2.1. Catalyst Characterization
The XRD pattern of γ-Al
2O
3 and Zn-Mn-Co modified γ-Al
2O
3 are shown in
Figure 1. The position of the diffraction peaks of the modified sample was well matched with that of the original sample, which indicated the modification of Zn-Mn-Co had no effects on the crystalline phase of γ-Al
2O
3. The intensity of the diffraction peaks became stronger, which suggested that the crystallinity of Zn-Mn-Co modified γ-Al
2O
3 was higher than that of γ-Al
2O
3. The improvement of crystallinity may be attributed to the fact that zinc, manganese, and cobalt formed a spinel solid solution with γ-Al
2O
3, which improved the stability of the catalyst in the reaction environment with water [
22]. Zinc, manganese, and cobalt oxides were not detected in the spectra, showing that zinc, manganese, and cobalt were highly dispersed on the catalyst surface. Research has suggested that manganese ions play an important role in improving the dispersion of other metal ions [
23]. To verify whether metal ions were loaded onto γ-Al
2O
3, the metal loadings of the modified catalyst were measured by the inductively coupled plasma atomic emission spectrometry (ICP-AES) method, and the loading amounts of zinc, manganese, and cobalt were 0.54%, 0.44%, and 0.23%, respectively.
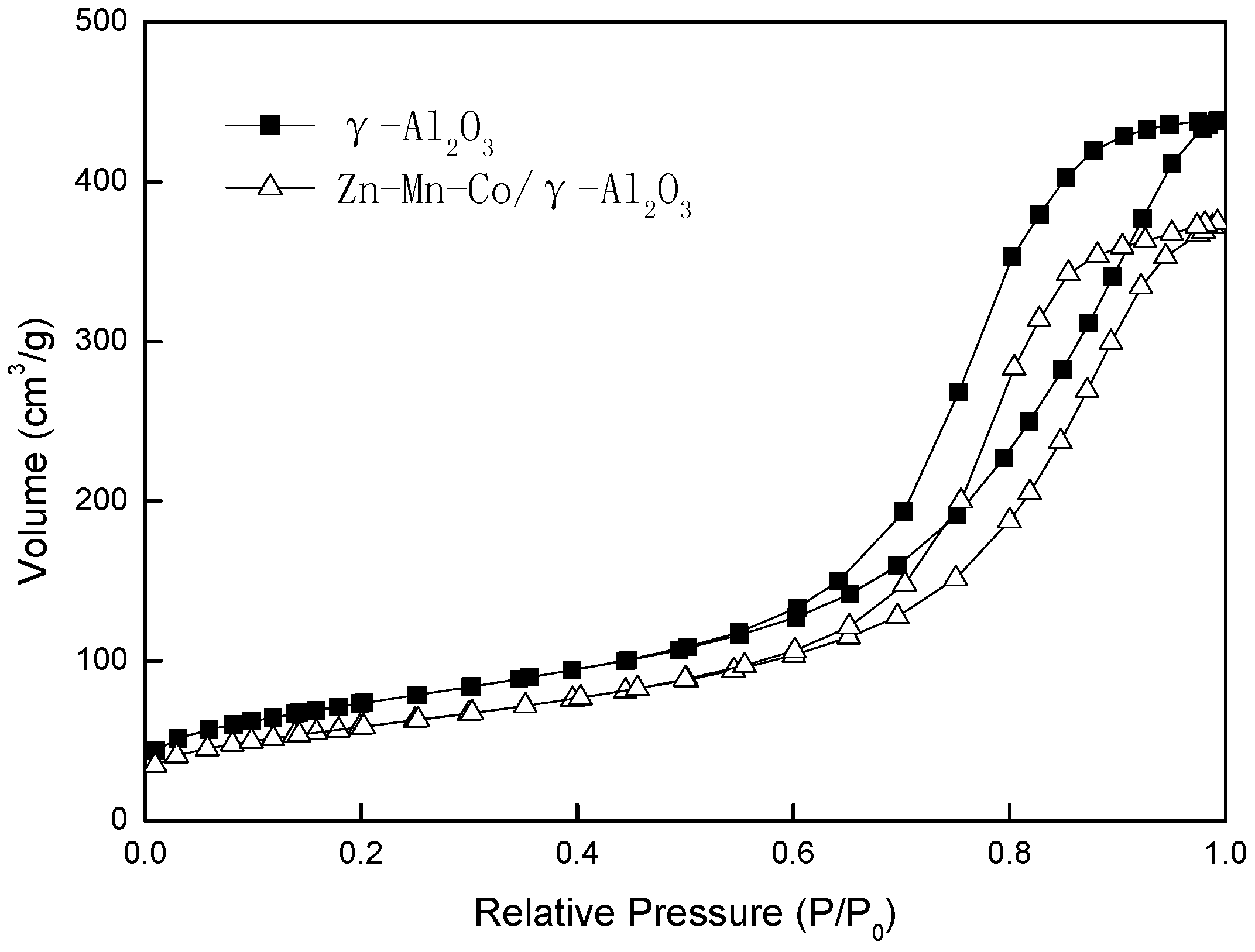
Figure 2 shows the nitrogen adsorption–desorption isotherms at liquid nitrogen temperature (77 K) for γ-Al
