Surface and Void Space Analysis of the Crystal Structures of Two Lithium Bis(pentafluoroethanesulfonyl)imide Salts
Abstract
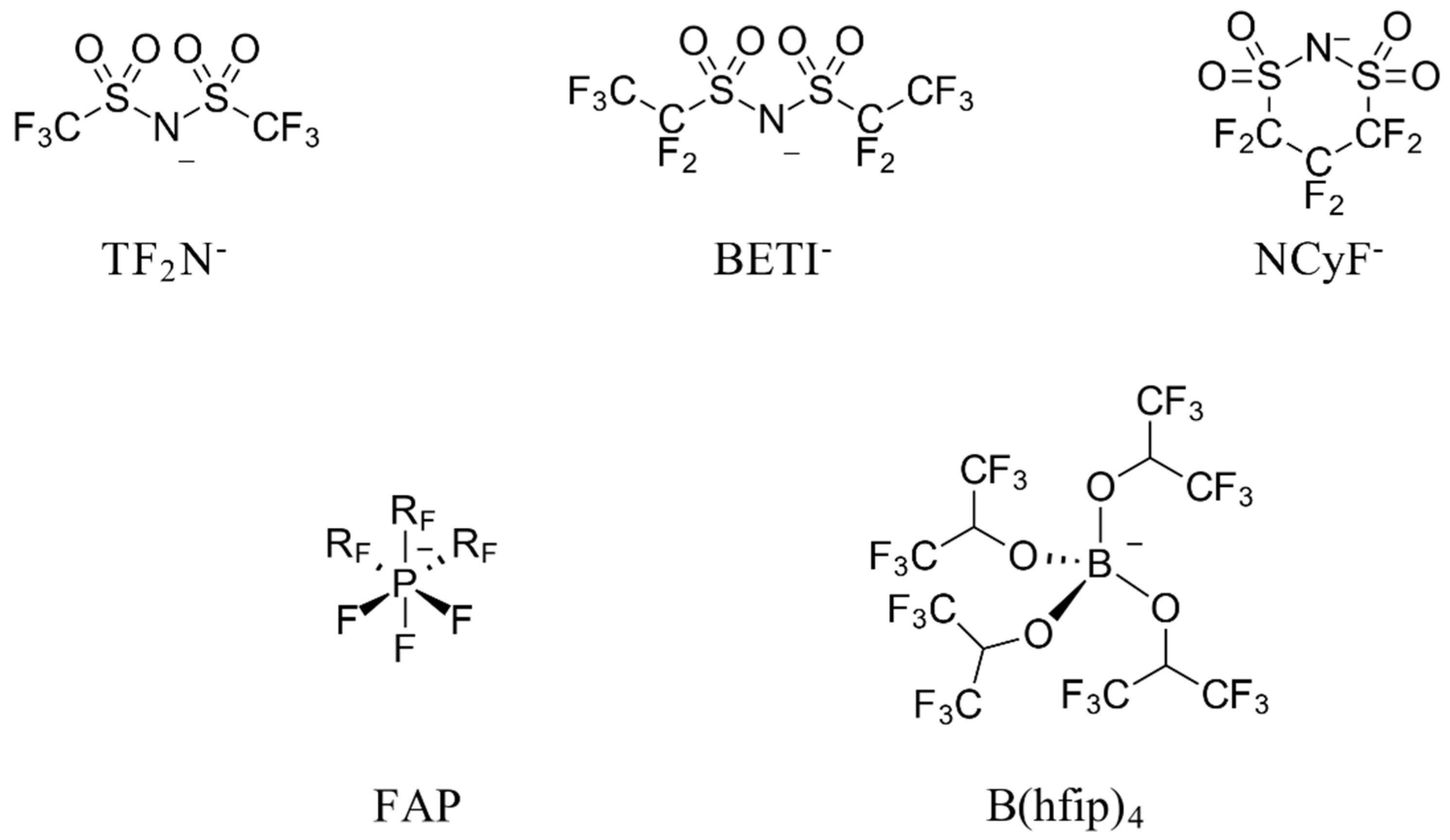
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Single Crystal Diffraction
2.3. Software
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- MacFarlane, D.R.; Kar, M.; Pringle, J.M. Fundamentals of Ionic Liquids; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017. [Google Scholar] [CrossRef]
- Wilkes, J.S.; Zaworotko, M.J. Air and Water Stable 1-Ethyl-3-Methylimidazolium Based Ionic Liquids. J. Chem. Soc. Chem. Commun. 1992, 13, 965–967. [Google Scholar] [CrossRef]
- Deetlefs, M.; Fanselow, M.; Seddon, K.R. Ionic Liquids: The View from Mount Improbable. RSC Adv. 2016, 6, 4280–4288. [Google Scholar] [CrossRef]
- Dupont, J. On the Solid, Liquid and Solution Structural Organization of Imidazolium Ionic Liquids. J. Braz. Chem. Soc. 2004, 15, 341–350. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Parvis, F.; Hossain, I.; Ma, K.; Jarošová, R.; Swain, G.M.; Blanchard, G.J. Local and Long-Range Organization in Room Temperature Ionic Liquids. Langmuir 2021, 37, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Welton, T. Ionic Liquids: A Brief History. Biophys. Rev. 2018, 10, 691–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuller, J.; Carlin, R.T.; De Long, H.C.; Haworth, D. Structure of 1-Ethyl-3-Methylimidazolium Hexafluorophosphate: Model for Room Temperature Molten Salts. J. Chem. Soc. Chem. Commun. 1994, 3, 299. [Google Scholar] [CrossRef]
- Bonhôte, P.; Dias, A.-P.; Papageorgiou, N.; Kalyanasundaram, K.; Grätzel, M. Hydrophobic, Highly Conductive Ambient-Temperature Molten Salts. Inorg. Chem. 1996, 35, 1168–1178. [Google Scholar] [CrossRef] [PubMed]
- Ignat’ev, N.V.; Welz-Biermann, U.; Kucheryna, A.; Bissky, G.; Willner, H. New Ionic Liquids with Tris(Perfluoroalkyl)Trifluorophosphate (FAP) Anions. J. Fluor. Chem. 2005, 126, 1150–1159. [Google Scholar] [CrossRef]
- Philippi, F.; Welton, T. Targeted Modifications in Ionic Liquids–from Understanding to Design. Phys. Chem. Chem. Phys. 2021, 23, 6993–7021. [Google Scholar] [CrossRef]
- Kaliner, M.; Rupp, A.; Krossing, I.; Strassner, T. Tunable Aryl Alkyl Ionic Liquids with Weakly Coordinating Tetrakis((1,1,1,3,3,3-Hexafluoropropan-2-Yl)Oxy)Borate [B(Hfip)4] Anions. Chem.–Eur. J. 2016, 22, 10044–10049. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 2016, 72, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Hashmi, A.S.K.; Braun, I.; Rudolph, M.; Rominger, F. The Role of Gold Acetylides as a Selectivity Trigger and the Importance of Gem-Diaurated Species in the Gold-Catalyzed Hydroarylating-Aromatization of Arene-Diynes. Organometallics 2012, 31, 644–661. [Google Scholar] [CrossRef]
- Cheng, Y.; Yang, J.; Hung, J.-H.; Patra, T.K.; Simmons, D.S. Design Rules for Highly Conductive Polymeric Ionic Liquids from Molecular Dynamics Simulations. Macromolecules 2018, 51, 6630–6644. [Google Scholar] [CrossRef]
