Chitosan/Polyvinyl Alcohol/Tea Tree Essential Oil Composite Films for Biomedical Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Composition of Tea Tree Essential Oil (TTEO)
2.3. Preparation and Characterization of CS/TTEO Emulsions
2.3.1. Particle Size
2.3.2. Viscosity Measurements
2.3.3. Total Solids
2.4. CS/PVA/TTEO Films Preparation
2.4.1. Characterization of CS/PVA and CS/PVA/TTEO Films
Fourier Transform Infrared Spectroscopy (FT-IR)
Scanning Electron Microscopy (SEM)
Analysis of Mechanical Properties
Thermal Stability of Films
Degradation in Simulated Biological Fluid
In Vivo Biomodel Evaluations
Statistical Analysis
3. Results
3.1. Tea Tree Essential Oil Characterization
3.2. Physical–Chemical Characterization of the Film-Forming Emulsions (FFE)
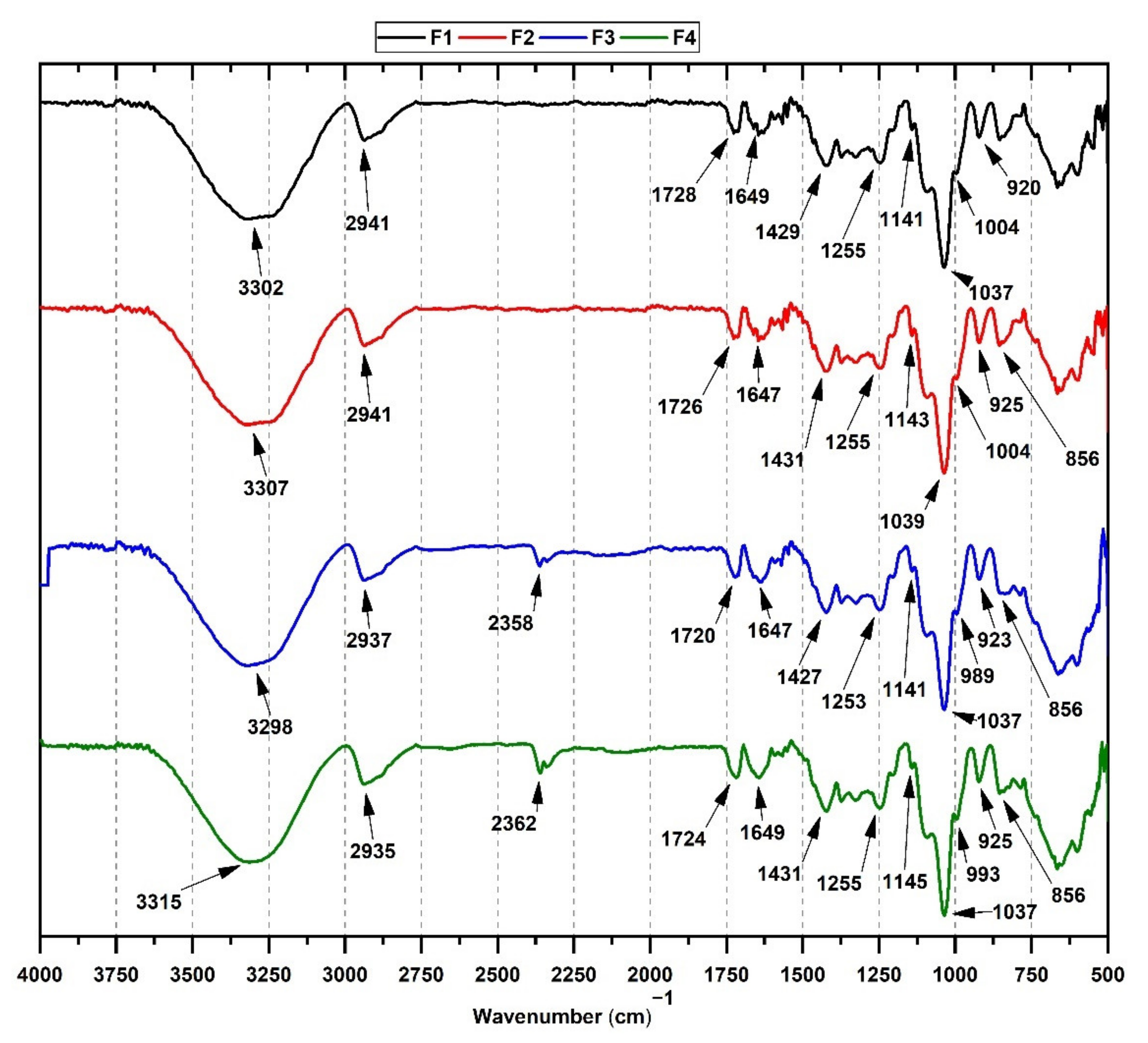
3.3. FT-IR from CS/PVA/TTEO Films
3.4. The Tensile Strength of the CS/PVA/TTEO Films
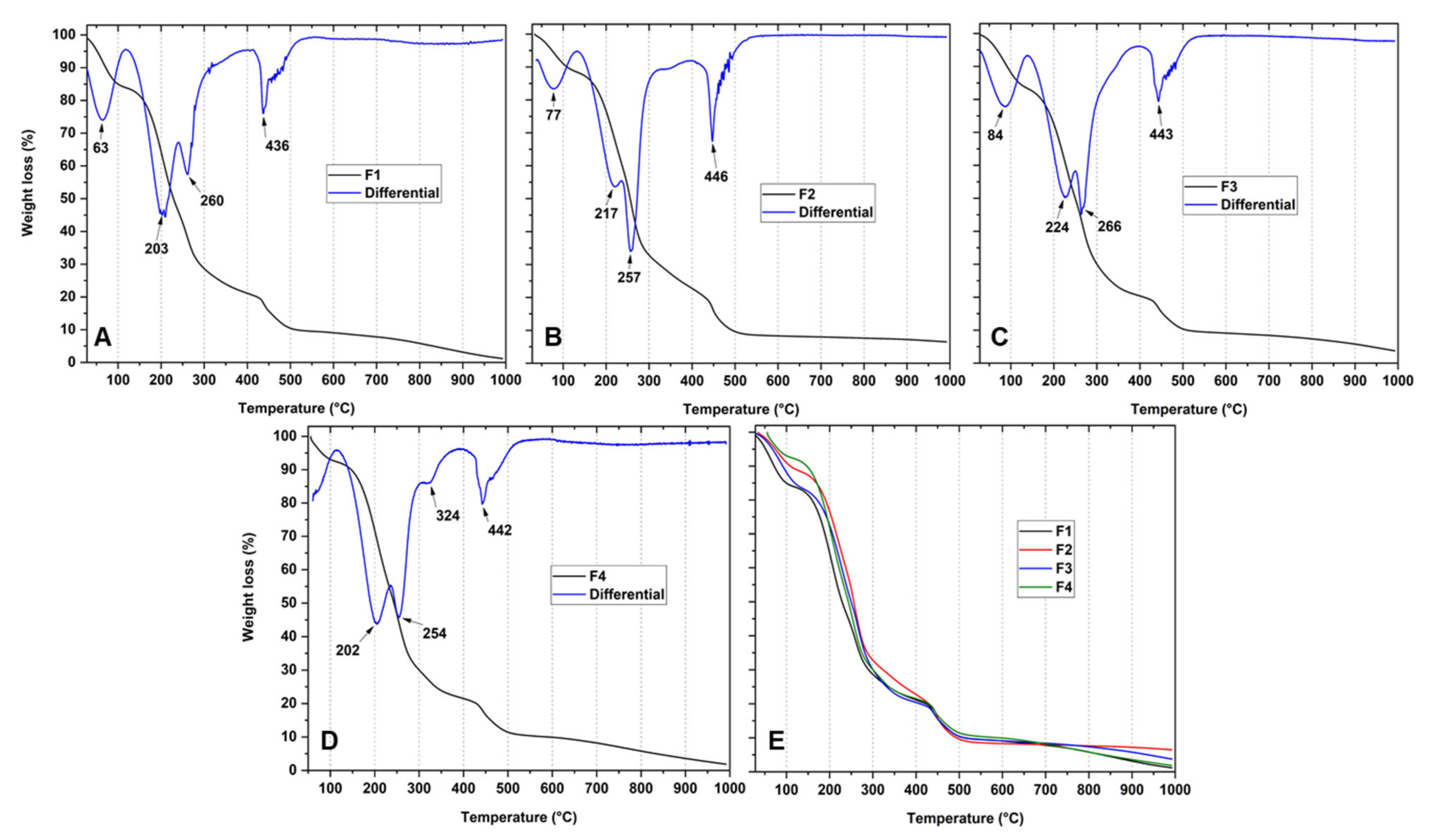
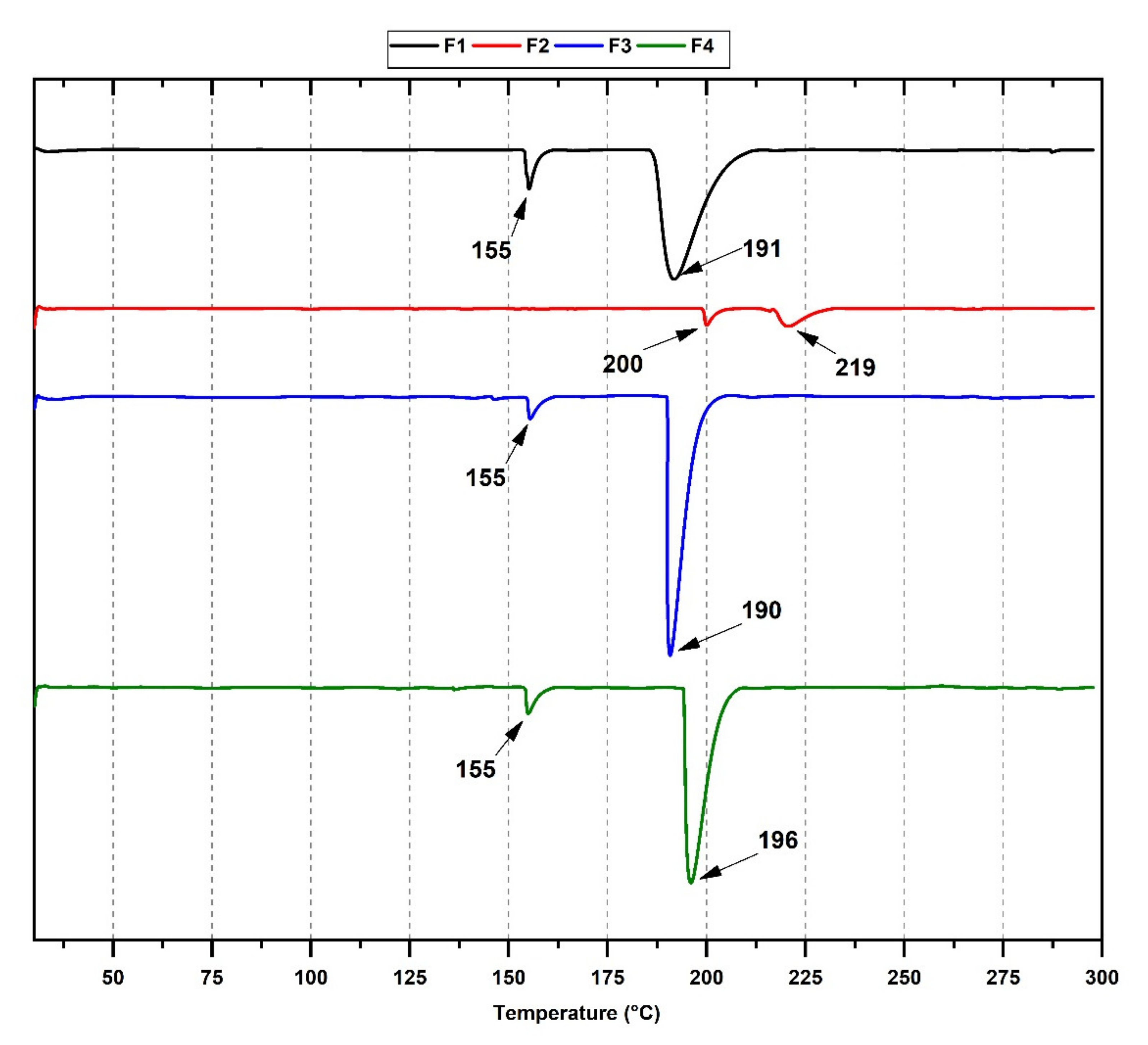
3.5. Thermal Analysis of CS/PVA/TTEO Films
3.6. Scanning Electron Microscopy (SEM) of CS/PVA/TTEO Films
3.7. CS/PVA/TTEO Film Degradation in Simulated Body Fluid (SBF)
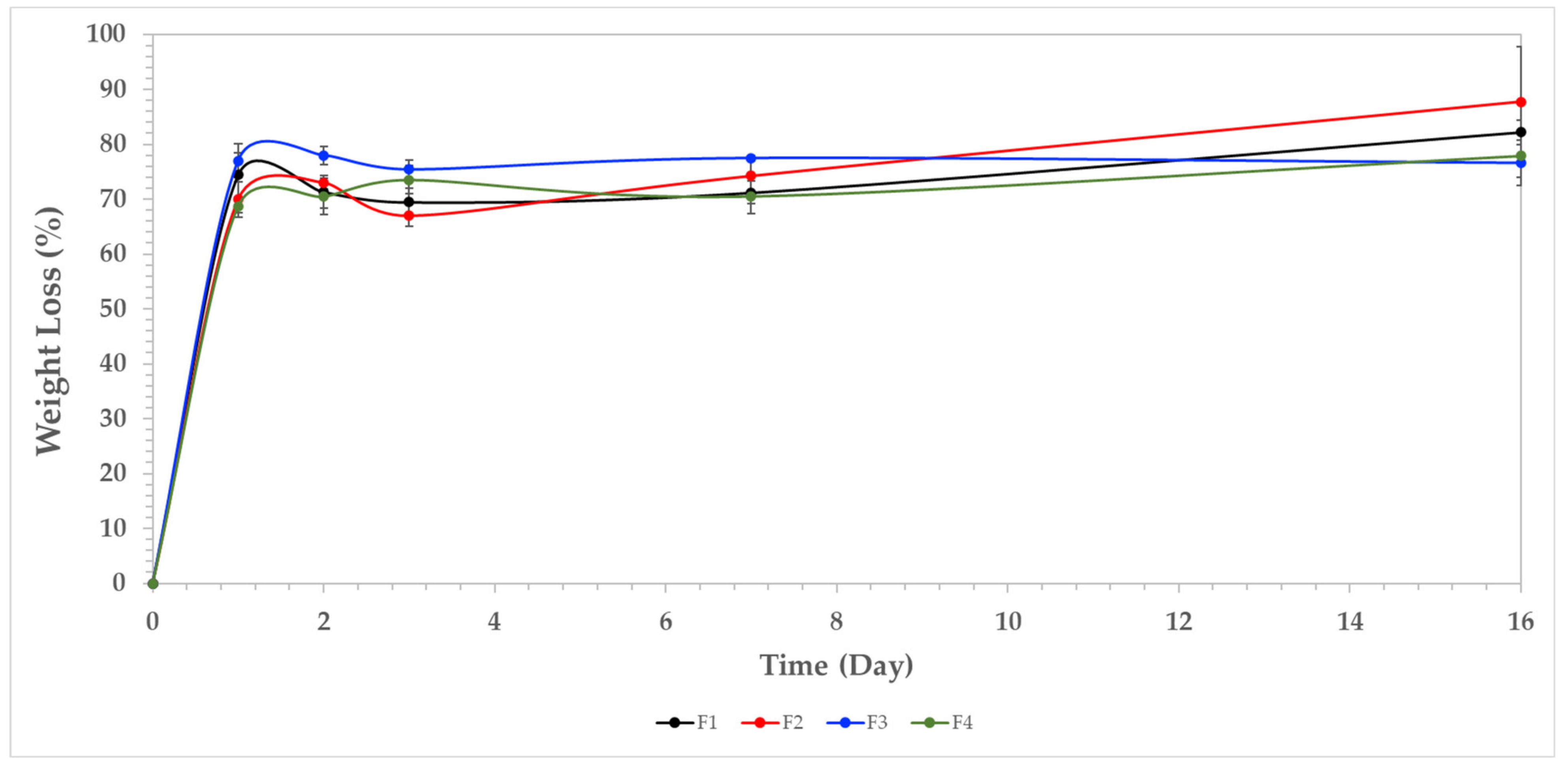
3.7.1. Weight Loss
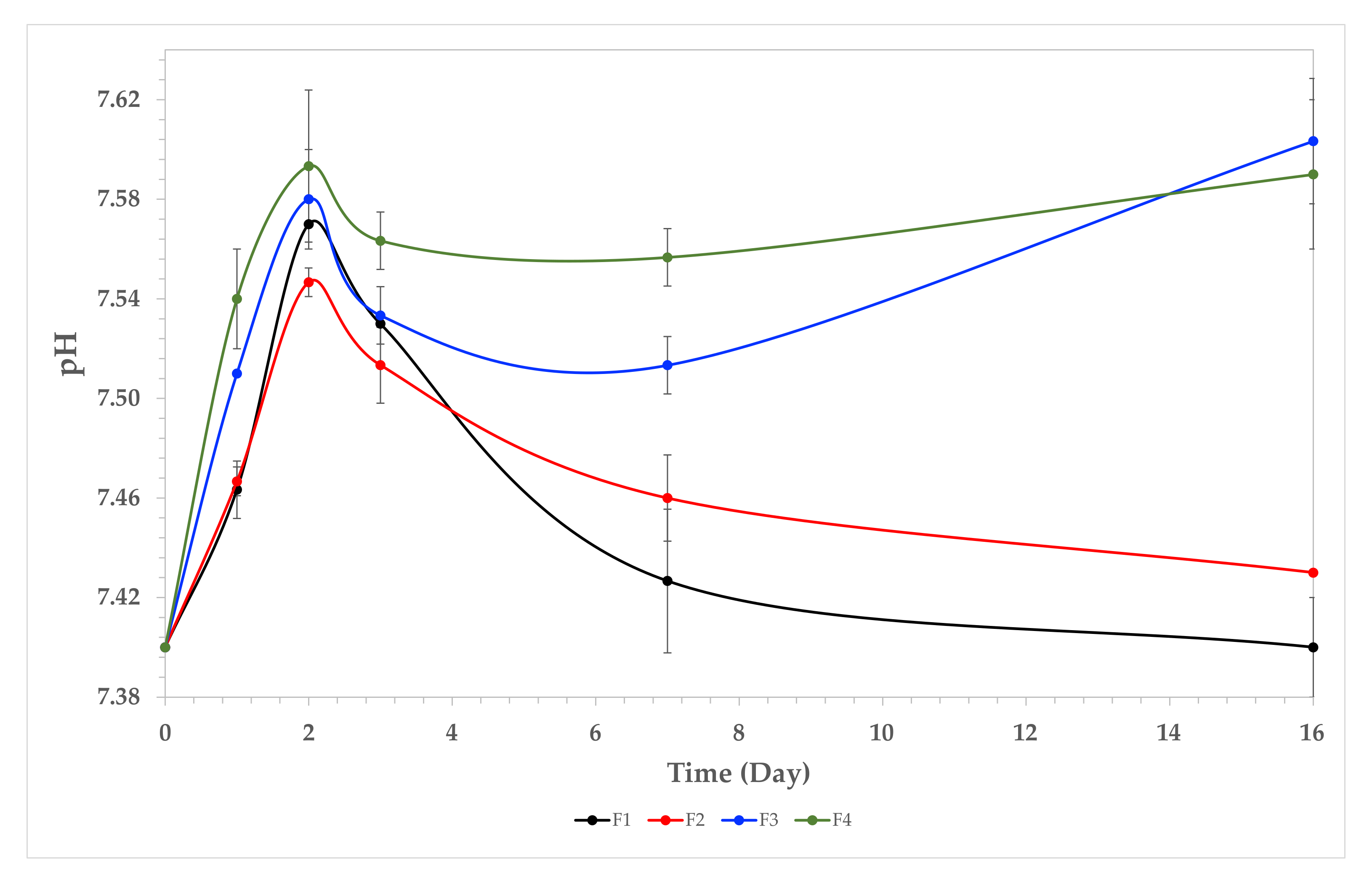
3.7.2. pH Changes
3.7.3. SEM of Films after Immersion in SBF
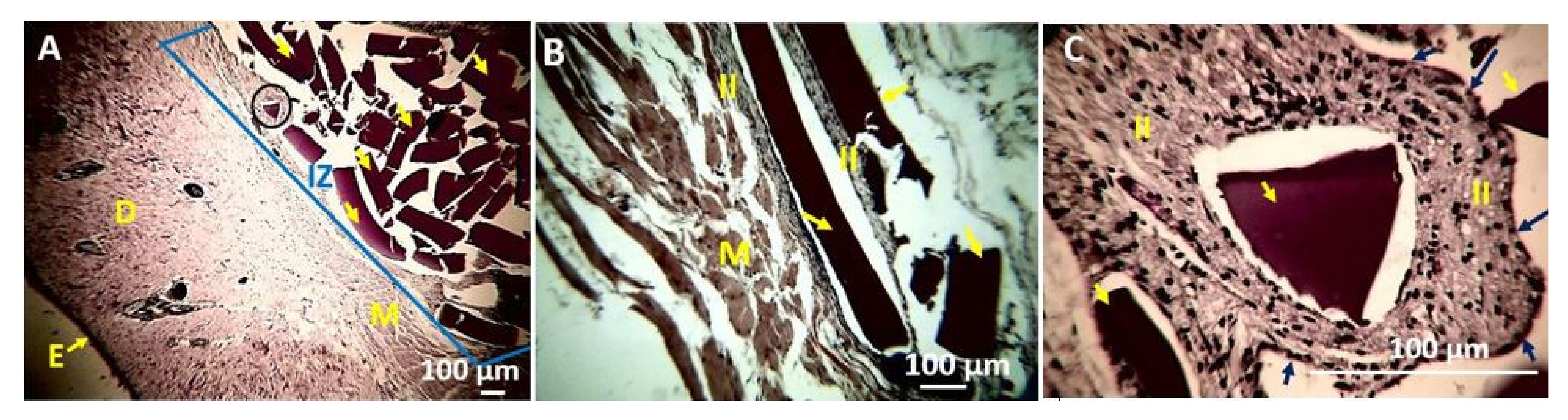
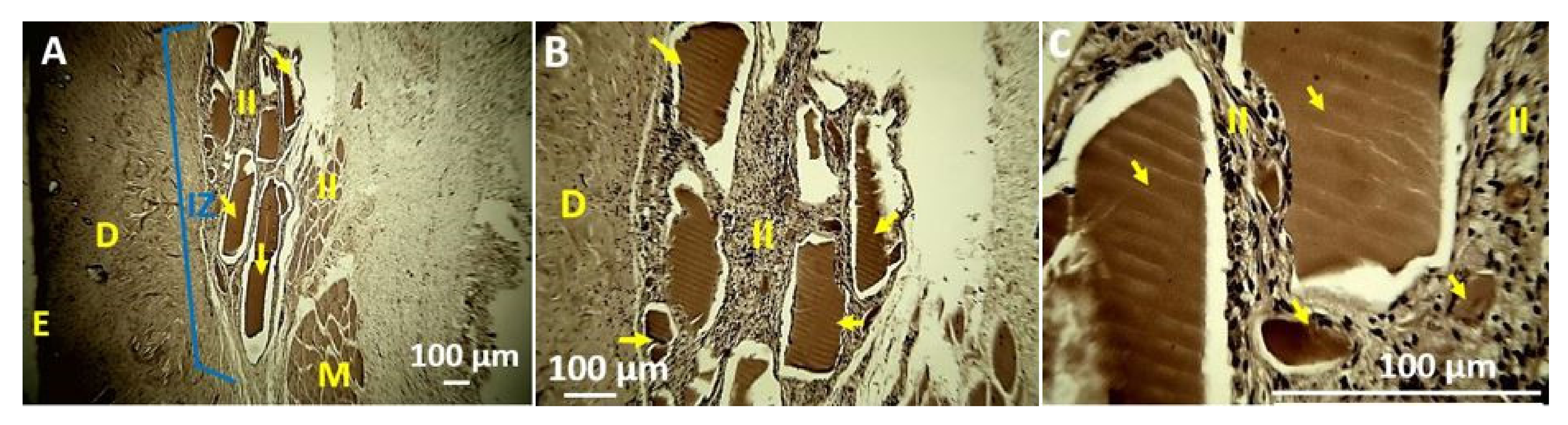
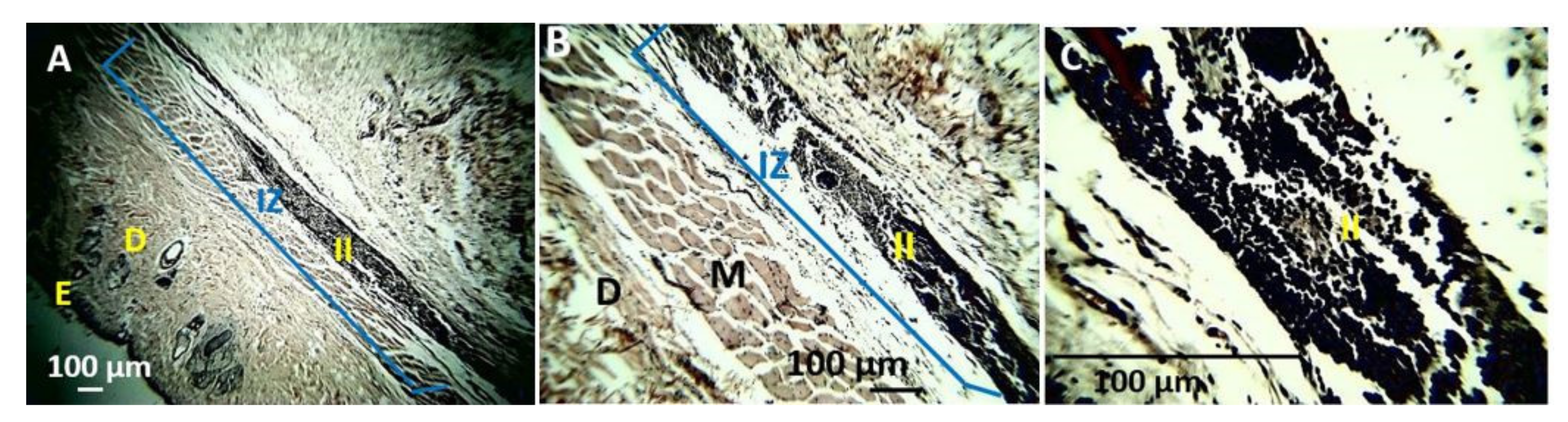
4. In Vivo Biocompatibility Tests of the CS/PVA/TTEO Films
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahlawat, J.; Kumar, V.; Gopinath, P. Carica papaya loaded poly (vinyl alcohol)-gelatin nanofibrous scaffold for potential application in wound dressing. Mater. Sci. Eng. C 2019, 103, 109834. [Google Scholar] [CrossRef] [PubMed]
- Semba, J.A.; Mieloch, A.A.; Rybka, J.D. Introduction to the state-of-the-art 3D bioprinting methods, design, and applications in orthopedics. Bioprinting 2020, 18, e00070. [Google Scholar] [CrossRef]
