Silica–Chitosan Composite Aerogels for Thermal Insulation and Adsorption
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of SAs
2.3. Preparation of CAs
2.4. Preparation of SCAs
2.5. Characterization
3. Results
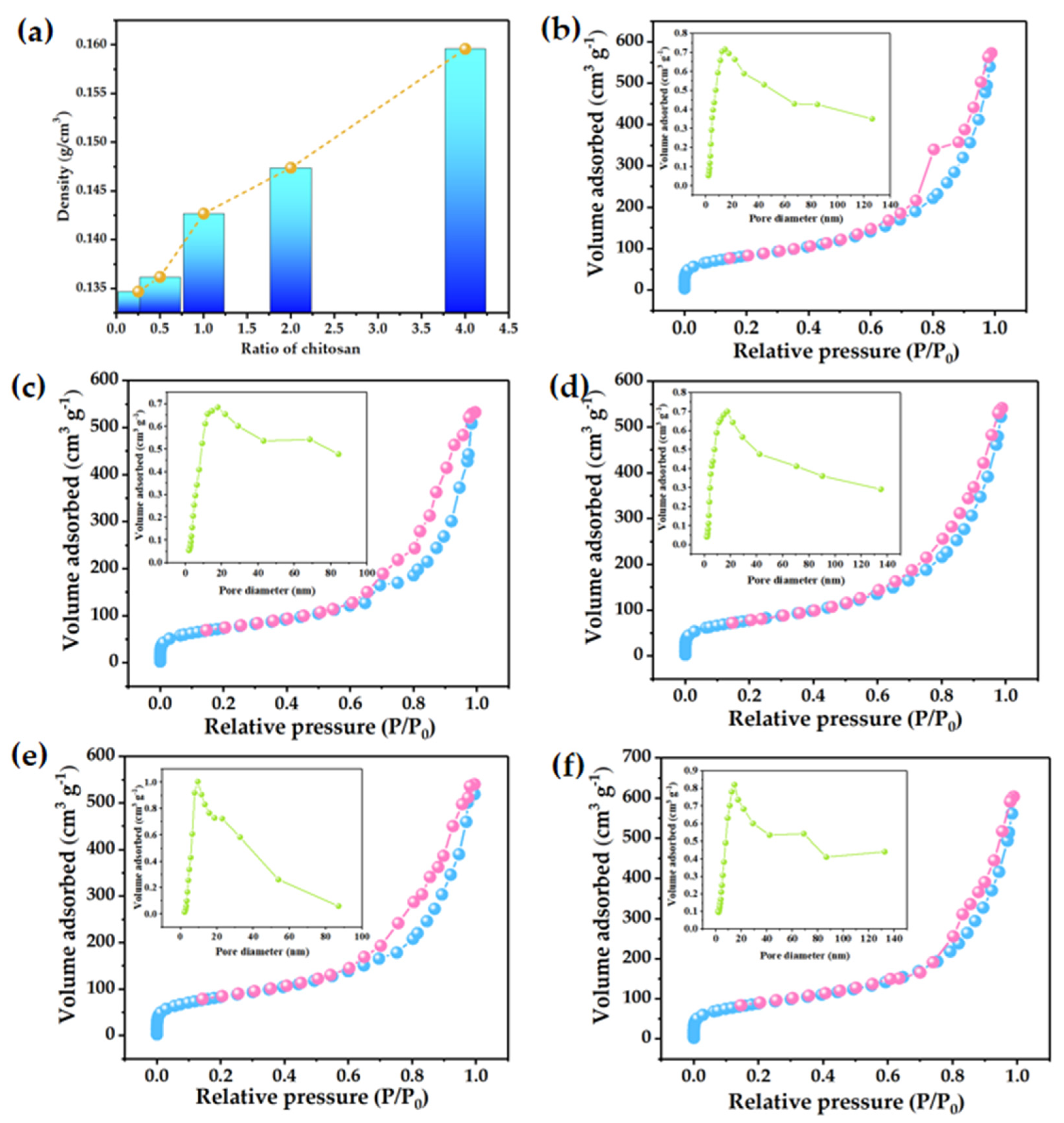
3.1. Morphology Analysis of SCAs
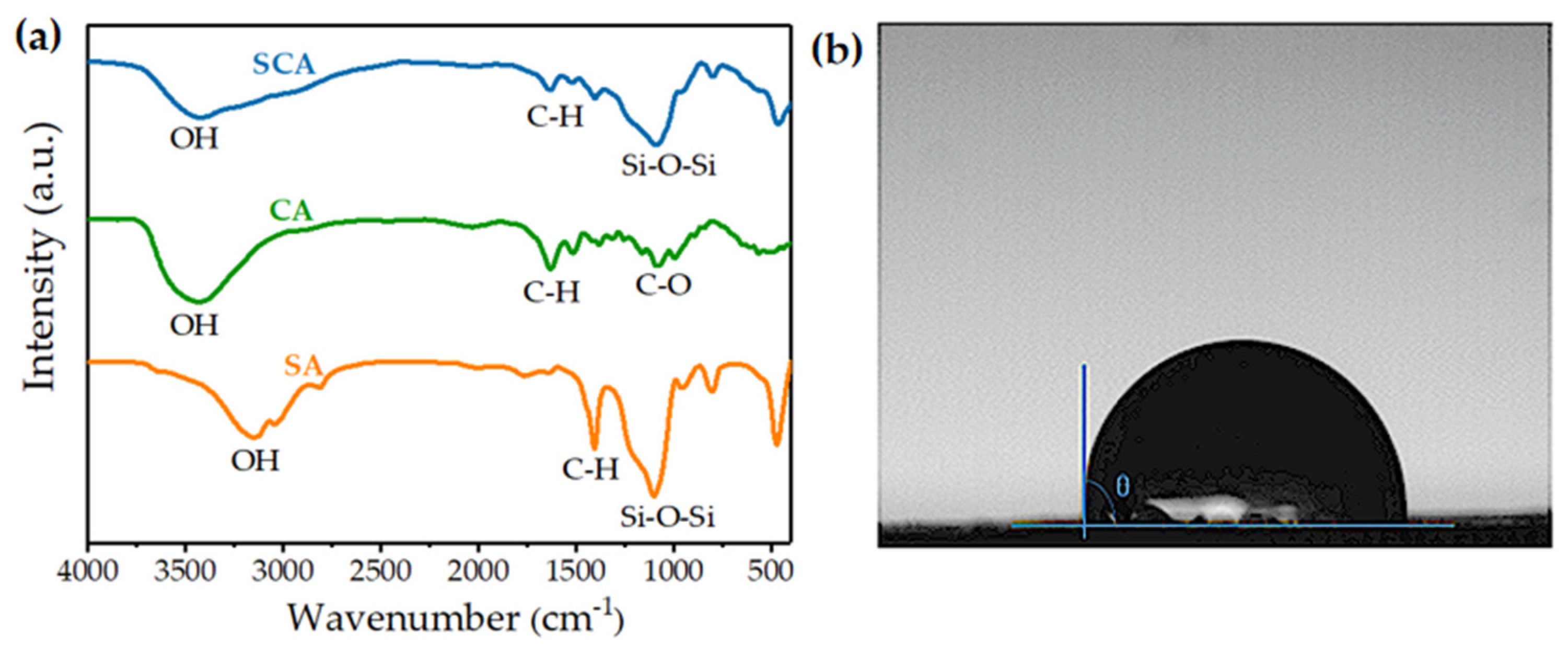
3.2. Structural Analysis of SCAs
3.3. Analysis of Heat Conduction, Heat Insulation and Thermal Stability of SCAs
3.4. Study on Hydrophilic/Hydrophobic Properties of SCA
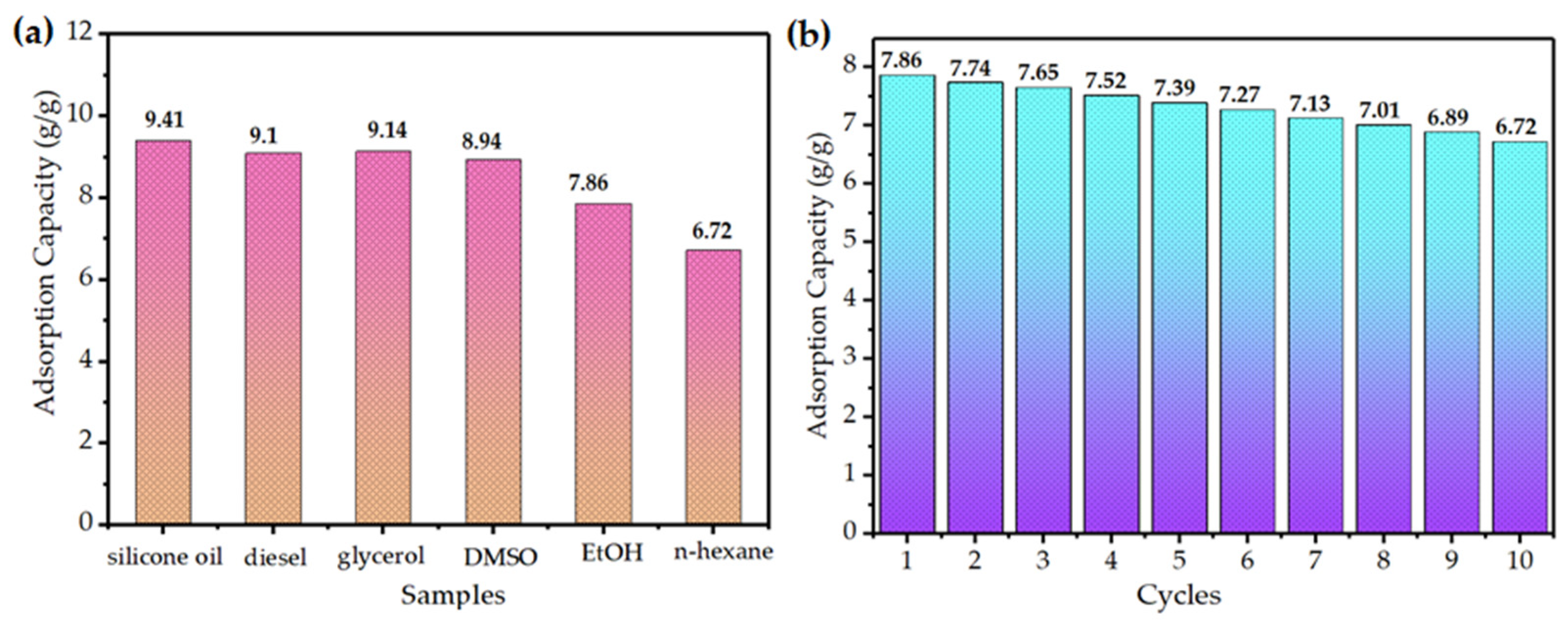
3.5. Adsorption Performance of SCA
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Baetens, R.; Jelle, B.P.; Gustavsen, A. Aerogel insulation for building applications: A state-of-the-art review. Energy Build. 2011, 43, 761–769. [Google Scholar] [CrossRef]
- Yang, J.; Wu, H.; Xu, X.; Huang, G.; Xu, T.; Guo, S.; Liang, Y. Numerical and experimental study on the thermal performance of aerogel insulating panels for building energy efficiency. Renew. Energy 2019, 138, 445–457. [Google Scholar] [CrossRef]
- Wang, L.; Feng, J.; Jiang, Y.; Zhang, S.; Li, L.; Feng, J. Facile fabrication of hydrophobic polyvinylpolysilsesquioxane aerogels with improved optical properties. J. Sol-Gel Sci. Technol. 2020, 94, 88–97. [Google Scholar] [CrossRef]
- Štandeker, S.; Novak, Z.; Knez, Ž. Adsorption of toxic organic compounds from water with hydrophobic silica aerogels. J. Colloid Interface Sci. 2007, 310, 362–368. [Google Scholar] [CrossRef]
- Hayase, G.; Kanamori, K.; Maeno, A.; Kaji, H.; Nakanishi, K. Dynamic spring-back behavior in evaporative drying of polymethylsilsesquioxane monolithic gels for low-density transparent thermal superinsulators. J. Non-Cryst. Solids 2016, 434, 115–119. [Google Scholar] [CrossRef]
