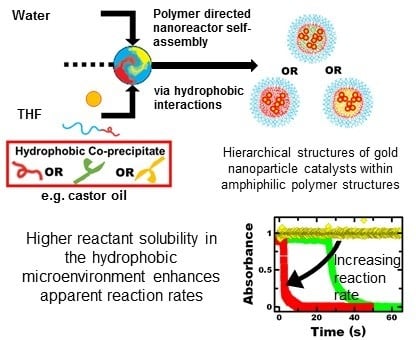
3.1. Nanoreactor Formulation and Characterization
To incorporate gold nanoparticle catalysts into hierarchical polymer nanostructures, we used polymer directed self-assembly driven by non-covalent, hydrophobic interactions. Specifically, we used flash nanoprecipitation. Flash nanoprecipitation is a rapid, scalable platform for polymer-directed self-assembly of colloidal nanoparticles with versatile materials selection [
16,
22,
23]; therefore, we aimed to examine the effect of various co-precipitants on self-assembly and catalytic performance.
To perform flash nanoprecipitation using castor oil as an initial co-precipitant, hydrophobic gold nanoparticles were dispersed with a dissolved amphiphilic block copolymer and castor oil co-precipitant in a water miscible organic solvent (tetrahydrofuran, THF). This solvent stream was rapidly mixed against water using a confined impinging jet mixer. Upon mixing, the rapid decrease in solvent quality caused the nanoparticles to aggregate, castor oil to precipitate and the block copolymer to micellize directing formation of the hierarchical polymer nanoparticle. This hybrid particle assembly ended when adsorption of the hydrophobic block of the block copolymer prevented further growth and colloidal aggregation. The self-assembled structure was sterically stabilized in water by the hydrophilic block of the block copolymer. Given the molecular weight of the block copolymer, dynamic exchange of the block copolymer did not occur, and the resulting structure was kinetically trapped [
2,
17,
24]. Successful polymer nanoreactor assembly resulted in homogenous dispersions with no macroscopic precipitation of the hydrophobic components, including the gold nanoparticles.
Further evidence of successful self-assembly of dispersed polymer systems was evident by dynamic light scattering (DLS). Specifically, DLS results confirmed that the composite nanoparticles were uniform with a single peak in the size intensity distribution measured by DLS indicating the hydrodynamic diameter was ~100 nm; representative data are shown in red in
Figure 1. The UV-vis absorbance of the nanoreactor dispersion was 526 nm, which is comparable to other self-assembled gold–polymer systems and has been attributed to the hydrophobicity of the surrounding environment and the close proximity of the incorporated gold nanoparticles [
16]. TEM of the nanoreactors is shown as an inset (
Figure 1), with a low magnification TEM image shown in
Figure S2 in Supplementary Materials. The nanoreactors appear spherical and the size seen in the images is comparable hydrodynamic diameters reported by DLS. Gold nanoparticles appear to be incorporated in the nanoreactor with multiple gold nanoparticles per nanoreactor. This result is consistent with previous reports [
16].
Since flash nanoprecipitation is a versatile platform in terms of materials selection, we prepared a number of other nanoreactor systems with various hydrophobic co-precipitants. We examined dodecane (DD), dodecylamine (DDA) and dodecanethiol (DDT). In the cases of dodecylamine and dodecanethiol, gold-core material interactions may affect both nanoreactor self-assembly as well as catalytic performance. For self-assembling systems that only involved non-covalent, hydrophobic interactions, we studied dodecane and polystyrene (
Mw 750 g/mol PS 750) in addition to castor oil. Using these various hydrophobic co-precipitants, nanoreactors with comparable hydrodynamic size and gold loading were achieved using the constant core volume approach [
16]. Specifically, since the densities of the core materials are comparable, using the same co-precipitant concentration in the organic stream during flash nanoprecipitation (11-mg/mL co-precipitant with 1-mg/mL gold nanoparticles and 12-mg/mL block copolymer stabilizer), the hydrodynamic size of the nanoreactors were comparable (
Table 1) [
16]. As shown in
Table 1, all systems formed nanoreactors around 130 nm with low polydispersity (<0.3 PDI) as measured by DLS. The nanoreactor size was stable for at least one week at room temperature, except for the dodecane system (which was not further analyzed).
