Pure and Fe-Doped Mesoporous Titania Catalyse the Oxidation of Acid Orange 7 by H2O2 under Different Illumination Conditions: Fe Doping Improves Photocatalytic Activity under Simulated Solar Light
Abstract
:1. Introduction
2. Results
2.1. Relevant Physico-Chemical Properties of the Prepared Samples
2.2. AO7 Degradation Tests: Preliminary Considerations
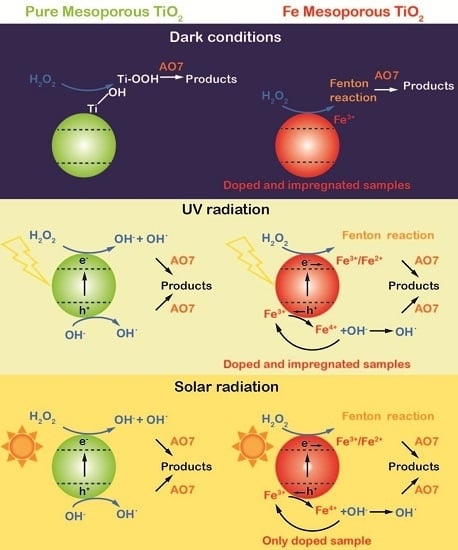
2.3. AO7 Degradation Tests: Catalytic Behavior of the Samples in Dark Conditions
2.4. AO7 Degradation Tests under UV Irradiation
2.5. AO7 Degradation Tests under Simulated Solar Light (1 SUN)
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.3. Catalytic Tests
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ge, M.; Cao, C.; Huang, J.; Li, S.; Chen, Z.; Zhang, K.-Q.; Al-Deyab, S.S.; Lai, Y. A review of one-dimensional TiO2 nanostructured materials for environmental and energy applications. J. Mater. Chem. A 2016, 4, 6772–6801. [Google Scholar] [CrossRef]
- Liao, J.-Y.; He, J.-W.; Xu, H.; Kuang, D.-B.; Su, C.-Y. Effect of TiO2 morphology on photovoltaic performance of dye-sensitized solar cells: Nanoparticles, nanofibers, hierarchical spheres and ellipsoid spheres. J. Mater. Chem. 2012, 22, 7910–7918. [Google Scholar] [CrossRef]
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: Design and applications. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Zhang, R.; Elzatahry, A.A.; Al-Deyab, S.S.; Zhao, D. Mesoporous titania: From synthesis to application. Nano Today 2012, 7, 344–366. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, N.; Schmuki, P. Photocatalysis with TiO2 Nanotubes: “Colorful” Reactivity and Designing Site-Specific Photocatalytic Centers into TiO2 Nanotubes. ACS Catal. 2017, 7, 3210–3235. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 Photocatalysis: Mechanisms and Materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, X.; Jia, Y.; Chen, X.; Han, H.; Li, C. Titanium Dioxide-Based Nanomaterials for Photocatalytic Fuel Generations. Chem. Rev. 2014, 114, 9987–10043. [Google Scholar] [CrossRef] [PubMed]
- Sauvage, F.; Chen, D.; Comte, P.; Huang, F.; Heiniger, L.-P.; Cheng, Y.-B.; Caruso, R.A.; Graetzel, M. Dye-Sensitized Solar Cells Employing a Single Film of Mesoporous TiO2 Beads Achieve Power Conversion Efficiencies Over 10%. ACS Nano 2010, 4, 4420–4425. [Google Scholar] [CrossRef] [PubMed]
- O’Regan, B.; Gratzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Roose, B.; Pathak, S.; Steiner, U. Doping of TiO2 for sensitized solar cells. Chem. Soc. Rev. 2015, 44, 8326–8349. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Qi, D.; Ding, H.; Wang, L.; Xing, M.; Shen, B.; Zhang, J. Carbon-doped titanum dioxide nanocrystals for highly efficient dye-sensitized solar cells. Catal. Today 2017, 281, 636–641. [Google Scholar] [CrossRef]
