Conversion of Sewage Water into H2 Gas Fuel Using Hexagonal Nanosheets of the Polyaniline-Assisted Deposition of PbI2 as a Nanocomposite Photocathode with the Theoretical Qualitative Ab-Initio Calculation of the H2O Splitting
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of PANI/PbI2 Nanocomposite
2.2. Materials
2.3. Characterization and Analyses
2.4. The Electrochemical Test
2.5. The Theoretical Calculation
3. Results and Discussion
3.1. Characterization of the Prepared Nanomaterials
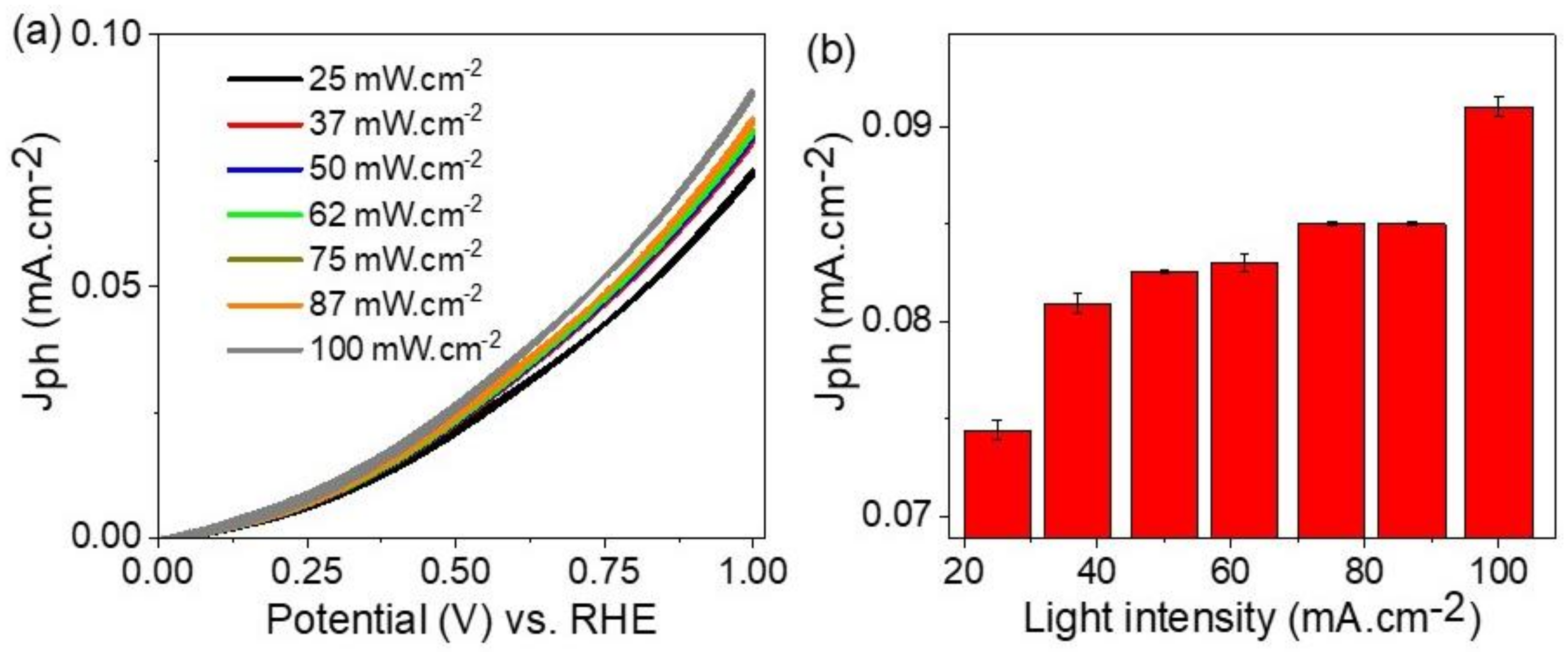
3.2. Photoelectrochemical Water-Splitting Reaction
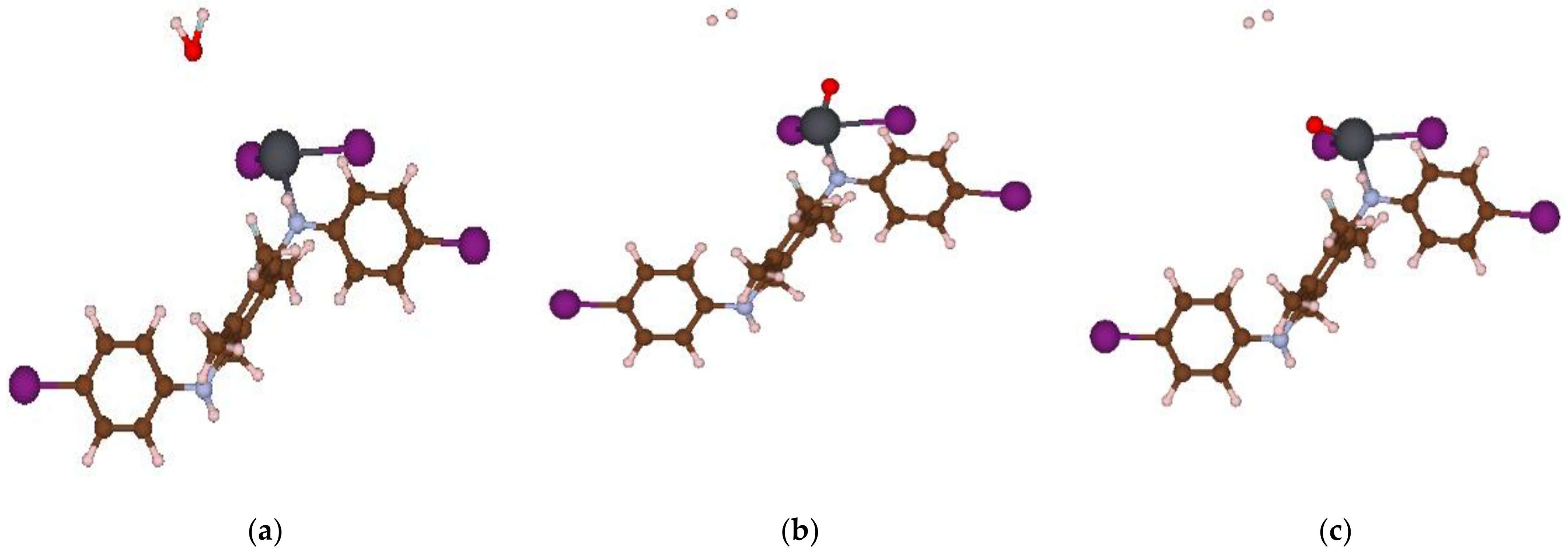
3.3. The Theoretical Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nishiyama, H.; Yamada, T.; Nakabayashi, M.; Maehara, Y.; Yamaguchi, M.; Kuromiya, Y.; Nagatsuma, Y.; Tokudome, H.; Akiyama, S.; Watanabe, T.; et al. Photocatalytic solar hydrogen production from water on a 100-m2 scale. Nature 2021, 598, 304–307. [Google Scholar] [CrossRef] [PubMed]
- Hisatomi, T.; Domen, K. Reaction systems for solar hydrogen production via water splitting with particulate semiconductor. Nat. Catal. 2019, 2, 387–399. [Google Scholar] [CrossRef]
- Pagliaro, M. Preparing for the future: Solar energy and bioeconomy in the United Arab Emirates. Energy Sci. Eng. 2019, 7, 1451–1457. [Google Scholar] [CrossRef] [Green Version]
- Takata, T. Photocatalytic water splitting with quantum efficiency of almost unity. Nature 2020, 581, 411–414. [Google Scholar] [CrossRef]
- Mohamed, F.; Rabia, M.; Shaban, M. Synthesis and characterization of biogenic iron oxides of different nanomorphologies from pomegranate peels for efficient solar hydrogen production. J. Mater. Res. Technol. 2020, 9, 4255–4271. [Google Scholar] [CrossRef]
- Shaban, M.; Ali, S.; Rabia, M. Design and application of nanoporous graphene oxide film for CO2, H2, and C2H2 gases sensing. J. Mater. Res. Technol. 2019, 8, 4510–4520. [Google Scholar] [CrossRef]
- Elsayed, A.M.; Rabia, M.; Shaban, M.; Aly, A.H.; Ahmed, A.M. Preparation of hexagonal nanoporous Al2O3/TiO2/TiN as a novel photodetector with high efficiency. Sci. Rep. 2021, 11, 17572. [Google Scholar] [CrossRef]
- Kang, Z.; Cheng, Y.; Zheng, Z.; Cheng, F.; Chen, Z.; Li, L.; Tan, X.; Xiong, L.; Zhai, T.; Gao, Y. MoS2-Based Photodetectors Powered by Asymmetric Contact Structure with Large Work Function Difference. Nano-Micro Lett. 2019, 11, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.H.; Lee, W.W.; Yang, D.W.; Chang, W.J.; Kwon, S.S.; Park, W. Il Anomalous Photovoltaic Response of Graphene-on-GaN Schottky Photodiodes. ACS Appl. Mater. Interfaces 2018, 10, 14170–14174. [Google Scholar] [CrossRef]
