1. Introduction
Rubber waste management is one of the challenges of materials science that has yet to be solved. With an estimated market of 30 million tons per year [
1,
2,
3], the rubber industry is a major producer of waste. Rubber products do not break down, nor do they melt, so conventional reprocessing techniques are not suitable for rubber recycling [
4]. Current recycling approaches are still similar to those developed in the 1850s, when Charles Goodyear first patented the addition of ground rubber waste into virgin rubber mixtures, and Hall patented the thermal plasticization of rubber shoe soles [
5]. The lack of true innovation in rubber recycling in the past 170 years has led to the current practice of incinerating or downcycling rubber waste [
5]. Devulcanisation has been proposed as a viable solution for this problem, as it would ideally convert rubber waste back into its original, uncured form. During this process, cross-links are selectively broken down, while the polymer chains retain their original length. In practice, true devulcanisation has never been achieved due to undesired chain degradation and oxidation. Ultimately, the side reactions compromise the mechanical properties of recycled rubber [
6].
EPDM (ethylene propylene diene monomer) rubber is mainly vulcanized by two methods: (a) with sulphur, creating sulphuric cross-links; or (b) with peroxides, linking polymer chains through C–C covalent bonds. Two main factors are generally mentioned in connection with devulcanisation. On the one hand, the bond energy of sulphuric cross-links (C–S and S–S covalent bonds) is significantly lower than that of C–C covalent bonds comprising the polymer backbone [
7]. Consequently, when a rubber vulcanisate is heated, sulphuric cross-links are more likely to break than bonds in the polymer chain. Still, considering the ratio of cross-links to other bonds in an elastomer, a large number of C–C bonds will break when exposed to heat only. On the other hand (according to Fukumori et al. [
8,
9]), the elastic constant of C–C bonds is about 30 times higher than that of S–S bonds, which means that high shearing forces would cause sulphuric cross-links to stretch to a greater extent than other bonds, making them more susceptible to cleavage. Both approaches imply that peroxide-cured rubbers cannot be effectively devulcanised, as their cross-links are made of the same covalent bonds as their polymer backbone. Thermomechanical stimuli would lead to the random degradation of peroxide-cured rubber, but they might be effective in the selective breakdown of sulphuric cross-links [
10].
Several devulcanisation strategies have been proposed and investigated in the academic literature. Microbiological treatment involves the use of microorganisms that consume the sulphuric components of organic materials to obtain energy [
11,
12]. Microwave devulcanisation harnesses the fact that microwaves can heat matter uniformly with little surface effect [
4,
13,
14,
15]. Thermomechanical devulcanisation ranges from extruders [
16] and high shear mixers [
17] to conventional rubber processing technologies (mills and internal mixers) [
18,
19]. During ultrasonic devulcanisation, ultrasound waves are used to break down rubber networks; this technology is often coupled with an extruder, combining the effects of thermomechanical and ultrasonic treatments [
20,
21]. Chemical devulcanisation is also widely studied, but its industrial significance is limited due to the need for solvent regeneration [
22,
23]. Currently, only the thermomechanical and microwave methods have high enough productivity for industrial use. Thermomechanical and ultrasonic devulcanisation technologies are preferable, due to their better scalability and higher selectivity for cross-link scission [
6,
24].
Sutanto et al. [
7] and Macsiniuc et al. [
25] devulcanised EPDM rubber in a batch internal mixer and achieved high degrees of devulcanisation (up to 75%). In general, temperature, shearing rate, fill ratio, and residence time were the most important factors impacting devulcanisation. The combination of high temperature and shearing rate often results in severe chain degradation, while at inadequate shearing rates, devulcanisation may not occur at all.
