang S, Tu J, Tuo Y, Liao W, et al. Carbon-Encapsulated Ni Nanoparticles Catalysts Derived from Ni-Hexamine Coordination Frameworks for Oxygen Reduction Reaction and Oxygen Evolution Reaction. Catalysts. 2025; 15(4):338. https://doi.org/10.3390/catal15040338
Chicago/Turabian StyleHuang, Huoxing, Jiaxing Huang, Guoyu Zhong, Shurui Xu, Hongwei Chen, Xiaobo Fu, Shimin Kang, Junling Tu, Yongxiao Tuo, Wenbo Liao, and et al. 2025. "Carbon-Encapsulated Ni Nanoparticles Catalysts Derived from Ni-Hexamine Coordination Frameworks for Oxygen Reduction Reaction and Oxygen Evolution Reaction" Catalysts 15, no. 4: 338. https://doi.org/10.3390/catal15040338
APA StyleHuang, H., Huang, J., Zhong, G., Xu, S., Chen, H., Fu, X., Kang, S., Tu, J., Tuo, Y., Liao, W., & Fang, B. (2025). Carbon-Encapsulated Ni Nanoparticles Catalysts Derived from Ni-Hexamine Coordination Frameworks for Oxygen Reduction Reaction and Oxygen Evolution Reaction. Catalysts, 15(4), 338. https://doi.org/10.3390/catal15040338