- Yin, S.; Zhang, W.; Zhang, Z.; Jiang, X. Recent Advances in Scaffold Design and Material for Vascularized Tissue-Engineered Bone Regeneration. Adv. Healthc. Mater. 2019, 8, 1–19. [Google Scholar] [CrossRef]
- Shelke, N.B.; James, R.; Laurencin, C.T. Polysaccharide biomaterials for drug delivery and regenerative engineering. Polym. Adv. Technol. 2014, 25, 448–460. [Google Scholar] [CrossRef]
- Medina, J.; Roche, Y.; Maldonado, O.; Hernández, J.C.; Zapata, C. Degradación hidrolítica y biodegradación de mezclas binarias de ácido poliláctico (PLA) con residuos plásticos. Rev. INGENIERÍA UC 2018, 25, 248–258. [Google Scholar]
- Jain, N.; Singh, V.K.; Chauhan, S. A review on mechanical and water absorption properties of polyvinyl alcohol based composites/films. J. Mech. Behav. Mater. 2017, 26, 213–222. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Freed, L.E.; Vunjak-Novakovic, G. Culture of organized cell communities. Adv. Drug Deliv. Rev. 1998, 33, 15–30. [Google Scholar] [CrossRef]
- Tovar, C.D.G.; Castro, J.I.; Valencia, C.H.; Porras, D.P.N.; Delgado-Ospina, J.; Valencia, M.E.; Hernandez, J.H.M.; Chaur, M.N. Synthesis, Characterization, and Histological Evaluation of Chitosan-Ruta Graveolens Essential Oil Films Carlos. Molecules 2020, 25, 1688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pereira dos Santos, E.; Nicácio, P.H.M.; Coêlho Barbosa, F.; Nunes da Silva, H.; Andrade, A.L.S.; Lia Fook, M.V.; Farias Leite, I. Chitosan/essential oils formulations for potential use as wound dressing: Physical and antimicrobial properties. Materials 2019, 12, 2223. [Google Scholar] [CrossRef] [Green Version]
- Antunes, J.C.; Tavares, T.D.; Teixeira, M.A.; Teixeira, M.O.; Homem, N.C.; Amorim, M.T.P.; Felgueiras, H.P. Eugenol-containing essential oils loaded onto chitosan/polyvinyl alcohol blended films and their ability to eradicate staphylococcus aureus or pseudomonas aeruginosa from infected microenvironments. Pharmaceutics 2021, 13, 195. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, S.; Rezaei, M.; Hadi, S.; Mohamad, S.; Hosseini, H. Development and evaluation of a novel biodegradable film made from chitosan and cinnamon essential oil with low affinity toward water. Food Chem. 2010, 122, 161–166. [Google Scholar] [CrossRef]