- Nourisabet, T.; Jamshidi Aval, H.; Shidpour, R.; Naji, L. Fabrication of a PEO-PVDF blend based polymer composite electrolyte with extremely high ionic conductivity via the addition of LLTO nanowires. Solid State Ion. 2022, 377, 115885. [Google Scholar] [CrossRef]
- Sarkar, J.; Ganguly, S. Investigation of the thermal properties of Cu–Ag core-shell nanowires using molecular dynamics simulation. Phys. B Condens. Matter 2022, 636, 413876. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, Z.; Wu, Q.; Liu, W.; Li, Y.; Zhang, H.; Ma, X.; Cong, L.; Wang, H.; Zhang, D.; et al. Effect of stacking faults on magnetic properties and magnetization reversal in Co nanowires. Mater. Charact. 2022, 187, 111861. [Google Scholar] [CrossRef]
- Abukhadra, M.R.; Rabia, M.; Shaban, M.; Verpoort, F. Heulandite/polyaniline hybrid composite for efficient removal of acidic dye from water; kinetic, equilibrium studies and statistical optimization. Adv. Powder Technol. 2018, 29, 2501–2511. [Google Scholar] [CrossRef]
- Shaban, M.; Abukhadra, M.R.; Rabia, M.; Elkader, Y.A.; Abd El-Halim, M.R. Investigation the adsorption properties of graphene oxide and polyaniline nano/micro structures for efficient removal of toxic Cr(VI) contaminants from aqueous solutions; kinetic and equilibrium studies. Rend. Lincei 2018, 29, 141–154. [Google Scholar] [CrossRef]
- Modibane, K.D.; Waleng, N.J.; Ramohlola, K.E.; Maponya, T.C.; Monama, G.R.; Makgopa, K.; Hato, M.J. Poly(3-aminobenzoic acid) Decorated with Cobalt Zeolitic Benzimidazolate Framework for Electrochemical Production of Clean Hydrogen. Polymers 2020, 12, 1581. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.Y.; Aroh, K.; Franson, N.; Satsangi, V.R.; Dass, S.; Ehrman, S. Copper oxide nanoparticle made by flame spray pyrolysis for photoelectrochemical water splitting—Part II. Photoelectrochemical study. Int. J. Hydrogen Energy 2011, 36, 15519–15526. [Google Scholar] [CrossRef]
- Guo, X.; Diao, P.; Xu, D.; Huang, S.; Yang, Y.; Jin, T.; Wu, Q.; Xiang, M.; Zhang, M. CuO/Pd composite photocathodes for photoelectrochemical hydrogen evolution reaction. Int. J. Hydrogen Energy 2014, 39, 7686–7696. [Google Scholar] [CrossRef]
