HPMA-Based Polymer Conjugates for Repurposed Drug Mebendazole and Other Imidazole-Based Therapeutics
Abstract
:1. Introduction
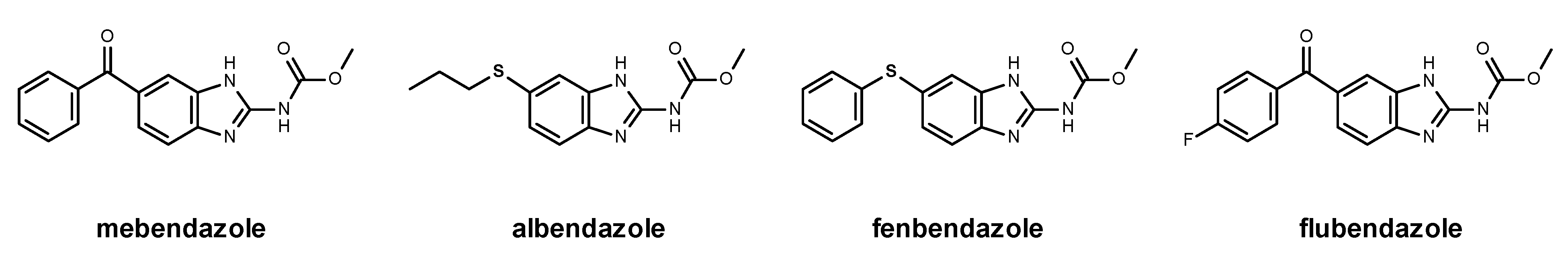
1.1. Benzimidazole Derivatives in Antitumor Therapy
1.2. Mebendazole Mechanism of Action and Antitumor Potential
1.3. Drug Delivery Systems Based on Synthetic Copolymers
1.4. Polymer-Drug Linkers Based on a Prodrug Strategy
2. Materials and Methods
2.1. Chemicals
2.2. Analytical Methods
2.3. Syntheses
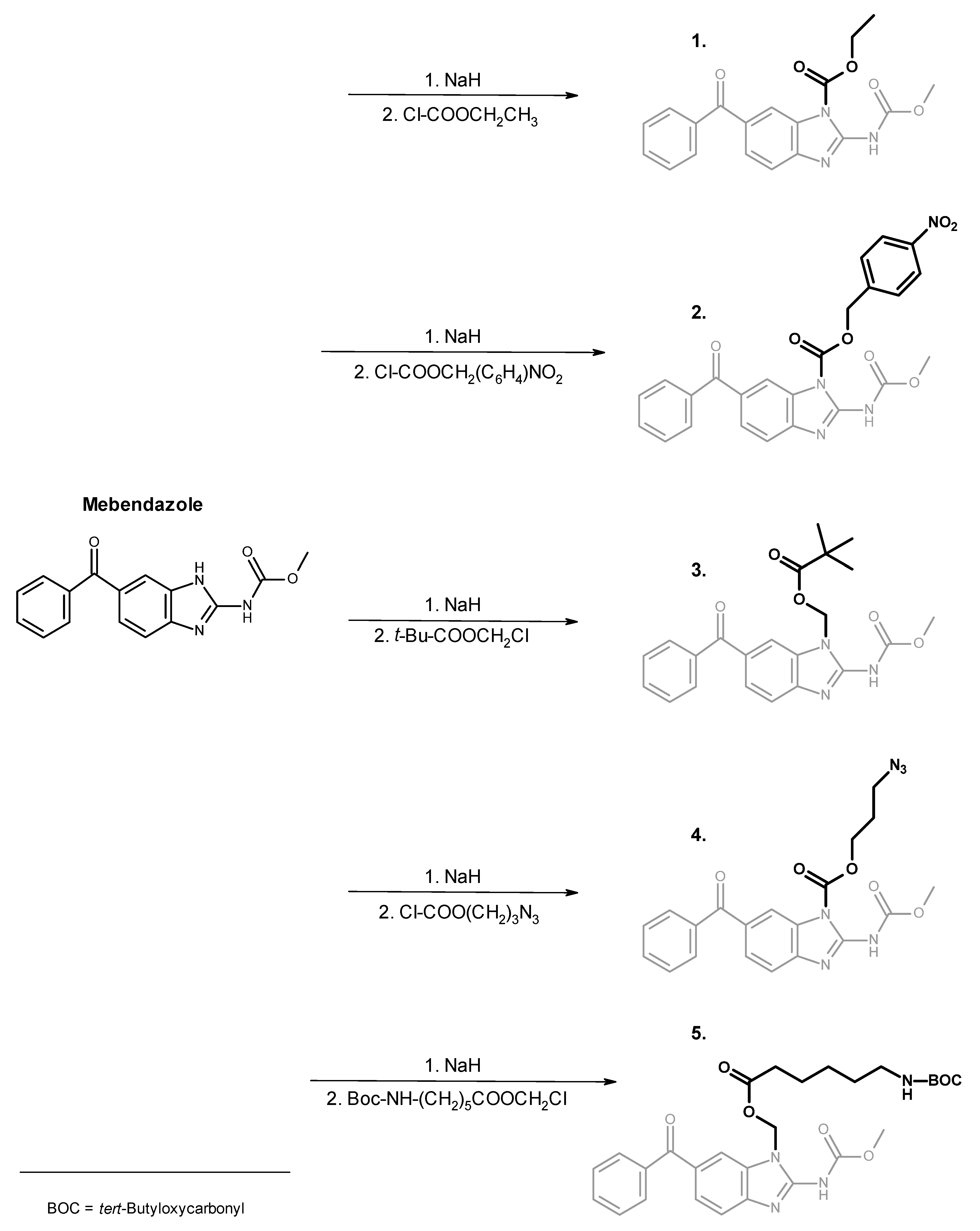
Mebendazole Derivatives 1–5
2.4. N-Ethoxycarbonyl Mebendazole (1)
2.5. N-(4-Nitrobenzyloxycarbonyl) Mebendazole (2)
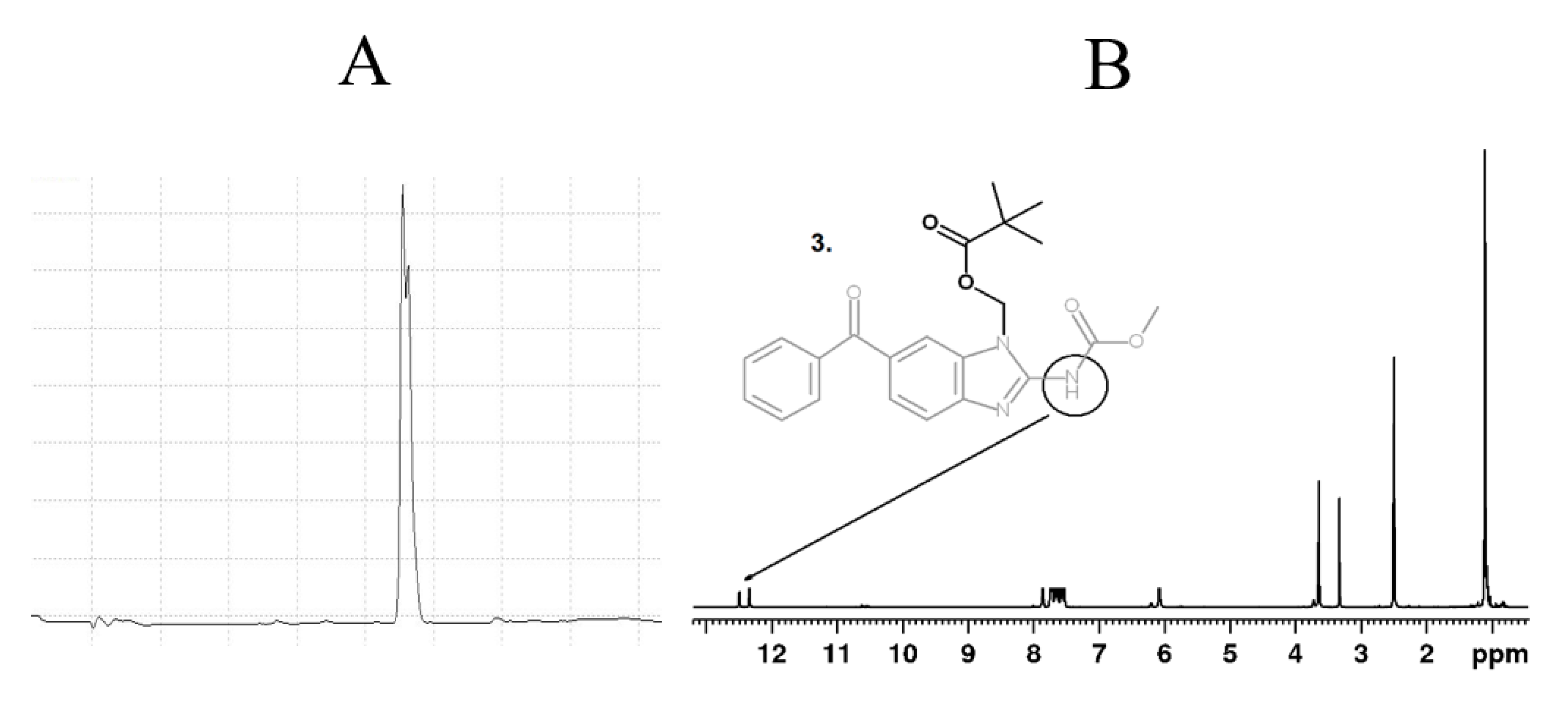
2.6. N-Pivaloyloxymethyl Mebendazole (3)
2.7. N-(3-Azidopropyloxycarbonyl) Mebendazole (4)
2.8. N-(6-(Boc-amino)hexanoyloxymethyl) Mebendazole (5)
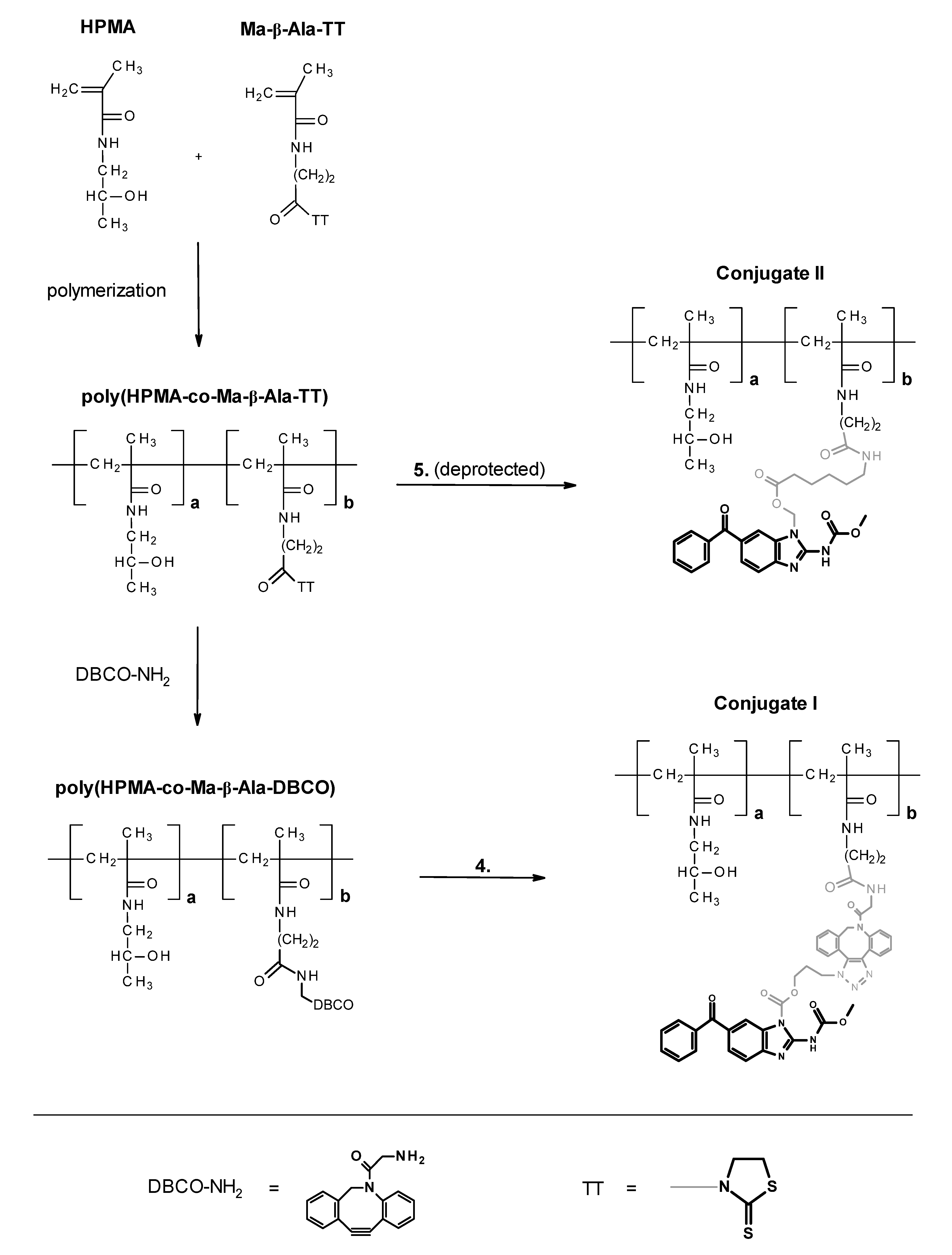
2.9. Polymer Conjugates of Derivatives 4 (Conjugate I) and 5 (Conjugate II)
2.10. N-(2-Hydroxypropyl)methacrylamide (HPMA)
2.11. 3-(3-Methacrylamidopropanoyl)thiazolidine-2-thione (Ma-β-Ala-TT)
2.12. poly(HPMA-co-Ma-β-Ala-TT)
2.13. poly(HPMA-co-Ma-β-Ala-DBCO)
2.14. Conjugate I (Polymer Bound Azido Derivative 4)
2.15. Conjugate II (Polymer Bound Amino Derivative 5)
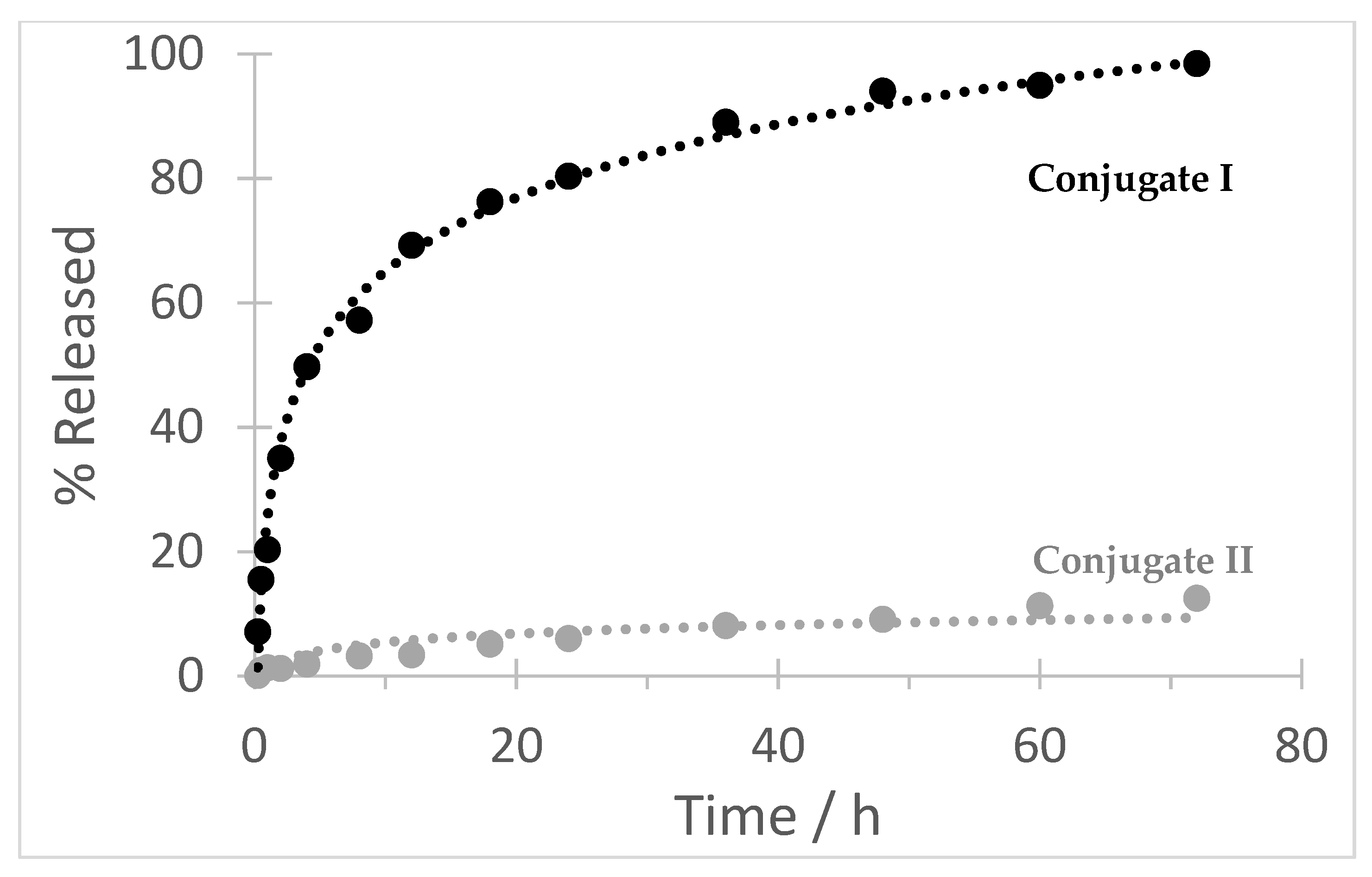
2.16. Hydrolytic Stability of the Conjugates