- Omranpour, H.; Motahari, S. Effects of processing conditions on silica aerogel during aging: Role of solvent, time and temperature. J. Non-Cryst. Solids 2013, 379, 7–11. [Google Scholar] [CrossRef]
- Chen, F.; Zhang, Y.; Liu, J.; Wang, X.; Chu, P.K.; Chu, B.; Zhang, N. Fly ash based lightweight wall materials incorporating expanded perlite/SiO2 aerogel composite: Towards low thermal conductivity. Constr. Build. Mater. 2020, 249, 118728. [Google Scholar] [CrossRef]
- Seo, J.; Bae, S.J.; Jang, D.I.; Park, S.; Yang, B.; Lee, H.K. Thermal behavior of alkali-activated fly ash/slag with the addition of an aerogel as an aggregate replacement. Cem. Concr. Compos. 2020, 106, 103462. [Google Scholar] [CrossRef]
- Long, E.; Zang, Z.; Ma, X. Are the energy conservation rates (RVRs) approximate in different cities for the same building with the same outer-wall thermal insulation measures. Build. Environ. 2005, 40, 537–544. [Google Scholar]
- Meng, X.; Yan, B.; Gao, Y.; Wang, J.; Zhang, W.; Long, E. Factors affecting the in situ measurement accuracy of the wall heat transfer coefficient using the heat flow meter method. Energy Build. 2015, 86, 754–765. [Google Scholar] [CrossRef]
- Zhao, F.; Zhu, J.; Peng, T.; Liu, H.; Ge, S.; Xie, H.; Xie, L.; Jiang, C. Preparation of functionalized halloysite reinforced polyimide composite aerogels with excellent thermal insu-lation properties. Appl. Clay Sci. 2021, 211, 160200. [Google Scholar] [CrossRef]
- Jung, S.B.; Park, S.W.; Yang, J.K.; Park, H.H.; Kim, H. Application of SiO2 aerogel film for interlayer dielectric on GaAs with a barrier of Si3N4. Thin Solid Film. 2004, 447–448, 580–585. [Google Scholar] [CrossRef]
- Liu, G.; Guo, Y.; Jian, Z.; Huang, M.; Zhao, T. Experimental Study on the Thermal Conductivity of Improved Graphite Composite Insulation Boards. Crystals 2023, 13, 102. [Google Scholar] [CrossRef]