- Teixeira, G.F.; Silva Junior, E.; Vilela, R.; Zaghete, M.A.; Colmati, F. Perovskite Structure Associated with Precious Metals: Influence on Heterogenous Catalytic Process. Catalysts 2019, 9, 721. [Google Scholar] [CrossRef] [Green Version]
- Mishra, M.; Chun, D.M. α-Fe2O3 as a photocatalytic material: A review. Appl. Catal. A Gen. 2015, 498, 126–141. [Google Scholar] [CrossRef]
- Acar, C.; Dincer, I.; Naterer, G.F. Review of photocatalytic water-splitting methods for sustainable hydrogen production. Int. J. Energy Res. 2016, 40, 1449–1473. [Google Scholar] [CrossRef]
- Liu, G.; Karuturi, S.K.; Chen, H.; Wang, D.; Ager, J.W.; Simonov, A.N.; Tricoli, A. Enhancement of the photoelectrochemical water splitting by perovskite BiFeO3 via interfacial engineering. Sol. Energy 2020, 202, 198–203. [Google Scholar] [CrossRef]
- Wang, Z.; Cao, D.; Wen, L.; Xu, R.; Obergfell, M.; Mi, Y.; Zhan, Z.; Nasori, N.; Demsar, J.; Lei, Y. Manipulation of charge transfer and transport in plasmonic-ferroelectric hybrids for photoelectrochemical applications. Nat. Commun. 2016, 7, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freeman, E.; Kumar, S.; Thomas, S.R.; Pickering, H.; Fermin, D.J.; Eslava, S. PrFeO3 Photocathodes Prepared Through Spray Pyrolysis. ChemElectroChem 2020, 7, 1365–1372. [Google Scholar] [CrossRef]
- Sherman, B.D.; Ashford, D.L.; Lapides, A.M.; Sheridan, M.V.; Wee, K.R.; Meyer, T.J. Light-Driven Water Splitting with a Molecular Electroassembly-Based Core/Shell Photoanode. J. Phys. Chem. Lett. 2015, 6, 3213–3217. [Google Scholar] [CrossRef]
- Neese, F.; Wiley, J. The ORCA program system. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2012, 2, 73–78. [Google Scholar] [CrossRef]
- Huzinaga, S.; Andzelm, J. Gaussian Basis Sets for Molecular Calculations; Elsevier: Amsterdam, The Netherlands, 1984; Volume 3, p. 426. [Google Scholar]
- Cossi, M.; Rega, N.; Scalmani, G.; Barone, V. Energies, structures, and electronic properties of molecules in solution with the C-PCM solvation model. J. Comput. Chem. 2003, 24, 669–681. [Google Scholar] [CrossRef]
- Saad, R.; Gamal, A.; Zayed, M.; Ahmed, A.M.; Shaban, M.; BinSabt, M.; Rabia, M.; Hamdy, H. Fabrication of ZnO/CNTs for Application in CO2 Sensor at Room Temperature. Nanomaterials 2021, 11, 3087. [Google Scholar] [CrossRef]