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cha, Y.; Erez, T.; Reynolds, I.J.; Kumar, D.; Ross, J.; Koytiger, G.; Kusko, R.; Zeskind, B.; Risso, S.; Kagan, E.; et al. Drug repurposing from the perspective of pharmaceutical companies. Br. J. Pharmacol. 2018, 175, 168–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mukhopadhyay, T.; Sasaki, J.; Ramesh, R.; Roth, J.A. Mebendazole elicits a potent antitumor effect on human cancer cell lines both in vitro and in vivo. Clin. Cancer Res. 2002, 8, 2963–2969. [Google Scholar] [PubMed]
- Chu, S.W.; Badar, S.; Morris, D.L.; Pourgholami, M.H. Potent inhibition of tubulin polymerisation and proliferation of paclitaxel-resistant 1A9PTX22 human ovarian cancer cells by albendazole. Anticancer Res. 2009, 29, 3791–3796. [Google Scholar]
- Dogra, N.; Kumar, A.; Mukhopadhyay, T. Fenbendazole acts as a moderate microtubule destabilizing agent and causes cancer cell death by modulating multiple cellular pathways. Sci. Rep. 2018, 8, 11926. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.J.; Luo, X.; Zhang, W.; Peng, F.; Cui, B.; Wu, S.J.; Zheng, F.M.; Xu, J.; Xu, L.Z.; Long, Z.J.; et al. Flubendazole, FDA-approved anthelmintic, targets breast cancer stem-like cells. Oncotarget 2015, 6, 6326–6340. [Google Scholar] [CrossRef] [Green Version]
- Sasaki, J.; Ramesh, R.; Chada, S.; Gomyo, Y.; Roth, J.A.; Mukhopadhyay, T. The anthelmintic drug mebendazole induces mitotic arrest and apoptosis by depolymerizing tubulin in non-small cell lung cancer cells. Mol. Cancer Ther. 2002, 1, 1201–1209. [Google Scholar]
