1. Introduction
There is considerable interest in the development of latent polymerization initiators that are inert in monomers or prepolymers under storage conditions. Latent thermal initiators must retain high activity under targeted temperatures while maintaining stability under storage conditions [
1,
2,
3,
4,
5,
6,
7,
8,
9,
10]. We have previously reported that primary alkylamines intercalated with α-zirconium phosphate (α-ZrP) can serve as latent thermal initiators in the reaction of glycidyl phenyl ether (GPE) [
11,
12] and that intercalation compounds of 1,4-diazabicyclo[2,2,2]octane (DABCO), 1,8-diazabicyclo[5,4,0]undec-7-ene (DBU), imidazole (Im), 2-methylimidazole (2MIm), and 2-ethyl-4-methylimidazole (2E4MIm) with α-ZrP (α-ZrP·DABCO, α-ZrP·DBU, α-ZrP·Im, α-ZrP·2MIm, and α-ZrP·2E4MIm, respectively) show good performance as latent thermal initiators in the reaction of GPE with hexahydro-4-methylphthalic anhydride (MHHPA) [
13,
14,
15].
Imidazoles are widely used in industry as curing agents for epoxy resins found in electric devices, laminated plates, semiconductor sealing agents, etc. The equivalents of imidazoles in intercalation compounds of α-ZrP are 0.78 (Im), 0.96 (2MIm), and 0.65 (2E4MIm). However, after reaction with the GPE-MHHPA system, 35% (α-ZrP·Im), 48% (α-ZrP·2MIm), and 37% (α-ZrP·2E4MIm) of the intercalated imidazoles were deintercalated from the layers of zirconium phosphate [
14]. Essentially, less than half of the imidazole was available for the reaction of GPE-MHHPA.
Benzimidazoles are known as good curing agents for epoxy-anhydride systems [
16]. Using benzimidazole as an intercalation compound, Costantino et al. [
17] reported a molar ratio of benzimidazole/zirconium phosphate of up to 1.9. Therefore, we anticipated that benzimidazole-intercalated α-ZrP would have better efficiency as a curing agent because it had a higher loading in α-ZrP. We prepared the intercalation compound of benzimidazole (Bim) and examined the capabilities of α-ZrP·Bim as a latent thermal initiator in the reaction of GPE with MHHPA. The release behavior of Bim from the interlayer of α-ZrP was studied in detail.
2. Results and Discussion
The intercalation of benzimidazole (Bim) into the layers of α-zirconium phosphate (α-ZrP) was carried out by slightly modifying a previously reported method [
14]. The α-ZrP was added to a solution of Bim in 1:1 water:methanol. The reaction mixture was stirred at 60 °C for 24 h. After the reaction, the intercalation compound was recovered by suction filtration. The ratio of C, H, and N of the product was C: 27.94%, H: 2.64%, and N: 9.28% and the composition was Zr(HPO
4)
2(C
7H
6N
2)
1.60·0.50H
2O as determined by elemental analysis. The interlayer distance of α-ZrP was calculated from the XRD patterns, which showed that pristine α-ZrP 7.6 Å (2
θ = 11.7°) was expanded to 20.3 Å (2
θ = 4.4°) as seen in
Figure 1a. Thus, the intercalation of Bim into the layers of α-ZrP (α-ZrP·Bim) was confirmed.
To evaluate the catalytic activity of α-ZrP·Bim, the copolymerization of glycidyl phenyl ether (GPE) and hexahydro-4-methylphthalic anhydride (MHHPA) was carried out. The conversion of GPE was 97% at 140 °C for 1 h as determined by
1H NMR analysis. The calculation of the conversion by
1H-NMR analysis determined was shwon in ref. [
14]. The intercalation compound α-ZrP·Bim showed good reactivity under heating conditions.
To evaluate the change of the layer structure after the reaction of α-ZrP·Bim, the product was washed with THF to remove the GPE-MHHPA products and the residue of α-ZrP (α-ZrP·Bim-RXN) was recovered. The interlayer distance of α-ZrP·Bim-RXN was increased to 22.9 Å (2
θ = 3.8°) and the peak intensity was decreased, as shown in
Figure 1b. This might cause the intercalation of the reaction products into the layers and the crystallinity of α-ZrP·Bim was decreased after the reaction.