2O
3 and Zn-Mn-Co modified γ-Al
2O
3 samples. Both of the isotherms belong to type IV, and the close hysteresis loops were classified as H
3 [
24]. The specific surface areas, average pore size, and total pore volume of the catalysts are presented in
Table 1. The specific surface area of Zn-Mn-Co/γ-Al
2O
3 was smaller than that of γ-Al
2O
3, and the total pore volume was reduced by 15% after modification. However, the average pore radius had a slight increase. This may be due to the modification of the metal ions: some of the micropores were blocked.
Figure 3 shows the pore size distribution and cumulative pore volume obtained for the catalysts. The pore sizes of both catalysts were mainly distributed in the range of 5–20 nm. The number of micropores in the modified catalyst was less than that in the original sample, which further proved that the modification of metal ions blocked some of the micropores.
The catalytic efficiency of the catalyst is intensively affected by the surface acid sites [
8]. In general, most Lewis acids react with water before reacting with the substrate in the presence of water, which results in the deactivation of the catalyst. However, it has been well established that surface Lewis acid sites of alumina catalyze the dehydration of alcohols. Phung et al. reported that the activity of alumina was observed in the dehydration of ethanol (in which water is a reaction product) because of its high surface acidity. Lewis acidity does not require dehydroxylation if the basic substrates can displace water. Substrates that have sufficient basicity to compete with water can also be activated by alumina in the presence of water [
14,
25].
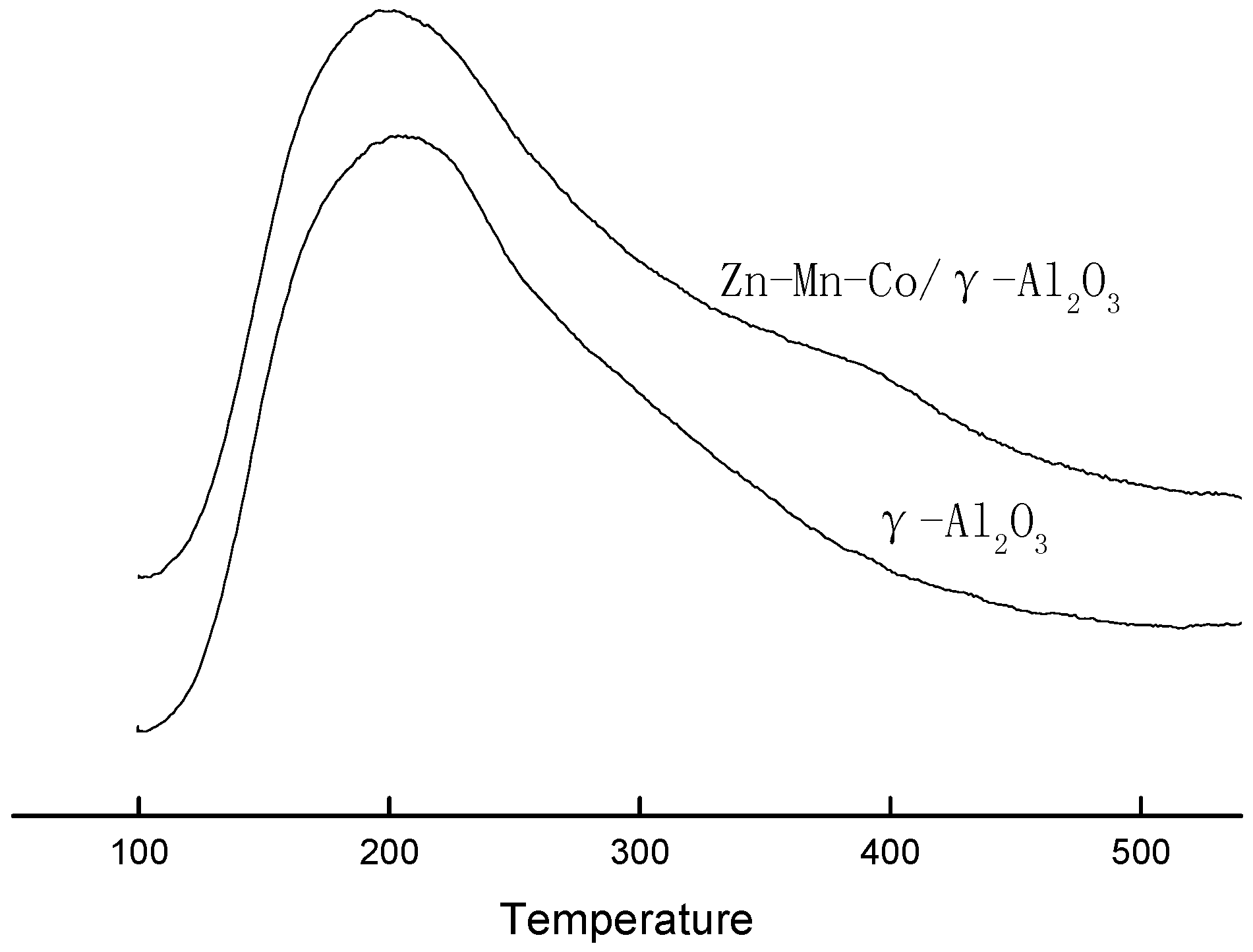
The pyridine temperature-programmed desorption (Py-TPD) profiles of γ-Al
2O
3 and Zn-Mn-Co modified γ-Al
2O
3 are shown in
Figure 4. The desorption peak at 200 °C was enhanced after modification, which proved that there were more weak acid sites on Zn-Mn-Co modified γ-Al
2O
3. A weak desorption peak appeared at 390 °C, indicating the increase of strong acid sites. The surface acidity values of γ-Al
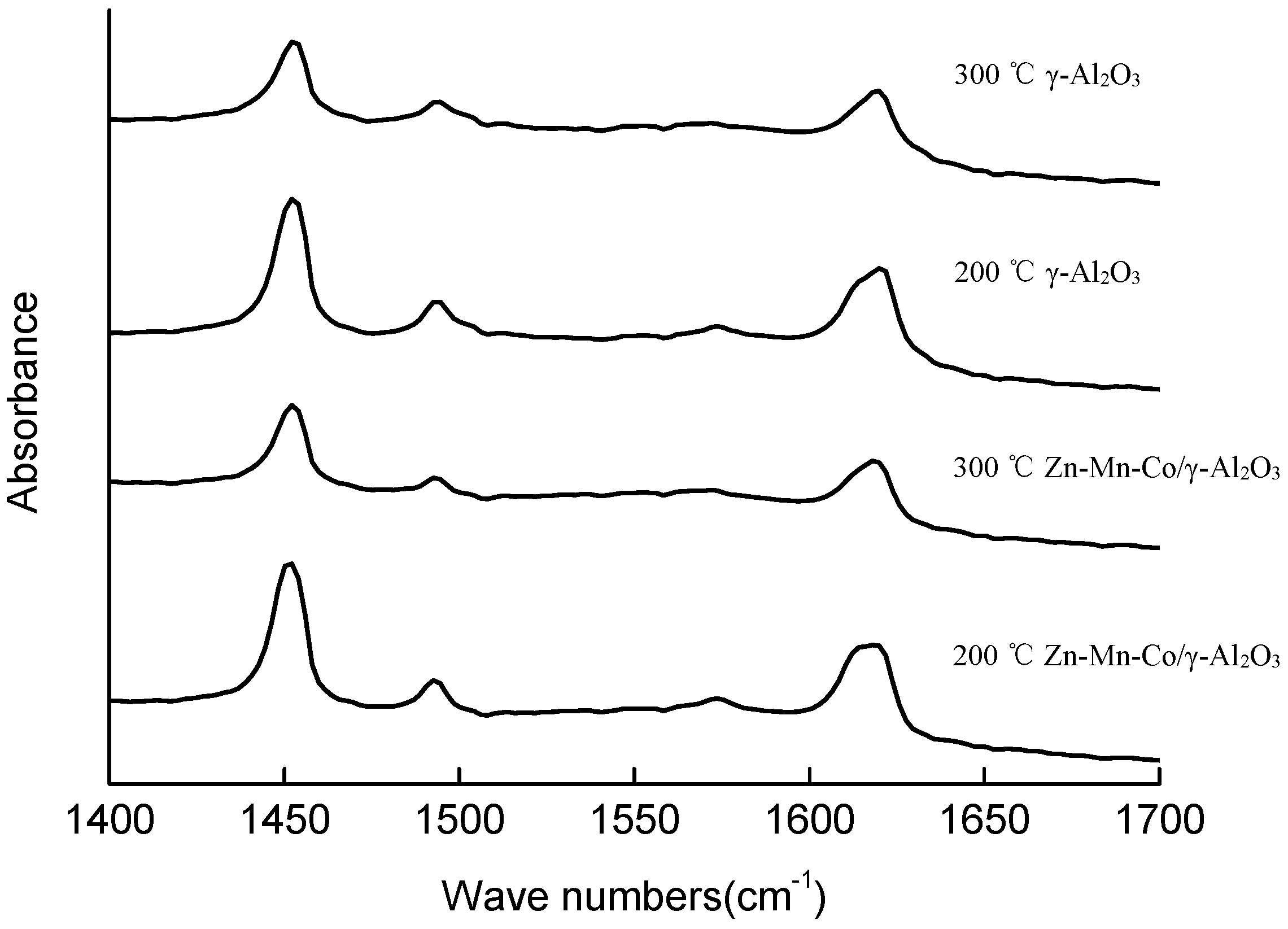
2O
3 and Zn-Mn-Co modified γ-Al
2O
3 were quantified by pyridine adsorption IR spectra (
Figure 5 and
Table 2). The bands distinguished at 1620 cm
−1 were assigned to pyridine coordinated to tetrahedral and octahedral Al
3+. The area of the 19b band at 1451 cm
−1 was used to quantify the total amount of Lewis acid sites (LAS) using its molar adsorption coefficient (ε = 1.5 cm/μmol) [
10,
26,
27,
28]. The amount of LAS is given in
Table 3. The acidic concentration of total acidity and the strong LAS were measured at 200 °C and 300 °C, respectively. After being modified by zinc, manganese, and cobalt, the total acid content of the catalyst increased by about 10.5%.
