1. Introduction
Lipases (E.C. 3.1.1.3) are widespread enzymes found mainly in the animal, plant and microbial kingdoms. They catalyze both the hydrolysis and synthesis of triglycerides containing long-chain fatty acids (C > 9) at the oil-water interface [
1], which has markedly spurred their demand in the enzyme industry. Lipases are also used in the dairy and beverage industry, oleochemical industry, cosmetic industry, biosensors industry, detergents formulations and paper manufacturing, production of the biodiesel and organic synthesis [
2]. Due to increasing demand for lipase across industries, the growth prospects of the global lipase market will remain positive. To date, several lipases from different genera of bacteria have been identified, purified and characterized [
3,
4].
To facilitate purification, remarkable research improvements have been reported in terms of sophistication and the variety of tags used for biological studies such as glutathione S-transferase, chitin-binding domain, Strep-tag, FLAG-tag, Thioredoxin (Trx) tag, self-cleaving intein, Elastin-like Polypeptide (ELP) or Polyhydroxybutyrate (PHB) [
5,
6,
7]. Tags play an ever-widening role as a form of tremendous advantage [
8]. However, for particular applications such as pharmaceutical industry or fundamental study, tags must be removed after the purification process. In some cases, the negative effects of additional tags to the target protein have been reported to result in alterations of the protein conformation, undesired flexibility in structural studies, changes in biological activity or activity inhibition, and toxicity [
9]. Based on the system, tags may be removed by chemical cleavage, self-cleavable protein linker or enzymatic digestion [
7].
Chemical cleavage is rather nonspecific when indicating the cutting side and may result in protein denaturation or cause a modification in the side chain of amino acids in the protein [
7]. Enzymatic digestion requires protease treatment; then, the protease must be efficiently removed by the second step of purification to recover the pure mature target protein and also to avoid the contamination of the sample by proteases. In brief, all these methods are complicated, expensive and could lead to problematic effects on the protein properties.
A new technique has been introduced to overcome the above-mention limitation to cleave the tag of fusion recombinant HZ lipase (rHZ lipase). The rHZ lipase gene, isolated from
Aneurinibacillus thermoaerophilus strain HZ, cloned into pET32b(+) plasmid and fused to the triple tags, Trx-His-S-tags, present in the plasmid [
10,
11]. The rHZ lipase and tags expressed as a single fusion protein. Fusion rHZ lipase has been purified using affinity chromatography. The tags can be removed using Enterokinase (EK) enzyme as plasmid contains a linker with a specific sequence, Asp-Asp-Asp-Asp-Lys-↓-X, Enterokinase site. Generally, one unit of Enterokinase is required to cleave 25 to 50 µg of the recombinant fusion proteins in a range of 16 to 20 h at 25 to 23 °C to 95% completion, for a total cost of 13,250 to 71,500 USD to cleave 1.0 g of the fusion protein. As the enzyme is expressed in a large amount, the huge cost of purification makes it unfavorable for industrial applications. Skala et al. [
12] reported an alternative economical cleavage enzyme for commercial Enterokinase, do-it-yourself EK, which was produced by the expression of pentahistidine-tagged EK in
E. coli and the refolding of the protein from bacterial inclusion bodies. However, using do-it-yourself EK requires another step of purification to remove the protease enzyme. To solve this problem, a simple and economical novel method was invented for tag removal from fusion rHZ lipase. The enzyme was further characterized to identify the properties of mature rHZ lipase.
3. Discussion
Protein purification is a necessary step to characterize the function, structure and interactions analysis of protein for fundamental study or industrial purposes [
14]. Such applications require a specific amount (from microgram to kilogram scales) of purified protein in different quality (e.g., partial or fully purified). The purification method could affect the quality and quantity of the protein, which must fit the intended use. For the majority of industrial purposes, the most important aspect is an economical, rapid, high yielding production process which could be shifted to large-scale operation. However, for fundamental studies, high purity of a protein is the first top priority, among others. To simplify the purification step(s) and detection, the expression of a recombinant protein attached to a tag of known size and biochemical properties, such as histidine tag, is one of the best options. The fusion His-tag proteins have the strong selective affinity toward metal ions such as Ni
2+, Co
2+, Cu
2+, and Zn
2+ which can be immobilized on chromatographic resin using chelating ligands [
15]. Nevertheless, a fusion tag linked to the protein enzyme could alter the catalytic properties of the enzyme [
16]; also, for crystallization purposes, only the mature protein is required so after affinity chromatography the tag needs to be cleaved.
