Role of Mg(NO3)2 as Defective Agent in Ameliorating the Electrical Conductivity, Structural and Electrochemical Properties of Agarose–Based Polymer Electrolytes
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of Polymer Electrolyte Films
2.3. Characterisation Techniques
2.3.1. Electrochemical Impedance Spectroscopy (EIS)
2.3.2. Fourier Transform Infrared (FTIR) Spectroscopy
2.3.3. X-ray Diffraction (XRD)
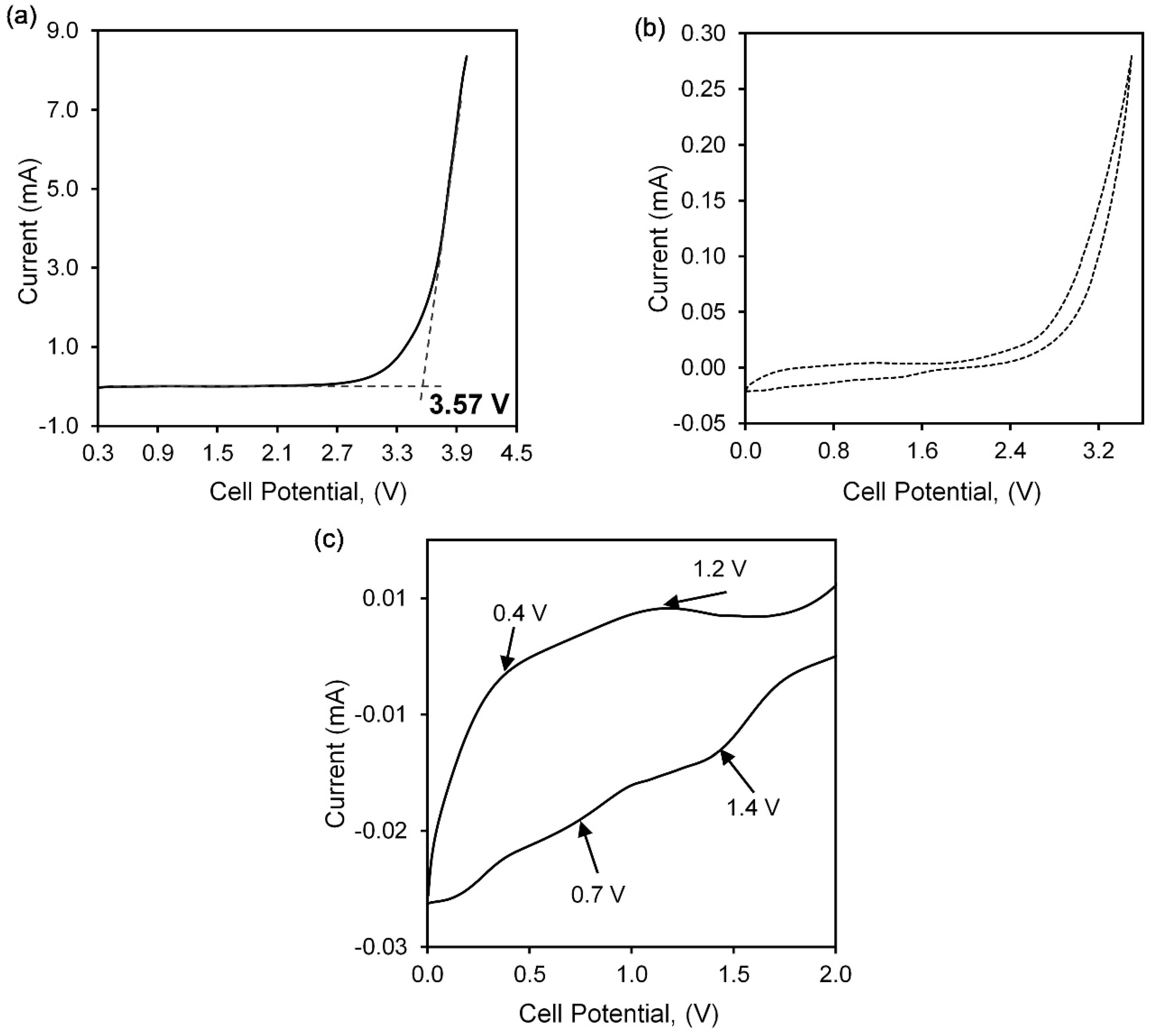
2.3.4. Electrochemical Characterisation
3. Results and Discussions
3.1. Impedance Analysis
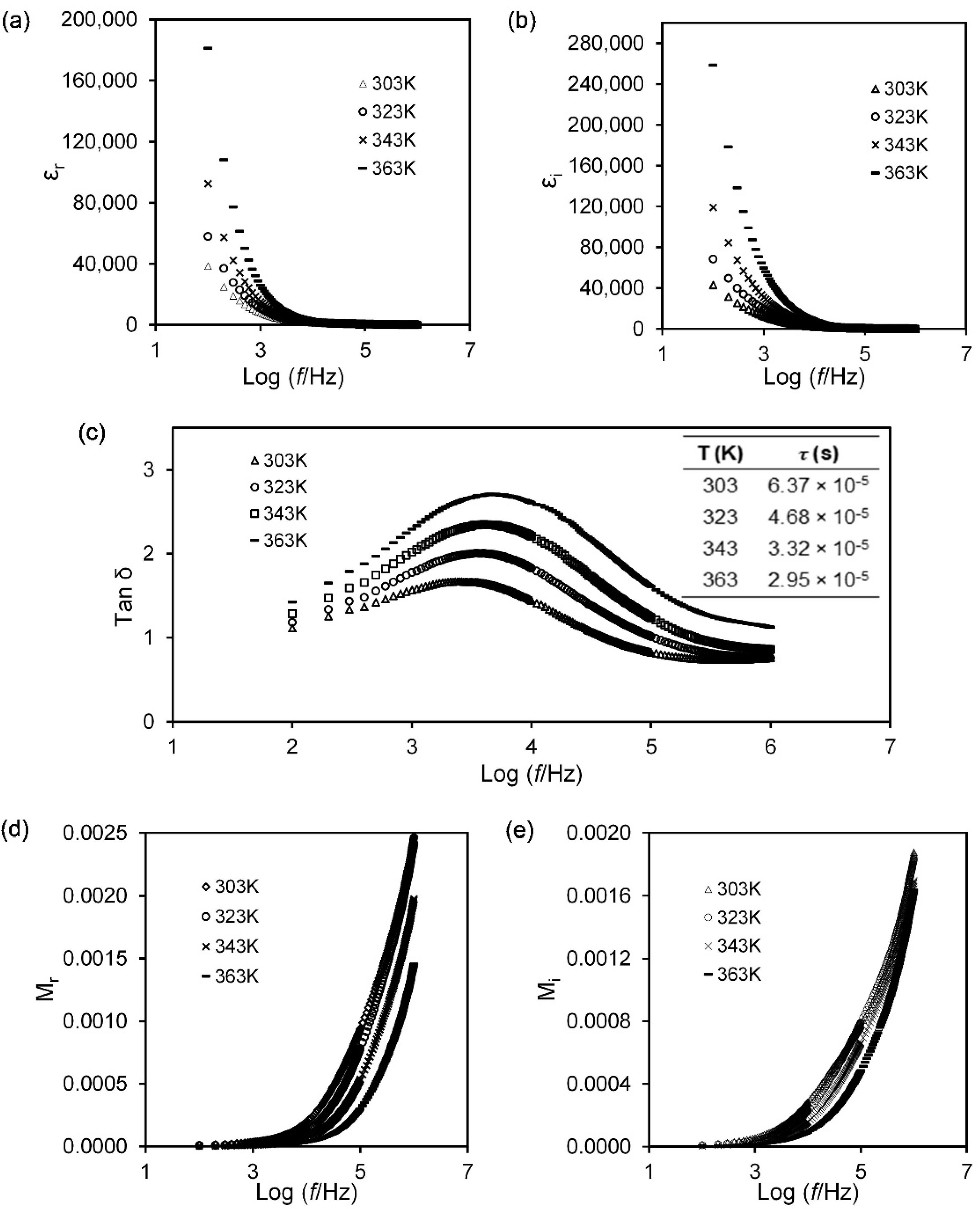
3.2. Dielectric Study
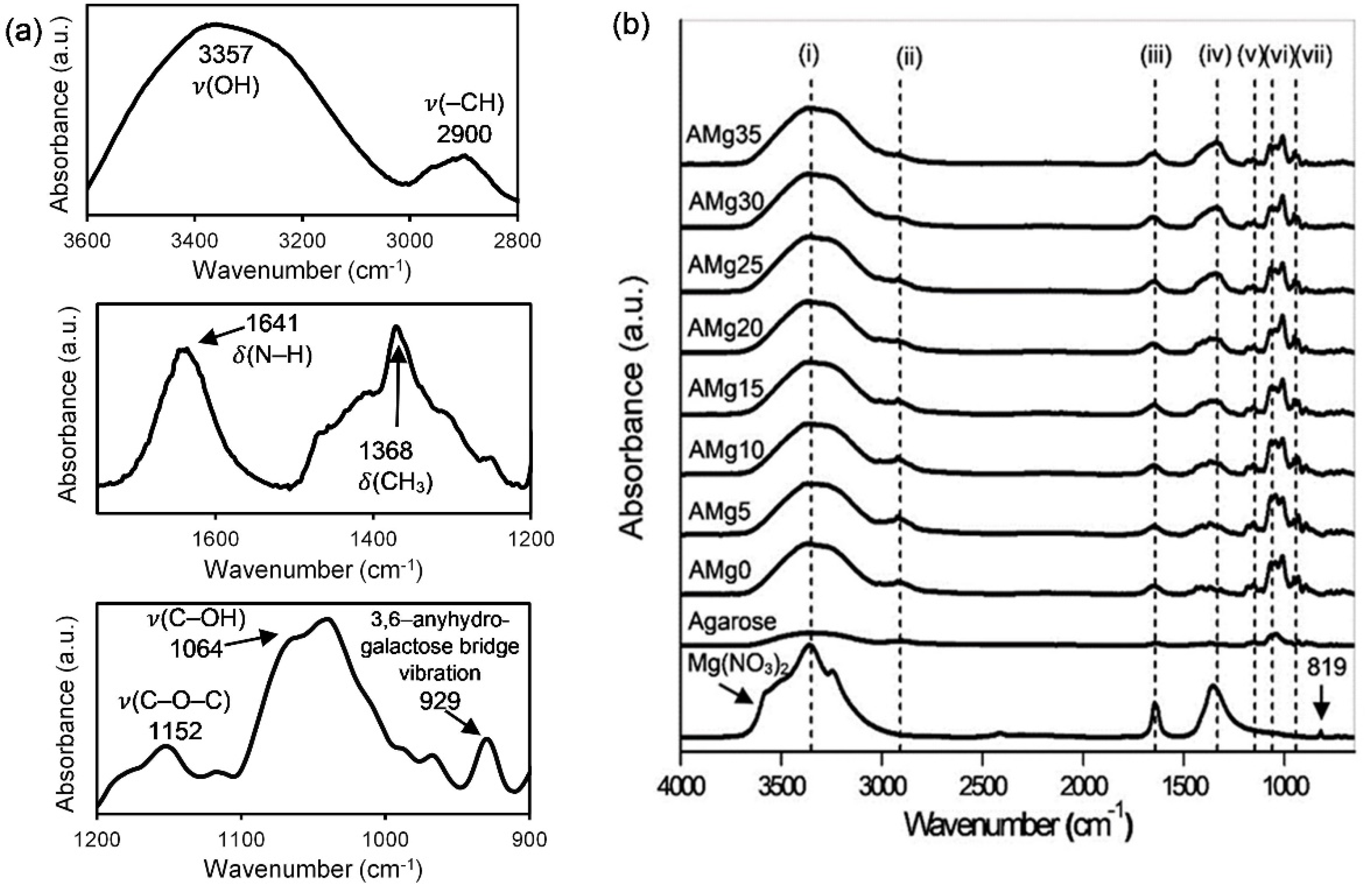
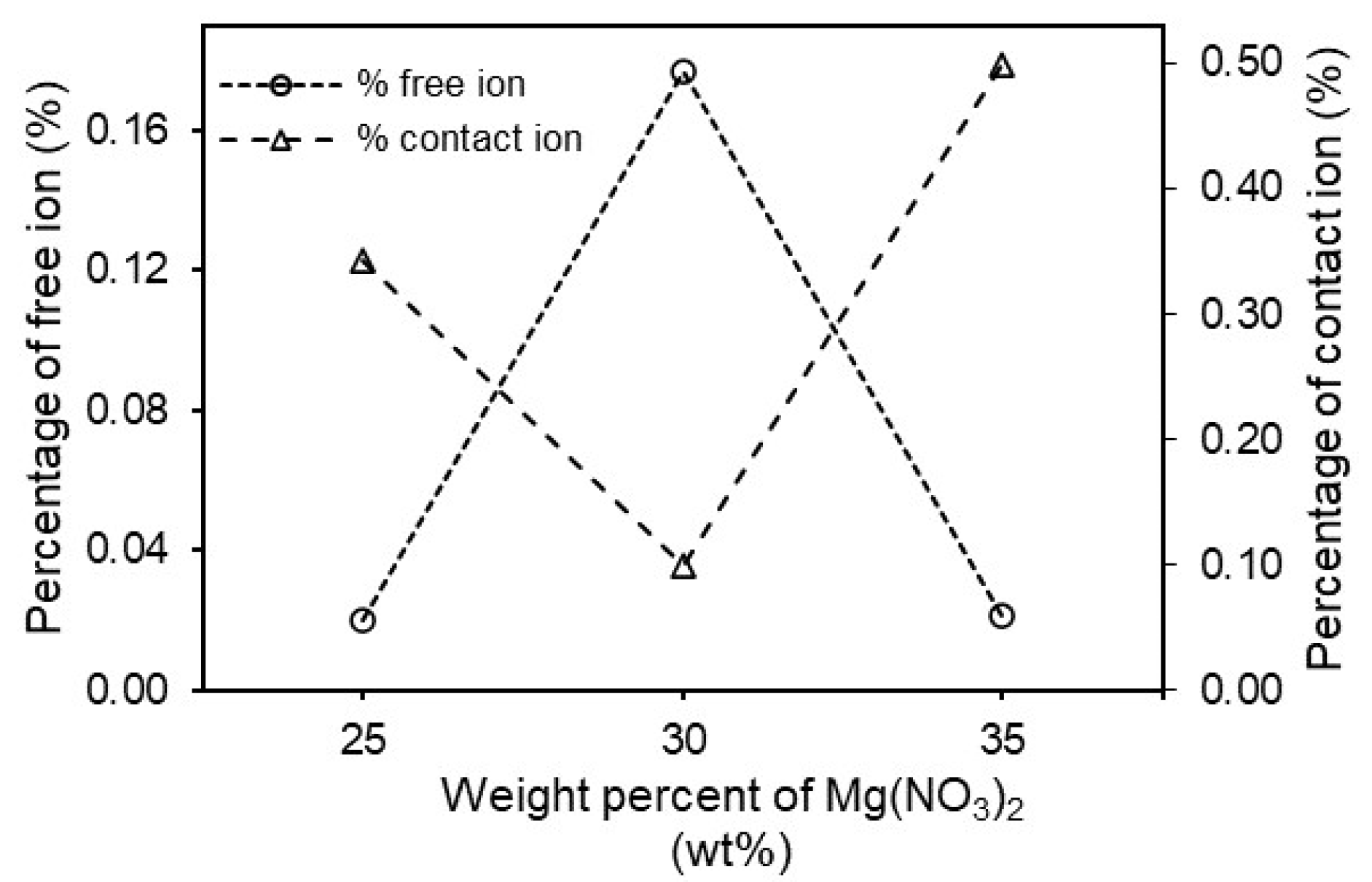
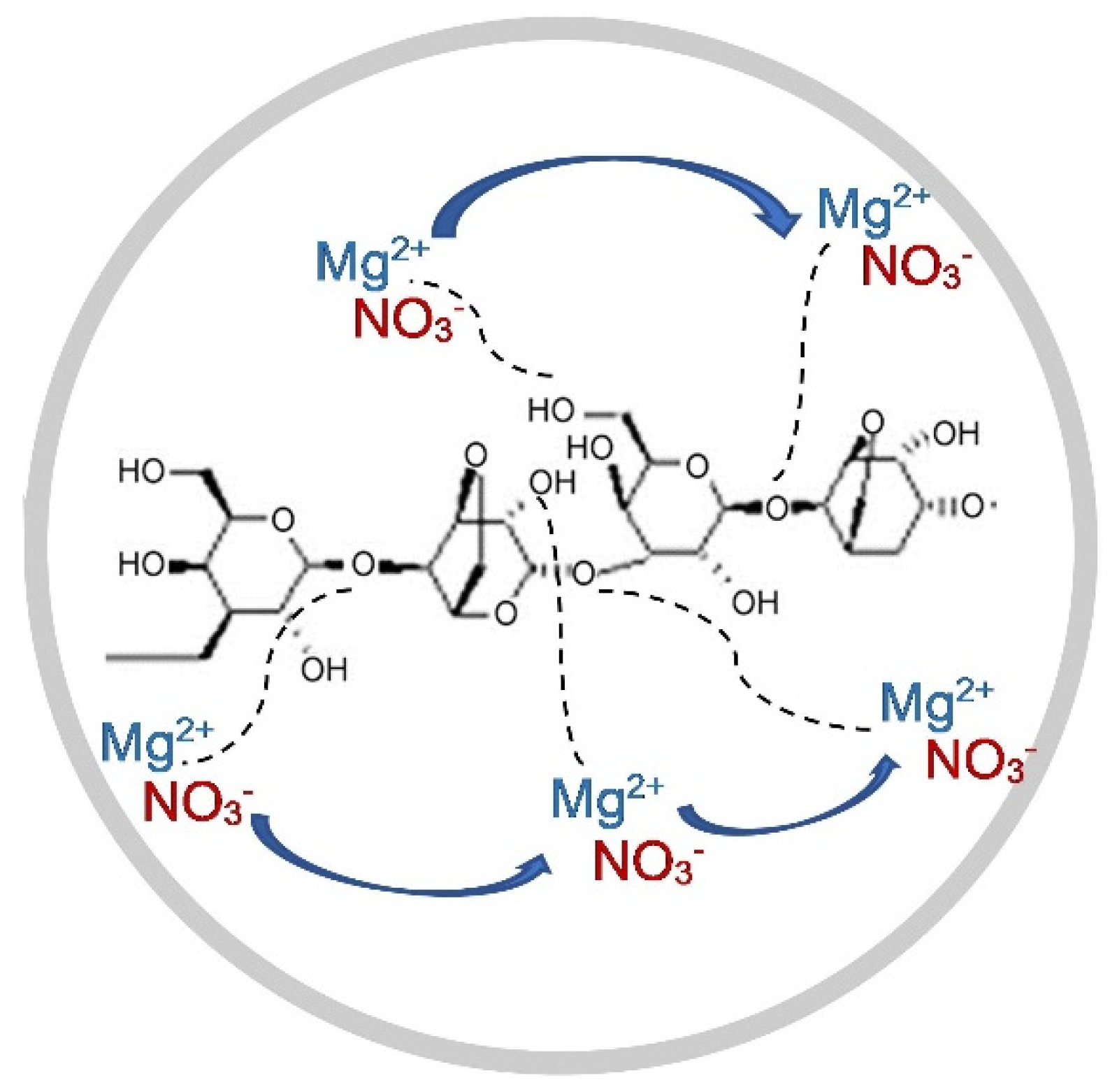
3.3. Fourier Transform Infrared (FTIR) Spectroscopy
3.4. XRD Analysis
3.5. Voltage Stability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, L.; Fan, X.; Hu, E.; Ji, X.; Chen, J.; Hou, S.; Deng, T.; Li, J.; Su, D.; Yang, X.; et al. Achieving high energy density through increasing the output voltage: A highly reversible 5.3 V battery. Chem 2019, 5, 896–912. [Google Scholar] [CrossRef]
- Aurbach, D.; Lu, Z.; Schechter, A.; Gofer, Y.; Gizbar, H.; Turgeman, R.; Cohen, Y.; Moshkovich, M.; Levi, E. Prototype systems for rechargeable magnesium batteries. Nature 2000, 407, 724–727. [Google Scholar] [CrossRef] [PubMed]
- Bucur, C.B.; Gregory, T.; Oliver, A.G.; Muldoon, J. Confession of a magnesium battery. J. Phys. Chem. Lett. 2015, 6, 3578–3591. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Zhang, W.; Lei, B.; Liu, H.; Li, W. Recent development of Mg ion solid electrolyte. Front. Chem. 2020, 8, 125. [Google Scholar] [CrossRef]
- Yoo, H.D.; Shterenberg, I.; Gofer, Y.; Gershinsky, G.; Pour, N.; Aurbach, D. Mg rechargeable batteries: An on-going challenge. Energy Environ. Sci. 2013, 6, 2265–2279. [Google Scholar] [CrossRef]