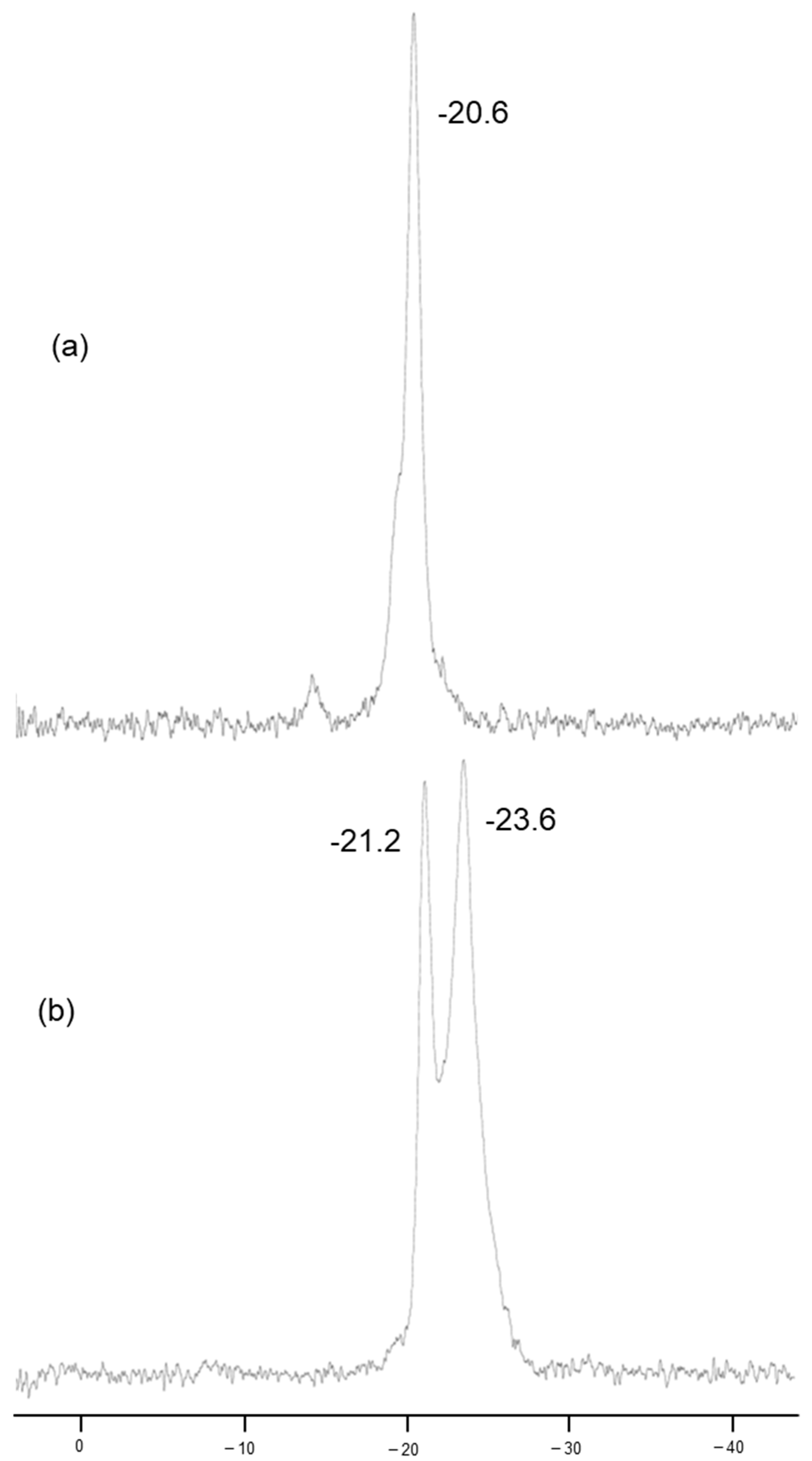
The
31P MAS NMR spectra of α-ZrP·Bim and α-ZrP·Bim-RXN are shown in
Figure 2a,b. The peak of pristine α-ZrP is observed at δ-20.1. As shown in
Figure 2a, the peak of α-ZrP·Bim is observed at δ-20.6. This chemical shift suggests that interactions between Bim and the HPO
4 group were not strong compared with that between alkylamines and the HPO
4 group [
18,
19]. In
Figure 2b for α-ZrP·Bim-RXN, the signal at δ-21.2 and -23.6 were observed. This shift of the signal from δ-20.6 to δ-21.2 and -23.6 might due to the separation of Bim from the phosphate groups [
11]. The
13C NMR spectra of α-ZrP·Bim and α-ZrP·Bim-RXN are shown in
Figure 3a,b. The 2-position of the imidazole ring at δ144.5 (N=
CH-NH) in
Figure 3a (assigned to 1) completely disappeared after the reaction (α-ZrP·Bim-RXN) and the corresponding peaks of the products of GPE-MHHPA appeared [
Figure 3b]. Therefore, Bim was completely deintercalated from the interlayers. This demonstrates that all of the intercalated Bim could be used to initiate the reaction of GPE-MHHPA. We have previously reported the intercalation compounds of α-ZrP·Im, α-ZrP·2MIm and α-ZrP·2E4MIm, showing that the deintercalation ratios after the reaction with GPE-MHHPA were 35%, 48%, and 37%, respectively [
14]. Moreover, the copolymer of GPE-MHHPA was not formed after the reaction of α-ZrP·Im-RXN, α-ZrP·2MIm-RXN, and α-ZrP·2E4MIm-RXN. Substances derived from GPE were present in the interlayer of these three intercalation compounds of α-ZrP. In the case of α-ZrP·Bim, the copolymer was confirmed by the presence of ester groups at δ173.2 in