One of the most widespread evaluation techniques to determine the effectiveness of devulcanisation was developed by Horikx in the 1950s [
26]. His method enables us to determine whether a polymer degradation is dominated by the selective breakdown of cross-links (devulcanisation), or chain degradation. He identified two distinct theoretical scenarios based on the Flory–Huggins theory [
27,
28]. The first scenario assumes that the polymer was degraded via a statistically random distribution of chain scission events. Equation (1) correlates the sol fraction and the cross-link density of the degraded polymer:
where
νi [mol/cm
3] stands for the initial cross-link density,
νf [mol/cm
3] stands for cross-link density after degradation,
si [–] stands for the initial sol fraction of the polymer, and
sf [–] stands for the post-treatment sol fraction of the polymer [
10,
26].
In the second scenario, it is assumed that the degradation process consists solely of the selective breakdown of cross-links, strictly corresponding with an ideal devulcanisation process. He devised Equation (2) with the same principle of expressing the polymer’s sol fraction as a function of its cross-link density:
where
γi [–] and
γf [–] stand for the initial and final cross-linking indices, respectively [
10]. The cross-linking index represents the average number of cross-links per polymer chain and can be determined according to Equation (3):
where
γx [–] is the cross-linking index,
νx [mol/cm
3] is the cross-link density,
Mn [g/mol] stands for the number-average molecular weight of the polymer, and
ρ [g/cm
3] is the density of the polymer [
26]. In this paper, the initial cross-linking index is estimated based on Equation (4) [
29]:
Devulcanisation on its own is not adequate; the application of devulcanisates is also a crucial step in rubber recycling. Three strategies can be distinguished, according to Forrest [
30] and Isayev [
6]. Under certain conditions, it is possible to produce thermoplastic elastomers by adding devulcanisates or ground waste rubber to conventional thermoplastic polymers [
31,
32,
33]. However, the circular economy of rubber is only achieved when the waste material is used in new rubber vulcanisates. Consequently, adding devulcanisates to virgin rubber mixtures can produce new rubber with some recycled content [
34,
35]. Revulcanising previously devulcanised rubber can produce 100% recycled rubber [
16,
36,
37].
Jacob et al. [
38] mixed ground EPDM vulcanisates into virgin rubber compounds. Their direct recycling method revealed that waste rubber would enhance the mechanical properties of the otherwise unfilled EPDM rubber, thanks to its carbon black content.
Seghar et al. [
39] followed similar principles and prepared revulcanised rubber and vulcanisates with various devulcanisate content. They achieved a 90% decrease in cross-link density, with a significant extent of degradative side reactions. Their revulcanisates with up to 10 wt% recycled content had no significant change in mechanical properties compared to virgin rubber. However, a 20 wt% recycled content resulted in a 20% decrease in the samples’ modulus and strength.
Yun et al. [
21] achieved an up to 85% decrease in cross-link density for unfilled EPDM via ultrasonic devulcanisation. However, even a 15 phr carbon black content would decrease the degree of devulcanisation to 70%. Additionally, they showed that the carbon black content would shift the devulcanisation process towards the random chain scission curve according to Horikx’s theory. They added the devulcanisates to virgin rubber compounds and prepared vulcanisates at various recycled contents. They revealed that the devulcanisate increased the Young’s modulus of the revulcanisates. The tensile strength and elongation at break of the revulcanisates decreased by around 35%, 45%, 50%, and 70% at 25 wt%, 50 wt%, 75 wt%, and 100 wt% recycled contents, respectively, compared to virgin rubber.
In this paper, we investigated the potential of batch devulcanisation of EPDM rubber on a two-roll mill by treating rubber samples at various temperatures and roll speeds. The use of traditional rubber processing equipment may encourage manufacturers to adopt this recycling technology. We identified the optimal devulcanisation parameters on the tested range using Horikx’s analysis. Subsequently, we revulcanised our devulcanisates and mixed the devulcanisates into virgin rubber mixtures of identical composition at various ratios. Finally, we tested the mechanical and morphological properties of the resulting, partially recycled rubbers.