2.2. Catalytic Activity of γ-Al2O3 and Zn-Mn-Co/γ-Al2O3
The catalytic activity of γ-Al
2O
3 and Zn-Mn-Co/γ-Al
2O
3 was investigated at different reaction temperatures (
Table 3 and
Table 4) and WSHV (
Table 5 and
Table 6), with a reaction time of 6 h. The higher reaction temperature had positive effects on the conversion of 1-butanol and the selectivity of butenes, but a too high reaction temperature worked against the selectivity of butenes. The conversion rates of 1-butanol were 71.21% and 82.63% when the reaction temperature was 300 °C and 350 °C, respectively, with γ-Al
2O
3 as a catalyst, and increased by 12.82% and 15.27% when Zn-Mn-Co/γ-Al
2O
3 was employed. When the reaction temperature increased from 300 °C to 450 °C, the selectivity of butenes increased to a maximum and then decreased. The highest selectivity of butenes (96.24%) was obtained at the reaction temperature of 400 °C when using Zn-Mn-Co/γ-Al
2O
3 as the catalyst. A lower reaction temperature led to the formation of dibutyl ether (DBE) and butene, which increased with the increase of the temperature. When the reaction temperature was 450 °C, there was no DBE formation, and some other hydrocarbons such as methane, ethylene, and propylene appeared. Furthermore, the selectivity of butene isomers was strongly influenced by the reaction temperature. More 1-butene was formed at a lower temperature, and higher temperatures led to increased isomerization of 1-butene.