- Rajh, T.; Dimitrijevic, N.M.; Bissonnette, M.; Koritarov, T.; Konda, V. Titanium Dioxide in the Service of the Biomedical Revolution. Chem. Rev. 2014, 114, 10177–10216. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Weng, Z.; Liu, X.; Yeung, K.W.K.; Chu, P.K. Functionalized TiO2 Based Nanomaterials for Biomedical Applications. Adv. Funct. Mater. 2014, 24, 5464–5481. [Google Scholar] [CrossRef]
- Bonelli, B.; Esposito, S.; Freyria, F.S. Mesoporous Titania: Synthesis, Properties and Comparison with Non-Porous Titania. In Titanium Dioxide; Janus, M., Ed.; Intech: Rijeka, Croatia, 2017. [Google Scholar]
- Su, J.; Guo, L.; Bao, N.; Grimes, C.A. Nanostructured WO3/BiVO4 Heterojunction Films for Efficient Photoelectrochemical Water Splitting. Nano Lett. 2011, 11, 1928–1933. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Weng, Y.; Zhou, X.; Wang, X.; Mi, Y.; Chong, R.; Han, H.; Li, C. Achieving overall water splitting using titanium dioxide-based photocatalysts of different phases. Energy Environ. Sci. 2015, 8, 2377–2382. [Google Scholar] [CrossRef]
- Kou, J.; Lu, C.; Wang, J.; Chen, Y.; Xu, Z.; Varma, R.S. Selectivity Enhancement in Heterogeneous Photocatalytic Transformations. Chem. Rev. 2017, 117, 1445–1514. [Google Scholar] [CrossRef] [PubMed]
- Lianos, P. Review of recent trends in photoelectrocatalytic conversion of solar energy to electricity and hydrogen. Appl. Catal. B Environ. 2017, 210, 235–254. [Google Scholar] [CrossRef]
- Freyria, F.S.; Armandi, M.; Compagnoni, M.; Ramis, G.; Rossetti, I.; Bonelli, B. Catalytic and Photocatalytic Processes for the Abatement of N-Containing Pollutants From Wastewater. Part 2: Organic Pollutants. J. Nanosci. Nanotechnol. 2017, 17, 3654–3672. [Google Scholar] [CrossRef]
- Piumetti, M.; Freyria, F.S.; Armandi, M.; Saracco, G.; Garrone, E.; Gonzalez, G.E.; Bonelli, B. Catalytic degradation of Acid Orange 7 by H2O2 as promoted by either bare or V-loaded titania under UV light, in dark conditions, and after incubating the catalysts in ascorbic acid. Catal. Struct. React. 2015, 1, 183–191. [Google Scholar] [CrossRef]
- Khataee, A.R.; Kasiri, M.B. Photocatalytic degradation of organic dyes in the presence of nanostructured titanium dioxide: Influence of the chemical structure of dyes. J. Mol. Catal. A Chem. 2010, 328, 8–26. [Google Scholar] [CrossRef]
- Rauf, M.A.; Meetani, M.A.; Hisaindee, S. An overview on the photocatalytic degradation of azo dyes in the presence of TiO2 doped with selective transition metals. Desalination 2011, 276, 13–27. [Google Scholar] [CrossRef]
- Piumetti, M.; Freyria, F.S.; Armandi, M.; Geobaldo, F.; Garrone, E.; Bonelli, B. Fe- and V-doped mesoporous titania prepared by direct synthesis: Characterization and role in the oxidation of AO7 by H2O2 in the dark. Catal. Today 2014, 227, 71–79. [Google Scholar] [CrossRef]
- Piumetti, M.; Freyria, F.S.; Armandi, M.; Saracco, G.; Garrone, E.; Bonelli, B. Anti-oxidant/pro-oxidant activity of ascorbic acid. Chem. Today 2015, 33, 13–15. [Google Scholar]