- Joubert, F.; Musa, O.M.; Hodgson, D.R.W.; Cameron, N.R. The preparation of graft copolymers of cellulose and cellulose derivatives using ATRP under homogeneous reaction conditions. Chem. Soc. Rev. 2014, 43, 7217–7235. [Google Scholar] [CrossRef] [PubMed]
- Yoda, S.; Takeshita, S.; Ono, T.; Tada, R.; Ota, H. Development of a new silica aerogel-polypropylene foam composite as a highly flexible thermal insulation material. Front. Mater. 2021, 8, 2296–8016. [Google Scholar] [CrossRef]
- Kucharek, M.; MacRae, W.; Yang, L. Investigation of the effects of silica aerogel part icles on thermal and mechanical properties of epoxy composites. Compos. Part A 2020, 139, 106108. [Google Scholar] [CrossRef]
- Liu, C.; Wu, S.; Yang, Z.; Sun, H.; Zhu, Z.; Liang, W.; Li, A. Mechanically robust and flame-retardant silicon aerogel elastomers for thermal insulation and efficient solar steam generation. ACS Omega 2020, 5, 8638–8646. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Le, D.; Nguyen, S.T.; Nien, V.T.C.; Jewell, D.; Duong, H.M. Silica cellulose hybrid aerogels for thermal and acoustic insulation applications. Colloids Surf. A 2016, 506, 298–305. [Google Scholar] [CrossRef]
- Jiang, S.; Zhang, M.; Li, M.; Liu, L.; Liu, L.F.; Yu, J. Cellulose nanofibril (CNF) based aerogels prepared by a facile process and the investigation of thermal insulation performance. Cellulose 2020, 27, 6217–6233. [Google Scholar] [CrossRef]
- Li, Z.; Cheng, X.; He, S.; Shi, X.; Gong, L.; Zhang, H. Aramid fibers reinforced silica aerogel composites with low thermal conductivity and improved mechanical performance. Compos. Part A 2016, 84, 316–325. [Google Scholar] [CrossRef]
- Alves, P.; Dias, D.A.; Pontinha, A.D.R. Silica Aerogel-Rubber Composite: A Sustainable Alternative for Buildings’ Thermal Insulation. Molecules 2022, 27, 7127. [Google Scholar] [CrossRef]
