Flame Retardant Submicron Particles via Surfactant-Free RAFT Emulsion Polymerization of Styrene Derivatives Containing Phosphorous
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
2.3. Synthesis of 4-Vinylbenzyl Alcohol (VBA)
2.4. Synthesis of Diethyl 4-Vinylphenyl Phosphate (DEVBP)
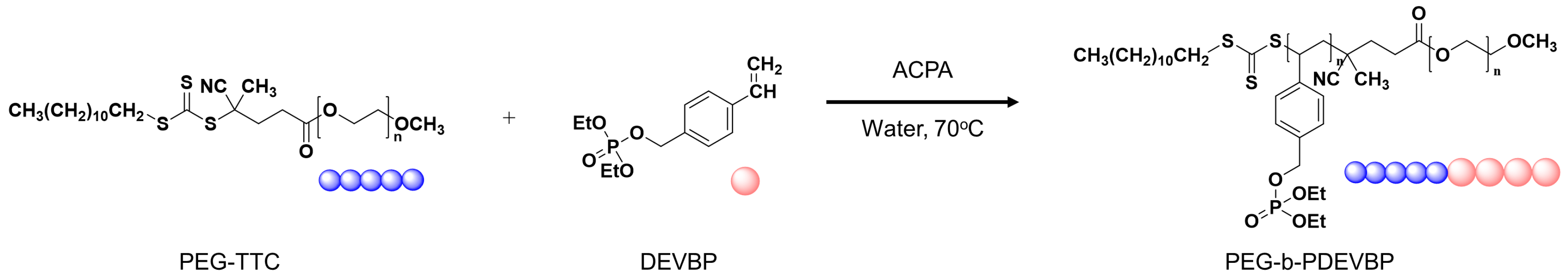
2.5. Synthesis of PEG-TTC (macro-CTA)
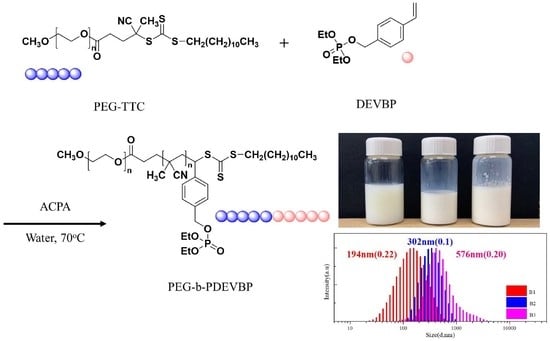
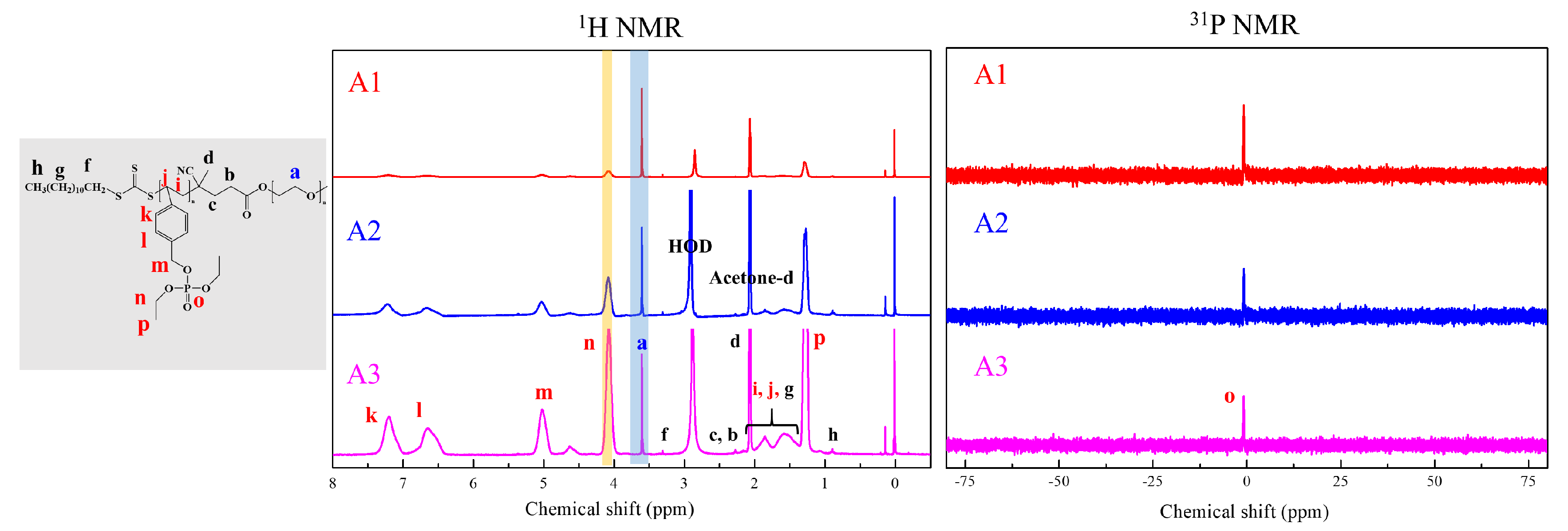
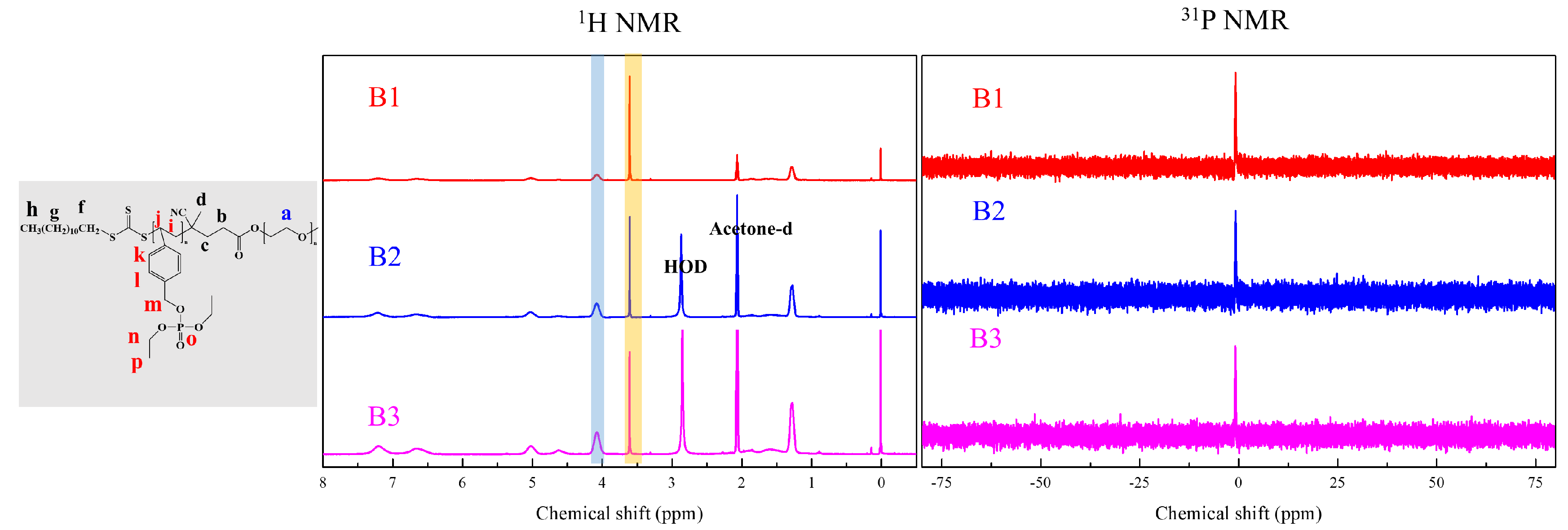
2.6. RAFT Emulsion Polymerization of Diethyl 4-Vinylphenyl Phosphate (DEVBP)
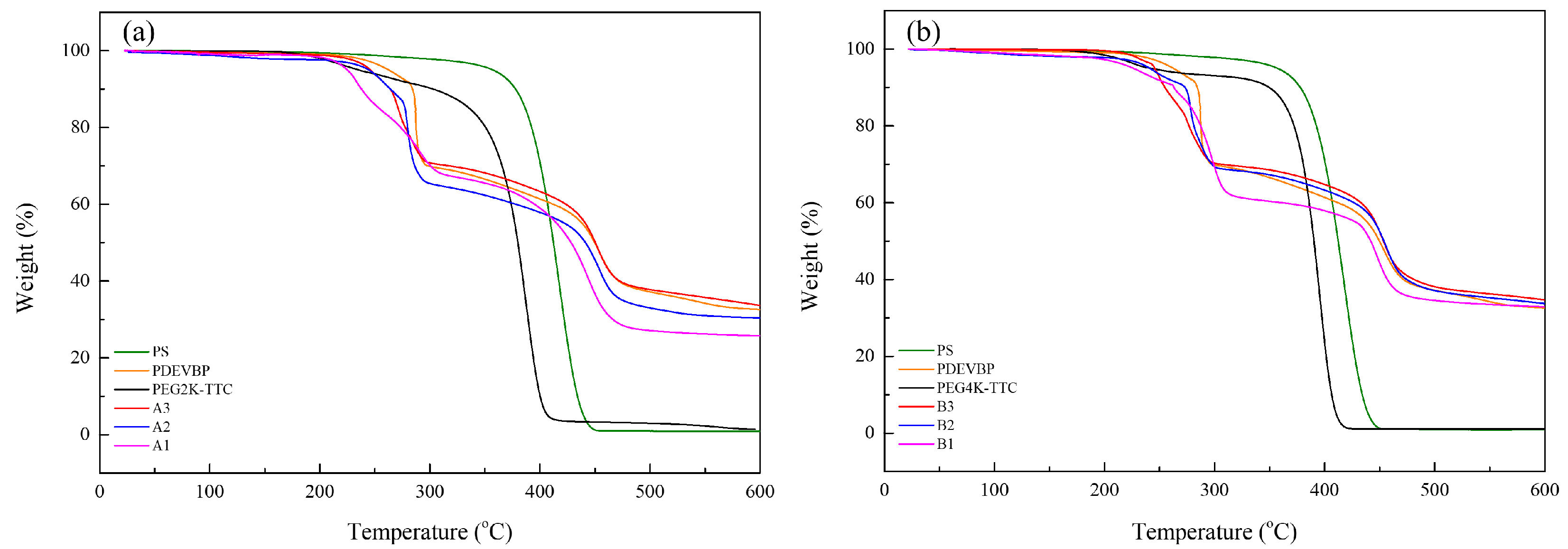
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Monge, S.; David, G.; Ilia, A.; Ishihara, K.; Fontaine, L.; Allcock, H.R.; Wentrup-Byrne, E.; Moszner, N.; Cuesta, J.L.; Popa, A. Phosphorus-Based Polymers: From Synthesis to Applications; Royal Society of Chemistry: London, UK, 2014. [Google Scholar]
- Zhang, Q.M.; Serpe, M.J. Synthesis, Characterization, and Antibacterial Properties of a Hydroxyapatite Adhesive Block Copolymer. Macromolecules 2014, 47, 8018–8025. [Google Scholar] [CrossRef]
- Brunklaus, G.; Schauff, S.; Markova, D.; Klapper, M.; Müllen, K.; Spiess, H.-W. Proton Mobilities in Phosphonic Acid-Based Proton Exchange Membranes Probed by 1H and 2H Solid-State NMR Spectroscopy. J. Phys. Chem. B 2009, 113, 6674–6681. [Google Scholar] [CrossRef]
- Quittmann, U.; Lecamp, L.; El Khatib, W.; Youssef, B.; Bunel, C. Synthesis of a New Phosphonated Dimethacrylate: Photocuring Kinetics in Homo- and Copolymerization, Determination of Thermal and Flame-Retardant Properties. Macromol. Chem. Phys. 2001, 202, 628–635. [Google Scholar] [CrossRef]
- Wang, H.; Niu, H.; Dong, J.-Y. Inherently flame retardant polypropylene copolymer. Polymer 2017, 126, 109–115. [Google Scholar] [CrossRef]