2. Materials and Methods
2.1. Materials
Table 1 shows the composition of the EPDM-based rubber mixture used in the experiments. The base polymer, Dutral TER 4047, has an intrinsic Mooney viscosity of 55 at 125 °C and contains 54 wt% ethylene, 41.5 wt% propylene, and 4.5 wt% ethylidene norbornene. The rubber mixture was prepared and provided by Palotás Mix Kft. (Kemeneshőgyész, Hungary), and contained large amounts of fillers: 85 phr of carbon black and 30 phr of dolomite. The rubber composition was created to mimic EPDM rubbers of general use: profiles, gaskets, etc.
Toluene was used as the general solvent for cross-link density and sol content measurements. It was supplied by Fisher Scientific UK (Loughborough, UK).
For the revulcanisation experiments, powdered zinc oxide was supplied by Werco Metal (Zlatna, Romania). Zinc stearate, TBTD, ZDBC, and sulphur were supplied by Ningbo Actmix Rubber Chemicals Co., Ltd. (Ningbo, China).
2.2. Preparation of Rubber Vulcanisates
We used a Collin Teach-Line Platen Press 200E (Dr. Collin GmbH, Ebersberg, Germany) hot press to cure the EPDM rubber. Rubber sheets with a 2 mm thickness were obtained. The plates of the press were heated up to 180 °C, and the ultimate pressure was 2.8 MPa. We determined the optimal curing time (t90, at which 90% curing is achieved) from rheometer measurements. We applied the pressure gradually to allow for degassing.
We used a Retsch ZM200 (Retsch GmbH, Haan, Germany) cryogenic mill to pulverise the cured rubber sheets. Initially, we cut the sheets into 10 mm × 10 mm pieces and cooled them in liquid nitrogen. The frozen rubber pieces were fed to the pre-cooled milling chamber one by one. The rotor speed was set to 12,000 rpm. The resulting particle size distribution of the ground EPDM rubber is shown in
Figure 1.
2.3. Devulcanisation
We devulcanised EPDM rubber on a Labtech LRM-SC-11/3E (Labtech Engineering Co. Ltd., Samutprakarn, Thailand) two-roll mill. We fed the material between the rolls in batches of 50 g and allowed the crumbs to pass through the gap 20 times. The rubber powder was allowed to cool between cycles to avoid heat buildup during the process. The temperature and roll speeds were varied according to
Table 2. We observed no deviation in the roll temperatures from the set values.
2.4. Revulcanisation
dev_80-1.33 was selected as the devulcanisate with the best properties, based on Horikx’s analysis, and hence it was used for revulcanisation experiments. We investigated the revulcanisability of dev_80-1.33 in two ways: (a) on its own, and (b) by adding the original curing system to the devulcanisate. We added the curing agents to the devulcanisate in a Brabender Plasti-Corder (Brabender Technologie GmbH & Co., Duisburg, Germany) internal mixer at a fill ratio of 70% in a 50 cm
3 mixing chamber at 70 °C for 20 min. The ratio at which the curing system was added to the devulcanisate is shown in
Table 3. The devulcanisate that contained additional curing agents was named dev_80-1.33_mix.
We prepared mixtures of the virgin EPDM rubber and dev_80-1.33_mix in the previously mentioned Brabender internal mixer with the same operating conditions. These mixtures, along with a reference virgin rubber and Sample 1, were tested in the rheometer and vulcanised in the compression mould to t
90 the same way the original rubber vulcanisate had been prepared.
Table 4 shows the composition of all revulcanisates.
2.5. Testing
We used a MonTech Monsanto R100S rheometer (MonTech Werkstoffprüfmaschinen GmbH, Buchen, Germany) to determine the curing properties of each rubber mixture. We ran the rheometer in isothermal (T = 180 °C) time sweep mode (1.667 Hz, 1° amplitude) for 30 min.
We measured the sol content of the rubber samples via Soxhlet extraction. We placed 10 g of rubber powder into cellulose thimbles and sealed them with cotton wool. We added 200 mL of toluene to the bottom distillation flask and ran the experiments for 20 h, throughout which the top column of the equipment was cooled with tap water. After the extraction, we dried the specimens at 120 °C for 6 h to remove the solvent.
