Melt Blending Modification of Commercial Polystyrene with Its Half Critical Molecular Weight, High Ion Content Ionomer, Poly(styrene–ran–cinnamic Acid) Zn Salt, toward Heat Resistance Improvement
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Suspension Free-Radical Copolymerization of Styrene and CA
2.3. Gel Permeation Chromatography (GPC)
2.4. Acid–Base Titration
2.5. Melt Neutralization of the Potential S with Excess ZnO
2.6. Fourier Transform Infrared (FTIR) Spectroscopy
2.7. 1H Nuclear Magnetic Resonance (NMR) Spectroscopy
2.8. Thermogravimetric Analysis (TGA)
2.9. Intrinsic Viscosity Measurement
2.10. Melt Blending of PS and the Zn-Salt SCA Ionomer
2.11. Oscillatory Shear Rheometry
2.12. Optical Microscopy
2.13. Differential Scanning Calorimetry (DSC)
2.14. Vicat Softening Temperature Testing
2.15. Mechanical Properties Testing
3. Results
3.1. Evidence of Success in Synthesizing Low (Half Critical) Molecular Weight High Ion Content Zn-salt SCA Ionomer
3.2. Minimization of a Processability Mismatch between PS and the SCA Zn Salt during their Melt Blending
3.3. An Effective Improvement in the Heat Resistance of PS upon its Blending Modification with a Significant Fraction of the SCA Zn Salt
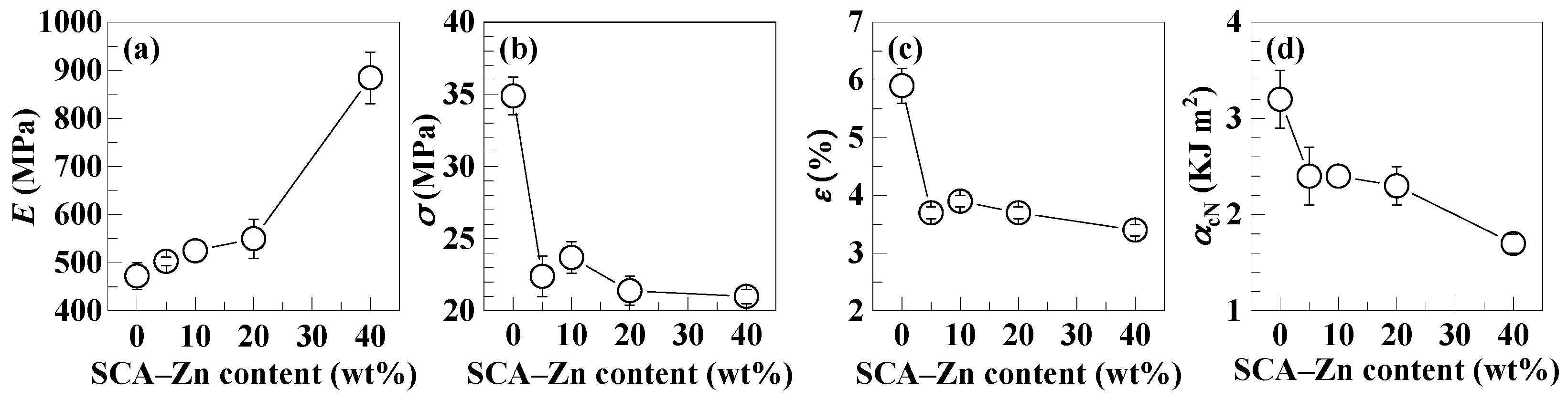
3.4. Deteriorations in the Mechanical Strengths of PS upon its Incorporation of the SCA Zn Salt
3.5. A Controlled Reduction in the Processability with Increasing the SCA Zn Salt Content of the PS/SCA Zn Salt Blends
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bastida, S.; Eguiazábal, J.I.; Nazábal, J. The Vicat softening temperature as a method to assess the phase behaviour of amorphous polymer blends. Polym. Test. 1993, 12, 233–242. [Google Scholar] [CrossRef]
- Aroğuz, A.Z.; Baysal, B.M. Thermal, mechanical, and morphological characterization studies of poly(2,6-dimethyl-1,4-phenylene oxide) blends with polystyrene and brominated polystyrene. J. Appl. Polym. Sci. 2000, 75, 225–231. [Google Scholar]
- Taşdemir, M.; Uluğ, E. Mechanical, morphological and thermal properties of SEBS, SIS and SBR-type thermoplastic elastomers toughened high impact polystyrene. Polym.-Plast. Tech. Eng. 2012, 51, 164–169. [Google Scholar] [CrossRef]