- Sánchez-González, L.; Cháfer, M.; Chiralt, A.; González-Martínez, C. Physical properties of edible chitosan films containing bergamot essential oil and their inhibitory action on Penicillium italicum. Carbohydr. Polym. 2010, 82, 277–283. [Google Scholar] [CrossRef]
- Siripatrawan, U.; Harte, B.R. Food Hydrocolloids Physical properties and antioxidant activity of an active fi lm from chitosan incorporated with green tea extract. Food Hydrocoll. 2010, 24, 770–775. [Google Scholar] [CrossRef]
- Zivanovic, S.; Chi, S.; Draughon, A.F. Antimicrobial Activity of Chitosan. Science 2005, 70, 45–51. [Google Scholar]
- Sánchez-González, L.; González-Martínez, C.; Chiralt, A.; Cháfer, M. Physical and antimicrobial properties of chitosan-tea tree essential oil composite films. J. Food Eng. 2010, 98, 443–452. [Google Scholar] [CrossRef]
- Hosseini, M.H.; Razavi, S.H.; Mousavi, M.A. Antimicrobial, physical and mechanical properties of chitosan-based films incorporated with thyme, clove and cinnamon essential oils. J. Food Process. Preserv. 2009, 33, 727–743. [Google Scholar] [CrossRef]
- Graziano, T.S.; Calil, C.M.; Sartoratto, A.; Franco, G.C.N.; Groppo, F.C.; Cogo-Müller, K. In vitro effects of Melaleuca alternifolia essential oil on growth and production of volatile sulphur compounds by oral bacteria. J. Appl. Oral Sci. 2016, 24, 582–589. [Google Scholar] [CrossRef] [Green Version]
- Barbosa, Q.; Lourenc, R.V.; Jos, P. Properties of gelatin-based films incorporated with chitosan-coated microparticles charged with rutin. Int. J. Biol. Macromol. 2017, 101, 643–652. [Google Scholar] [CrossRef]
- De Sá, C.; Mendes, H.; Figueiredo, D.; Lúcia, T.; Stamford, M.; Helena, L. International Journal of Food Microbiology Inhibition of Listeria monocytogenes by Melaleuca alternifolia (tea tree) essential oil in ground beef. Int. J. Food Microbiol. 2019, 293, 79–86. [Google Scholar] [CrossRef]