- Freyria, F.S.; Bonelli, B.; Sethi, R.; Armandi, M.; Belluso, E.; Garrone, E. Reactions of Acid Orange 7 with Iron Nanoparticles in Aqueous Solutions. J. Phys. Chem. C 2011, 115, 24143–24152. [Google Scholar] [CrossRef]
- Momeni, S.; Nematollahi, D. New insights into the electrochemical behavior of acid orange 7: Convergent paired electrochemical synthesis of new aminonaphthol derivatives. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Shafia, E.; Esposito, S.; Armandi, M.; Bahadori, E.; Garrone, E.; Bonelli, B. Reactivity of bare and Fe-doped alumino-silicate nanotubes (imogolite) with H2O2 and the azo-dye Acid Orange 7. Catal. Today 2016, 277, 89–96. [Google Scholar] [CrossRef]
- Sun, D.; Zhang, X.; Wu, Y.; Liu, X. Adsorption of anionic dyes from aqueous solution on fly ash. J. Hazard. Mater. 2010, 181, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Moon, B.H.; Park, Y.B.; Park, K.H. Fenton oxidation of Orange II by pre-reduction using nanoscale zero-valent iron. Desalination 2011, 268, 249–252. [Google Scholar] [CrossRef]
- Ramirez, J.H.; Maldonado-Hódar, F.J.; Pérez-Cadenas, A.F.; Moreno-Castilla, C.; Costa, C.A.; Madeira, L.M. Azo-dye Orange II degradation by heterogeneous Fenton-like reaction using carbon-Fe catalysts. Appl. Catal. B Environ. 2007, 75, 312–323. [Google Scholar] [CrossRef]
- Di Paola, A.; Marcì, G.; Palmisano, L.; Schiavello, M.; Uosaki, K.; Ikeda, S.; Ohtani, B. Preparation of Polycrystalline TiO2 Photocatalysts Impregnated with Various Transition Metal Ions: Characterization and Photocatalytic Activity for the Degradation of 4-Nitrophenol. J. Phys. Chem. B 2002, 106, 637–645. [Google Scholar] [CrossRef]
- Pesci, F.M.; Wang, G.; Klug, D.R.; Li, Y.; Cowan, A.J. Efficient Suppression of Electron–Hole Recombination in Oxygen-Deficient Hydrogen-Treated TiO2 Nanowires for Photoelectrochemical Water Splitting. J. Phys. Chem. C 2013, 117, 25837–25844. [Google Scholar] [CrossRef] [PubMed]
- Sachs, M.; Pastor, E.; Kafizas, A.; Durrant, J.R. Evaluation of Surface State Mediated Charge Recombination in Anatase and Rutile TiO2. J. Phys. Chem. Lett. 2016, 7, 3742–3746. [Google Scholar] [CrossRef] [PubMed]
- Guayaquil-Sosa, J.F.; Serrano-Rosales, B.; Valadés-Pelayo, P.J.; de Lasa, H. Photocatalytic hydrogen production using mesoporous TiO2 doped with Pt. Appl. Catal. B Environ. 2017, 211, 337–348. [Google Scholar] [CrossRef]
- Ren, F.; Li, H.; Wang, Y.; Yang, J. Enhanced photocatalytic oxidation of propylene over V-doped TiO2 photocatalyst: Reaction mechanism between V5+ and single-electron-trapped oxygen vacancy. Appl. Catal. B Environ. 2015, 176–177, 160–172. [Google Scholar] [CrossRef]