- Parres, F.; Crespo, J.E. Degradation of high-impact polystyrene with processing and its recovery via the addition of styrene–butadiene rubber and styrene–ethylene–butylene–styrene block copolymer. J. Appl. Polym. Sci. 2001, 121, 574–581. [Google Scholar] [CrossRef]
- Taşdemir, M.; Yildirim, H. Effect of styrene-butadiene-styrene addition on polystyrene/high-density polyethylene blends. J. Appl. Polym. Sci. 2002, 83, 2967–2975. [Google Scholar] [CrossRef]
- Dolores, S.M.; Patricia, A.M.; Santiago, F.; Juan, L. Influence of biodegradable materials in the recycled polystyrene. J. Appl. Polym. Sci. 2014, 131, 41161. [Google Scholar] [CrossRef]
- Bouslah, N.; Hammachin, R.; Amrani, F. Study of the compatibility of poly[styrene-co-(cinnamic acid)]/poly[(ethyl methacrylate)-co-(2-dimethylaminoethyl methacrylate)] blends. Macromol. Chem. Phys. 1999, 200, 678–682. [Google Scholar] [CrossRef]
- Branston, R.E.; Plaumann, H.P.; Sendorek, J.J. Emulsion copolymers of α-methylstyrene and styrene. J. Appl. Polym. Sci. 1990, 40, 1149–1162. [Google Scholar] [CrossRef]
- Yang, L.T.; Zhang, L.H.; Liu, G.D.; Gao, J.G.; Yang, S.; Yang, M.S.; Jin, R.G. Synthesis and thermoanalysis of emulsion terpolymers of N-phenylmaleimide, styrene, and acrylonitrile. J. Appl. Polym. Sci. 2001, 79, 1067–1073. [Google Scholar] [CrossRef]
- So, Y.H.; Hahn, S.F.; Li, Y.F.; Reinhard, M.T. Styrene 4-vinylbenzocyclobutene copolymer for microelectronic applications. J. Polym. Sci. Part A: Polym. Chem. 2008, 46, 2799–2806. [Google Scholar] [CrossRef]
- Narkis, M.; Kohn, D.H. Copolymerization of styrene. III. Physical and mechanical properties of copolymers with styrene derivatives containing nitrile Groups in the side chain. J. Polym. Sci. Part A: Polym. Chem. 1967, 5, 1049–1057. [Google Scholar] [CrossRef]
- Moore, E.R.; Plckelman, D.M. Synthesis of styrene/maleimide copolymers and physical properties thereof. Ind. Eng. Chem. Prod. Res. Dev. 1986, 25, 603–609. [Google Scholar] [CrossRef]