- Vazquez, J.A.; Zawawi, A.A. Efficacy of Alcohol-Based and Alcohol-Free Melaleuca Oral Solution for the Treatment of Fluconazole-Refractory Oropharyngeal Candidiasis in Patients with AIDS. HIV Clin. Trials 2002, 3, 379–385. [Google Scholar] [CrossRef]
- Bagg, J.; Jackson, M.S.; Sweeney, M.P.; Ramage, G.; Davies, A.N. Susceptibility to Melaleuca alternifolia (tea tree) oil of yeasts isolated from the mouths of patients with advanced cancer. Oral Oncol. 2006, 42, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M.; Milas, M.; Le Dung, P. Characterization of chitosan. Influence of ionic strength and degree of acetylation on chain expansion. Int. J. Biol. Macromol. 1993, 15, 281–285. [Google Scholar] [CrossRef]
- Sanchez-Gonzalez, L.; Pastor, C.; Vargas, M.; Chiralt, A.; Gonzalez-Martinez, C.; Chafer, M. Effect of hydroxypropylmethylcellulose and chitosan coatings with and without bergamot essential oil on quality and safety of cold-stored grapes. Postharvest Biol. Technol. 2011, 60, 57–63. [Google Scholar] [CrossRef] [Green Version]
- Martínez, K.; Ortiz, M.; Albis, A.; Castañeda, C.G.G.; Valencia, M.E.; Tovar, C.D.G. The effect of edible chitosan coatings incorporated with thymus capitatus essential oil on the shelf-life of strawberry (Fragaria × ananassa) during cold storage. Biomolecules 2018, 8, 155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, R.M. Particle size analysis by laser diffraction: ISO 13320, standard operating procedures, and Mie theory. Am. Lab. 2003, 35, 44–47. [Google Scholar]
- Steffe, J.F. Rheological Methods in Food Process Engineering; Freeman Press: East Lansing, MI, USA, 1996; ISBN 0963203614. [Google Scholar]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- Shellie, R.; Marriott, P.; Zappia, G.; Mondello, L.; Dugo, G. Interactive use of linear retention indices on polar and apolar columns with an MS-library for reliable characterization of australian tea tree and other Melaleuca sp. Oils. J. Essent. Oil Res. 2003, 15, 305–312. [Google Scholar] [CrossRef]