We also compared the various nanoreactor systems in terms of the amount of gold successfully incorporated into the nanoreactor during self-assembly, which we quantify with the incorporation efficiency (IE). To remove any hydrophobic gold nanoparticles in the dispersion that were not incorporated into the nanoreactors, we performed extraction with ether to selectively remove unincorporated gold nanoparticles while preventing disruption of the nanoreactors (as the PEG stabilizing the nanoreactors was insoluble). The incorporation efficiency for gold nanoparticles via flash nanoprecipitation was greater than 77% ± 5% for all systems compared to 5% for dodecanethiol-capped gold nanoparticles in water (
Table S1 in Supplementary Materials). Co-precipitates that interact with gold had the highest incorporation efficiencies. Both dodecanethiol and dodecylamine had incorporation efficiencies greater than 95% (
Table S2 in Supplementary Materials). Performing DLS analysis after extraction, we confirmed that the nanoreactors remain intact through the extraction process which we attribute to the insolubility of the amphiphilic polymer, PS–
b–PEG, in diethyl ether. These results demonstrate that use of co-precipitants with relatively strong interactions with gold (e.g., thiols and amines) increase incorporation efficiency of the gold nanoparticles into the hybrid polymer nanoparticles compared to assemblies that rely solely on noncovalent interactions.
3.2. Analysis of Apparent Reaction Kinetics
We began by assessing the catalytic performance of the castor oil nanoreactors using the reduction of 4-nitrophenol by sodium borohydride as a model reaction. Because this reaction proceeds at room temperature without side products and byproducts, it is a well-established model reaction for kinetic studies using gold nanoparticles [
16,
18,
20,
21,
25,
26,
27]. We also note that reduction of 4-nitrophenol is important in waste water treatment [
28,
29] and the product resulting from the model reaction, 4-aminophenol, has several applications, e.g., a corrosion inhibitor as well as an intermediate in the pharmaceutical industry [
30]. A schematic of the nanoreactor structure and these potential catalytic applications are illustrated in
Figure S3 in Supplementary Materials. We confirmed that the nanoreactors were stable following reaction. Some minor swelling was observed; the size increased from 113 ± 10 (PDI 0.184 ± 0.011) before reaction to 154 ± 8 (PDI 0.220 ± 0.012) after reaction.
To facilitate pseudo-first order rate kinetics where the reaction rate is independent of sodium borohydride concentration, we first examined the effect of sodium borohydride concentration on reaction kinetics [
20,
21]. For the CO NRs prepared via flash nanoprecipitation, we observe the apparent reaction rate per surface area of gold (
k1) increased with sodium borohydride concentration until it plateaus at a sodium borohydride concentration of 1.0 M sodium borohydride (
Figure 2A). Thus, the bulk sodium borohydride concentration in the reaction medium was 1.0 M to achieve pseudo-first order rate kinetics i.e., reaction is independent of sodium borohydride concertation. These results are similar to previous reports for gold nanoparticle-polymer nanoparticle systems [
21]. This plateau has been attributed to active site blocking predicted by Langmuir-Hinshelwood kinetics when one reagent predominantly occupies the catalyst surface [
21,
31,
32]. Interestingly, the sodium borohydride concentration required to achieve a plateau for the nanoreactors was a 10-fold increase from previous reports [
21,
32] which could suggest differences in localized reagent concentrations compared to other systems, i.e., a difference between the bulk sodium borohydride concentration and the concentration at the catalyst surface due to partitioning and diffusion.
We also note that increasing the concentration of sodium borohydride decreases the induction time (
Figure 2B). These results are consistent with the trends in which changes in induction time and reaction kinetics were inversely proportional to one another reported by Ballauff [
21]. The change in induction time that we observe in this case appears to be related to an increase in sodium borohydride concentration which has recently been attributed to consumption of oxygen by sodium borohydride [
33].
To further understand the potential effect of hierarchical structure of the self-assembled, amphiphilic particles achieved during flash nanoprecipitation on catalytic performance, we compared the catalytic performance of the castor oil nanoreactors to gold nanoparticles added to preformed castor oil nanoparticles. We further compared the results with gold nanoparticles with hydrophilic stabilizers (PEG-coated gold nanoparticles and citrate-stabilized gold) at equivalent gold concentrations measured at 1.0-M NaBH
4, 0.1-mM 4-nitrophenol. Thus, castor oil nanoparticles of comparable hydrodynamic size were formulated (
Table S2). Following flash nanoprecipitation, hydrophobic gold nanoparticles dispersed in THF were added to the dispersed polymer nanoparticles. No macroscopic aggregation of the gold nanoparticles in the aqueous phase was observed. Rather, the gold nanoparticles associated with the polymer nanoparticle via nonspecific adsorption (presumably with the PEG surface). In this study, we were interested in comparing the catalytic performance of the castor oil nanoreactors to gold nanoparticles added to preformed castor oil nanoparticles to PEG-coated gold nanoparticles and citrate-stabilized gold nanoparticles (hydrophilic stabilizers) to determine the effect of the hierarchical, amphiphilic nanoreactor structure achieved by flash nanoprecipitation.