- Roberts, R.D.; Kwok, J.C. Styrene–maleic anhydride copolymer foam for heat resistant packaging. J. Cell. Plast. 2007, 43, 135–143. [Google Scholar] [CrossRef]
- Ko, K.H.; Luqman, M.; Kim, Y.W.; Kim, J.S. Effects of Na-sulfopropyl groups on the mechanical properties and morphology of polystyrene-co-methacrylate ionomers. J. Polym. Sci. Part B: Polym. Phys. 2016, 54, 1043–1053. [Google Scholar] [CrossRef]
- Jaymand, M. Synthesis and characterization of syndiotactic polystyrene-graft-poly(methyl methacrylate) via free radical polymerization. Polym.-Plast. Tech. Eng. 2012, 51, 514–520. [Google Scholar] [CrossRef]
- Peppas, N.A.; Bussing, W.R. Friedel-crafts crosslinking methods for polystyrene modification: 2. Tg, TII and T’II transitions of grated polystyrene. Polymer 1983, 24, 898–902. [Google Scholar] [CrossRef]
- Taşdemir, M.; Gülsoy, Ö. Physical and mechanical properties of iron powder filled polystyrene composites. Polym.-Plast. Tech. Eng. 2006, 45, 1207–1211. [Google Scholar] [CrossRef]
- Wang, Z.B.; Pang, H.T.; Li, G.C.; Zhang, Z.K. Glass transition and free volume of high impact polystyrene/TiO2 nanocomposites determined by dilatometry. J. Macromol. Sci. Part B: Phys. 2006, 45, 689–697. [Google Scholar] [CrossRef]
- Thomas, P.S.; Thomas, S.; Bandyopadhyay, S.; Wurm, A.; Schick, C. Polystyrene/calcium phosphate nanocomposites: Dynamic mechanical and differential scanning calorimetric studies. Compos. Sci. Technol. 2008, 68, 3220–3229. [Google Scholar] [CrossRef]
- Munhoz Junior, A.H.; Pacheco, C.V.; Melo, H.T.T.S.; Peres, R.M.; Silva, L.G.A.; Miranda, L.F.; Filho, M.R. Characterization of a composite of high-impact polystyrene, pseudoboehmite and graphene oxide. In Characterization of Minerals, Metals, and Materials 2019; Li, B., Li, J., Ikhmayies, S., Zhang, M.M., Kalay, Y.E., Carpenter, J.S., Hwang, J.Y., Monteiro, S.N., Bai, C.G., Escobedo-Diaz, J.P., et al., Eds.; The Minerals, Metals & Materials Series; Springer: Cham, Switzerland, 2019; pp. 511–522. [Google Scholar]
- Chen, G.M.; Liu, S.H.; Chen, S.J.; Qi, Z.N. FTIR spectra, thermal properties, and dispersibility of a polystyrene/montmorillonite nanocomposite. Macromol. Chem. Phys. 2001, 202, 1189–1193. [Google Scholar] [CrossRef]