- Martínez Vargas, D.X.; Rivera De la Rosa, J.; Lucio-Ortiz, C.J.; Hernández-Ramirez, A.; Flores-Escamilla, G.A.; Garcia, C.D. Photocatalytic degradation of trichloroethylene in a continuous annular reactor using Cu-doped TiO2 catalysts by sol–gel synthesis. Appl. Catal. B Environ. 2015, 179, 249–261. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Irie, H.; Ohwaki, T. Nitrogen-Doped Titanium Dioxide as Visible-Light-Sensitive Photocatalyst: Designs, Developments, and Prospects. Chem. Rev. 2014, 114, 9824–9852. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.; Xiong, F.; Ganeshraja, A.S.; Feng, X.; Wang, C.; Thomas, T.; Yang, M. Visible light photocatalysts (Fe, N):TiO2 from ammonothermally processed, solvothermal self-assembly derived Fe-TiO2 mesoporous microspheres. Mater. Chem. Phys. 2017, 195, 259–267. [Google Scholar] [CrossRef]
- Gole, J.L.; Prokes, S.M.; Glembocki, O.J.; Wang, J.; Qiu, X.; Burda, C. Study of concentration-dependent cobalt ion doping of TiO2 and TiO2-xNx at the nanoscale. Nanoscale 2010, 2, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Xiang, Q.; Zhou, M. Preparation, characterization and visible-light-driven photocatalytic activity of Fe-doped titania nanorods and first-principles study for electronic structures. Appl. Catal. B Environ. 2009, 90, 595–602. [Google Scholar] [CrossRef]
- Tong, T.; Zhang, J.; Tian, B.; Chen, F.; He, D. Preparation of Fe3+-doped TiO2 catalysts by controlled hydrolysis of titanium alkoxide and study on their photocatalytic activity for methyl orange degradation. J. Hazard. Mater. 2008, 155, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Yu, J.; Cheng, B.; Yu, H. Preparation and photocatalytic activity of Fe-doped mesoporous titanium dioxide nanocrystalline photocatalysts. Mater. Chem. Phys. 2005, 93, 159–163. [Google Scholar] [CrossRef]
- Ohno, T.; Sarukawa, K.; Tokieda, K.; Matsumura, M. Morphology of a TiO2 Photocatalyst (Degussa, P25) Consisting of Anatase and Rutile Crystalline Phases. J. Catal. 2001, 203, 82–86. [Google Scholar] [CrossRef]
- Pignatello, J.J.; Oliveros, E.; MacKay, A. Advanced Oxidation Processes for Organic Contaminant Destruction Based on the Fenton Reaction and Related Chemistry. Crit. Rev. Environ. Sci. Technol. 2006, 36, 1–84. [Google Scholar] [CrossRef]
- Schwenzer, B.; Wang, L.; Swensen, J.S.; Padmaperuma, A.B.; Silverman, G.; Korotkov, R.; Gaspar, D.J. Tuning the Optical Properties of Mesoporous TiO2 Films by Nanoscale Engineering. Langmuir 2012, 28, 10072–10081. [Google Scholar] [CrossRef] [PubMed]
- Grosvenor, A.P.; Kobe, B.A.; Biesinger, M.C.; McIntyre, N.S. Investigation of multiplet splitting of Fe 2p XPS spectra and bonding in iron compounds. Surf. Interface Anal. 2004, 36, 1564–1574. [Google Scholar] [CrossRef]
- Fan, J.-X.; Wang, Y.-J.; Fan, T.-T.; Dang, F.; Zhou, D.-M. Effect of aqueous Fe(II) on Sb(V) sorption on soil and goethite. Chemosphere 2016, 147, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Hayes, P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
- Mielczarski, J.A.; Atenas, G.M.; Mielczarski, E. Role of iron surface oxidation layers in decomposition of azo-dye water pollutants in weak acidic solutions. Appl. Catal. B Environ. 2005, 56, 289–303. [Google Scholar] [CrossRef]