Interestingly, we observed that the reaction rate normalized per surface area per gold (
k1) for the gold nanoparticles added to the preformed gold nanoparticles were comparable to the PEG-coated gold nanoparticles and citrate-stabilized gold nanoparticles (
Table 2). Notably, the
k1 of the castor oil nanoreactors was over 2.7-fold faster than these other systems. These results suggest that the hierarchical, amphiphilic structure of the nanoreactors achieved by self-assembly with the amphiphilic polymer, castor oil and gold nanoparticles is beneficial for catalytic performance compared to gold nanoparticles with hydrophilic stabilizers.
3.3. Analysis of Intrinsic Reaction Kinetics via Partition Coefficient Analysis and Langmuir–Hinshelwood Kinetics
Rate acceleration in self-assembled amphiphilic systems has been previously observed and attributed to increased local concentrations of the reactants [
34]. To quantify this difference in concentration in this system, we measured the partition coefficient of 4-nitrophenol between water and castor oil. The castor oil:water partition coefficient was 7.81 ± 0.16 indicating an increased local concentration within the nanoreactor was possible. We used this information to next examine the intrinsic kinetics of the 4-nitrophenol reduction on the gold nanoparticle catalyst surface based on the Langmuir–Hinshelwood mechanism [
21,
35]. For nanoreactors, the concentration in 4-nitrophenol was calculated based on the bulk concentration and the experimentally determined core material:water partition coefficient. For gold nanoparticles added to preformed polymer nanoparticles, the 4-nitrophenol concentration was taken to be the concentration in the bulk aqueous phase. Initially, we used a two-step reaction model involving reduction of 4-nitrophenol to 4-hydroxylaminophenol and the rate limiting step of 4-hydroxylaminophenol to 4-aminophenol previously used for gold-polymer systems [
21,
32]. The two-step reaction is evident by a change in reaction rate typically when the conversion of 4-nitrophenol is above 30% [
21]. While we observed a change in reaction rate, it generally occurred around 70% conversion of 4-nitrophenol. Fitting data up until 70% conversion resulted in non-real solutions. Poor fits and large error (greater than 100%) were observed when fitting data up to 30%.
Therefore, we used an alternative model. Specifically, the data up to 30% conversion was modeled using a single reaction step and no observed change in reaction rate based on previous reports of polymer–gold systems [
18], according to:
where
cnip is the bulk aqueous phase concentration of 4-nitrophenol,
k0 is the reaction rate constant of the single step reaction,
S is the reaction solution specific catalyst surface area concentration,
Pnip is the core material:water partition coefficient of 4-nitrophenol,
n is the reaction order and
Knip and
KBH4 are the Langmuir–Hinshelwood adsorption constants of 4-nitrophenol and sodium borohydride, respectively.
Using the adsorption parameters previously reported [
21], the calculated rate constants are reported in
Table 3. Notably, the rate constants for castor oil nanoreactors and gold nanoparticles added to preformed castor oil nanoparticles were comparable. Further, the measured intrinsic kinetic parameters were comparable to PEG-coated gold nanoparticles and citrate-stabilized gold nanoparticles. These results suggest that the intrinsic kinetics of the catalysts for all the systems are comparable once the partition coefficient was used to estimate the localized 4-nitrophenol concentration. Thus, incorporating the gold nanoparticles into the nanoreactors via self-assembly does not adversely impact the intrinsic catalytic performance compared to adding gold nanoparticles to preformed castor oil nanoparticles or citrate-stabilized gold nanoparticles. Therefore, the difference in apparent catalytic performance can be attributed to an increase in local reactant concentration.