- Saha, P.; Datta, M.K.; Velikokhatnyi, O.I.; Manivannan, A.; Alman, D.; Kumta, P.N. Rechargeable magnesium battery: Current status and key challenges for the future. Prog. Mater. Sci. 2014, 66, 1–86. [Google Scholar] [CrossRef]
- Muldoon, J.; Bucur, C.B.; Oliver, A.G.; Sugimoto, T.; Matsui, M.; Kim, H.S.; Allred, G.D.; Zajicek, J.; Kotanie, Y. Electrolyte roadblocks to a magnesium rechargeable battery. Energy Environ. Sci. 2012, 5, 5941–5950. [Google Scholar] [CrossRef]
- Pan, W.; Guan, W.; Jiang, Y. Research advances in polyanion-type cathodes for sodium-ion batteries. Acta Phys. Chim. Sin. 2020, 36, 1905017. [Google Scholar] [CrossRef]
- Bella, F.; Luca, S.D.; Fagiolari, L.; Versaci, D.; Amici, J.; Francia, C.; Bodoardo, S. An overview on anodes for magnesium batteries: Challenges towards a promising storage solution for renewables. Nanomaterials 2021, 11, 810. [Google Scholar] [CrossRef]
- Polu, A.R.; Kumar, R.; Rhee, H.-W. Magnesium ion conducting solid polymer blend electrolyte based on biodegradable polymers and application in solid-state batteries. Ionics 2014, 21, 125–132. [Google Scholar] [CrossRef]
- Asmara, S.N.; Kufian, M.Z.; Majid, S.R.; Arof, A.K. Preparation and characterization of magnesium ion gel polymer electrolytes for application in electrical double layer capacitors. Electrochim. Acta 2011, 57, 91–97. [Google Scholar] [CrossRef]
- Sim, L.N.; Majid, S.R.; Arof, A.K. FTIR studies of PEMA/PVdF-HFP blend polymer electrolyte system incorporated with LiCF3 SO3 salt. Vib. Spectrosc. 2012, 58, 57–66. [Google Scholar] [CrossRef]
- Ponraj, T.; Ramalingam, A.; Selvasekarapandian, S.; Srikumar, S.R.; Manjuladevi, R. Plasticized solid polymer electrolyte based on triblock copolymer poly (vinylidene chloride-co-acrylonitrile-co-methyl methacrylate) for magnesium ion batteries. Polym. Bull. 2020, 78, 35–57. [Google Scholar] [CrossRef]