- Sayyah, S.M.; Azooz, R.E. Electrosynthesis and characterization of adherent poly(2-aminobenzothiazole) on Pt-electrode from acidic solution. Arab. J. Chem. 2016, 9, S576–S586. [Google Scholar] [CrossRef] [Green Version]
- Sayyah, S.M.; Shaban, M.; Rabia, M. A High-Sensitivity Potentiometric Mercuric Ion Sensor Based on m-Toluidine Films. IEEE Sens. J. 2016, 16, 1541–1548. [Google Scholar] [CrossRef]
- Basak, M.; Rahman, M.L.; Ahmed, M.F.; Biswas, B.; Sharmin, N. The use of X-ray diffraction peak profile analysis to determine the structural parameters of cobalt ferrite nanoparticles using Debye-Scherrer, Williamson-Hall, Halder-Wagner and Size-strain plot: Different precipitating agent approach. J. Alloy. Compd. 2022, 895, 162694. [Google Scholar] [CrossRef]
- Burton, A.W.; Ong, K.; Rea, T.; Chan, I.Y. On the estimation of average crystallite size of zeolites from the Scherrer equation: A critical evaluation of its application to zeolites with one-dimensional pore systems. Microporous Mesoporous Mater. 2009, 117, 75–90. [Google Scholar] [CrossRef]
- Almohammedi, A.; Shaban, M.; Mostafa, H.; Rabia, M. Nanoporous TiN/TiO2/Alumina Membrane for Photoelectrochemical Hydrogen Production from Sewage Water. Nanomaterials 2021, 11, 2617. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Huang, S.; Jiang, Q.; Yu, C.; Zhou, X. Experimental study of hydrogen generation from in-situ heavy oil gasification. Fuel 2022, 313, 122640. [Google Scholar] [CrossRef]
- Aazam, E.S.; Zaheer, Z. Silver-Cobalt bimetallic nanoparticles to the generation of hydrogen from formic acid decomposition. Arab. J. Chem. 2022, 15, 103795. [Google Scholar] [CrossRef]
- Xiao, F.; Yang, R.; Liu, Z. Active aluminum composites and their hydrogen generation via hydrolysis reaction: A review. Int. J. Hydrogen Energy 2022, 47, 365–386. [Google Scholar] [CrossRef]
- Abdelazeez, A.A.A.; El-Fatah, G.A.; Shaban, M.; Ahmed, A.M.; Rabia, M. ITO/Poly-3-Methylaniline/Au Electrode for Electrochemical Water Splitting and Dye Removal. ECS J. Solid State Sci. Technol. 2021, 10, 123009. [Google Scholar] [CrossRef]
- Kenfoud, H.; Nasrallah, N.; Baaloudj, O.; Belabed, C.; Chaabane, T.; Trari, M. Opto-electrochemical characteristics of synthesized BaFe2O4 nanocomposites: Photocatalytic degradation and hydrogen generation investigation. Int. J. Hydrogen Energy 2022, 47, 12039–12051. [Google Scholar] [CrossRef]