- Rushworth, L.K.; Hewit, K.; Munnings-Tomes, S.; Somani, S.; James, D.; Shanks, E.; Dufes, C.; Straube, A.; Patel, R.; Leung, H.Y. Repurposing screen identifies mebendazole as a clinical candidate to synergise with docetaxel for prostate cancer treatment. Br. J. Cancer 2020, 122, 517–527. [Google Scholar] [CrossRef] [Green Version]
- Poruchynsky, M.S.; Komlodi-Pasztor, E.; Trostel, S.; Wilkerson, J.; Regairaz, M.; Pommier, Y.; Zhang, X.; Maity, T.K.; Robey, R.; Burotto, M.; et al. Microtubule-targeting agents augment the toxicity of DNA-damaging agents by disrupting intracellular trafficking of DNA repair proteins. Proc. Natl. Acad. Sci. USA 2015, 112, 1571–1576. [Google Scholar] [CrossRef] [Green Version]
- Celestino Pinto, L.; Moreira-Nunes, C.D.F.A.; Moreira Soares, B.; Rodrigues Burbano, R.M.; Rodrigues de Lemos, J.A.; Carvalho Montenegro, R. Mebendazole, an antiparasitic drug, inhibits drug transporters expression in preclinical model of gastric peritoneal carcinomatosis. Toxicol. In Vitro 2017, 43, 87–91. [Google Scholar] [CrossRef]
- Dawson, M.; Braithwaite, P.A.; Roberts, M.S.; Watson, T.R. The pharmacokinetics and bioavailability of a tracer dose of [3H]-mebendazole in man. Br. J. Clin. Pharmacol. 1985, 19, 79–86. [Google Scholar] [CrossRef] [Green Version]
- Dawson, M.; Allan, R.J.; Watson, T.R. The pharmacokinetics and bioavailability of mebendazole in man: A pilot study using [3H]-mebendazole. Br. J. Clin. Pharmacol. 1982, 14, 453–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dayan, A.D. Albendazole, mebendazole and praziquantel. Review of non-clinical toxicity and pharmacokinetics. Acta Trop. 2003, 86, 141–159. [Google Scholar] [CrossRef]