- Osman, Z.; Ghazali, M.I.M.; Othman, L.; Isa, K.B. AC ionic conductivity and DC polarization method of lithium ion transport in PMMA—LiBF 4 gel polymer electrolytes. Results Phys. 2012, 2, 1–4. [Google Scholar] [CrossRef]
- Sivakumar, M.; Subadevi, R.; Rajendran, S.; Wu, N.-L.; Lee, J.-Y. Electrochemical studies on [(1−x) PVA—x PMMA] solid polymer blend electrolytes complexed with LiBF 4. Mater. Chem. Phys. 2006, 97, 330–336. [Google Scholar] [CrossRef]
- Mohamed, N.S.; Arof, A.K. Investigation of electrical and electrochemical properties of PVDF-based polymer electrolytes. J. Power Sources 2004, 132, 229–234. [Google Scholar] [CrossRef]
- Li, Y.; Yerian, J.A.; Khan, S.A.; Fedkiw, P.S. Crosslinkable fumed silica-based nanocomposite electrolytes for rechargeable lithium batteries. J. Power Sources 2006, 161, 1288–1296. [Google Scholar] [CrossRef]
- Anilkumar, K.M.; Jinisha, B.; Manoj, M.; Jayalekshmi, S. Poly(ethylene oxide) (PEO)—poly(vinyl pyrrolidone) (PVP) blend polymer based solid electrolyte membranes for developing solid state magnesium ion cells. Eur. Polym. J. 2017, 89, 249–262. [Google Scholar] [CrossRef]
- Varshney, P.K.; Gupta, S. Natural polymer-based electrolytes for electrochemical devices: A review. Ionics 2011, 17, 479–483. [Google Scholar] [CrossRef]
- Yang, Y.; Cui, J.; Yi, P.; Zheng, X.; Guo, X.; Wang, W. Effects of nanoparticle additives on the properties of agarose polymer electrolytes. J. Power Sources 2014, 248, 988–993. [Google Scholar] [CrossRef]
- Yang, Y.; Hu, H.; Zhou, C.-H.; Xu, S.; Sebo, B.; Zhao, X.-Z. Novel agarose polymer electrolyte for quasi-solid state dye-sensitized solar cell. J. Power Sources 2011, 196, 2410–2415. [Google Scholar] [CrossRef]