- Thomas, S.; Thomas, R.; Zachariah, A.K.; Kumar, R. Thermal and Rheological Measurement Techniques for Nanomaterials Characterization; Elsevier: Amsterdam, The Netherlands, 2017; Volume 3, ISBN 032346145X. [Google Scholar]
- Kale, S.; Deore, S. Emulsion Microemulsion and Nanoemulsion. Syst. Rev. Pharm. 2017, 8, 39–47. [Google Scholar] [CrossRef] [Green Version]
- Goodarzi, F.; Zendehboudi, S. A Comprehensive Review on Emulsions and Emulsion Stability in Chemical and Energy Industries. Can. J. Chem. Eng. 2019, 97, 281–309. [Google Scholar] [CrossRef] [Green Version]
- Bonilla, J.; Atarés, L.; Vargas, M.; Chiralt, A. Effect of essential oils and homogenization conditions on properties of chitosan-based films. Food Hydrocoll. 2012, 26, 9–16. [Google Scholar] [CrossRef]
- Abdollahi, M.; Rezaei, M.; Farzi, G. Improvement of active chitosan film properties with rosemary essential oil for food packaging. Int. J. Food Sci. Technol. 2012, 47, 847–853. [Google Scholar] [CrossRef]
- Myong Kim, K.; Hwa Son, J.; Kim, S. Properties of Chitosan Films as a Function of pH and Solvent Type. J. Food Sci. 2006, 71, 1–6. [Google Scholar]
- Cazón, P.; Antoniewska, A.; Rutkowska, J.; Manuel, V. Evaluation of easy-removing antioxidant films of chitosan with Melaleuca alternifolia essential oil. Int. J. Biol. Macromol. 2021, 186, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Kalaycıoğlu, Z.; Torlak, E.; Akın-Evingür, G.; Özen, İ.; Erim, F.B. Antimicrobial and physical properties of chitosan films incorporated with turmeric extract. Int. J. Biol. Macromol. 2017, 101, 882–888. [Google Scholar] [CrossRef]
- Tahir, P.M.; Owolabi, F.A.T.; Shawkataly, A.K.H.P.; Alkarkhi, A.F.M.; Nwakaego, E.G.; Kamilu, O.K.F.; Chima, I.C.; Rizal, S.; Ghazali, A. Pulp and Paper Potentials of Alkaline Peroxide Pre-Treated of Oil Palm Waste and Industrial Application. In Palm Oil; IntechOpen: London, UK, 2018. [Google Scholar]