There are a few reports that the fusion tag has been cleaved using protease treatment such as PF2001Δ60 lipase from
Pyrococcus furiosus and T1 lipase from
Geobacillus zalihae that cloned into pET-32a and pGEX plasmids, respectively [
17,
18]. In pET-32b vector using Enterokinase enzyme, tags can be cleaved from mature protein after lysine residue at its recognition site: DDDDK. However, the cost of the Enterokinase is prohibitively expensive for the production of one gram of mature protein.
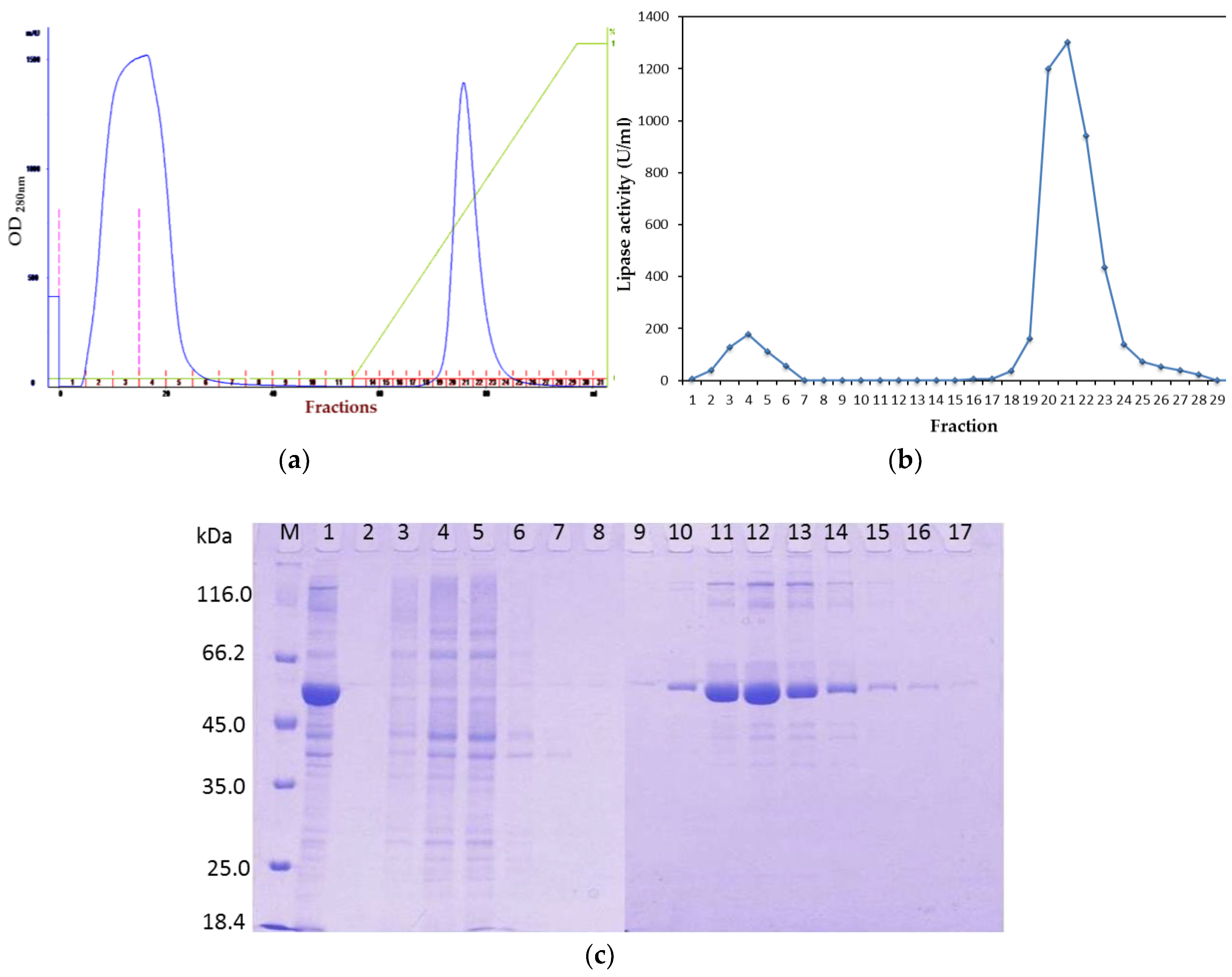
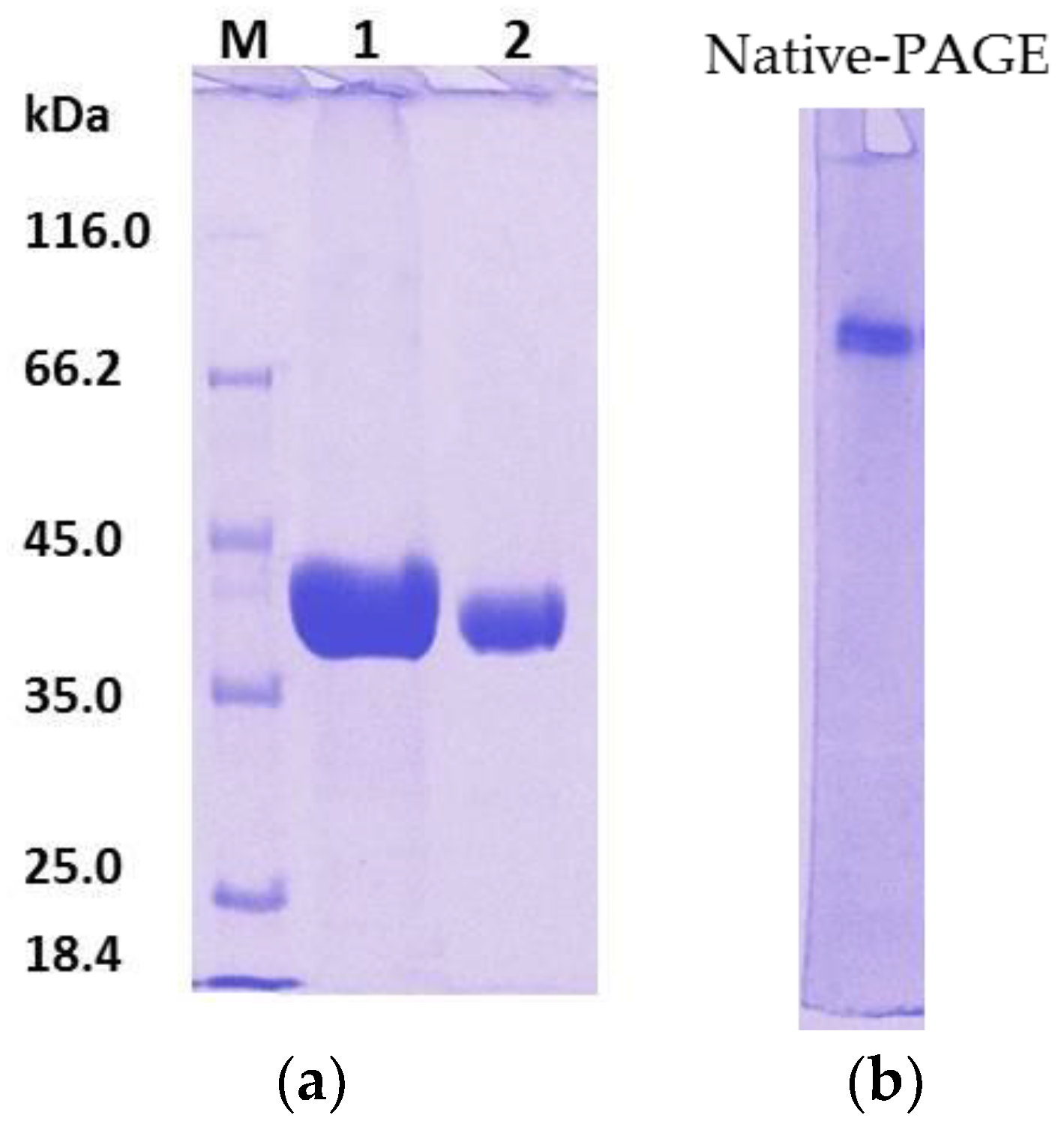
To overcome this problem, after affinity the chromatography step, treatment of rHZ lipase at 20 °C was a straightforward and efficient technique that presented a non-enzymatic cleavage and non-chromatographic purification procedure. This method induced specific auto-cleavage of the target protein and precipitation of few undesirable
E. coli proteins that eluted along with rHZ lipase. N-terminal sequencing confirmed that 17.4 kDa tag has been successfully removed from one amino acid before the methionine start amino acid, DDDDKAM↓DMQ, which is an advantage of this new technique. The obtained final yield was 79% which was higher than the purified T1 lipase expressed in
E. coli systems with a final recovery of 51.5% [
17], the purified BTL2 lipase expressed in a
Pichia pastoris system with a final recovery of 18% [
19], P1 lipase which purified through ultrafiltration and Q Hyper D column with a final recovery of 71% [
20], and recombinant His-tagged L2 lipase that purified by single-step affinity chromatography with a final recovery of 76.1% [
21].