- Singh, R.; Jadhav, N.A.; Majumder, S.; Bhattacharya, B.; Singh, P.K. Novel biopolymer gel electrolyte for dye-sensitized solar cell application. Carbohydr. Polym. 2013, 91, 682–685. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Guo, X.; Yang, Y. Lithium iodide effect on the electrochemical behavior of agarose based polymer electrolyte for dye-sensitized solar cell. Electrochim. Acta 2011, 56, 7347–7351. [Google Scholar] [CrossRef]
- Aziz, S.B.; Woo, T.J.; Kadir, M.F.Z.; Ahmed, H.M. A conceptual review on polymer electrolytes and ion transport models. J. Sci. Adv. Mater. Devices 2018, 3, 1–17. [Google Scholar] [CrossRef]
- Brza, M.A.; Aziz, S.B.; Anuar, H.; Ali, F. Structural, ion transport parameter and electrochemical properties of plasticized polymer composite electrolyte based on PVA: A novel approach to fabricate high performance EDLC devices. Polym. Test. 2020, 91, 106813. [Google Scholar] [CrossRef]
- Aziz, S.B.; Brza, M.A.; Dannoun, E.M.A.; Hamsan, M.H.; Hadi, J.M.; Kadir, M.F.Z.; Abdulwahid, R.T. The study of electrical and electrochemical properties of magnesium ion conducting CS: PVA based polymer blend electrolytes: Role of lattice energy of magnesium salts on EDLC performance. Molecules 2020, 25, 4503. [Google Scholar] [CrossRef]
- Polu, A.R.; Kumar, R. Impedance spectroscopy and FTIR studies of PEG—based polymer electrolytes. E J. Chem. 2011, 8, 347–353. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdullah, O.G.; Hussein, S.A.; Ahmed, H.M. Effect of PVA blending on structural and ion transport properties of CS: AgNt-based polymer. Polymers 2017, 9, 622. [Google Scholar] [CrossRef]
- Hafiza, M.N.; Isa, M.I.N. Solid polymer electrolyte production from 2-hydroxyethyl cellulose: Effect of NH4NO3 composition on its structural properties. Carbohydr. Polym. 2017, 165, 123–131. [Google Scholar] [CrossRef]
- Sim, L.N.; Yahya, R.; Arof, A.K. Blend polymer electrolyte films based on poly (ethyl methacrylate)/poly (vinylidene fluoride-co-hexa fluoropropylene) incorporated with 1-butyl-3-methyl imidazolium iodide ionic liquid. Solid State Ionics 2016, 291, 26–32. [Google Scholar] [CrossRef]