Figure 3b (assigned to 1). Therefore, the products of GPE-MHHPA can be intercalated in the layers of α-ZrP·Bim. The intercalation compound of Bim (α-ZrP·Bim) was efficiently utilized in the reaction of GPE-MHHPA. FT-IR spectra of α-ZrP·Bim and α-ZrP·Bim-RXN are shown in
Figure 4a,b. The peak due to the Bim (ν C=N) and α-ZrP (ν P–O) were detected at 1622 and 1004 cm
−1 in
Figure 3a. The carbonyl groups (ν C=O at 1736 cm
−1), aromatics (ν C–C at 1600, 1497 and 1458 cm
−1), and ether groups (ν C–O–C at 1249 cm
−1) in the products of GPE-MHHPA were clearly observed in
Figure 3b.
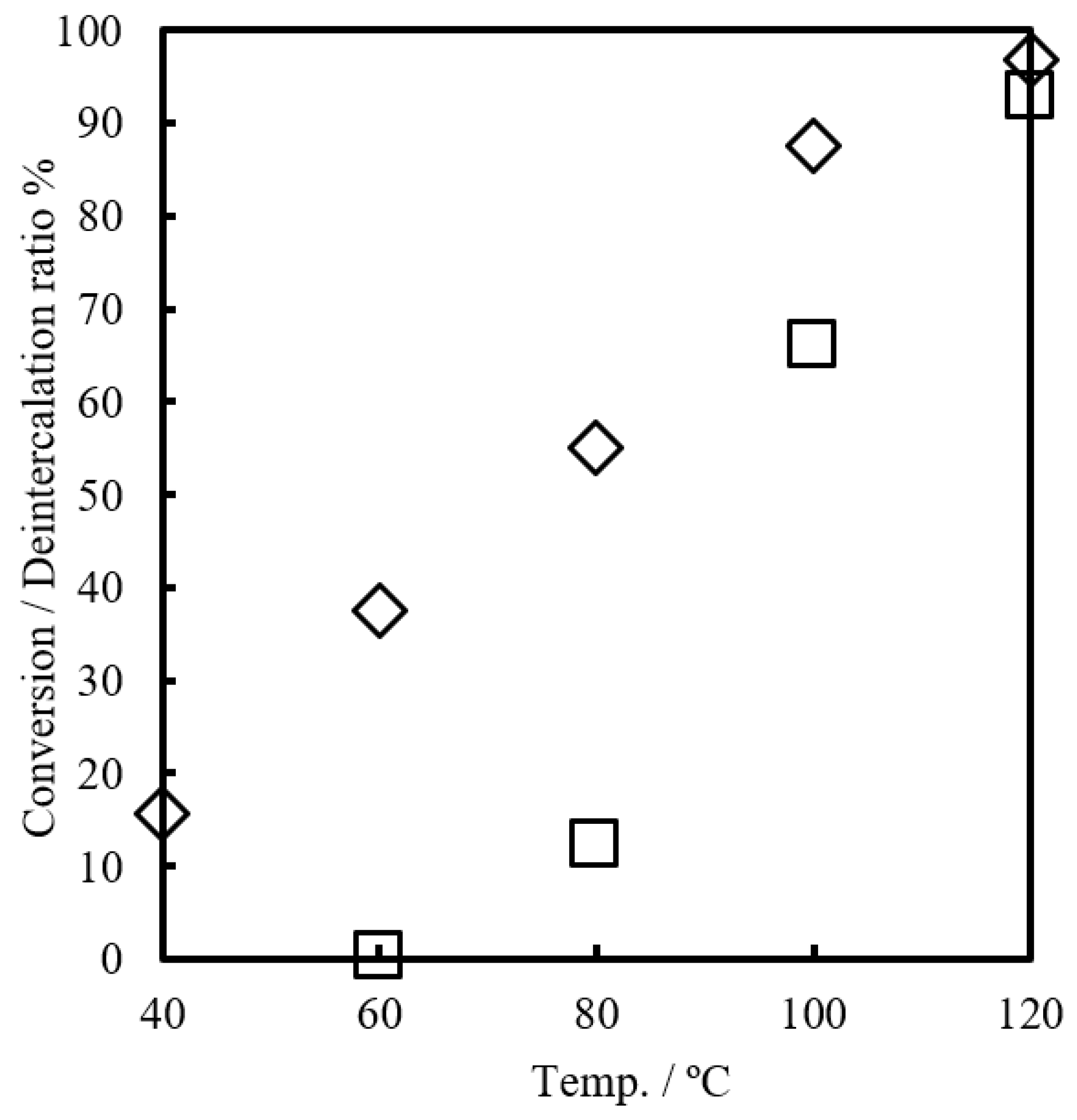
The capabilities of Bim as a latent thermal initiator were examined in the reaction of GPE and MHHPA. The conversion of GPE and the deintercalation ratio of Bim from the layers of α-ZrP containing 3 mol% of Bim were measured at varying temperatures for 1 h, as shown in
Figure 5. The deintercalation ratio of Bim from α-ZrP·Bim were calculated by decreasing the N content of α-ZrP·Bim by elemental analyses. The conversion did not occur at 60 °C after 1 h. Upon increasing the reaction temperature to 120 °C, the conversion improved to 97%. The deintercalation ratio increased with increasing reaction temperature. At 120 °C, the deintercalation ratio became quantitative, (i.e., all of the Bim in the interlayer of α-ZrP deintercalated). At 60 °C, the deintercalation ratio was 38% and the reaction did not proceed in 1 h.
The changes in the interlayer distances of α-ZrP were examined after the reaction at varying temperatures, as shown in
Figure 6. At 60 °C for 1 h, the interlayer distance of 19.9 Å (2
θ = 4.4°) was almost the same as that of α-ZrP·Bim (20.2 Å, 2