- Canadell, J.; Hunt, B.J.; Cook, A.G.; Mantecón, A.; Cádiz, V. Flame retardance and shrinkage reduction of polystyrene modified with acrylate-containing phosphorus and crosslinkable spiro-orthoester moieties. Polym. Degrad. Stab. 2007, 92, 1482–1490. [Google Scholar] [CrossRef]
- Wang, J.S.; Matyjaszewski, K. Controlled/”living” radical polymerization. atom transfer radical polymerization in the presence of transition-metal complexes. J. Am. Chem. Soc. 1995, 117, 5614–5615. [Google Scholar] [CrossRef]
- Hawker, C.J.; Bosman, A.W.; Harth, E. New Polymer Synthesis by Nitroxide Mediated Living Radical Polymerizations. Chem. Rev. 2001, 101, 3661–3688. [Google Scholar] [CrossRef]
- Perrier, S. 50th Anniversary Perspective: RAFT Polymerization—A User Guide. Macromolecules 2017, 50, 7433–7447. [Google Scholar] [CrossRef]
- Kim, T.; Mays, J.; Chung, I. Porous poly(ε-caprolactone) microspheres via UV photodegradation of block copolymers prepared by RAFT polymerization. Polymer 2018, 158, 198–203. [Google Scholar] [CrossRef]
- McLeary, J.B.; Klumperman, B. RAFT mediated polymerisation in heterogeneous media. Soft Matter 2006, 2, 45–53. [Google Scholar] [CrossRef]
- Zhou, J.; Yao, H.; Ma, J. Recent advances in RAFT-mediated surfactant-free emulsion polymerization. Polym. Chem. 2018, 9, 2532–2561. [Google Scholar] [CrossRef]
- Truong, N.P.; Quinn, J.F.; Anastasaki, A.; Rolland, M.; Vu, M.N.; Haddleton, D.M.; Whittaker, M.R.; Davis, T.P. Surfactant-free RAFT emulsion polymerization using a novel biocompatible thermoresponsive polymer. Polym. Chem. 2017, 8, 1353–1363. [Google Scholar] [CrossRef]
- Khor, S.Y.; Vu, M.N.; Pilkington, E.H.; Johnston, A.P.R.; Whittaker, M.R.; Quinn, J.F.; Truong, N.P.; Davis, T.P. Elucidating the Influences of Size, Surface Chemistry, and Dynamic Flow on Cellular Association of Nanoparticles Made by Polymerization-Induced Self-Assembly. Small 2018, 14, 1801702. [Google Scholar] [CrossRef]