A high kosmotropic salt (sodium, potassium, or ammonium sulfates) concentration enhances the hydrophobic interaction and protein precipitation due to higher “salting-out” or molar surface tension increment effects, while lowering the salt concentration weakens the hydrophobic interaction [
22]. The fusion rHZ lipase was purified in 500 mM NaCl concentration; the salt was then removed by dialysis, which could be harsh to the Trx-His-S-tags protein. In addition, it is possible that the aggregation of the triple tag could not be reversed by removing the salt. Therefore, dialysis by incubation of the protein at 20 °C for a longer period (48 h) promoted the cleavage of the tag. However, the thermostable rHZ lipase structure was rigid enough not to undergo any conformational changes during dialysis and treatment at 20 °C.
The differences in the lipase activity of fusion and mature rHZ lipase (
Figure 2d) showed that the presence of 17.4 kDa tag has a considerable effect on the active site of the enzyme. So, the biochemical characteristics of mature rHZ lipase which is the first member of subfamily I.9 true lipase from
Aneurinibacillus thermoaerophilus strain HZ have been studied. Based on the gene sequence analysis, rHZ lipase is close to mesophilic
Bacillus lipases. The predicted structure also revealed some differences between rHZ lipase and other thermostable lipases [
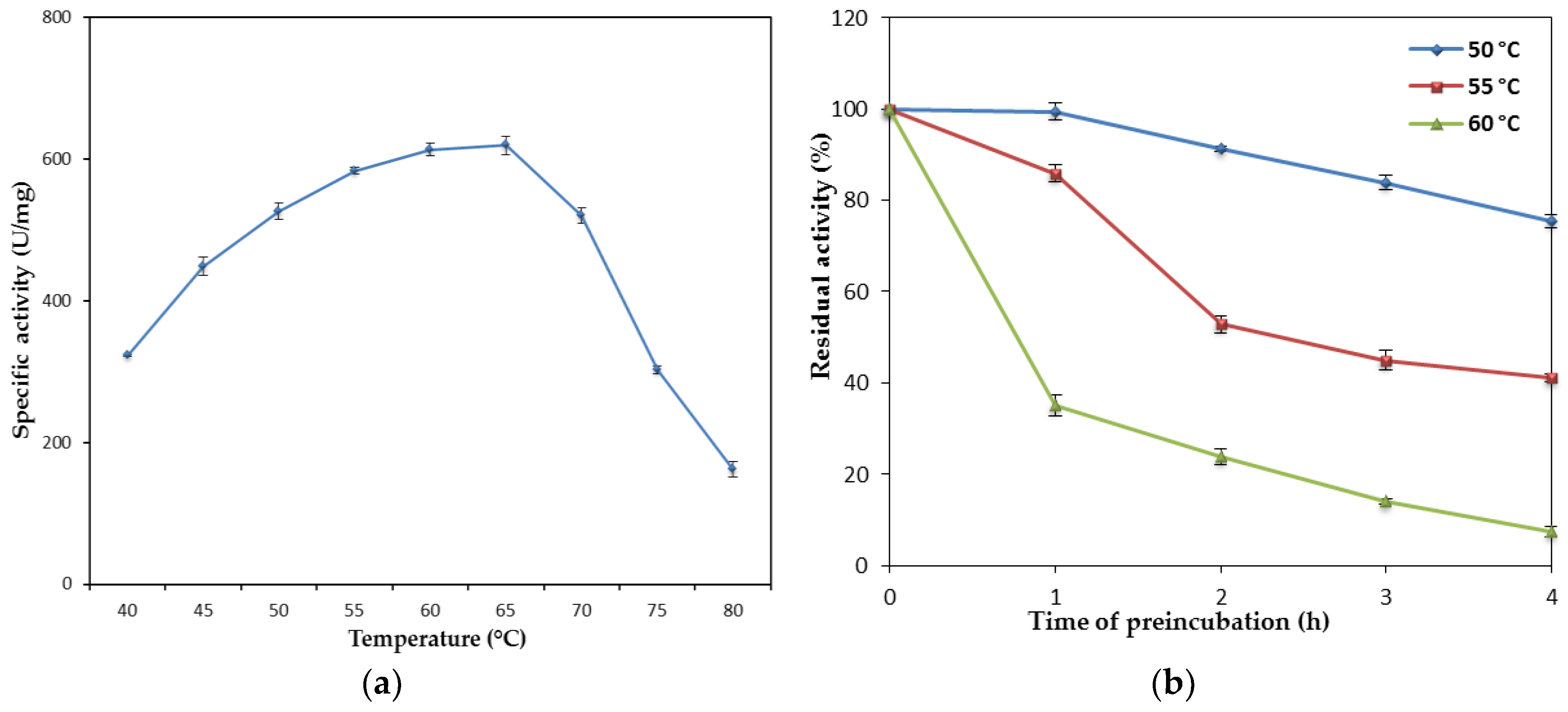
10]. However, a study on the effect of temperature of rHZ lipase showed that the enzyme has high optimum activity and moderate stability at elevated temperatures which are comparable with thermostable lipases. The optimum reaction temperatures of 60–65 °C for rHZ lipase were higher than the majority of thermostable lipases, which had optimum temperatures of around 45–50 °C [
23]. In addition, the rHZ lipase showed good stability at 50 °C which is lower than its optimum activity, but it maintained half of its activity for 45 min at 60 °C. The wild-type HZ lipase (wHZ lipase) has also an optimum temperature of 65 °C, but higher thermostability compared to the rHZ lipase, the half-life of 3 h 10 min at 65 °C [
24]. Several lipases have been to be unstable at their optimum temperatures such as lipase from