- Pinto, L.C.; Moreira Soares, B.; Viana Pinheiro, J.J.; Riggins, G.J.; Pimentel Assumpcao, P.; Rodriguez Burbano, R.M.; Carvalho Montenegro, R. The anthelmintic drug mebendazole inhibits growth, migration and invasion in gastric cancer cell model. Toxicol. In Vitro 2015, 29, 2038–2044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, R.Y.; Staedke, V.; Aprhys, C.M.; Gallia, G.L.; Riggins, G.J. Antiparasitic mebendazole shows survival benefit in 2 preclinical models of glioblastoma multiforme. Neuro Oncol. 2011, 13, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Simbulan-Rosenthal, C.M.; Dakshanamurthy, S.; Gaur, A.; Chen, Y.S.; Fang, H.B.; Abdussamad, M.; Zhou, H.; Zapas, J.; Calvert, V.; Petricoin, E.F.; et al. The repurposed anthelmintic mebendazole in combination with trametinib suppresses refractory NRASQ61K melanoma. Oncotarget 2017, 8, 12576–12595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blom, K.; Senkowsky, W.; Jarvius, M.; Berglund, M.; Rubin, J.; Lenhammar, L.; Parrow, V.; Andersson, C.; Loskog, A.; Fryknas, M.; et al. The anticancer effect of mebendazole may be due to M1 monocyte/macrophage activation via ERK1/2 and TLR8-dependent inflammasome activation. Immunopharmacol. Immunotoxicol. 2017, 39, 199–210. [Google Scholar] [CrossRef]
- Blom, K.; Rubin, J.; Berglund, M.; Jarvius, M.; Lenhammar, L.; Parrow, V.; Andersson, C.; Loskog, A.; Fryknas, M.; Nygren, P.; et al. Mebendazole-induced M1 polarisation of THP-1 macrophages may involve DYRK1B inhibition. BMC Res. Notes 2019, 12, 234. [Google Scholar] [CrossRef]