- Xie, W.; Hwu, J.M.; Jiang, G.J.; Buthelezi, T.M.; Pan, W.P. A study of the effect of surfactants on the properties of polystyrene-montmorillonite nanocomposites. Polym. Eng. Sci. 2003, 43, 214–222. [Google Scholar] [CrossRef]
- Xiao, M.; Sun, L.Y.; Liu, J.J.; Li, Y.; Gong, K.C. Synthesis and properties of polystyrene/graphite nanocomposites. Polymer 2002, 43, 2245–2248. [Google Scholar] [CrossRef]
- Hu, H.T.; Wang, X.B.; Wang, J.C.; Wan, L.; Liu, F.M.; Zheng, H.; Chen, R.; Xu, C.H. Preparation and properties of graphene nanosheets–polystyrene nanocomposites via in situ emulsion polymerization. Chem. Phys. Lett. 2010, 484, 247–253. [Google Scholar] [CrossRef]
- Dave, V.J.; Patel, H.S. Synthesis and characterization of interpenetrating polymer networks from transesterified castor oil based polyurethane and polystyrene. J. Saudi Chem. Soc. 2013, 21, 18–24. [Google Scholar] [CrossRef]
- Khutia, M.; Joshi, G.M.; Deshmukh, K.; Pandey, M. Optimization of dielectric constant of polycarbonate/polystyrene modified blend by ceramic metal oxide. Polym.-Plast. Tech. Eng. 2015, 54, 383–389. [Google Scholar] [CrossRef]
- Bellinger, M.A.; Sauer, J.A.; Hara, M. Tensile fracture properties of rigid–rigid blends made of sulfonated polystyrene ionomer and polystyrene. Macromolecules 1994, 27, 6147–6155. [Google Scholar] [CrossRef]
- Hara, M.; Bellinger, M.; Sauer, J.A. Blends of sulphonated polystyrene and polystyrene: Morphology and deformation modes. Polym. Int. 1991, 26, 137–141. [Google Scholar] [CrossRef]
- Marshall, A.S.; Petrie, S.E.B. Rate-determining factors for enthalpy relaxation of glassy polymers. Molecular weight. J. Appl. Phys. 1975, 46, 4223–4230. [Google Scholar] [CrossRef]
- Kim, S.H.; Son, W.K.; Kim, Y.J.; Kang, E.; Kim, D.; Park, C.W.; Kim, W.; Kim, H. Synthesis of polystyrene/poly(butyl acrylate) core–shell latex and its surface morphology. J. Appl. Polym. Sci. 2003, 88, 595–601. [Google Scholar] [CrossRef]
- Thakur, V.K.; Tan, E.J.; Lin, M.F.; Lee, P.S. Polystyrene grafted polyvinylidenefluoride copolymers with high capacitive performance. Polym. Chem. 2011, 2, 2000–2009. [Google Scholar] [CrossRef]