θ = 4.4°). A peak of 23.3 Å (2
θ = 3.8°) was newly observed after the reaction at 80 °C, and the peak of 19.9 Å (2
θ = 4.4°) completely disappeared at 100 °C. The interlayer distance gradually increased from 19.9 Å (2
θ = 4.4°) at 60 °C to 23.1 Å (2
θ = 3.8°) at 100 °C.
To study the reaction behavior of GPE-MHHPA, the effect of the layer distances of ZrP using GPE and MHHPA at 100 °C for 1 h was investigated. In the case of GPE, the XRD pattern is shown in
Figure 7a. The interlayer distance increased from 20.3 Å (2
θ = 4.4°) to 26.5 Å (2
θ = 3.3°), suggesting that the monomer of GPE might be intercalated into the interlayer of α-ZrP·Bim. The ratio of C, H, and N of the product was C: 48.42, H: 4.43, and N: 1.52, and the composition was Zr(HPO
4)
2·(C
9H
10O
2)
3.5·(C
7H
6N
2)
0.47, as determined by elemental analysis. 71% of intercalated Bim was deintercalated from α-ZrP·Bim and the molar ratio of 3.5 of GPE to Zr was intercalated into the layer of α-ZrP. Similarly, upon treatment of α-ZrP with MHHPA, the interlayer distance was slightly decreased from 20.3 Å (2
θ = 4.4°) to 19.3 Å (2
θ = 4.6°), showing that deintercalation of Bim from the interlayers of α-ZrP·Bim occurred. The ratio of C, H, and N of the product was C: 42.67, H: 4.61, and N: 3.87 and the composition was Zr(HPO
4)
2·(C
9H
12O
3)
2.3·(C
7H
6N
2)
1.1 as determined by elemental analysis. 31% of intercalated Bim was deintercalated from α-ZrP·Bim and the molar ratio of 2.3 of MHHPA to Zr was detected. The interlayer distance after the treatment of α-ZrP·Bim with MHHPA slightly decreased, indicating that MHHPA was immobilized on the surface of α-ZrP.