- Liu, Z.; Xiao, F.; Tang, W.; Cong, K.; Li, J.; Yang, R.; Hao, J. Study on the hydrogen generation performance and hydrolyzates of active aluminum composites. Int. J. Hydrogen Energy 2022, 47, 1701–1709. [Google Scholar] [CrossRef]
- Keshipour, S.; Asghari, A. A review on hydrogen generation by phthalocyanines. Int. J. Hydrogen Energy 2022, 47, 12865–12881. [Google Scholar] [CrossRef]
- Elsayed, A.M.; Shaban, M.; Aly, A.H.; Ahmed, A.M.; Rabia, M. Preparation and characterization of a high-efficiency photoelectric detector composed of hexagonal Al2O3/TiO2/TiN/Au nanoporous array. Mater. Sci. Semicond. Process. 2022, 139, 106348. [Google Scholar] [CrossRef]
- Liu, J.; Yuan, Q.; Huang, W.; Song, X. A novel nanoporous Mg-Li material for efficient hydrogen generation. J. Magnes. Alloy. 2021. [Google Scholar] [CrossRef]
- Srimurugan, V.; Jothiprakash, C.G.; Souparnika, V.; Prasanth, R. Photocorrosion-less stable heterojunction photoanode for efficient visible-light driven solar hydrogen generation. Int. J. Hydrogen Energy 2022, 47, 12515–12527. [Google Scholar] [CrossRef]
- Ivanenko, I.; Ruda, A.; Povazhnyi, V. Cobalt-nitrogen-doped activated carbons for hydrogen generation. Mater. Today Proc. 2022, 7, 15. [Google Scholar] [CrossRef]
- Ali, I.; Imanova, G.T.; Mbianda, X.Y.; Alharbi, O.M.L. Role of the radiations in water splitting for hydrogen generation. Sustain. Energy Technol. Assess. 2022, 51, 101926. [Google Scholar] [CrossRef]
- Ouyang, L.; Liu, M.; Chen, K.; Liu, J.; Wang, H.; Zhu, M.; Yartys, V. Recent progress on hydrogen generation from the hydrolysis of light metals and hydrides. J. Alloy. Compd. 2022, 910, 164831. [Google Scholar] [CrossRef]
- Hadia, N.M.A.; Abdelazeez, A.A.A.; Alzaid, M.; Shaban, M.; Mohamed, S.H.; Hoex, B.; Hajjiah, A.; Rabia, M. Converting Sewage Water into H2 Fuel Gas Using Cu/CuO Nanoporous Photocatalytic Electrodes. Materials 2022, 15, 1489. [Google Scholar] [CrossRef]
- Rabia, M.; Mohamed, S.H.; Zhao, H.; Shaban, M.; Lei, Y.; Ahmed, A.M. TiO2/TiOxNY hollow mushrooms-like nanocomposite photoanode for hydrogen electrogeneration. J. Porous Mater. 2020, 27, 133–139. [Google Scholar] [CrossRef]
- Ahmed, A.M.; Rabia, M.; Shaban, M. The structure and photoelectrochemical activity of Cr-doped PbS thin films grown by chemical bath deposition. RSC Adv. 2020, 10, 14458–14470. [Google Scholar] [CrossRef]
- Rabia, M.; Shaban, M.; Adel, A.; Abdel-Khaliek, A.A. Effect of plasmonic au nanoparticles on the photoactivity of polyaniline/indium tin oxide electrodes for water splitting. Environ. Prog. Sustain. Energy 2019, 38, 13171. [Google Scholar] [CrossRef]