B. stearothermophilus MC7 [
25] and BTL2 lipase from
B. thermocatenulatus BTL2 [
19]. To pursue any strategy to stabilize an enzyme such as preventing or decelerating of losing activity, it must be considered that enzyme inactivation may be related directly to shifting the native state (N) ⇄ unfolded enzyme (U) equilibrium or preventing the important irreversible process that happens after formation of unfolded protein [
26]. To prevent these process and stabilize the enzyme, there are some soluble additives such as substrates, low molecular weight organic solutes, salts, polymeric solutes and synthetic polymers [
27].
The maximum hydrolytic activity of mature rHZ lipase was noted at pH (65 °C) 7.0 using Tris-HCl buffer and the enzyme retained > 75% of its activity at the pH range of 6.0 to 10.0 after 30 min pre-incubation at 50 °C indicating that the purified enzyme is a neutral lipase. The wHZ lipase preferred potassium phosphide buffer pH (65 °C) 7.0 as an optimum buffer [
24] while rHZ lipase lost more than 50% of its activity in the presence of potassium phosphide buffer pH (65 °C) 7.0 (
Figure 6a). The optimal lipases activity of several thermostable lipases found in the moderate to highly alkaline pH range from 8.5–10.0, such as lipases from
G. zalihae sp. T1 with the optimum pH at 9.0 and more than 60% stability at pH 9 to 11 [
17], and
B.
stearothermophilus L1 [
28] with optimum pH at 9–10 and 24 h stability at pH 5 to 11. On the other hand, two thermostable lipases from
B. stearothermophilus SB-1 [
29] and
Bacillus megaterium CCOC-P2637 [
30] showed optimum activity in a pH range of 3.0–6.0, lower than reports of optimum pH for
Bacillus lipases. Meanwhile, using multiple buffer systems to cover a wide range of pH in this study enabled to detect the effect of buffer compound on the activity of the enzyme. Overlapping assay on a specific pH was done, when the buffer system was changed. The result indicated that the compound of buffers had also effect on the hydrolytic activity of rHZ lipase such as MES and phosphate buffers (Na
+ and K
+) at pH 6.0 and those other overlap buffers at pH 7.0 to 9.0 (
Figure 6).