- Baes, A.U.; Bloom, P.R. Diffuse reflectance and transmission Fourier transform infrared (DRIFT) spectroscopy of humic and fulvic acids. Soil Sci. Soc. Am. J. 1989, 53, 695–700. [Google Scholar] [CrossRef]
- Kutsumizu, S.; Hara, H.; Tachino, H.; Shimabayashi, K.; Yano, S. Infrared spectroscopic study of the binary blends of sodium and zinc salt ionomers produced from poly(ethylene-co-methacrylic acid). Macromolecules 1999, 32, 6340–6347. [Google Scholar] [CrossRef]
- Ro, A.J.; Huang, S.J.; Weiss, R.A. Synthesis and thermal properties of telechelic poly(lactic acid) ionomers. Polymer 2008, 49, 422–431. [Google Scholar] [CrossRef]
- Dreveni, I.; Berkesi, O.; Janovics, I.; Andor, J.A. Fourier transform infrared spectroscopic study on the molecular structure of bis(butanoato) zinc(II) and μ4-oxohexa-μ-butanoatotetrazinc(II) complexes. Vibr. Spectrosc. 1991, 2, 205–209. [Google Scholar] [CrossRef]
- Fulmer, G.R.; Miller, A.J.M.; Sherden, N.H.; Gottlieb, H.E.; Nudelman, A.; Stoltz, B.M.; Bercaw, J.E.; Goldberg, K.I. NMR chemical shifts of trace impurities: Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist. Organometallics 2010, 29, 2176–2179. [Google Scholar] [CrossRef]
- Crowther, M.W.; Cabasso, I.; Levy, G.C. An NMR study of miscible blends in concentrated solution. 1. Poly(vinyl methyl ether)/polystyrene. Macromolecules 1988, 21, 2924–2928. [Google Scholar] [CrossRef]
- Junk, T.; Catallo, W.J. Hydrogen isotope exchange reactions involving C–H (D, T) bonds. Chem. Soc. Rev. 1997, 26, 401–406. [Google Scholar] [CrossRef]
- Deng, F.G.; Wang, L.S.; Zhou, Y.; Gong, X.H.; Zhao, X.P.; Hu, T.; Wu, C.G. Effect of nanosilica content on the corrosion inhibition of composite coatings of a filled epoxy resin grafted with a hydrophobic fluoroalkylsilane: A dual critical concentrations interpretation. RSC Adv. 2017, 7, 48876–48893. [Google Scholar] [CrossRef]
- McNeill, I.C.; Sadeghi, S.M.T. Thermal stability and degradation mechanisms of poly(acrylic acid) and its salts: Part 1—poly(acrylic acid). Polym. Degrad. Stabil. 1990, 29, 233–246. [Google Scholar] [CrossRef]