- Fan, F.-L.; Qin, Z.; Cao, S.-W.; Tan, C.-M.; Huang, Q.-G.; Chen, D.-S.; Wang, J.-R.; Yin, X.-J.; Xu, C.; Feng, X.-G. Highly Efficient and Selective Dissolution Separation of Fission Products by an Ionic Liquid [Hbet][Tf2N]: A New Approach to Spent Nuclear Fuel Recycling. Inorg. Chem. 2019, 58, 603–609. [Google Scholar] [CrossRef]
- Xue, L.; Padgett, C.W.; DesMarteau, D.D.; Pennington, W.T. Synthesis and Structures of Alkali Metal Salts of Bis[(Trifluoromethyl)Sulfonyl]Imide. Solid State Sci. 2002, 4, 1535–1545. [Google Scholar] [CrossRef]
- Nockemann, P.; Thijs, B.; Pittois, S.; Thoen, J.; Glorieux, C.; Van Hecke, K.; Van Meervelt, L.; Kirchner, B.; Binnemans, K. Task-Specific Ionic Liquid for Solubilizing Metal Oxides. J. Phys. Chem. B 2006, 110, 20978–20992. [Google Scholar] [CrossRef] [Green Version]
- Fujii, K.; Fujimori, T.; Takamuku, T.; Kanzaki, R.; Umebayashi, Y.; Ishiguro, S. Conformational Equilibrium of Bis(Trifluoromethanesulfonyl) Imide Anion of a Room-Temperature Ionic Liquid: Raman Spectroscopic Study and DFT Calculations. J. Phys. Chem. B 2006, 110, 8179–8183. [Google Scholar] [CrossRef]
- McOwen, D.W.; Seo, D.M.; Borodin, O.; Vatamanu, J.; Boyle, P.D.; Henderson, W.A. Concentrated Electrolytes: Decrypting Electrolyte Properties and Reassessing Al Corrosion Mechanisms. Energy Environ. Sci. 2014, 7, 416–426. [Google Scholar] [CrossRef]
- Holbrey, J.D.; Reichert, W.M.; Rogers, R.D. Crystal Structures of Imidazolium Bis(Trifluoromethanesulfonyl)Imide ‘Ionic Liquid’ Salts: The First Organic Salt with a Cis-TFSI Anion Conformation. Dalton Trans. 2004, 15, 2267–2271. [Google Scholar] [CrossRef]
- Dean, P.M.; Pringle, J.M.; Forsyth, C.M.; Scott, J.L.; MacFarlane, D.R. Interactions in Bisamide Ionic Liquids—Insights from a Hirshfeld Surface Analysis of Their Crystalline States. New J. Chem. 2008, 32, 2121. [Google Scholar] [CrossRef] [Green Version]
- Mochida, T.; Funasako, Y.; Inagaki, T.; Li, M.-J.; Asahara, K.; Kuwahara, D. Crystal Structures and Phase-Transition Dynamics of Cobaltocenium Salts with Bis(Perfluoroalkylsulfonyl)Amide Anions: Remarkable Odd-Even Effect of the Fluorocarbon Chains in the Anion. Chem.-Eur. J. 2013, 19, 6257–6264. [Google Scholar] [CrossRef] [PubMed]
- Traver, J.; Chenard, E.; Zeller, M.; Guillet, G.L.; Lynch, W.E.; Hillesheim, P.C. Directing Cation-Cation Interactions in Thiamine Compounds: Analysis of a Series of Organic Salts Based on Vitamin B1. J. Mol. Struct. 2021, 1232, 130046. [Google Scholar] [CrossRef]
- Siegel, D.J.; Anderson, G.I.; Cyr, N.; Lambrecht, D.S.; Zeller, M.; Hillesheim, P.C.; Mirjafari, A. Molecular Design Principles of Ionic Liquids with a Sulfonyl Fluoride Moiety. New J. Chem. 2021, 45, 2443–2452. [Google Scholar] [CrossRef]