- Anker, M.; Stading, M.; Hermansson, A.M. Mechanical Properties, Water Vapor Permeability, and Moisture Contents of β-Lactoglobulin and Whey Protein Films Using Multivariate Analysis. J. Agric. Food Chem. 1998, 46, 1820–1829. [Google Scholar] [CrossRef]
- Srinivasa, P.C.; Ramesh, M.N.; Tharanathan, R.N. Effect of plasticizers and fatty acids on mechanical and permeability characteristics of chitosan films. Food Hydrocoll. 2007, 21, 1113–1122. [Google Scholar] [CrossRef]
- Vargas, M.; Albors, A.; Chiralt, A.; González-Martínez, C. Characterization of chitosan-oleic acid composite films. Food Hydrocoll. 2009, 23, 536–547. [Google Scholar] [CrossRef]
- Altiok, D.; Altiok, E.; Tihminlioglu, F. Physical, antibacterial and antioxidant properties of chitosan films incorporated with thyme oil for potential wound healing applications. J. Mater. Sci. Mater. Med. 2010, 21, 2227–2236. [Google Scholar] [CrossRef] [Green Version]
- Sheikh, Z.; Brooks, P.J.; Barzilay, O.; Fine, N.; Glogauer, M. Macrophages, foreign body giant cells and their response to implantable biomaterials. Materials 2015, 8, 5671–5701. [Google Scholar] [CrossRef] [Green Version]
- Park, S.I.; Zhao, Y. Incorporation of a High Concentration of Mineral or Vitamin into Chitosan-Based Films. J. Agric. Food Chem. 2004, 52, 1933–1939. [Google Scholar] [CrossRef]
- Shen, Z.; Kamdem, D.P. Development and characterization of biodegradable chitosan films containing two essential oils. Int. J. Biol. Macromol. 2015, 74, 289–296. [Google Scholar] [CrossRef]
- Brugnerotto, J.; Lizardi, J.; Goycoolea, F.M.; Argüelles-Monal, W.; Desbrières, J.; Rinaudo, M. An infrared investigation in relation with chitin and chitosan characterization. Polymer 2001, 42, 3569–3580. [Google Scholar] [CrossRef]
- Depan, D.; Shah, J.S.; Misra, R.D.K. Degradation mechanism and increased stability of chitosan-based hybrid scaffolds cross-linked with nanostructured carbon: Process–structure–functional property relationship. Polym. Degrad. Stab. 2013, 98, 2331–2339. [Google Scholar] [CrossRef]