- Cox, W.P.; Merz, E.H. Correlation of dynamic and steady flow viscosities. J. Polym. Sci. 1958, 28, 619. [Google Scholar] [CrossRef]
- Han, C.D.; Kim, J. Rheological Technique for determining the crder-disorder transition of block copolymers. J. Polym. Sci. Part B: Polym. Phys. 1987, 25, 1741–1764. [Google Scholar] [CrossRef]
- Han, C.D. Influence of molecular weight distribution on the linear viscoelastic properties of polymer blends. J. Appl. Polym. Sci. 1988, 35, 167–213. [Google Scholar] [CrossRef]
- Eisenberg, A.; Hird, B.; Moore, R.B. A new multiplet-cluster model for the morphology of random ionomers. Macromolecules 1990, 23, 4098–4107. [Google Scholar] [CrossRef]
- Brekner, M.J.; Schneider, H.A.; Cantow, H.J. Approach to the composition dependence of the glass transition temperature of compatible polymer blends: 1. Polymer 1988, 29, 78–85. [Google Scholar] [CrossRef]
- Takidis, G.; Bikiaris, D.N.; Papageorgiou, G.Z.; Achilias, D.S.; Sideridou, I. Compatibility of low-density polyethylene/poly(ethylene-co-vinyl acetate) binary blends prepared by melt mixing. J. Appl. Polym. Sci. 2003, 90, 841–852. [Google Scholar] [CrossRef]

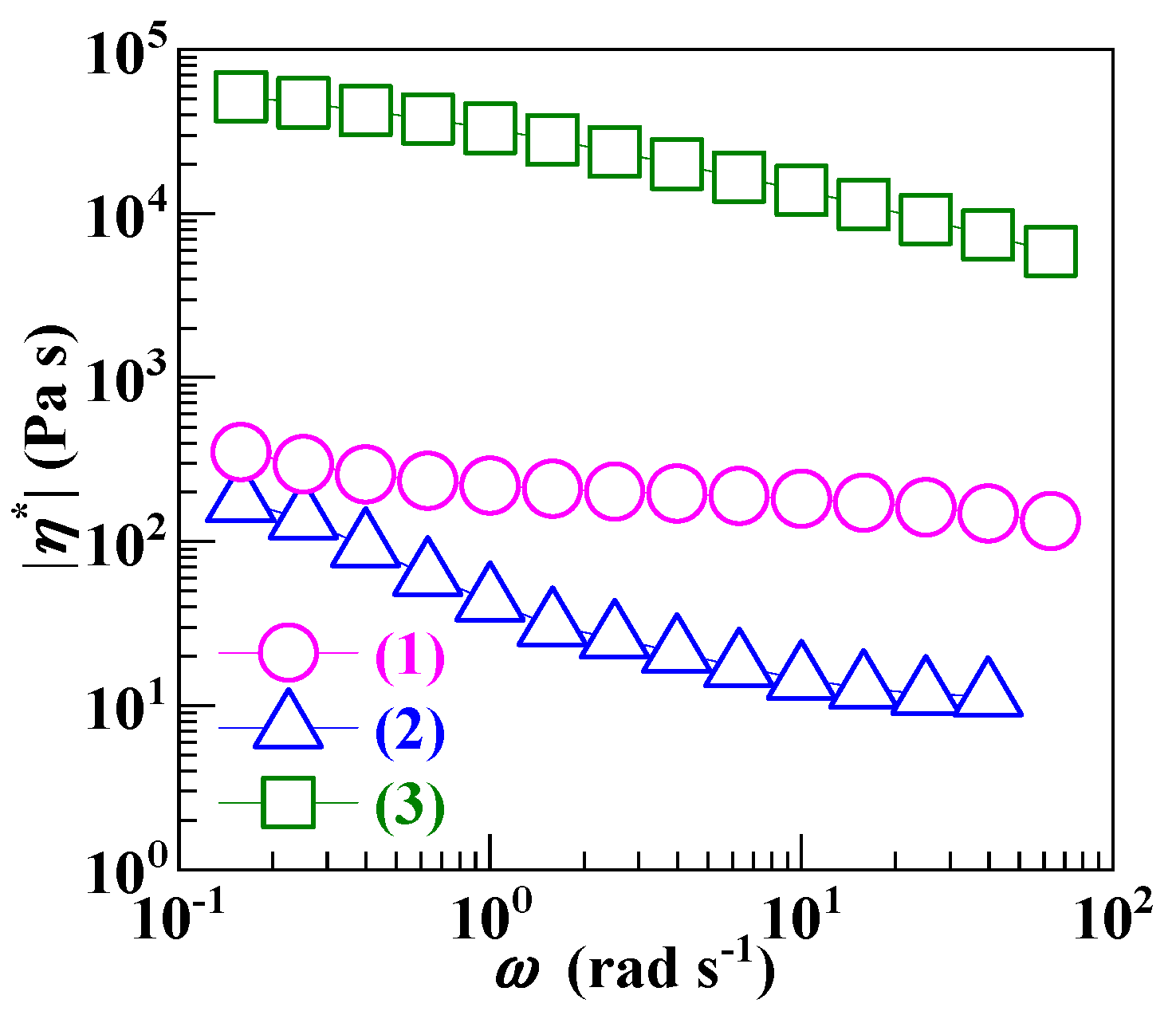


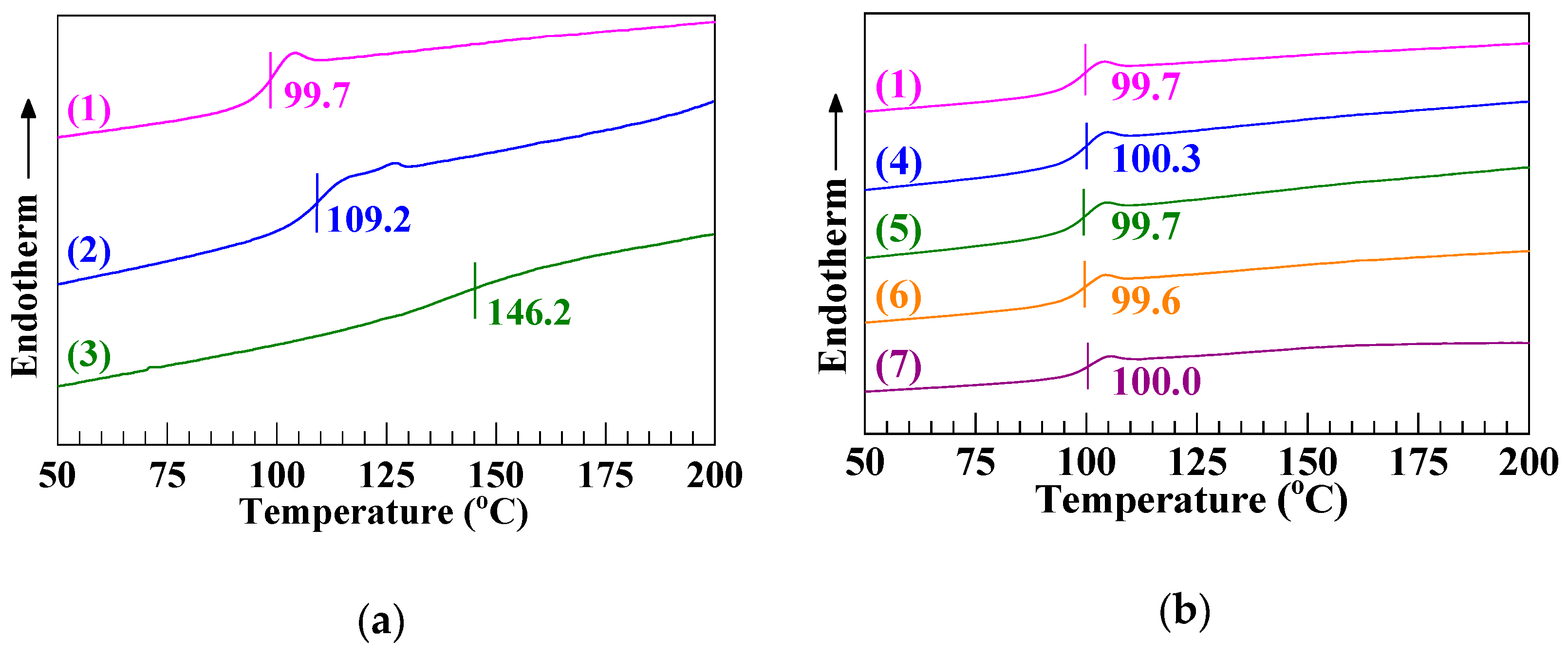


| Resin | (g mol−1) b | (g mol−1) | (g mol−1) c | |
|---|---|---|---|---|
| (a) (potential) SCA | 12,000 | 21,000 | 19,000 | 1.8 |
| (b) PS | 57,000 | 180,000 | 130,000 | 3.2 |
| Resin | CA Content (mol %) a |
|---|---|
| (a) (potential) as-synthesized SCA | cas = 13.6 ± 0.1 |
| (b) (potential) purified SCA | cCA = 10.8 ± 0.3 |
| H atom no. | δ (ppm) | ||
|---|---|---|---|
| (1) PS | (2) Potential SCA | (3) Potential SCA–Zn | |
| 1 | 1.45 | 1.47 | 1.46 |
| 2 | 1.83 | 1.83 | 1.83 |
| 3 | 6.45, 6.56 | 6.45, 6.58 | 6.48, 6.60 |
| 4 | 7.07, 7.12 | 7.07, 7.12 | 7.07, 7.13 |
| 5 | – a | – b | – b |
| 6 | – a | – b | – b |
| 7 | – a | – a | 1.62 |
| Resin | [η] (dL g−1) a |
|---|---|
| (a) (probable) SCA | 0.197 |
| (b) (probable) SCA–Zn | 0.162 |
| Composition | VST (°C) |
|---|---|
| (a) PS | 97.7 ± 0.5 |
| (b) SCA | 108.2 ± 0.4 |
| (c) SCA–Zn | 153.1 ± 0.5 |