- Huang, J.; Matyjaszewski, K. Atom Transfer Radical Polymerization of Dimethyl(1-ethoxycarbonyl)vinyl Phosphate and Corresponding Block Copolymers. Macromolecules 2005, 38, 3577–3583. [Google Scholar] [CrossRef]
- Mukumoto, K.; Zhong, M.; Matyjaszewski, K. Atom transfer radical polymerization of dimethyl(methacryloyloxymethyl) phosphonate. Eur. Polym. J. 2014, 56, 11–16. [Google Scholar] [CrossRef]
- Britze, A.; Moosmann, K.; Jähne, E.; Adler, H.-J.; Kuckling, D. Synthesis of Block Copolymers Modified with Phosphonate Ester Groups Using Nitroxide-Mediated Radical Polymerization. Macromol. Rapid Commun. 2006, 27, 1906–1912. [Google Scholar] [CrossRef]
- Hanisch, A.; Yang, P.; Kulak, A.N.; Fielding, L.A.; Meldrum, F.C.; Armes, S.P. Phosphonic Acid-Functionalized Diblock Copolymer Nano-Objects via Polymerization-Induced Self-Assembly: Synthesis, Characterization, and Occlusion into Calcite Crystals. Macromolecules 2016, 49, 192–204. [Google Scholar] [CrossRef]
- Canniccioni, B.; Monge, S.; David, G.; Robin, J.-J. RAFT polymerization of dimethyl(methacryloyloxy)methyl phosphonate and its phosphonic acid derivative: A new opportunity for phosphorus-based materials. Polym. Chem. 2013, 4, 3676–3685. [Google Scholar] [CrossRef]
- Furukawa, J.; Kobayashi, E.; Wakui, T. Phosphorus-Containing Polystyrene Derivatives as Flame Resistance. Polym. J. 1980, 12, 277–285. [Google Scholar] [CrossRef] [Green Version]
- Dumitrascu, A.; Howell, B.A. Flame-retarding vinyl polymers using phosphorus-functionalized styrene monomers. Polym. Degrad. Stab. 2011, 96, 342–349. [Google Scholar] [CrossRef]
- Schartel, B. Phosphorus-based Flame Retardancy Mechanisms-Old Hat or a Starting Point for Future Development? Materials (Basel) 2010, 3, 4710–4745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schartel, B.; Balabanovich, A.I.; Braun, U.; Knoll, U.; Artner, J.; Ciesielski, M.; Döring, M.; Perez, R.; Sandler, J.K.W.; Altstädt, V.; et al. Pyrolysis of epoxy resins and fire behavior of epoxy resin composites flame-retarded with 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide additives. J. Appl. Polym. Sci. 2007, 104, 2260–2269. [Google Scholar] [CrossRef]