- Shaban, M.; Rabia, M.; Eldakrory, M.G.; Maree, R.M.; Ahmed, A.M. Efficient photoselectrochemical hydrogen production utilizing of APbI3 (A = Na, Cs, and Li) perovskites nanorods. Int. J. Energy Res. 2020, 45, 7436–7446. [Google Scholar] [CrossRef]
- Ragupathi, V.; Raja, M.A.; Panigrahi, P.; Ganapathi Subramaniam, N. CuO/g-C3N4 nanocomposite as promising photocatalyst for photoelectrochemical water splitting. Optik 2020, 208, 164569. [Google Scholar] [CrossRef]
- Huang, X.; Zhang, M.; Sun, R.; Long, G.; Liu, Y.; Zhao, W. Enhanced hydrogen evolution from CuOx-C/TiO2 with multiple electron transport pathways. PLoS ONE 2019, 14, e0215339. [Google Scholar] [CrossRef] [PubMed]
- Naldoni, A.; Guler, U.; Wang, Z.; Marelli, M.; Malara, F.; Meng, X.; Besteiro, L.V.; Govorov, A.O.; Kildishev, A.V.; Boltasseva, A.; et al. Broadband Hot-Electron Collection for Solar Water Splitting with Plasmonic Titanium Nitride. Adv. Opt. Mater. 2017, 5, 1601031. [Google Scholar] [CrossRef] [Green Version]
- Abdelazeez, A.A.A.; Hadia, N.M.A.; Alzaid, M.; Shaban, M.; Mourad, A.H.I.; Fernández, S.; Rabia, M. Development of CuO nanoporous material as a highly efficient optoelectronic device. Appl. Phys. A Mater. Sci. Process. 2022, 128, 1–10. [Google Scholar] [CrossRef]
- Becke, A.D. A new mixing of Hartree–Fock and local density-functional theories. J. Chem. Phys. 1998, 98, 1372. [Google Scholar] [CrossRef]
- Tönsing, C.; Timmer, J.; Kreutz, C. Optimal Paths Between Parameter Estimates in Non-linear ODE Systems Using the Nudged Elastic Band Method. Front. Phys. 2019, 7, e00149. [Google Scholar] [CrossRef]








| Band Position (cm−1) | Assignment | |
|---|---|---|
| PANI/PbI2 | PANI | |
| 3424 | 3401 | N–H group [29] |
| 2923 2856 | 2918 | C–H aromatic ring group [29] |
| 1470 | 1467 | C=C of the quinoid ring [30] |
| 1375 | - | heteropolar diatomic molecules |
| 1291 | 1301 | C=C of benzenoid rings [30] |
| 1108 | 1105 | C–N |
| 1010 | 1049 | Heteropolar diatomic molecules of PbI2 |
| 792 | 789 | C–H in-plane [29] |
| 590 | 587 | Para disubstituted aromatic rings |
| Material or Element | Concentration (mg/L) |
|---|---|
| Phenols | 0.015 |
| F− | 1.0 |
| Al3+ | 3.0 |
| NH3 | 5.0 |
| Hg2+ | 0.005 |
| Pb2+ | 0.5 |
| Cd3+ | 0.05 |
| As3+ | 0.05 |
| Cr3+ | 1.0 |
| Cu2+ | 1.5 |
| Ni3+ | 0.1 |
| Fe3+ | 1.5 |
| Mn2+ | 1.0 |