Equation (5) was used to compute the sol content:
where
s [–] stands for sol content, and
Mi [g] and
Mf [g] stand for the mass of rubber before and after the extraction, respectively.
The sol content obtained from Equation (5) stands for the rubber mixture’s overall sol content. However, Horikx’s theory uses the sol content of the polymer part only. It was safe to assume that the oil, lubricant, and PEG components would be completely extracted from the mixture. Consequently, we devised Equation (6) to solve this issue:
We measured the cross-link density of the rubber samples according to the ASTM D6814 standard. We immersed the rubber samples in toluene for 72 h (until equilibrium swelling was achieved). We dabbed the swollen rubber powder dry with paper towels and weighed them. We then dried the samples at 120 °C for 6 h and weighed the samples again. Cross-link densities were evaluated using the Flory–Rehner Equation (7):
where
νx [mol/cm
3] denotes the cross-link density,
χ [–] means the polymer–solvent interaction parameter (which equals 0.496 for an EPDM-toluene system),
V1 [cm
3/mol] is the molar volume of the solvent (106.13 cm
3/mol for toluene), and
vr [–] stands for the volume fraction of rubber in the toluene-swollen rubber sample [
10,
25]. We applied Equation (8) to estimate
vr from our experimental data, as suggested in the ASTM D6814-02 (2018) standard:
where
mr [g] stands for the mass of the dried rubber sample,
ms [g] denotes the mass of the solvent absorbed by the swollen rubber (the difference between the masses of the swollen and dry rubber samples),
ρr [g/cm
3] is the density of the rubber samples (measured to be 1.23 g/cm
3), and
ρs [g/cm
3] means the density of the solvent (0.867 g/cm
3 for toluene) [
10,
25].
We used a ball press to cut tensile and tear strength test specimens out of the compression moulded sheets. We performed tensile tests on a Zwick Z020 (ZwickRoell GmbH, Ulm, Germany) tensile tester with a 20 kN load cell, according to ISO 37:2017. The clamping distance was 60 mm, and the crosshead speed was 500 mm/min. We used the same tensile tester for tear strength tests with a clamping distance of 56 mm and a crosshead speed of 500 mm/min, according to ISO 34-1:2015.
The Shore A hardness of the rubber sheets was determined with a Zwick H04.3150.000 hardness tester, according to ISO 48-4:2018.
We took scanning electron micrographs (SEM) of the fracture surfaces of the tensile test specimens. Surfaces were first sputter-coated with gold and then placed into a Jeol JSM-6380LA (Jeol Lt., Tokyo, Japan) microscope.
4. Conclusions
We devulcanised a sulphur-cured EPDM rubber of known composition on a two-roll mill at moderate temperatures. We tested three temperature settings (80, 100, and 120 °C) and three friction settings (1.33, 1.67, and 2.00). We achieved a 53% decrease in cross-link density at 80 °C and 1.33 friction, while keeping polymer degradation at a minimum, which is supported by Horikx’s analysis.
We decided that the effectiveness of devulcanisation should be further analysed by evaluating the applicability of the devulcanisate. Hence, we tried revulcanising the devulcanisate on its own. Though the rheometer showed a small increase in modulus during this recuring process, the resulting rubber sheet disintegrated into crumbs. Based on this experience, we added curing agents to the devulcanisate and prepared rubber mixtures containing various amounts of devulcanisate and virgin EPDM rubber.