- Maruyama, M.; Ito, M. In vitro properties of a chitosan-bonded self-hardening paste with hydroxyapatite granules. J. Biomed. Mater. Res. 1996, 32, 527–532. [Google Scholar] [CrossRef]
- Grande, C.D.; Mangadlao, J.; Fan, J.; De Leon, A.; Delgado-Ospina, J.; Rojas, J.G.; Rodrigues, D.F.; Advincula, R. Chitosan Cross-Linked Graphene Oxide Nanocomposite Films with Antimicrobial Activity for Application in Food Industry. Macromol. Symp. 2017, 374, 1–8. [Google Scholar] [CrossRef]
- Suvik, A.; Effendy, A.W. The use of modified Masson’s trichrome staining in collagen evaluation in wound healing study. Malays. J. Vet. Res. 2012, 3, 39–47. [Google Scholar]
- Valencia, A.M.; Valencia, C.H.; Zuluaga, F.; Grande-Tovar, C.D. Synthesis and fabrication of films including graphene oxide functionalized with chitosan for regenerative medicine applications. Heliyon 2021, 7, e07058. [Google Scholar] [CrossRef]
- Chandorkar, Y.; Basu, B. The foreign body response demystified. ACS Biomater. Sci. Eng. 2018, 5, 19–44. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarritt, M.E.; Londono, R.; Badylak, S.F. Host response to implanted materials and devices: An overview. Immune Response Implant. Mater. Devices 2017, 1–14. [Google Scholar]
- Intini, C.; Elviri, L.; Cabral, J.; Mros, S.; Bergonzi, C.; Bianchera, A.; Flammini, L.; Govoni, P.; Barocelli, E.; Bettini, R. 3D-printed chitosan-based scaffolds: An in vitro study of human skin cell growth and an in-vivo wound healing evaluation in experimental diabetes in rats. Carbohydr. Polym. 2018, 199, 593–602. [Google Scholar] [CrossRef] [PubMed]







| Sample | Solids (%) * | pH * | Apparent Viscosity (cP) (1/100) * | Density (g/mL) | Particle Size * | |
|---|---|---|---|---|---|---|
| D50 (µm) | D98 (µm) | |||||
| CS | 1.66 b ± 0.03 | 7.43 a ± 0.009 | 106 a ± 0.000 | 1.00 | N/A | N/A |
| CS/TTEO 0.5% | 2.46 a ± 0.04 | 7.44 a ± 0.007 | 74 ab ± 0.444 | 1.00 | 1.29 a ± 0.140 | 3.16 a ± 0.160 |