Changing the pH could alter the ionic bonds in the enzyme structure whereas these bonds are important to structural stability and help to determine the 3D feature and functionality of the protein enzyme. The solubility of the protein increases at pH values farther away from its isoelectric point due to greater electrostatic repulsions [
31]. Therefore, rHZ lipase was less active at a pH near or far below/above its pI (5.74) as the enzyme might be inactivated or formed aggregates during treatment. These results help in choosing the appropriate buffer for enzyme crystallization.
Characteristic of mature rHZ lipase revealed that various metal ions had different impacts on the activity of the enzyme. Similar to mature rHZ lipase, most
Bacillus lipases maintained their initial activities or were slightly activated (5–20% higher than the maximum activity) in the presence of up to 10 mM potassium ion. Calcium ions have been reported to increase the activity of most
Bacillus lipases 10 to 50% compared to the maximum (
Table S2), and the same results were also observed for rHZ and wHZ lipases [
24]. Additionally, several reports have illustrated that calcium ion could maintain the activity of the lipases at elevated temperatures [
23]. In addition, a Ca
2+-binding motif, GXXGXD, reported in the protein sequence of
Staphylococcus epidermidis lipase [
32] and lipA from
Acinetobacter sp. RAG-1 [
33] was found in the gene sequence of rHZ lipase unlike the other mesophilic or thermostable
Bacillus lipases. This motif shows the rHZ lipase can be a Ca
2+-dependent lipase. The mature rHZ lipase lost its activity in the presence of most of the heavy metal ions tested in this study similar to the majority of thermostable
Bacillus and
Geobacillus lipases [
23]. The inhibition of rHZ lipase activity in the presence of heavy metal ions might be due to the toxic effect of the excess amount of these ions in the reaction medium. The ionization of surface charged amino acid residues on the enzyme by the interaction of them with salt ions could significantly change the enzyme conformation followed by its activity [
34].
Generally, some structural features of enzymes can be demonstrated by studying the impact of inhibitors. Unlike wHZ lipase, the complete activity inhibition of mature rHZ lipase by EDTA (metal-chelating agent) suggests that the enzyme is a metalloenzyme which supported that Ca
2+-dependently of rHZ lipase. Moreover, the addition of DTT and 2-mercaptoethanol (reducing agents) to the rHZ lipase reaction promoted this activity which, suggests that there is no disulfide bond in rHZ lipase structure. Guncheva and Zhiryakova [
23] reported that reducing agents decreased the activity of lipases from subfamily I.5, since disulfide bridges not only support the active site conformation, also play important roles on the thermostability of these enzymes. Hence, the activation of thermostable rHZ lipase by reducing agents can explain the structural differences of this enzyme and its lower thermostability compared to the other thermostable lipases belonging to subfamily I.5 of true lipases. The mature rHZ lipase activity inhibition by PMSF indicated the presence of a serine residue at the catalytic triad of the active site. The enzyme might belong to serine hydrolase. PMSF strongly inhibited the activity of the most of lipases from
Bacillus genus by irreversible binding to the catalytic serine residue. However, it has also been reported that PMSF did not inhibit the activity of a thermostable lipase B from
G. thermoleovorans ID-1 [
35].