- Moad, G.; Rizzardo, E.; Thang, S.H. Living Radical Polymerization by the RAFT Process. Aust. J. Chem. 2005, 58, 379. [Google Scholar] [CrossRef]
- Zheng, X.; Wilkie, C.A. Flame retardancy of polystyrene nanocomposites based on an oligomeric organically-modified clay containing phosphate. Polym. Degrad. Stab. 2003, 81, 539–550. [Google Scholar] [CrossRef] [Green Version]
- Rieger, J.; Stoffelbach, F.; Bui, C.; Alaimo, D.; Jérôme, C.; Charleux, B. Amphiphilic Poly(ethylene oxide) Macromolecular RAFT Agent as a Stabilizer and Control Agent in ab Initio Batch Emulsion Polymerization. Macromolecules 2008, 41, 4065–4068. [Google Scholar] [CrossRef]
- dos Santos, A.M.; Pohn, J.; Lansalot, M.; D’Agosto, F. Combining Steric and Electrostatic Stabilization Using Hydrophilic MacroRAFT Agents in an Ab Initio Emulsion Polymerization of Styrene. Macromol. Rapid Commun. 2007, 28, 1325–1332. [Google Scholar] [CrossRef]
- Rieger, J.; Osterwinter, G.; Bui, C.; Stoffelbach, F.; Charleux, B. Surfactant-Free Controlled/Living Radical Emulsion (Co)polymerization of n-Butyl Acrylate and Methyl Methacrylate via RAFT Using Amphiphilic Poly(ethylene oxide)-Based Trithiocarbonate Chain Transfer Agents. Macromolecules 2009, 42, 5518–5525. [Google Scholar] [CrossRef]
- Truong, N.P.; Dussert, M.V.; Whittaker, M.R.; Quinn, J.F.; Davis, T.P. Rapid synthesis of ultrahigh molecular weight and low polydispersity polystyrene diblock copolymers by RAFT-mediated emulsion polymerization. Polym. Chem. 2015, 6, 3865–3874. [Google Scholar] [CrossRef]