- Orozco, S.L.; Bandala, E.R.; Arancibia-Bulnes, C.A.; Serrano, B.; Suárez-Parra, R.; Hernández-Pérez, I. Effect of iron salt on the color removal of water containing the azo-dye reactive blue 69 using photo-assisted Fe(II)/H2O2 and Fe(III)/H2O2 systems. J. Photochem. Photobiol. A Chem. 2008, 198, 144–149. [Google Scholar] [CrossRef]
- Nie, Y.; Hu, C.; Qu, J.; Zhou, L.; Hu, X. Photoassisted Degradation of Azodyes over FeOxH2x-3/Fe° in the Presence of H2O2 at Neutral pH Values. Environ. Sci. Technol. 2007, 41, 4715–4719. [Google Scholar] [CrossRef] [PubMed]
- Gutowska, A.; Kałuzna-Czaplińska, J.; Jóźwiak, W.K. Degradation mechanism of Reactive Orange 113 dye by H2O2/Fe2+ and ozone in aqueous solution. Dyes Pigment. 2006, 74, 41–46. [Google Scholar] [CrossRef]
- Lin, W.; Frei, H. Photochemical and FT-IR Probing of the Active Site of Hydrogen Peroxide in Ti Silicalite Sieve. J. Am. Chem. Soc. 2002, 124, 9292–9298. [Google Scholar] [CrossRef] [PubMed]
- Fenton, H.J.H. LXXIII.-Oxidation of tartaric acid in presence of iron. J. Chem. Soc. Trans. 1894, 65, 899–910. [Google Scholar] [CrossRef]
- Bandara, J.; Morrison, C.; Kiwi, J.; Pulgarin, C.; Peringer, P. Degradation/decoloration of concentrated solutions of Orange II. Kinetics and quantum yield for sunlight induced reactions via Fenton type reagents. J. Photochem. Photobiol. A Chem. 1996, 99, 57–66. [Google Scholar] [CrossRef]
- Liang, X.; Zhong, Y.; Zhu, S.; Zhu, J.; Yuan, P.; He, H.; Zhang, J. The decolorization of Acid Orange II in non-homogeneous Fenton reaction catalyzed by natural vanadium–titanium magnetite. J. Hazard. Mater. 2010, 181, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Neyens, E.; Baeyens, J. A review of classic Fenton’s peroxidation as an advanced oxidation technique. J. Hazard. Mater. 2003, 98, 33–50. [Google Scholar] [CrossRef]
- Lopez, T.; Moreno, J.A.; Gomez, R.; Bokhimi, X.; Wang, J.A.; Yee-Madeira, H.; Pecchi, G.; Reyes, P. Characterization of iron-doped titania sol-gel materials. J. Mater. Chem. 2002, 12, 714–718. [Google Scholar] [CrossRef]
- Fallet, M.; Gschwind, R.; Bauer, P. Oxidation States of Iron in Doped TiO2-SiO2 Sol-Gel Powders: A 57Fe Mössbauer Study. J. Sol-Gel Sci. Technol. 2003, 27, 167–173. [Google Scholar] [CrossRef]
- Shafia, E.; Esposito, S.; Manzoli, M.; Chiesa, M.; Tiberto, P.; Barrera, G.; Menard, G.; Allia, P.; Freyria, F.S.; Garrone, E.; Bonelli, B. Al/Fe isomorphic substitution versus Fe2O3 clusters formation in Fe-doped aluminosilicate nanotubes (imogolite). J. Nanoparticle Res. 2015, 17, 336. [Google Scholar] [CrossRef]
- Kho, Y.K.; Iwase, A.; Teoh, W.Y.; Mädler, L.; Kudo, A.; Amal, R. Photocatalytic H2 Evolution over TiO2 Nanoparticles. The Synergistic Effect of Anatase and Rutile. J. Phys. Chem. C 2010, 114, 2821–2829. [Google Scholar] [CrossRef]