The conversion of 1-butanol dropped rapidly with the increase of WHSV. When the WHSV increased from 1 h−1 to 4 h−1, the conversion rate of 1-butanol decreased from 98.01% to 51.04% with γ-Al2O3 as the catalyst. The same trend was obtained when the reaction was catalyzed by Zn-Mn-Co/γ-Al2O3, and the conversion of 1-butanol was much higher than that when γ-Al2O3 was used as the catalyst. With the increase of WHSV from 1 h−1 to 4 h−1, the proportion of 1-butene in the product increased, and the proportion of butylene isomers decreased. WHSV had little effect on the total selectivity of butenes, which was about 80% and 90% when using γ-Al2O3 and Zn-Mn-Co/γ-Al2O3, respectively.
The catalytic performance of Zn-Mn-Co/γ-Al
2O
3 was much better than that of γ-Al
2O
3. The high conversion rate of 1-butanol and the selectivity of butenes indicated that Zn-Mn-Co/γ-Al
2O
3 exhibited excellent activity at low temperature. Zinc, manganese, and cobalt oxides exhibited high activity in alcohol dehydration, as already proven [
15,
16,
17,
29]. The modification of metal ions improved the acidity of the catalyst, and the Lewis acid sites played an important role in the catalytic dehydration of alcohols [
30]. A synergistic effect between different metal ions and catalyst carriers improved the catalytic efficiency [
23,
31].
2.3. Optimization of Temperature and WHSV for Butanol Dehydration
To optimize the temperature and WHSV of the 1-butanol dehydration process, experiments were performed in accordance with the conditions listed in
Table 7, which was designed by a central composite design. The values of butene yield (Y) obtained in the 1-butanol dehydration experiments at different temperatures and WHSV are shown in
Table 7. The results of ANOVA for a fitted quadratic polynomial model are shown in
Table 8. The ‘‘Model F-value” of 642.51 and the lowest