| Zn2+ | 5.0 |
| Ag+ | 0.1 |
| Ba3+ | 2.0 |
| Co2+ | 2.0 |
| Other cations | 0.1 |
| Pesticides | 0.2 |
| CN−1 | 0.1 |
| Industrial washing | 0.5 |
| Coli groups | 4000/100 cm3 |
| Photoelectrode | Electrolyte | Jph (mA/cm2) |
|---|---|---|
| Ni/PANI [20] | H2SO4 | 0.091 |
| PANI/MoS2 [22] | H2SO4 | 0.09 |
| Poly(3-aminobenzoic acid) frame [15] | H2SO4 | 0.08 |
| g–C3N4–CuO [52] | NaOH | 0.01 |
| CuO–C/TiO2 [53] | glycerol | 0.012 |
| TiN–TiO2 [54] | NaOH | 3.0 × 10−4 |
| BiFeO3 [21] | NaOH | 0.09 |
| Au/Pb(Zr, Ti)O3 [55] | NaOH | 0.06 |
| ATO/PMT/PbI2 (present work) | Sewage water | 0.095 |
| PbI2-Polyaniline | Polyaniline | |
|---|---|---|
| EHOMO, eV | −7.9119 | −7.8566 |
| ELUMO, eV | 0.6318 | 3.1765 |
| Electronegativity (χ), eV | 3.64005 | 2.34005 |
| Global hardness (η), eV | 4.27185 | 5.51655 |
| Electrophilicity (ω), eV | 1.5509 | 0.49631 |
| Total dipole moment, Debye | 12.56297 | 0.00019 |
| Gibbs free energy (G), Ha | −2332.91 | −1547.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hadia, N.M.A.; Khalafalla, M.A.H.; Abdel Salam, F.M.; Ahmed, A.M.; Shaban, M.; Almuqrin, A.H.; Hajjiah, A.; Hanafi, H.A.; Alruqi, M.; Mourad, A.-H.I.; et al. Conversion of Sewage Water into H2 Gas Fuel Using Hexagonal Nanosheets of the Polyaniline-Assisted Deposition of PbI2 as a Nanocomposite Photocathode with the Theoretical Qualitative Ab-Initio Calculation of the H2O Splitting. Polymers 2022, 14, 2148. https://doi.org/10.3390/polym14112148
Hadia NMA, Khalafalla MAH, Abdel Salam FM, Ahmed AM, Shaban M, Almuqrin AH, Hajjiah A, Hanafi HA, Alruqi M, Mourad A-HI, et al. Conversion of Sewage Water into H2 Gas Fuel Using Hexagonal Nanosheets of the Polyaniline-Assisted Deposition of PbI2 as a Nanocomposite Photocathode with the Theoretical Qualitative Ab-Initio Calculation of the H2O Splitting. Polymers. 2022; 14(11):2148. https://doi.org/10.3390/polym14112148
Chicago/Turabian StyleHadia, N. M. A., Mohammed A. H. Khalafalla, Fatma M. Abdel Salam, Ashour M. Ahmed, Mohamed Shaban, Aljawhara H. Almuqrin, Ali Hajjiah, H. A. Hanafi, Mansoor Alruqi, Abdel-Hamid I. Mourad, and et al. 2022. "Conversion of Sewage Water into H2 Gas Fuel Using Hexagonal Nanosheets of the Polyaniline-Assisted Deposition of PbI2 as a Nanocomposite Photocathode with the Theoretical Qualitative Ab-Initio Calculation of the H2O Splitting" Polymers 14, no. 11: 2148. https://doi.org/10.3390/polym14112148