- Yoshida, Y.; Saito, G. Ionic Liquids Based on Diethylmethyl(2-Methoxyethyl)Ammonium Cations and Bis(Perfluoroalkanesulfonyl)Amide Anions: Influence of Anion Structure on Liquid Properties. Phys. Chem. Chem. Phys. 2011, 13, 20302–20310. [Google Scholar] [CrossRef] [PubMed]
- Henderson, W.A.; McKenna, F.; Khan, M.A.; Brooks, N.R.; Young, V.G.; Frech, R. Glyme–Lithium Bis(Trifluoromethanesulfonyl)Imide and Glyme–Lithium Bis(Perfluoroethanesulfonyl)Imide Phase Behavior and Solvate Structures. Chem. Mater. 2005, 17, 2284–2289. [Google Scholar] [CrossRef]
- Siraj, N.; Hasan, F.; Das, S.; Kiruri, L.W.; Steege Gall, K.E.; Baker, G.A.; Warner, I.M. Carbazole-Derived Group of Uniform Materials Based on Organic Salts: Solid State Fluorescent Analogues of Ionic Liquids for Potential Applications in Organic-Based Blue Light-Emitting Diodes. J. Phys. Chem. C 2014, 118, 2312–2320. [Google Scholar] [CrossRef]
- Yim, J.-H.; Oh, B.-K.; Lim, J.S. Solubility Measurement and Correlation of CO 2 in Bis(Pentafluoroethylsulfonyl)Imide ([BETI]) Anion-Based Ionic Liquids: [EMIM][BETI], [BMIM][BETI], [HMIM][BETI]. J. Chem. Eng. Data 2020, 65, 4378–4386. [Google Scholar] [CrossRef]
- Sun, X.; Luo, H.; Dai, S. Mechanistic Investigation of Solvent Extraction Based on Anion-Functionalized Ionic Liquids for Selective Separation of Rare-Earth Ions. Dalton Trans. 2013, 42, 8270–8275. [Google Scholar] [CrossRef]
- Tokuda, H.; Hayamizu, K.; Ishii, K.; Susan, A.B.H.; Watanabe, M. Physicochemical Properties and Structures of Room Temperature Ionic Liquids. 1. Variation of Anionic Species. J. Phys. Chem. B 2004, 108, 16593–16600. [Google Scholar] [CrossRef]
- Siu, B.; Cassity, C.G.; Benchea, A.; Hamby, T.; Hendrich, J.; Strickland, K.J.; Wierzbicki, A.; Sykora, R.E.; Salter, E.A.; O’Brien, R.A.; et al. Thermally Robust: Triarylsulfonium Ionic Liquids Stable in Air for 90 Days at 300 °C. RSC Adv. 2017, 7, 7623–7630. [Google Scholar] [CrossRef] [Green Version]
- Benchea, A.; Siu, B.; Soltani, M.; McCants, J.H.; Salter, E.A.; Wierzbicki, A.; West, K.N.; Davis, J.H.J. An Evaluation of Anion Suitability for Use in Ionic Liquids with Long-Term, High-Temperature Thermal Stability. New J. Chem. 2017, 41, 7844–7848. [Google Scholar] [CrossRef]
- Pulido, A.; Chen, L.; Kaczorowski, T.; Holden, D.; Little, M.A.; Chong, S.Y.; Slater, B.J.; McMahon, D.P.; Bonillo, B.; Stackhouse, C.J.; et al. Functional Materials Discovery Using Energy–Structure–Function Maps. Nature 2017, 543, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Bruker. Apex3 V2019.1-0, SAINT V8.40A; Bruker AXS Inc.: Madison, WI, USA, 2019. [Google Scholar]
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of Silver and Molybdenum Microfocus X-ray Sources for Single-Crystal Structure Determination. J. Appl. Crystallogr. 2015, 48, 3–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bruker. SHELXTL Suite of Programs, Version 6.14, 2000–2003, Bruker Advanced X-ray Solutions; Bruker AXS Inc.: Madison, WI, USA, 2000–2003. [Google Scholar]