p value of less than 0.0001 implied that the model was highly significant and there was only a 0.01% chance that a “Model F-value” this large could occur because of noise. The value of
R2 for Y was 0.9978. Butene yield was significantly affected by reaction temperature and WHSV, which could be deduced from the higher absolute value of F and the lower
p value (
Table 8). The yield of butanol can be predicted by the following equation:
where X
1, X
2 are the temperature and WHSV, respectively.
X
1X
2 was significant, suggesting that temperature and WHSV interacted.
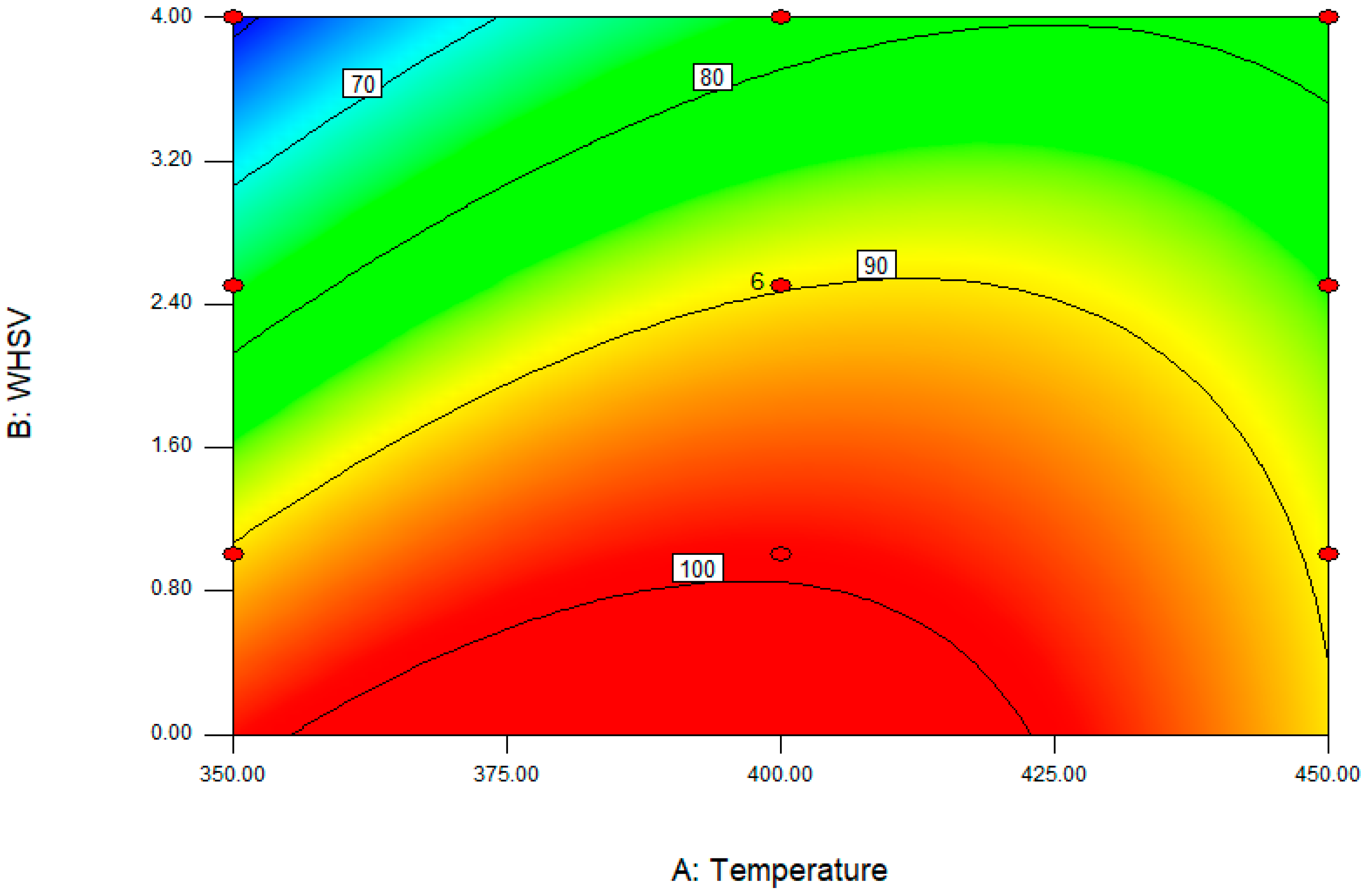
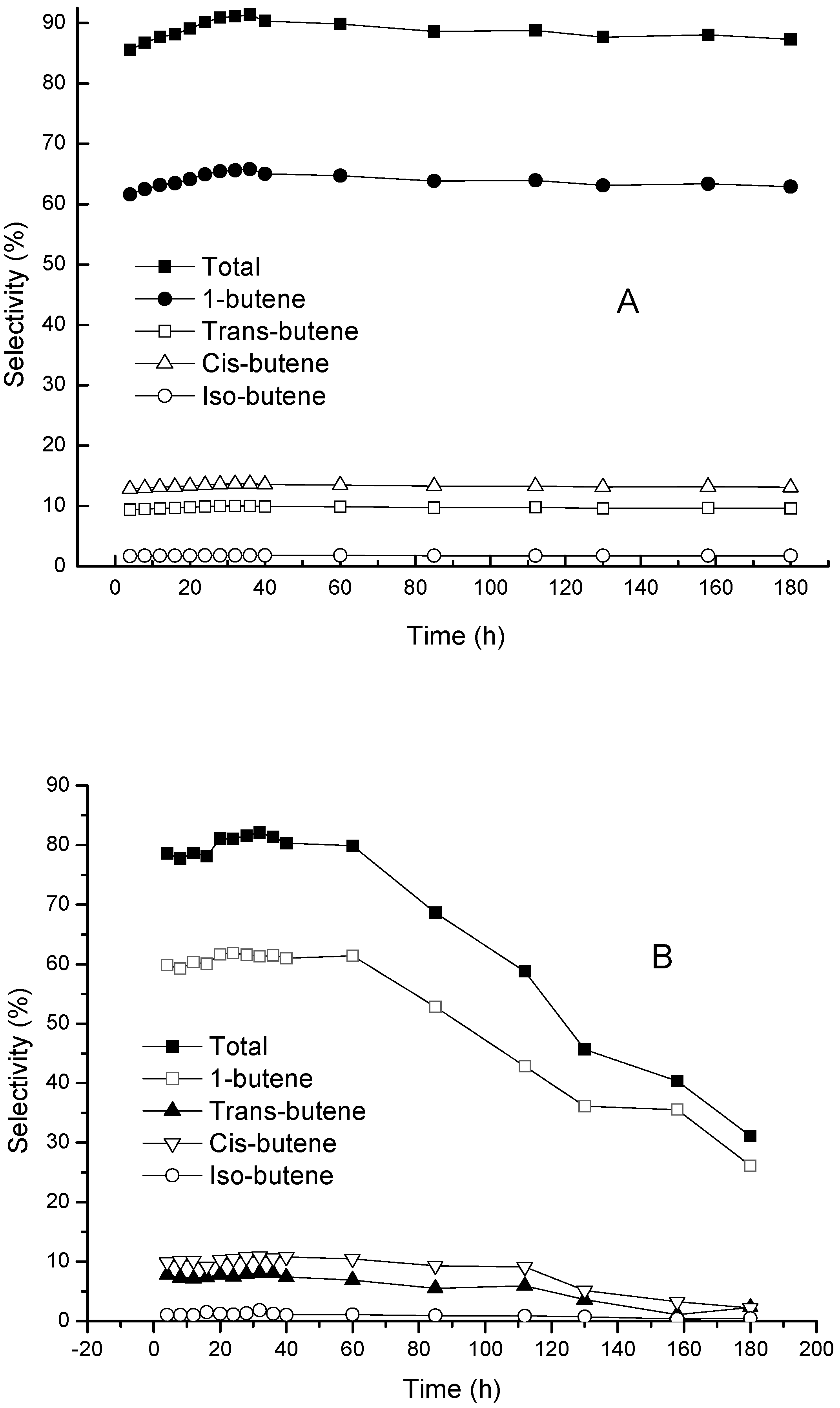
Figure 6 shows the response surface diagram of temperature and WHSV interactions for the total yield of butenes. Butene yield was enhanced as the temperature increased but reduced as WHSV increased. The temperature and WHSV were optimized by the model with the hypothetical conditions of minimum temperature, maximum WHSV, and maximum butene selectivity. The optimum conditions were found to be 375.78 °C and 1.67 h
−1 of WHSV and, under the optimum conditions, the predicted value of total yield of butenes was 92.61%. In a real catalytic reaction of 1-butanol dehydration, the total yield of butenes was 90% at optimum conditions, and the conversion of 1-butanol reached 100%, which were 10% and 6% higher than that when using unmodified γ-Al
2O
3 in the first 60 h of the reaction. After 60 h of reaction, the unmodified γ-Al
2O
3 deactivated rapidly, while the Zn-Mn-Co modified γ-Al
2O
3 showed high stability and long life (
Figure 7). Rehydration of γ-Al
2O
3 is the main reason of deactivation in the presence of water. The rehydration of γ-Al
2O
3 results in a decrease of the crystallinity of the catalyst and in a significant decrease in the specific surface area of the catalyst [