- Hamdan, H.; Halim, Z.A.A.; Ahmad, N.; Yajid, M.A.M. Thermal insulation performance of silicone rubber/silica aerogel composite. Mater. Chem. Phys. 2022, 276, 125359. [Google Scholar]
- Merillas, B.; Lamy-Mendes, A.; Villafañe, F.; Durães, L.; Rodríguez-Pérez, M.Á. Polyurethane foam scaffold for silica aerogels: Effect of cell size on the mechanical properties and thermal insulation. Mater. Today Chem. 2022, 26, 101257. [Google Scholar] [CrossRef]
- Merillas, B.; Lamy-Mendes, A.; Villafañe, F.; Durães, L.; Rodríguez-Pérez, M.Á. Silica-Based Aerogel Composites Reinforced with Reticulated Polyurethane Foams: Thermal and Mechanical Properties. Gels 2022, 8, 392. [Google Scholar] [CrossRef]
- Linhares, T.; Amorim, M.T.P.; Durães, L. Silica aerogel composites with embedded fibres: A review on their preparation, properties and applications. J. Mater. Chem. 2019, 7, 22768–22802. [Google Scholar] [CrossRef]
- Xiao, W.; Wang, P.; Song, X.; Liao, B.; Yan, K.; Zhang, J. Facile fabrication of anisotropic chitosan aerogel with hydrophobicity and thermal superinsulation for advanced thermal management. ACS Sustain. Chem. Eng. 2021, 9, 9348–9357. [Google Scholar] [CrossRef]
- Cui, H.; Wu, N.; Ma, X.; Niu, F. Superior intrinsic flame-retardant phosphorylated chitosan aerogel as fully sustainable thermal insulation bio-based material. Polym. Degrad. Stab. 2023, 207, 110213. [Google Scholar] [CrossRef]
- Zhang, M.; Jiang, S.; Han, F.; Li, M.; Wang, N.; Liu, L. Anisotropic cellulose nanofiber/chitosan aerogel with thermal management and oil absorption properties. Carbohyd. Polym. 2021, 264, 0144–8617. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Qiu, C.; Zhang, Y.; Zhang, X.; Yuan, Z.; Tan, H.; Wang, L.; de Hoop, C.F.; Qi, J.; Huang, X. Effective Adsorption of Diclofenac Sodium from Wastewater by Nanocellulose/Alum/Chitosan/Polyethyleneimine Biohybrid Aerogel Beads Adsorbent. J. Polym. Environ. 2023, 31, 1129–1143. [Google Scholar] [CrossRef]