| (d) PS/SCA–Zn (95/5) | 97.6 ± 0.2 |
| (e) PS/SCA–Zn (90/10) | 98.2 ± 0.4 |
| (f) PS/SCA–Zn (80/20) | 99.1 ± 0.5 |
| (g) PS/SCA–Zn (60/40) | 111.1 ± 0.6 |
| Composition | E (MPa) | σ (MPa) | ε (%) | αcN (kJ m−2) |
|---|---|---|---|---|
| (a) PS | 472.6 ± 27.8 | 34.9 ± 1.3 | 5.9 ± 0.3 | 3.2 ± 0.3 |
| (b) PS/SCA–Zn (95/5) | 503.0 ± 8.9 | 22.4 ± 1.4 | 3.7 ± 0.1 | 2.4 ± 0.3 |
| (c) PS/SCA–Zn (90/10) | 525.1 ± 22.9 | 23.7 ± 1.1 | 3.9 ± 0.1 | 2.4 ± 0.0 |
| (d) PS/SCA–Zn (80/20) | 550.0 ± 40.7 | 21.4 ± 1.0 | 3.7 ± 0.1 | 2.3 ± 0.2 |
| (e) PS/SCA–Zn (60/40) | 884.6 ± 53.5 | 21.0 ± 0.5 | 3.4 ± 0.1 | 1.7 ± 0.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Z.; Wang, J.; Li, P.; Ding, D.; Zheng, X.; Hu, C.; Gao, Z.; Hu, T.; Gong, X.; Wu, C. Melt Blending Modification of Commercial Polystyrene with Its Half Critical Molecular Weight, High Ion Content Ionomer, Poly(styrene–ran–cinnamic Acid) Zn Salt, toward Heat Resistance Improvement. Polymers 2020, 12, 584. https://doi.org/10.3390/polym12030584
Yu Z, Wang J, Li P, Ding D, Zheng X, Hu C, Gao Z, Hu T, Gong X, Wu C. Melt Blending Modification of Commercial Polystyrene with Its Half Critical Molecular Weight, High Ion Content Ionomer, Poly(styrene–ran–cinnamic Acid) Zn Salt, toward Heat Resistance Improvement. Polymers. 2020; 12(3):584. https://doi.org/10.3390/polym12030584
Chicago/Turabian StyleYu, Zixin, Jie Wang, Peihua Li, Dachuan Ding, Xuan Zheng, Chuanqun Hu, Zhinan Gao, Tao Hu, Xinghou Gong, and Chonggang Wu. 2020. "Melt Blending Modification of Commercial Polystyrene with Its Half Critical Molecular Weight, High Ion Content Ionomer, Poly(styrene–ran–cinnamic Acid) Zn Salt, toward Heat Resistance Improvement" Polymers 12, no. 3: 584. https://doi.org/10.3390/polym12030584
APA StyleYu, Z., Wang, J., Li, P., Ding, D., Zheng, X., Hu, C., Gao, Z., Hu, T., Gong, X., & Wu, C. (2020). Melt Blending Modification of Commercial Polystyrene with Its Half Critical Molecular Weight, High Ion Content Ionomer, Poly(styrene–ran–cinnamic Acid) Zn Salt, toward Heat Resistance Improvement. Polymers, 12(3), 584. https://doi.org/10.3390/polym12030584