| Entry | Monomer wt. % | Mn (PEG) | [DEVBP]0/[PEG-TTC]0 | Ptheory % | Conversion a % | Molecular Weight | Size | Residual Weight at 500 °C (%) TGA analysis | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Theory b | GPC (Mw/Mn) | 1H NMR | Dz (nm) c (Ð) d | |||||||
| A1 | 5 | 2K | 32 | 9.0 | 89 | 10,100 | 5200 (1.08) | 9800 | 216 (0.22) | 27.1 |
| A2 | 9 | 2K | 65 | 10.1 | 85 | 17,300 | 9400 (1.08) | 16,800 | 1056 (0.68) | 33 |
| A3 | 16 | 2K | 130 | 10.7 | 92 | 31,500 | 16,100 (1.07) | 34,200 | - | 37.8 |
| B1 | 5 | 4K | 32 | 7.6 | 91 | 12,300 | 6700 (1.20) | 12,200 | 194 (0.22) | 34.5 |
| B2 | 9 | 4K | 65 | 9.1 | 82 | 18,800 | 11,000 (1.35) | 18,100 | 302 (0.10) | 37.1 |
| B3 | 16 | 4K | 130 | 10.2 | 87 | 35,000 | 14,100 (1.17) | 33,200 | 576 (0.20) | 38.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.; Song, J.-H.; Back, J.-H.; Seo, B.; Lim, C.-S.; Paik, H.-J.; Lee, W. Flame Retardant Submicron Particles via Surfactant-Free RAFT Emulsion Polymerization of Styrene Derivatives Containing Phosphorous. Polymers 2020, 12, 1244. https://doi.org/10.3390/polym12061244
Kim T, Song J-H, Back J-H, Seo B, Lim C-S, Paik H-J, Lee W. Flame Retardant Submicron Particles via Surfactant-Free RAFT Emulsion Polymerization of Styrene Derivatives Containing Phosphorous. Polymers. 2020; 12(6):1244. https://doi.org/10.3390/polym12061244
Chicago/Turabian StyleKim, Taeyoon, Joo-Hyun Song, Jong-Ho Back, Bongkuk Seo, Choong-Sun Lim, Hyun-Jong Paik, and Wonjoo Lee. 2020. "Flame Retardant Submicron Particles via Surfactant-Free RAFT Emulsion Polymerization of Styrene Derivatives Containing Phosphorous" Polymers 12, no. 6: 1244. https://doi.org/10.3390/polym12061244
APA StyleKim, T., Song, J.-H., Back, J.-H., Seo, B., Lim, C.-S., Paik, H.-J., & Lee, W. (2020). Flame Retardant Submicron Particles via Surfactant-Free RAFT Emulsion Polymerization of Styrene Derivatives Containing Phosphorous. Polymers, 12(6), 1244. https://doi.org/10.3390/polym12061244