- Zhang, W.; Huang, T.; Ren, Y.; Yang, S.; Zhao, X.; Yuan, M.; Wang, J.; Tu, Q. A multifunctional chitosan composite aerogel for PPCPs adsorption. Carbohydr. Polym. 2022, 298, 120102. [Google Scholar] [CrossRef]
- Guo, Q.; Amendola, E.; Lavorgna, M.; Li, Z.; Feng, H.; Wu, Y.; Fei, G.; Wang, Z.; Xia, H. Robust and recyclable graphene/chitosan composite aerogel microspheres for adsorption of oil pollutants from water. Carbohydr. Polym. 2022, 290, 119416. [Google Scholar] [CrossRef] [PubMed]
- Jaxel, J.; Markevicius, G.; Rigacci, A.; Budtova, T. Thermal superinsulating silica aerogels reinforced with short man-made cellulose fibers. Compos. Part A 2014, 103, 113–121. [Google Scholar] [CrossRef]
- Appostolopoulou-Kalkavoura, V.; Munier, P.; Bergström, L. Thermally insulating nanocellulose-based materials. Adv. Mate. 2021, 33, 20001839. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, X.; Shen, J. Effect of organic-inorganic crosslinking degree on mechanical and thermal properties of aerogel skeleton. Inorg. Mate. 2020, 35, 454–460. [Google Scholar]
- Hamann, T.W.; Martinson, A.B.F.; Elam, J.W.; Elam, J.W.; Pellin, M.J.; Hupp, J.T. Atomic layer deposition of TiO2 on aerogel templates: New photoanodes for dye-sensitized solar cells. J. Phys. Chem. 2008, 112, 10303–10307. [Google Scholar] [CrossRef]
- Lou, F.; Dong, S.; Zhu, K.; Chen, X.; Ma, Y. Thermal Insulation Performance of Aerogel Nano-Porous Materials: Characterization and Test Methods. Gels 2023, 9, 220. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Z.; Zhang, J.; Ai, S.; Tang, H. Ultralight and thermal insulation carbon foam/SiO2 aerogel composites. J. Porous Mater. 2019, 26, 1305–1312. [Google Scholar] [CrossRef]