Comparing the rate constants from the Langmuir–Hinshelwood model to previous literature, we found that the rate constants for the systems prepared by flash nanoprecipitation (
Table 3) were at least two orders of magnitude greater than previously reported gold–polymer systems [
18]. Their system and their analysis were comparable to the gold nanoparticles added to preformed polymer particles. These results further indicate that flash nanoprecipitation is a promising platform for producing nanoreactors while preserving the intrinsic activity of the catalyst. Furthermore, the catalytic performance can be affected by carefully considering solubility of the reactants when selecting materials for the nanoreactor.
3.4. Comparison of Catalytic Performance
Next, we were interested in comparing the apparent catalytic performance of the castor oil nanoreactors to the other nanoreactor systems. We began by comparing the castor oil to other nanoreactor systems including PS 750 nanoparticles without gold (less than 1 ppm by ICP-OES) as a negative control (
Table 4). We used 1.0-M sodium borohydride concentration and 0.1-mM 4-nitrophenol concentration to achieve pseudo-first-order rate kinetics. Interacting core materials such as dodecanethiol and dodecylamine did not catalyze the 4-nitrophenol reduction, likely due to active site blocking. While gold-core material interactions were beneficial for improving incorporation efficiency, there was a trade off with catalytic performance. Therefore, nanoreactors prepared via flash nanoprecipitation, which can be driven by hydrophobic interactions, may offer advantages over self-assembled systems that rely on gold–polymer interactions [
18,
31].
Notably, at these reaction conditions the apparent reaction rate constant per surface area of gold (
k1) for both PS 750 NR and CO NR were at least 2-fold greater than ligand-free gold nanoparticles [
32] and approximately 10-fold greater than gold nanoparticles functionalized with pH responsive poly(acrylic acid) [
26].
For the non-interacting core materials, the apparent reaction rate for CO NRs was over 8-fold greater than PS 750 NRs with apparent reaction rate constant per surface area of gold of 5.7 ± 0.7 L m
−2 s
−1 for CO NRs compared to 0.7 ± 0.1 L m
−2 s
−1 for PS 750 NRs (
Table 4). We further investigated the difference between the PS 750 and CO nanoreactor systems. Based on the apparent kinetics, we considered potential mass transfer limitations using scaling analysis and the second Damkohler number (
DaII), i.e., a ratio of the reaction and diffusion rates given by,
where
n is the reaction order,
β is the mass transport coefficient (which is a quotient of the diffusion coefficient and the characteristic length of the system) and
a is the interfacial area. Using a previously established NMR method [
16], the diffusion coefficient for 4-nitrophenol in CO NR was determined to be 1.70 ± 0.02 × 10
−8 m
2/s (
Figure S1 in Supplementary Materials) which is slightly lower than for polystyrene 1.91 ± 0.01 × 10
−8 m
2/s [
16]. Based on these experimentally determined diffusion coefficients, the second Damkohler number was found for all systems to be on the order of 10
−4 or smaller, signifying there were no diffusion limitations for either the CO NRs or PS 750 NRs.
Based on our results, we posited that the difference in apparent catalytic performance may be attributed to differences in local reactant concentration and specifically 4-nitrophenol concentration. Hanson solubility parameters suggest that castor oil is a better solvent for 4-nitrophenol than polystyrene (
Table S3 in Supplementary Materials) and the effective 4-nitrophenol concentration within CO NRs would be higher than PS 750 NRs. In order to confirm this difference in solubility of 4-nitrophenol in different nanoreactor microenvironments, we measured the partition coefficient of 4-nitrophenol between water and castor oil or toluene (to mimic PS). As seen in
Table 5, the core material:water partition coefficient for castor oil was 7.81 ± 0.16 compared to 0.09 ± 0.01 for toluene. The higher core material:water partition coefficient for castor oil compared to toluene suggests that the effective concentration of 4-nitrophenol in the CO NRs would be higher than a PS NR contributing to enhanced apparent catalytic performance. Taken together, these results indicate that 8-fold enhancement in catalytic activity with the castor oil nanoreactors compared to the PS 750 nanoreactors could be attributed to the increased solubility of the reactants rather than changes in the intrinsic catalytic properties or differences in mass transfer. Broadly, ability to enhance the apparent catalytic performance through selection of the core material (i.e., choosing a core material that the reactant is highly soluble in) is promising approach for rational design of nanoreactors. Thus, we note Hansen solubility parameter distance (RA
2) may be a useful tool for future nanoreactor design.