- Jornet, D.; Bosca, F.; Andreu, J.M.; Domingo, L.R.; Tormos, R.; Miranda, M.A. Analysis of mebendazole binding to its target biomolecule by laser flash photolysis. J. Photochem. Photobiol. B 2016, 155, 1–6. [Google Scholar] [CrossRef]
- Doudican, N.; Rodriguez, A.; Osman, I.; Orlow, S.J. Mebendazole induces apoptosis via Bcl-2 inactivation in chemoresistant melanoma cells. Mol. Cancer Res. 2008, 6, 1308–1315. [Google Scholar] [CrossRef] [Green Version]
- Sung, S.J.; Kim, H.K.; Hong, Y.K.; Joe, Y.A. Autophagy is a potential target for enhancing the anti-angiogenic effect of mebendazole in endothelial cells. Biomol. Ther. 2019, 27, 117–125. [Google Scholar] [CrossRef]
- Williamson, T.; Bai, R.Y.; Staedtke, V.; Huso, D.; Riggins, G.J. Mebendazole and a non-steroidal anti-inflammatory combine to reduce tumor initiation in a colon cancer preclinical model. Oncotarget 2016, 7, 68571–68584. [Google Scholar] [CrossRef] [Green Version]
- Zhang, F.; Li, Y.; Zhang, H.; Huang, E.; Gao, L.; Luo, W.; Wei, Q.; Fan, J.; Song, D.; Liao, J.; et al. Anthelmintic mebendazole enhances cisplatin’s effect on suppressing cell proliferation and promotes differentiation of head and neck squamous cell carcinoma (HNSCC). Oncotarget 2017, 8, 12968–12982. [Google Scholar] [CrossRef]