- Sheldrick, G.M. A Short History of SHELX. Acta Crystallogr. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [Green Version]
- Hübschle, C.B.; Sheldrick, G.M.; Dittrich, B. ShelXle: A Qt Graphical User Interface for SHELXL. J. Appl. Crystallogr. 2011, 44, 1281–1284. [Google Scholar] [CrossRef] [Green Version]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Spackman, P.R.; Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer: A Program for Hirshfeld Surface Analysis, Visualization and Quantitative Analysis of Molecular Crystals. J. Appl. Crystallogr. 2021, 54, 1006–1011. [Google Scholar] [CrossRef]
- Spackman, M.A.; McKinnon, J.J. Fingerprinting Intermolecular Interactions in Molecular Crystals. CrystEngComm 2002, 4, 378–392. [Google Scholar] [CrossRef]
- Spackman, M.A.; McKinnon, J.J.; Jayatilaka, D. Electrostatic Potentials Mapped on Hirshfeld Surfaces Provide Direct Insight into Intermolecular Interactions in Crystals. CrystEngComm 2008, 10, 377–388. [Google Scholar] [CrossRef]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; van de Streek, J.; Wood, P.A. Mercury CSD 2.0—New Features for the Visualization and Investigation of Crystal Structures. J. Appl. Crystallogr. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Spackman, M.A.; Jayatilaka, D. Hirshfeld Surface Analysis. CrystEngComm 2009, 11, 19–32. [Google Scholar] [CrossRef]
- McKinnon, J.J.; Spackman, M.A.; Mitchell, A.S. Novel Tools for Visualizing and Exploring Intermolecular Interactions in Molecular Crystals. Acta Crystallogr. B 2004, 60, 627–668. [Google Scholar] [CrossRef] [PubMed]
- Rybalova, T.V.; Bagryanskaya, I.Y. C-F…π, F…H, and F…F Intermolecular Interactions and F-Aggregation: Role in Crystal Engineering of Fluoroorganic Compounds. J. Struct. Chem. 2009, 50, 741–753. [Google Scholar] [CrossRef]
- Arkhipov, D.E.; Lyubeshkin, A.V.; Volodin, A.D.; Korlyukov, A.A. Molecular Structures Polymorphism the Role of F…F Interactions in Crystal Packing of Fluorinated Tosylates. Crystals 2019, 9, 242. [Google Scholar] [CrossRef] [Green Version]
- Bauzá, A.; Frontera, A. Electrostatically Enhanced F⋯F Interactions through Hydrogen Bonding, Halogen Bonding and Metal Coordination: An Ab Initio Study. Phys. Chem. Chem. Phys. 2016, 18, 20381–20388. [Google Scholar] [CrossRef] [Green Version]
- Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. The Halogen Bond. Chem. Rev. 2016, 116, 2478–2601. [Google Scholar] [CrossRef] [Green Version]
- Varadwaj, P.; Varadwaj, A.; Marques, H. Halogen Bonding: A Halogen-Centered Noncovalent Interaction Yet to Be Understood. Inorganics 2019, 7, 40. [Google Scholar] [CrossRef] [Green Version]
- Scheiner, S. F-Halogen Bond: Conditions for Its Existence. J. Phys. Chem. A 2020, 124, 7290–7299. [Google Scholar] [CrossRef]