- Chen, Y.X.; Klima, K.m.; Brouwers, H.j.H.; Yu, Q. Effect of silica aerogel on thermal insulation and acoustic absorption of geopolymer foma composites: The role of aerogel particle size. Composites Part B 2022, 242, 110048. [Google Scholar] [CrossRef]
- Yan, K.; Zhang, Z.; Liu, Y.; Li, S.; Chen, D.; Li, Z. Superhydrophobic tough hierarchical porous thermal insulation composites prepared by in situ formation. J. Appl. Polym. Sci. 2022, 139, 52806. [Google Scholar]
- Laskowski, J.; Milow, B.; Ratke, L. Aeerogel-aerogel composites for normal temperature range thermal insulation. J. Non-Cryst. Solids. 2016, 441, 42–48. [Google Scholar] [CrossRef]
- Wei, G.; Liu, Y.; Zhang, X.; Yu, F.; Du, X. Thermal conductivities study on silica aerogel and its composite insulation materials. Int. J. Heat Mass Transfer. 2011, 54, 2355–2366. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mei, X.; Li, S.; Chen, Y.; Huang, X.; Cao, Y.; Guro, V.P.; Li, Y. Silica–Chitosan Composite Aerogels for Thermal Insulation and Adsorption. Crystals 2023, 13, 755. https://doi.org/10.3390/cryst13050755
Mei X, Li S, Chen Y, Huang X, Cao Y, Guro VP, Li Y. Silica–Chitosan Composite Aerogels for Thermal Insulation and Adsorption. Crystals. 2023; 13(5):755. https://doi.org/10.3390/cryst13050755
Chicago/Turabian StyleMei, Xueli, Shihao Li, Yaoyao Chen, Xueli Huang, Yali Cao, Vitaliy P. Guro, and Yizhao Li. 2023. "Silica–Chitosan Composite Aerogels for Thermal Insulation and Adsorption" Crystals 13, no. 5: 755. https://doi.org/10.3390/cryst13050755
APA StyleMei, X., Li, S., Chen, Y., Huang, X., Cao, Y., Guro, V. P., & Li, Y. (2023). Silica–Chitosan Composite Aerogels for Thermal Insulation and Adsorption. Crystals, 13(5), 755. https://doi.org/10.3390/cryst13050755