- Fernández-Bañares, F.; Gonzalez-Huix, F.; Xiol, X.; Catala, I.; Miro, J.; Lopez, N.; Casais, L. Marrow aplasia during high dose mebendazole treatment. Am. J. Trop. Med. Hyg. 1986, 35, 350–351. [Google Scholar] [CrossRef] [Green Version]
- Colle, I.; Naegels, S.; Hoorens, A.; Hautekeete, M. Granulomatous hepatitis due to mebendazole. J. Clin. Gastroenterol. 1999, 28, 44–45. [Google Scholar] [CrossRef]
- Duncan, R. Development of HPMA copolymer–anticancer conjugates: Clinical experience and lessons learnt. Adv. Drug Deliv. Rev. 2009, 61, 1131–1148. [Google Scholar] [CrossRef]
- Kopeček, J. Polymer-drug conjugates: Origins, progress to date and future directions. Adv. Drug Deliv. Rev. 2013, 65, 49–59. [Google Scholar] [CrossRef] [Green Version]
- Rihova, B.; Kovar, M. Immunogenicity and immunomodulatory properties of HPMA-based polymers. Adv. Drug Deliv. Rev. 2010, 62, 184–191. [Google Scholar] [CrossRef]
- Sirova, M.; Kabesova, M.; Kovar, L.; Etrych, T.; Strohalm, J.; Ulbrich, K.; Rihova, B. HPMA copolymer-bound doxorubicin induces immunogenic tumor cell death. Curr. Med. Chem. 2013, 20, 4815–4826. [Google Scholar] [CrossRef]
- Maeda, H. Tumor-selective delivery of macromolecular drugs via the EPR effect: Background and future prospects. Bioconjug. Chem. 2010, 21, 797–802. [Google Scholar] [CrossRef]
- Taurin, S.; Nehoff, H.; Greish, K. Anticancer nanomedicine and tumor vascular permeability; Where is the missing link? J. Control. Release 2012, 164, 265–275. [Google Scholar] [CrossRef]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar]