- Hafiza, M.N.; Isa, M.I.N. Analyses of ionic conductivity and dielectric behavior of solid polymer electrolyte based 2-hydroxyethyl cellulose doped ammonium nitrate plasticized with ethylene carbonate. AIP Conf. Proc. 2017, 1885, 020098. [Google Scholar] [CrossRef]
- Aji, M.P.; Bijaksana, S.; Abdullah, M. Electrical and magnetic properties of polymer electrolyte (PVA: LiOH) Containing in situ dispersed Fe3O4 nanoparticles. Int. Sch. Res. Not. 2012, 2012, 795613. [Google Scholar] [CrossRef][Green Version]
- Priya, S.S.; Karthika, M.; Selvasekarapandian, S.; Manjuladevi, R. Preparation and characterization of polymer electrolyte based on biopolymer I-Carrageenan with magnesium nitrate. Solid State Ionics 2018, 327, 136–149. [Google Scholar] [CrossRef]
- Ramalingaiah, S.; Reddy, D.S.; Reddy, M.J.; Laxminarsaiah, E.; Rao, U.V.S. Conductivity and discharge characteristic studies of novel polymer electrolyte based on PEO complexed with Mg(NO3)2 salt. Mater. Lett. 1996, 29, 285–289. [Google Scholar] [CrossRef]
- Mohamed, A.S.; Shukur, M.F.; Kadir, M.F.Z.; Yusof, Y.M. Ion conduction in chitosan-starch blend based polymer electrolyte with ammonium thiocyanate as charge provider. J. Polym. Res. 2020, 27, 1–14. [Google Scholar] [CrossRef]
- Fuzlin, A.F.; Bakri, N.A.; Sahraoui, B.; Samsudin, A.S. Study on the effect of lithium nitrate in ionic conduction properties based alginate biopolymer electrolytes. Mater. Res. Express 2019, 7, 015902. [Google Scholar] [CrossRef]
- Ponraj, T.; Ramalingam, A.; Selvasekarapandian, S.; Srikumar, S.R.; Manjuladevi, R. Mg-ion conducting triblock copolymer electrolyte based on poly (VdCl-co-AN-co-MMA) with magnesium nitrate. Ionics 2019, 26, 789–800. [Google Scholar] [CrossRef]