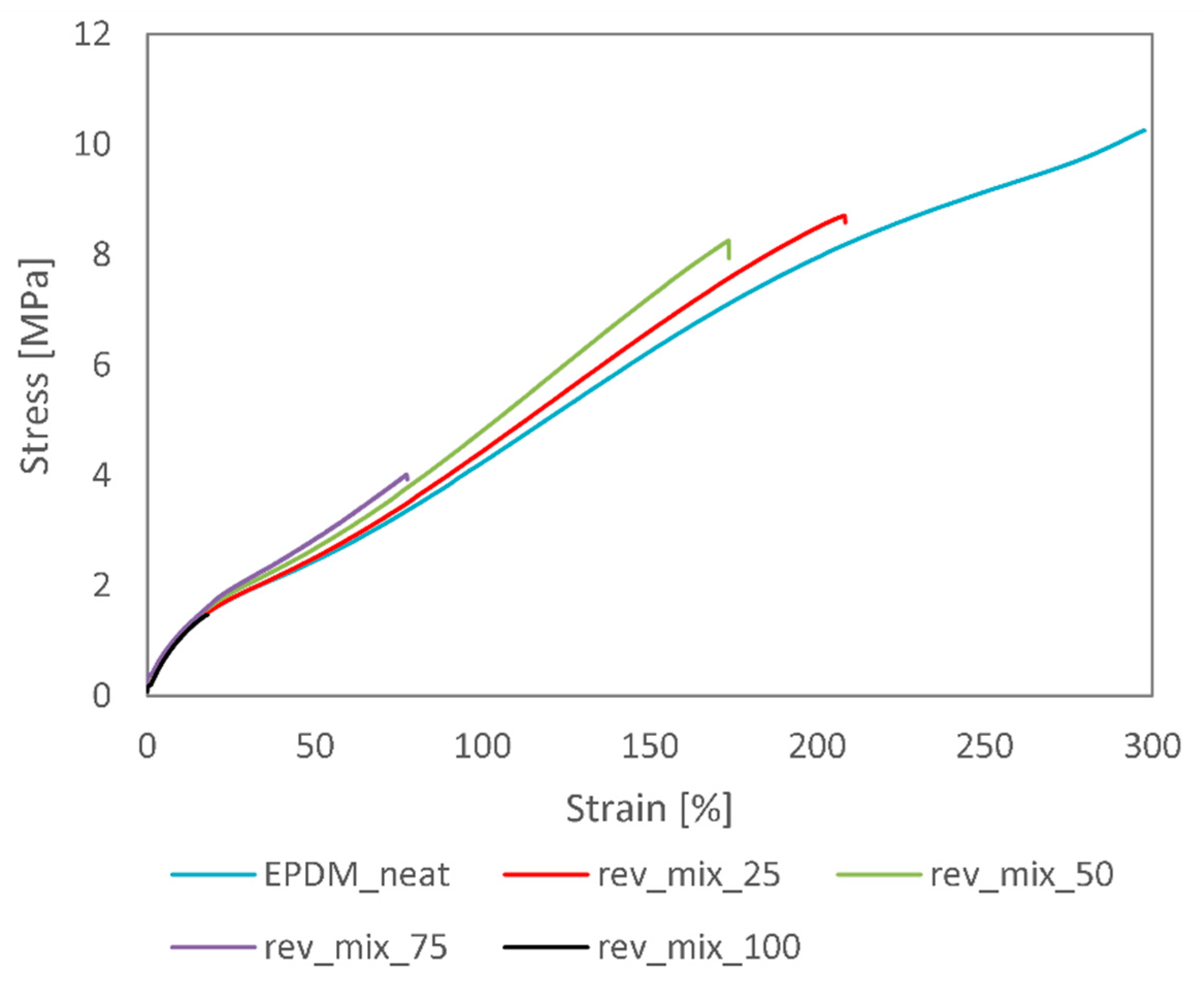
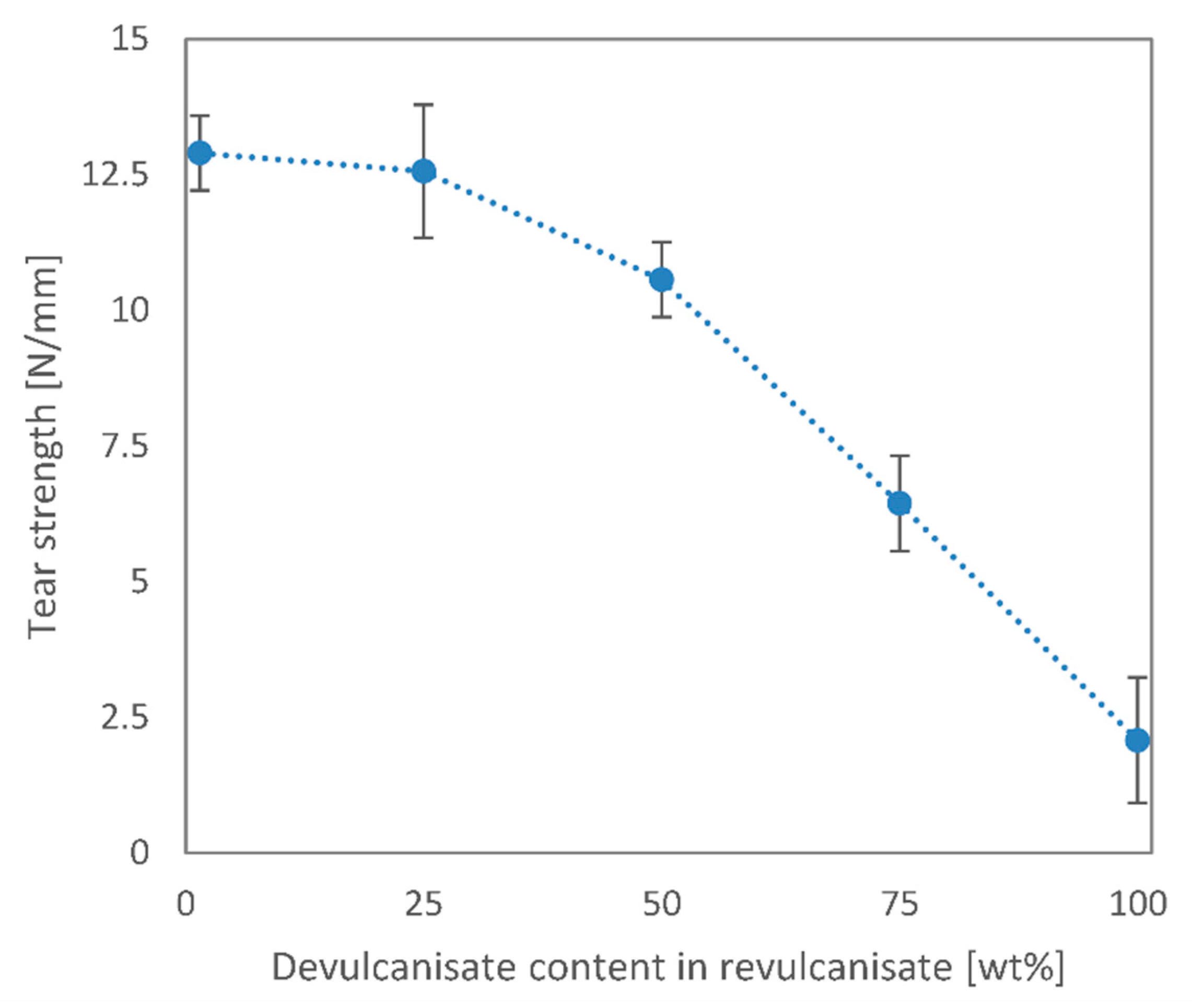
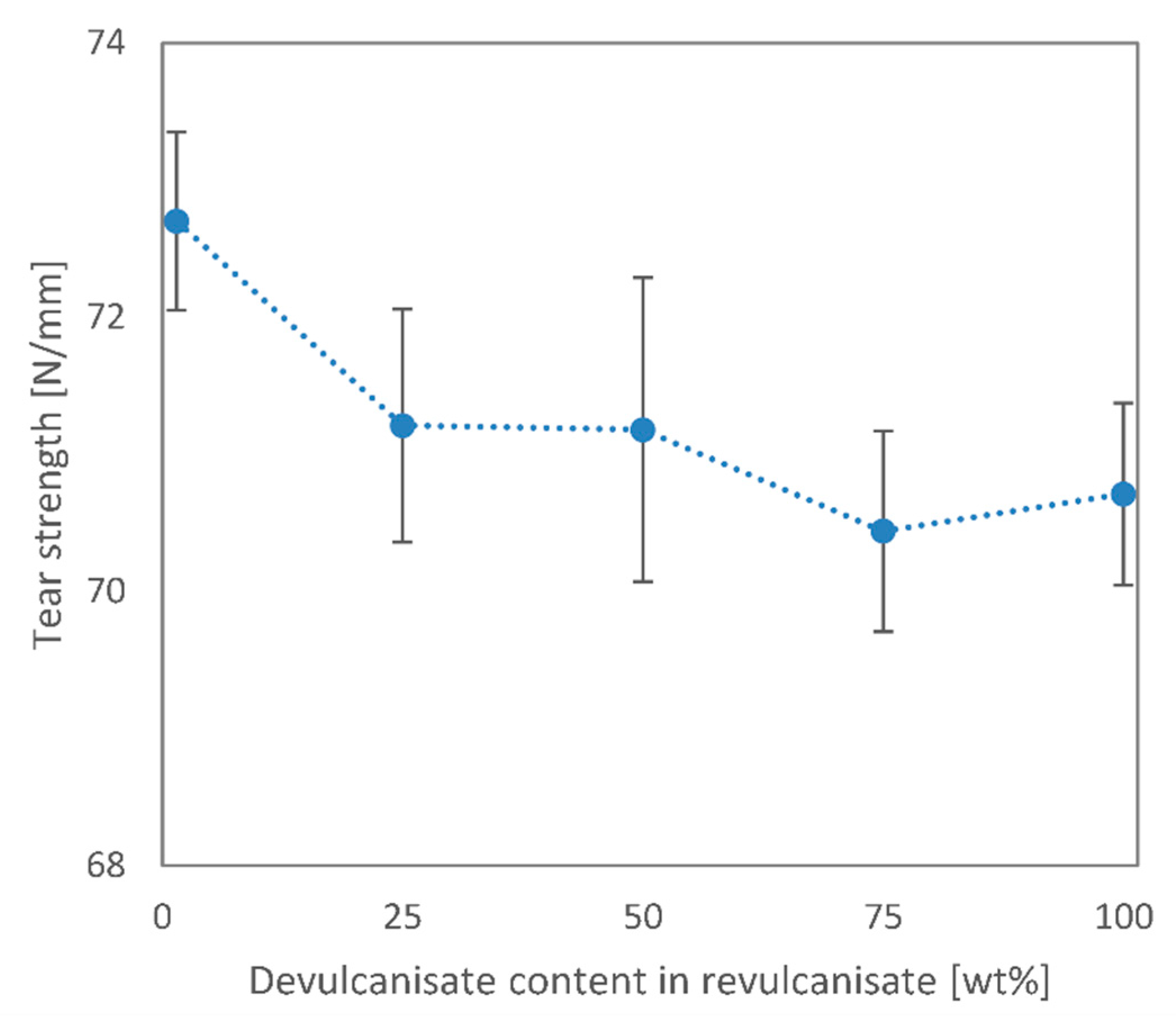
We performed mechanical and morphological tests on these rubber blends and found that most mechanical and structural properties may be retained up to a 50 wt% devulcanisate content. The elongation at break reduced from around 300% to 180%, while the tensile strength only decreased by 20%. Tear strength and hardness values also barely deteriorated at 50 wt% recycled content. We analysed the tensile test specimens’ fracture surfaces with a scanning electron microscope and found that a relatively smooth fracture surface can be achieved only up to 50 wt% recycled content. At higher devulcanisate contents, the virgin EPDM rubber cannot surround the devulcanised rubber crumbs, which did not stick sufficiently to one another.
Overall, the recycling method presented in this paper is indeed suitable for the recycling of EPDM rubber with a mild reduction in product quality at 25 and 50 wt% recycled contents. The addition of extra curing agents to the devulcanisate is crucial in achieving uniform product quality.