- Seymour, L.W.; Miyamoto, Y.; Maeda, H.; Brereton, M.; Strohalm, J.; Ulbrich, K.; Duncan, R. Influence of molecular weight on passive tumour accumulation of a soluble macromolecular drug carrier. Eur. J. Cancer 1995, 5, 766–770. [Google Scholar] [CrossRef]
- Simplício, A.L.; Clancy, J.M.; Gilmer, J.F. Prodrugs for amines. Molecules 2008, 13, 519–547. [Google Scholar] [CrossRef] [Green Version]
- Zimmermann, S.C.; Tichy, T.; Vavra, J.; Dash, R.P.; Slusher, C.E.; Gadiano, A.J.; Wu, Y.; Jancarik, A.; Tenora, L.; Monincova, L.; et al. N-substituted prodrugs of mebendazole provide improved aqueous solubility and oral bioavailability in mice and dogs. J. Med. Chem. 2018, 61, 3918–3929. [Google Scholar] [CrossRef]
- Studenovsky, M.; Pola, R.; Pechar, M.; Etrych, T.; Ulbrich, K.; Kovar, L.; Kabesova, M.; Rihova, B. Polymer carriers for anticancer drugs targeted to EGF receptor. Macromol. Biosci. 2012, 12, 1714–1720. [Google Scholar] [CrossRef]
- Pola, R.; Janouskova, O.; Etrych, T. The pH-dependent and enzymatic release of cytarabine from hydrophilic polymer conjugates. Physiol. Res. 2016, 65 (Suppl. 2), S225–S232. [Google Scholar] [CrossRef]
- Luttringhaus, A.; Dirksen, H.W. Tetramethylurea as a solvent and reagent. Angew. Chem. Int. Ed. 1964, 3, 260–269. [Google Scholar] [CrossRef]
- Graeme, M.; Rizzardo, E.; Thang, S.H. Radical addition–fragmentation chemistry in polymer synthesis. Polymer 2008, 49, 1079–1131. [Google Scholar]