- Bernstein, J.; Davis, R.E.; Shimoni, L.; Chang, N.-L. Patterns in Hydrogen Bonding: Functionality and Graph Set Analysis in Crystals. Angew. Chem. Int. Ed. Engl. 1995, 34, 1555–1573. [Google Scholar] [CrossRef]
- Bissantz, C.; Kuhn, B.; Stahl, M. A Medicinal Chemist’s Guide to Molecular Interactions. J. Med. Chem. 2010, 53, 5061–5084. [Google Scholar] [CrossRef]
- Dunitz, J.D.; Gavezzotti, A. How Molecules Stick Together in Organic Crystals: Weak Intermolecular Interactions. Chem. Soc. Rev. 2009, 38, 2622. [Google Scholar] [CrossRef] [PubMed]
- Scilabra, P.; Terraneo, G.; Resnati, G. The Chalcogen Bond in Crystalline Solids: A World Parallel to Halogen Bond. Acc. Chem. Res. 2019, 52, 1313–1324. [Google Scholar] [CrossRef] [PubMed]










| Li{BETI} | Li{BETI}(H2O)2 | |
|---|---|---|
| Chemical formula | C4F10LiNO4S2 | C4H4F10LiNO6S2 |
| Mr | 387.11 | 423.14 |
| Crystal system, space group | Monoclinic, C2/c | Triclinic, P |
| Temperature (K) | 150 | 150 |
| a, b, c (Å) | 42.733 (5), 5.4927 (7), 10.4751 (14) | 6.4581 (3), 8.0725 (4), 13.0495 (5) |
| α (°) | 90 | 95.160 (3) |
| β (°) | 99.632 (11) | 103.406 (3) |
| γ (°) | 90 | 91.403 (3) |
| V (Å3) | 2424.0 (5) | 658.37 (5) |
| Z | 8 | 2 |
| Radiation type | Cu Kα | Mo Kα |
| μ (mm−1) | 5.50 | 0.56 |
| Crystal size (mm) | 0.21 × 0.15 × 0.02 | 0.20 × 0.05 × 0.03 |
| Data collection | ||
| Diffractometer | Bruker AXS D8 Quest diffractometer with PhotonIII_C14 charge-integrating and photon counting pixel array detector | Bruker AXS D8 Quest diffractometer with PhotonII charge-integrating pixel array detector (CPAD) |
| Absorption correction | Multi-scan SADABS 2016/2 | Multi-scan SADABS 2016/2 |
| Tmin, Tmax | 0.503, 0.754 | 0.676, 0.747 |
| No. of measured, independent and observed [I > 2σ(I)] reflections | 7332, 2443, 1991 | 21544, 4985, 3337 |
| Rint | 0.097 | 0.066 |
| (sin θ/λ)max (Å−1) | 0.638 | 0.770 |
| Refinement | ||
| R[F2 > 2σ(F2)], wR(F2), S | 0.042, 0.112, 1.05 | 0.042, 0.091, 1.01 |
| No. of reflections | 2443 | 4985 |
| No. of parameters | 199 | 229 |
| Δρmax, Δρmin (e Å−3) | 0.36, −0.38 | 0.49, −0.54 |
| [Li{BETI}] | [Li{BETI}(H2O)2] | |
|---|---|---|
| F···All | 69.5% | 54.9% |
| O···All | 19.9% | 21.2% |
| N···All | 2.5% | 2.4% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Collart, A.; Zeller, M.; Hillesheim, P.C. Surface and Void Space Analysis of the Crystal Structures of Two Lithium Bis(pentafluoroethanesulfonyl)imide Salts. Crystals 2022, 12, 701. https://doi.org/10.3390/cryst12050701
Collart A, Zeller M, Hillesheim PC. Surface and Void Space Analysis of the Crystal Structures of Two Lithium Bis(pentafluoroethanesulfonyl)imide Salts. Crystals. 2022; 12(5):701. https://doi.org/10.3390/cryst12050701
Chicago/Turabian StyleCollart, Anne, Matthias Zeller, and Patrick C. Hillesheim. 2022. "Surface and Void Space Analysis of the Crystal Structures of Two Lithium Bis(pentafluoroethanesulfonyl)imide Salts" Crystals 12, no. 5: 701. https://doi.org/10.3390/cryst12050701