- Manjuladevi, R.; Thamilselvan, M.; Selvasekarapandian, S.; Mangalam, R.; Premalatha, M.; Monisha, S. Mg-ion conducting blend polymer electrolyte based on poly (vinyl alcohol)—poly (acrylonitrile) with magnesium perchlorate. Solid State Ionics 2017, 308, 90–100. [Google Scholar] [CrossRef]
- Singh, R.; Bhattacharya, B.; Tomar, S.K.; Singh, V.; Singh, P.K. Electrical, optical and electrophotochemical studies on agarose based biopolymer electrolyte towards dye sensitized solar cell application. Measurement 2017, 102, 214–219. [Google Scholar] [CrossRef]
- Hafiza, M.N.; Bashirah, A.N.A.; Bakar, N.Y.; Isa, M.I.N. Electrical properties of carboxyl methylcellulose/chitosan dual-blend green polymer doped with ammonium bromide. Int. J. Polym. Anal. Charact. 2014, 19, 151–158. [Google Scholar] [CrossRef]
- Selvalakshmi, S.; Vijaya, N.; Selvasekarapandian, S.; Premalatha, M. Biopolymer agar-agar doped with NH4SCN as solid polymer electrolyte for electrochemical cell application. J. Appl. Polym. Sci. 2017, 134, 1–10. [Google Scholar] [CrossRef]
- Guo, X.; Yi, P.; Yang, Y.; Cui, J.; Xiao, S.; Wang, W. Effects of surfactants on agarose-based magnetic polymer electrolyte for dye-sensitized solar cells. Electrochim. Acta 2013, 90, 524–529. [Google Scholar] [CrossRef]
- Zarei, Z.; Akhlaghinia, B. Cu (II) immobilized on Fe3O4@Agarose nanomagnetic catalyst functionalized with ethanolamine phosphate—salicylaldehyde Schiff base: A magnetically reusable nanocatalyst for preparation of 2-substituted imidazolines, oxazolines, and thiazolines. Turk. J. Chem. 2018, 42, 170–191. [Google Scholar] [CrossRef]
- Wang, Y.; Li, C.; Liu, P.; Ahmed, Z.; Xiao, P.; Bai, X. Physical characterization of exopolysaccharide produced by Lactobacillus plantarum KF5 isolated from Tibet Kefir. Carbohydr. Polym. 2010, 82, 895–903. [Google Scholar] [CrossRef]