Most lipases contain a mobile lid domain placed over the active site which is also responsible for catalytic activity. The movements of amino acids in the lid are different in closed or open conformations; hence, the amino acid sequence of the lid defines the activity and specificity of lipases [
36]. The mature rHZ lipase showed preference to hydrolyze the long chain triacylglycerides among all tested oils with the highest affinity to olive oil while wHZ lipase has 30% higher preference toward the sunflower oil compared to the olive oil [
24]. Similarly, the lipases from
G. zalihae sp. T1 and
Bacillus sp. L2 exhibited a preference for natural oils such as olive, corn, palm, and soybean oils that mostly are contained unsaturated fatty acids [
17,
37] even though the sequence of amino acids in the lid domain of rHZ lipase and thermostable lipases from subfamily I.5 are different. The high activity of mature rHZ lipase to hydrolyze the natural oils can make it suitable for modification and improvement of physicochemical properties and nutritional value of the natural oils and formation of an extensive variety of saturated and polyunsaturated fatty acids [
23]. Mesophilic and thermostable lipases from subfamily I.4 and I.5 have shown a different preference for
p-nitrophenyl esters substrate, which could be due to their different protein amino acid sequences and biochemical characteristics. Lipase BTL2, Lip B and P1 lipase are thermostable enzymes which shown higher preference toward the
p-nitrophenyl decanoate (C10) compared to the other
p-nitrophenyl esters (
Table S2). The highest activity of mature rHZ lipase towards
p-nitrophenyl decanoate (C10) and olive oil was 947.7 and 620 U mg
−1 at 65 °C, respectively, strongly indicating that rHZ lipase is a true lipase [
38]. Besides, the enzyme showed more than 75% of hydrolysis activity toward both shorter (4-nitrophenyl acetate (C2)) and longer acyl chain (4-nitrophenyl palmitate (C16)) lengths
p-nitrophenyl esters which was in line with the result of molecular docking study. Molecular docking helps to predict the possible position and orientation of ligand in protein binding site. The docking results of rHZ lipase showed that apart from the size of acyl chain length, the conformation of active site and the amino acid sequences were important parameters to determine the preference of rHZ lipase toward of
p-nitrophenyl esters substrate. This makes the rHZ lipase an ideal catalyst for organic synthesis in aqueous and non-aqueous conditions.
Most enzymes applied in industrial activities have a range of
Km values between 10
−1 to 10
−5 M when acting on biotechnological important substrates [
39]. It has been reported that the
Km and
Vmax values of thermostable lipase BTID-B for the hydrolysis of tricaprylin was 6.24 mM and 63.3 μmol min
−1 mg
−1, respectively [
35]. However, Massadeh et al., [
40] reported that a lipase from
Bacillus stearothermophilus HU1 had a
Km and
Vmax value of 0.235 mM and 161.2 nmol min
−1, respectively when using
p-NP-Palmitate as substrate. A similar study using
p-NP-Palmitate was conducted on a purified thermostable lipase from
Bacillus stearothermophilus MC7, which was found to have a
Km and
Vmax values of 0.33 mM and 188 μmol min
−1 mg
−1, respectively [
25]. This suggests that the rHZ lipase had a high potential in industrial applications as it has a
Km value between the required range.
In the presence of organic solvents, native enzymes generally show low activities and stabilities limiting enzymatic reactions [
41]. Lipase performance in different solvents is associated with its efficiency in hydrolytic and synthetic reactions [
23]. Exposure of the mature rHZ lipase to various organic solvents for 30 min ascertained that the enzyme was stable to both very polar and non-polar organic solvents. Polar water-miscible organic solvents destabilize the enzymes by eliminating the solvation water from the enzyme surface area [
42] but increasing the activity of mature rHZ lipase in the presence of 25% DMSO could be due to the prevention of enzyme aggregating in the reaction mixture [
30]. Lee et al. [
35] reported that the activity of thermostable lipase BTID-B enhanced in the presence of 1% DMSO in the reaction medium. Nonpolar solvents are often less harmful to the enzymes because they contain two phases in water + water-immiscible organic solvent system; an immiscible organic solvent phase and an aqueous phase. The enzymatic reaction proceeds in the aqueous phase, which contains a dissolved enzyme. In the biphasic system, the enzyme is relatively stable due to the less direct contact of the enzyme with an organic solvent [
43]. This phenomenon indicates the presence of more charged amino acid residues on the surface of rHZ lipase which is in an agreement with a report on the analysis of the predicted structure of the enzyme [
10]. On the other hand, alcohol organic solvents decreased or inactivated the mature rHZ lipase. Guncheva and Zhiryakova [
23] reported that most
Bacillus lipases were inactivated by polar alcohol solvents. The majority of thermostable lipase from subfamily I.5 could not tolerate the presence of organic solvents or they tolerate a certain polarity of solvents. Interestingly, wHZ lipase has a closely similar organic solvent activity profile with rHZ lipase, except it stimulated wHZ lipase activity in the presence of methanol. In terms of the stability profile of the rHZ lipase for a wide polarity range of organic solvents, it showed a promising potential for industrial chemical processes.