| CS/TTEO 1.0% | 2.05 a ± 0.01 | 7.44 a ± 0.009 | 66 ab ± 0.000 | 1.00 | 1.43 a ± 0.031 | 3.44 a ± 0.069 |
| CS/TTEO 1.5% | 2.10 a ± 0.06 | 4.55 a ± 0.013 | 28 b ± 0.444 | 1.00 | 1.60 a ± 0.024 | 3.38 a ± 0.049 |
| Formulation | Young’s Modulus (Pa) * | Tensile Strength (Pa) * | Deformation (%) * |
|---|---|---|---|
| F1 | 1.6 a ± 0.3 | 5.5 a ± 1.0 | 362.0 a ± 25.5 |
| F2 | 2.0 b ± 0.2 | 6.3 a ± 0.7 | 280.9 b ± 12.9 |
| F3 | 1.4 a ± 0.1 | 6.0 a ± 2.3 | 269.7 b ± 52.3 |
| F4 | 1.6 a ± 0.1 | 4.6 a ± 0.6 | 188.2 c ± 13.8 |
| Formulations | Td3% (°C) |
|---|---|
| F1 | 42 |
| F2 | 61 |
| F3 | 56 |
| F4 | 68 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro, J.I.; Valencia-Llano, C.H.; Valencia Zapata, M.E.; Restrepo, Y.J.; Mina Hernandez, J.H.; Navia-Porras, D.P.; Valencia, Y.; Valencia, C.; Grande-Tovar, C.D. Chitosan/Polyvinyl Alcohol/Tea Tree Essential Oil Composite Films for Biomedical Applications. Polymers 2021, 13, 3753. https://doi.org/10.3390/polym13213753
Castro JI, Valencia-Llano CH, Valencia Zapata ME, Restrepo YJ, Mina Hernandez JH, Navia-Porras DP, Valencia Y, Valencia C, Grande-Tovar CD. Chitosan/Polyvinyl Alcohol/Tea Tree Essential Oil Composite Films for Biomedical Applications. Polymers. 2021; 13(21):3753. https://doi.org/10.3390/polym13213753
Chicago/Turabian StyleCastro, Jorge Iván, Carlos Humberto Valencia-Llano, Mayra Eliana Valencia Zapata, Yilmar Joan Restrepo, José Herminsul Mina Hernandez, Diana Paola Navia-Porras, Yamid Valencia, Cesar Valencia, and Carlos David Grande-Tovar. 2021. "Chitosan/Polyvinyl Alcohol/Tea Tree Essential Oil Composite Films for Biomedical Applications" Polymers 13, no. 21: 3753. https://doi.org/10.3390/polym13213753
APA StyleCastro, J. I., Valencia-Llano, C. H., Valencia Zapata, M. E., Restrepo, Y. J., Mina Hernandez, J. H., Navia-Porras, D. P., Valencia, Y., Valencia, C., & Grande-Tovar, C. D. (2021). Chitosan/Polyvinyl Alcohol/Tea Tree Essential Oil Composite Films for Biomedical Applications. Polymers, 13(21), 3753. https://doi.org/10.3390/polym13213753