- Segneanu, A.E.; Orbeci, C.; Lazau, C.; Sfirloaga, P.; Vlazan, P.; Bandas, C.; Grozescu, I. Waste Water Treatment Methods. In Water Treatment; Elshorbagy, W., Chowdhury, R.K., Eds.; InTech: Rijeka, Croatia, 2013. [Google Scholar]
- Feng, J.; Wong, R.S.K.; Hu, X.; Yue, P.L. Discoloration and mineralization of Orange II by using Fe3+-doped TiO2 and bentonite clay-based Fe nanocatalysts. Catal. Today 2004, 98, 441–446. [Google Scholar] [CrossRef]
- Mangayayam, M.; Kiwi, J.; Giannakis, S.; Pulgarin, C.; Zivkovic, I.; Magrez, A.; Rtimi, S. FeOx magnetization enhancing E. coli inactivation by orders of magnitude on Ag-TiO2 nanotubes under sunlight. Appl. Catal. B Environ. 2017, 202, 438–445. [Google Scholar] [CrossRef]








| Sample | Preparation | Crystallites Size (nm) | SSA (m2·g−1) a | Metal Content (wt.%) b | Surface Metal Content (wt.%) c | Metal Density (Atoms nm−2) d |
|---|---|---|---|---|---|---|
| MT | Soft-template synthesis | 14 ± 3 (Anatase) | 150 | - | - | - |
| P25 | Commercial | 19 ± 3 (Anatase) 23 ± 4 (Rutile) | 56 | - | - | - |
| Fe2.5-MTd | Direct Synthesis | 11 ± 3 (Anatase) | 147 | 2.5 | 1.33 ± 0.20 | 1.8 (0.97 c) |
| IFe0.8-IT | Impregnation of P25 | 21 ± 4 (Anatase) 28 ± 2 (Rutile) | 56 | 0.80 | 1.10 ± 0.20 | 1.5 (2.1 c) |
| Fe2.5-MTi | Impregnation of MT | 11 ± 1 (Anatase) | 106 | 2.6 | 2.30 ± 0.20 | 2.7 (2.4 c) |
| Fe2.5-IT | Impregnation of P25 | - | 50 | 2.8 | - | 6.0 |
| Sample | Initial Rate with 0.030 M H2O2 (M·s−1)∙10−8 | Final Conversion with 0.030 M H2O2 (%) | Initial Rate with 0.80 M H2O2 (M·s−1)∙10−8 | Final Conversion with 0.80 M H2O2 (%) |
|---|---|---|---|---|
| MT | 3.1 | 9 | 5.8 | 66 |
| P25 | 2.7 | 5.2 | 1.0 | 25 |
| Fe2.5-MTd | 3.1 | 27 | 4.2 | 90 |
| Fe0.8-IT | 2.3 | 9.6 | 2.4 | 98 |
| Fe2.5-MTi | 5.6 | 31 | 17 | 100 |
| Fe2.5-IT | 2.1 | 7.8 | 1.0 | 79 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freyria, F.S.; Compagnoni, M.; Ditaranto, N.; Rossetti, I.; Piumetti, M.; Ramis, G.; Bonelli, B. Pure and Fe-Doped Mesoporous Titania Catalyse the Oxidation of Acid Orange 7 by H2O2 under Different Illumination Conditions: Fe Doping Improves Photocatalytic Activity under Simulated Solar Light. Catalysts 2017, 7, 213. https://doi.org/10.3390/catal7070213
Freyria FS, Compagnoni M, Ditaranto N, Rossetti I, Piumetti M, Ramis G, Bonelli B. Pure and Fe-Doped Mesoporous Titania Catalyse the Oxidation of Acid Orange 7 by H2O2 under Different Illumination Conditions: Fe Doping Improves Photocatalytic Activity under Simulated Solar Light. Catalysts. 2017; 7(7):213. https://doi.org/10.3390/catal7070213
Chicago/Turabian StyleFreyria, Francesca S., Matteo Compagnoni, Nicoletta Ditaranto, Ilenia Rossetti, Marco Piumetti, Gianguido Ramis, and Barbara Bonelli. 2017. "Pure and Fe-Doped Mesoporous Titania Catalyse the Oxidation of Acid Orange 7 by H2O2 under Different Illumination Conditions: Fe Doping Improves Photocatalytic Activity under Simulated Solar Light" Catalysts 7, no. 7: 213. https://doi.org/10.3390/catal7070213