- Lee, J.Y.; Bhattacharya, B.; Kim, D.-W.; Park, J.-K. Poly (ethylene oxide)/poly (dimethylsiloxane) blend solid polymer electrolyte and its dye-sensitized solar cell applications. J. Phys. Chem. C 2008, 112, 12576–12582. [Google Scholar] [CrossRef]
- Manjuladevi, R.; Selvasekarapandian, S.; Thamilselvan, M.; Mangalam, R.; Monisha, S.; Selvin, P.C. A study on blend polymer electrolyte based on poly (vinyl alcohol) -poly (acrylonitrile) with magnesium nitrate for magnesium battery. Ionics 2018, 24, 3493–3506. [Google Scholar] [CrossRef]
- Wahab, A.; Mahiuddin, S.; Hefter, G.; Kunz, W.; Minofar, B.; Jungwirth, P. Ultrasonic velocities, densities, viscosities, electrical conductivities, raman spectra, and molecular dynamics simulations of aqueous solutions of Mg(OAc)2 and Mg(NO3)2: Hofmeister effects and ion pair formation. J. Phys. Chem. B 2005, 109, 24108–24120. [Google Scholar] [CrossRef]
- Wahab, A.; Mahiuddin, S. Density, ultrasonic velocity, electrical conductivity, viscosity, and raman spectra of methanolic Mg(ClO4)2, Mg(NO3)2, and Mg(OAc)2 solutions. J. Chem. Eng. Data 2009, 54, 436–443. [Google Scholar] [CrossRef]
- Béléké, A.B.; Mizuhata, M.; Kajinami, A.; Deki, S. Diffuse reflectance FT-IR spectroscopic study of interactions of α -Al2O3/molten alkali nitrate coexisting systems. J. Colloid Interface Sci. 2003, 268, 413–424. [Google Scholar] [CrossRef]
- Sulaiman, M.; Rahman, A.A.; Mohamed, N.S. Structural, thermal and conductivity studies of magnesium nitrate—alumina composite solid electrolytes prepared via sol-gel method. Int. J. Electrochem. Sci. 2013, 8, 6647–6655. [Google Scholar] [CrossRef]
- Hafiza, M.N.; Isa, M.I.N. Correlation between structura, ion transport and ionic conductivity of plasticized 2-hydroxyethyl cellulose based solid biopolymer electrolyte. J. Memb. Sci. 2020, 597, 117176. [Google Scholar] [CrossRef]