32]. In addition, there is a higher level of by-products of DBE generated with γ-Al
2O
3 as the catalyst. The DBE may be adsorbed on the surface of the catalyst which leads to the decrease of catalytic efficiency and deactivation of γ-Al
2O
3. The crystallinity of γ-Al
2O
3 was enhanced after modification by Zn-Mn-Co (
Figure 1), which improves the stability of the catalyst in the reaction environment with water [
22]. The enhancement of surface acidity reduces DBE formation, which also prolongs the life of the catalyst.
The application of γ-Al
2O
3 for the dehydration of 1-butanol has been reported, and the process temperature appeared to play a decisive role in the distribution of olefins and ethers in the products [
12]. The proportion of ethers in the product was larger than that of olefin when the reaction temperature was below 300 °C, and olefin superseded ethers with an increasing temperature [
13]. The range of reaction temperature in this study was 350–450 °C, and the main products were butenes, which is consistent with the literature. WHSV mainly affected the retention time of 1-butanol in the catalyst micro-reactor system, and high WHSV was not conducive to the adsorption of butanol by a catalyst, which resulted in a decline of the conversion rate of 1-butanol. Zinc, manganese, and cobalt oxides exhibited excellent catalytic activity in the dehydration reaction of alcohol [
15,
16,
17] and they are also used as active components to be loaded onto a catalyst carrier [
16,
19]. In this study, zinc, manganese, and cobalt were loaded onto γ-Al
2O
3, and the modified catalyst showed superior catalytic activity, which was attributed to the catalytic ability of the active components and the increase of the catalyst’s acidity. In addition, the enhancement of the crystallinity and acidity of the catalyst made it stable and gave it a long lifetime.