- Kostka, L.; Subr, V.; Laga, R.; Chytil, P.; Ulbrich, K.; Seymour, L.W.; Etrych, T. Nanotherapeutics shielded with a pH responsive polymeric layer. Physiol. Res. 2015, 64, S29–S44. [Google Scholar] [CrossRef]

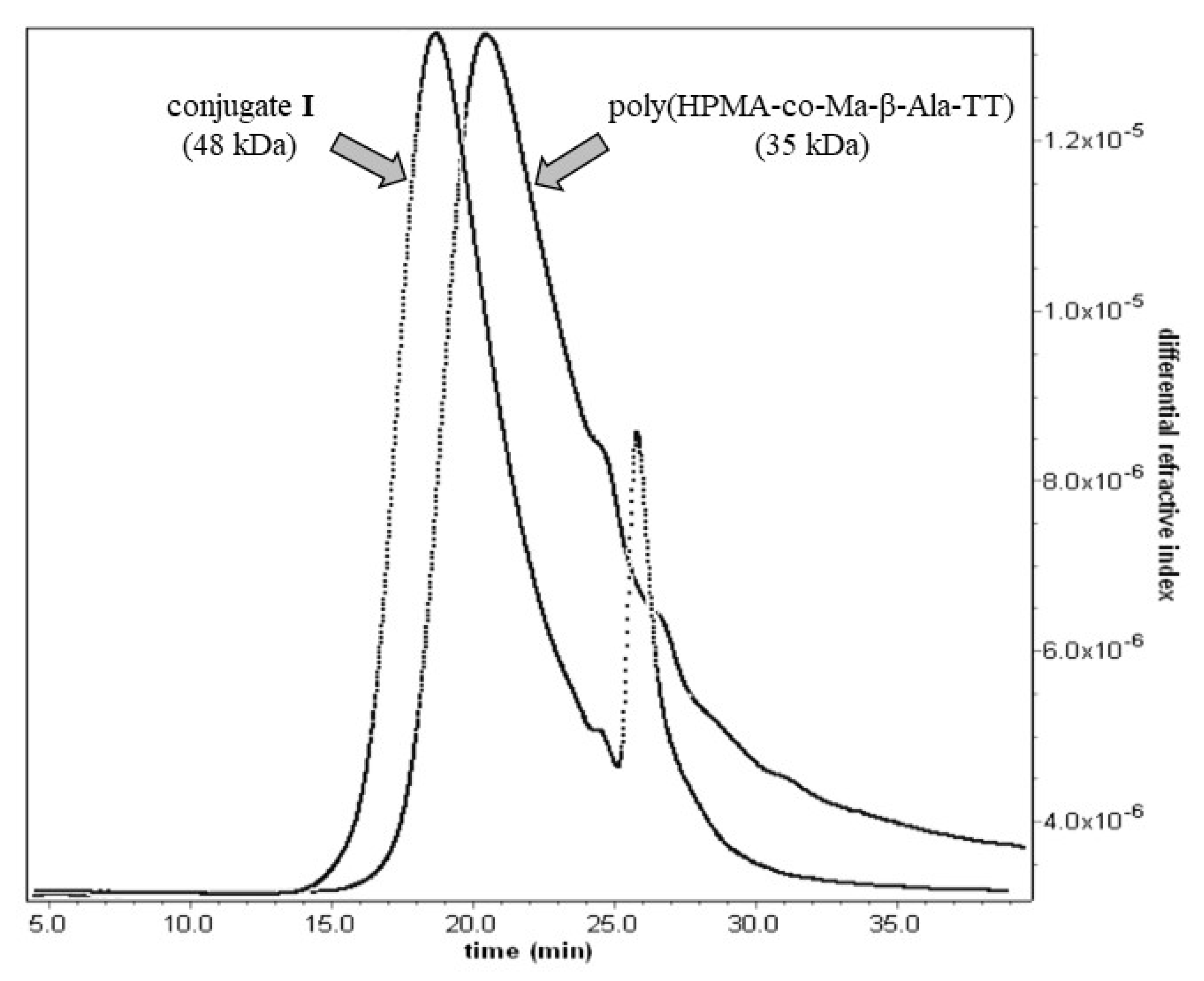
| Sample | Mw kDa | Mw/Mn | Size (Dh) nm | Functionality mmolg−1 | Group Type |
|---|---|---|---|---|---|
| poly(HPMA-co-Ma-β-Ala-TT) | 35 | 1.1 | 8.6 | 0.35 | TT |
| poly(HPMA-co-Ma-β-Ala-DBCO) | 40 | 1.2 | 10.5 | 0.32 | DBCO |
| Conjugate I | 48 | 1.2 | 16.3 | 0.31 | MBZ |
| Conjugate II | 37 | 1.1 | 10.3 | 0.22 | MBZ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Studenovský, M.; Rumlerová, A.; Kostka, L.; Etrych, T. HPMA-Based Polymer Conjugates for Repurposed Drug Mebendazole and Other Imidazole-Based Therapeutics. Polymers 2021, 13, 2530. https://doi.org/10.3390/polym13152530
Studenovský M, Rumlerová A, Kostka L, Etrych T. HPMA-Based Polymer Conjugates for Repurposed Drug Mebendazole and Other Imidazole-Based Therapeutics. Polymers. 2021; 13(15):2530. https://doi.org/10.3390/polym13152530
Chicago/Turabian StyleStudenovský, Martin, Anna Rumlerová, Libor Kostka, and Tomáš Etrych. 2021. "HPMA-Based Polymer Conjugates for Repurposed Drug Mebendazole and Other Imidazole-Based Therapeutics" Polymers 13, no. 15: 2530. https://doi.org/10.3390/polym13152530
APA StyleStudenovský, M., Rumlerová, A., Kostka, L., & Etrych, T. (2021). HPMA-Based Polymer Conjugates for Repurposed Drug Mebendazole and Other Imidazole-Based Therapeutics. Polymers, 13(15), 2530. https://doi.org/10.3390/polym13152530