It is important to maintain stability under storage conditions. The stabilities were examined at 25 °C and 40 °C in the GPE-MHHPA system. The conversion of GPE was 51% for Bim and 22% for α-ZrP·Bim at 25 °C for 14 days, as shown in
Figure 8. At 40 °C, the conversion was 50% for Bim and 21% for α-ZrP·Bim for 7 days, as shown in
Figure 9. The storage stability for α-ZrP·Bim was maintained for 14 days (2 weeks) at 25 °C.
Accordingly, α-ZrP·Bim can serve as a latent thermal initiator in the reaction of epoxy-acid anhydride systems. In the reaction of GPE-MHHPA with α-ZrP·Bim, the conversion reached 97% at 140 °C for 1 h, and the storage stability was maintained for 2 weeks at 25 °C. All of the intercalated Bim could be deintercalated at 120 °C for 1 h.
3. Experimental Section
3.1. Materials
Zr(HPO4)2·H2O (CZP-100) was purchased from Daiichi Kigenso Kagaku Kogyo Co., Ltd. (Osaka, Japan). GPE was purchased from Aldrich Chemical Co., Inc. (Tokyo, Japan). Benzimidazole and MHHPA were purchased from Tokyo Chemical Industries, Co., Ltd. (Tokyo, Japan). Solvents were used as received without further purification.
3.2. Measurements
X-ray diffraction (XRD) patterns were obtained using a Rigaku RINT2200 (Tokyo, Japan) with Cu Kα radiation over a scan range of 3–40° at a rate of 2° min−1. NMR spectra in solution were recorded on a Varian Unity-300 spectrometer (Palo Alto, CA, USA) and a JEOL JNM-ECZS (400 MHz) spectrometer (Tokyo, Japan) using tetramethylsilane (TMS) as an internal standard. The 31P MAS NMR and 13C CPMAS NMR spectra were recorded on a JEOL ECA-600 NMR spectrometer (Tokyo, Japan). The contents of benzimidazole and water in the intercalation compounds of α-ZrP were measured using a PerkinElmer 2400II (Waltham, MA, USA). Gel permeation chromatography (GPC) was carried out on a Shodex GPC-101 (LF804*3 and KF-800RF*3, THF as eluent) (Showa Denko Co. Ltd., Tokyo, Japan) using polystyrene standards. The Fourier transform infrared spectroscopy (FT-IR) measurements were carried out with an ALPHA spectrometer (Billerica, MA, USA).
3.3. Preparation of Benzimidazole-Intercalated α-ZrP (α-ZrP·Bim)
The intercalation of benzimidazole into the layers of Zr(HPO
4)
2·H
2O (α-ZrP) was carried out using a previously reported method [
14]. α-ZrP (10.54 g) was added to 117 mL of a 11.78 g (99.7 mmol) benzimidazole methanol/water = 1/1 solution. The reaction mixture was then allowed to stand at 60 °C for 24 h before the product was collected by filtration and washed with methanol several times. The resulting residue was dried at 40 °C under vacuum (Yield: 15.7 g). The composition determined by elemental analysis was Zr(HPO
4)
2(C
7H
6N
2)
1.60·0.50 H
2O (C: 27.94%, H: 2.64%, N: 9.28%).
3.4. Typical Polymerization Procedure
A mixture of GPE (150 mg, 1.0 mmol), MHHPA (168 mg, 1.0 mmol), and benzimidazole intercalation compound with α-ZrP (α-ZrP·Bim) (9.0 mg, 0.019 mmol, content of benzimidazole: 0.030 mmol) was heated at 120 °C for 1 h. A small aliquot of the reaction mixture was dissolved in CDCl3, and its 1H-NMR spectrum was acquired to determine the extent of the conversion of GPE and MHHPA. At 40 °C, a small aliquot of the sample was collected at determined times.
3.5. Polymerization Procedure for Poly(GPE-co-MHHPA) with α-ZrP·Bim and Recovery of α-ZrP·Bim (α-ZrP·Bim-RXN) after the Reaction
A mixture of GPE (4.51 g, 30.0 mmol), MHHPA (5.05 g, 30.0 mmol), and α-ZrP·Bim (271 mg, 0.56 mmol, content of benzimidazole: 0.90 mmol) was heated at 120 °C for 1 h. After the reaction, tetrahydrofuran (THF) was added to the mixture. The solution was filtered off, and the residue, α-ZrP·Bim-RXN, was rinsed, dried under vacuum, and analyzed by XRD. The polymer was precipitated from the THF solution with methanol and dried under vacuum. The molecular weight was determined by GPC analysis (Mn = 1130, Mw/Mn = 1.6).