- Pandey, G.P.; Agrawal, R.C.; Hashmi, S.A. Performance studies on composite gel polymer electrolytes for rechargeable magnesium battery application. J. Phys. Chem. Solids 2011, 72, 1408–1413. [Google Scholar] [CrossRef]
- Aziz, A.A.; Tominaga, Y. Magnesium ion-conductive poly (ethylene carbonate) electrolytes. Ionics 2018, 24, 3475–3481. [Google Scholar] [CrossRef]




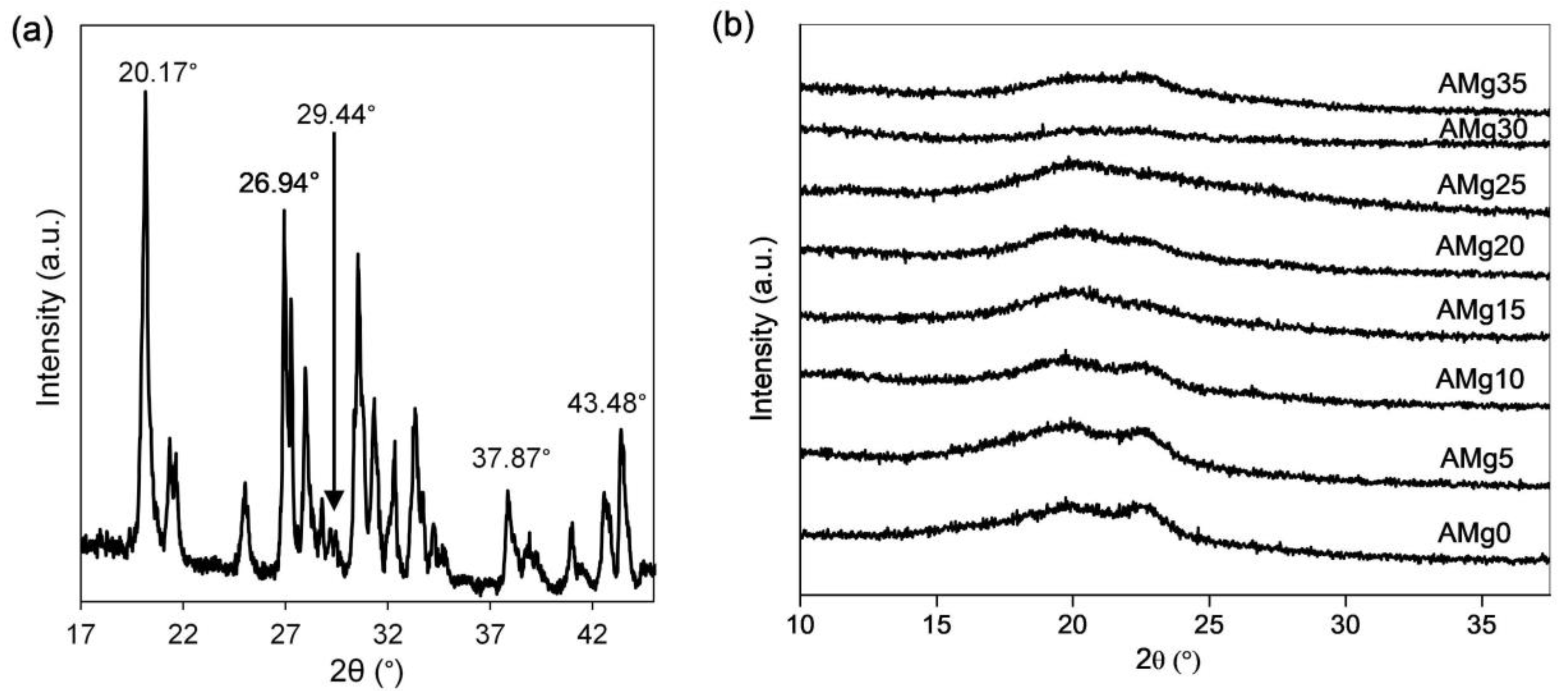
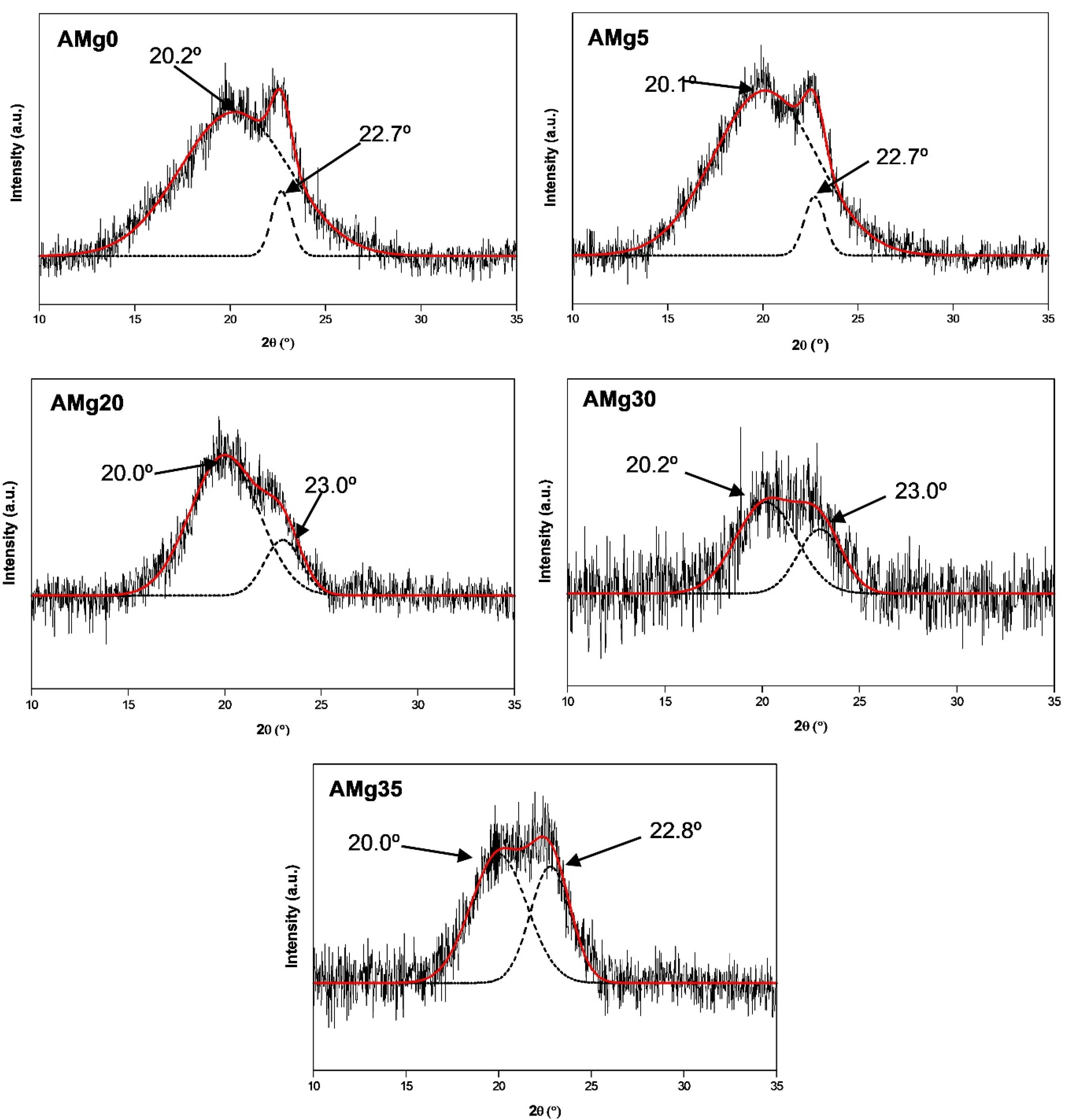
| Mg(NO2)3 Content (wt%) | Degree of Crytallinity, Xc (%) | FWHM (rad) | Crystalline Size, L (nm) |
|---|---|---|---|
| 0 | 38.58 | 0.0214 | 6.62 |
| 5 | 36.69 | 0.0225 | 6.28 |
| 20 | 29.72 | 0.0392 | 3.61 |
| 30 | 11.29 | 0.0468 | 3.03 |
| 35 | 37.94 | 0.0446 | 3.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, N.I.; Abidin, S.Z.Z.; Majid, S.R.; Jaafar, N.K. Role of Mg(NO3)2 as Defective Agent in Ameliorating the Electrical Conductivity, Structural and Electrochemical Properties of Agarose–Based Polymer Electrolytes. Polymers 2021, 13, 3357. https://doi.org/10.3390/polym13193357
Ali NI, Abidin SZZ, Majid SR, Jaafar NK. Role of Mg(NO3)2 as Defective Agent in Ameliorating the Electrical Conductivity, Structural and Electrochemical Properties of Agarose–Based Polymer Electrolytes. Polymers. 2021; 13(19):3357. https://doi.org/10.3390/polym13193357
Chicago/Turabian StyleAli, N. I., S. Z. Z. Abidin, S. R. Majid, and N. K. Jaafar. 2021. "Role of Mg(NO3)2 as Defective Agent in Ameliorating the Electrical Conductivity, Structural and Electrochemical Properties of Agarose–Based Polymer Electrolytes" Polymers 13, no. 19: 3357. https://doi.org/10.3390/polym13193357
APA StyleAli, N. I., Abidin, S. Z. Z., Majid, S. R., & Jaafar, N. K. (2021). Role of Mg(NO3)2 as Defective Agent in Ameliorating the Electrical Conductivity, Structural and Electrochemical Properties of Agarose–Based Polymer Electrolytes. Polymers, 13(19), 3357. https://doi.org/10.3390/polym13193357