In addition to comparing the PS 750 and CO nanoreactor systems to each other, comparisons to previously reported nanoparticle-based catalyst systems were also made. The turnover frequency (TOF) is related to apparent reaction rate constant, the initial amount of 4-nitrophenol and the amount of gold catalyst and is a useful metric for comparing across various catalytic systems [
36]. The turnover frequency (TOF) was calculated according to:
where
k is the apparent reaction rate constant, while
nAu and
n0,AU are the molar amounts of gold and initial 4-nitrophenol in the reaction, respectively [
36]. Based on the observed reaction rate constants, the TOFs for CO and PS 750 NRs were approximately 300,000 and 30,000 min
−1, respectively (
Table 6). These TOFs were higher than typically reported for gold-based catalysts (~1–2 min
−1) [
37,
38,
39]. The TOF of the self-assembled nanoreactors were also higher than gold–polymer systems previous reported such as gold nanoparticles coated on poly(glycidyl methacrylate) microspheres modified with poly(allylamine hydrochloride) (PAH) and negatively charged preformed gold nanoparticles (AuNPs) with a TOF ~15,000 min
−1 [
40], as well as dendrimer encapsulated gold nanoparticle catalysts TOF ~2,000 min
−1 [
41], as well as gold nanoparticles on N-containing polymer nanospheres TOF ~17,000 min
−1 [
42]. These results suggest that self-assembled nanoreactors produced via flash nanoprecipitation have promising catalytic performance. Furthermore, these results indicate the catalytic performance is highly influenced by the self-assembled polymer microenvironment.
3.5. Nanoreactor Reuse
Finally, we consider the ability to use the nanoreactors for multiple reactions as an initial step to understanding the robustness of the castor oil nanoreactors. Importantly, full conversion of the 4-nitrophenol was achieved for the CO nanoreactor systems within 3 min using 1-M NaBH
4 for at least 10 sequential reactions. This indicates that the nanoparticle catalysts incorporated in the kinetically trapped polymer nanoreactor systems resist irreversible aggregation that would result in a complete loss of activity [
27].
When performing sequential reactions, a decrease in activity was seen when sequentially adding 4-nitrophenol into the reaction mixture under 1-M NaBH
4 conditions (
Figure 3). Generally, the greatest decrease in activity occurred after the first reaction, with less significant changes with additional reactions. For example, CO NR undergo an immediate 5-fold decrease in activity from the initial 4-nitrophenol reduction to the first sequential reaction. Following the first reaction, the tenth sequential reaction is 6-fold slower than the first sequential reaction. Overall, the tenth reaction is 30-fold slower than the initial reaction, with a final minimum activity of 0.18 ± 0.01 L m
−2 s
−1. Similarly, the largest decrease in reaction rate for the citrate-stabilized and PEG-coated nanoparticles occurred after the first reaction (
Figure 3). Interestingly, the CO NR and citrate-stabilized gold nanoparticles plateaued to the same value following the first reaction. This result indicated that the decrease in rate was likely inherent to the gold nanoparticle. Decreases in reaction rate constant with multiple reactions has been previously observed and attributed to partial gold nanoparticle aggregation in the presence of sodium borohydride [
26,
27] and/or competitive adsorption on the catalyst surface [
46].
We note that that the PEG-stabilized gold nanoparticles better retained their activity with sequential reactions compared to the other systems. The CO NR had a greater initial activity than PEG-stabilized, 5.7 ± 0.7 L m−2 s−1 compared to 2.1 ± 0.2 L m−2 s−1, respectively. After 10 sequential additions the PEG AuNP had 3-fold greater activity than the CO NR (0.56 ± 0.8 and 0.18 ± 0.01 L m−2 s−1, respectively). These results may indicate a trade off between stability and initial apparent kinetics with hydrophilic stabilizers utilizing gold–thiol interactions for self-assembly and the amphiphilic nanoreactors produced here via polymer directed self-assembly leveraging non-covalent interactions.
Notably, even after 10 sequential reactions, the remaining catalytic activity of the castor oil nanoreactors exceeds that observed in previously reported gold nanoparticle systems. For example, the apparent reaction rate constant
k1 of CO NR after 10 sequential additions was 3-orders of magnitude greater than previously reported gold nanoparticle–polymer brushes systems [
47] and roughly equivalent to ligand free gold nanoparticles [
32]. Further, after 10 sequential additions (despite the 3-fold decrease in activity), the TOF was 23,000 min
−1 which was higher than typically reported for gold-based catalysts (~1–2 min
−1) [
37,
38,
39] and comparable to previously reported gold–polymer based systems [
40,
42].