Artificial Intelligence-Empowered 3D and 4D Printing Technologies toward Smarter Biomedical Materials and Approaches
Abstract
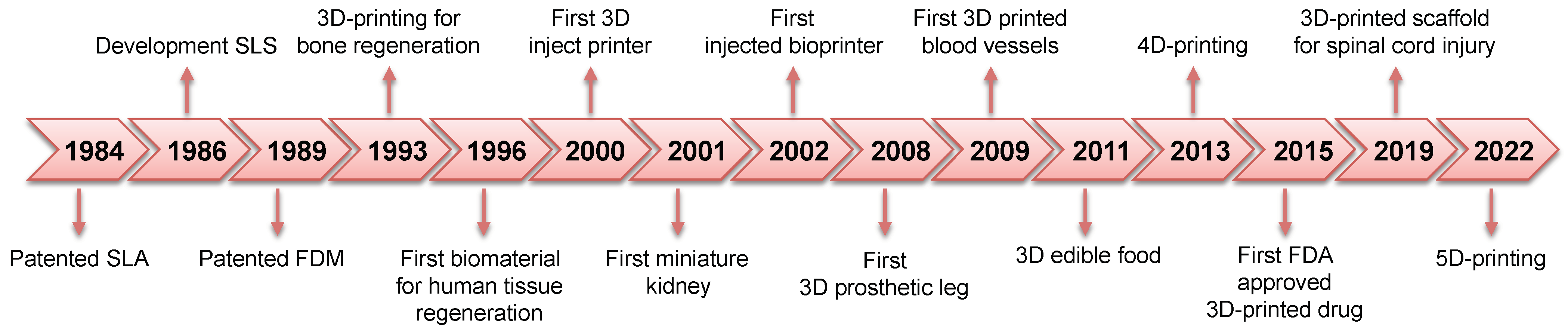
1. Introduction
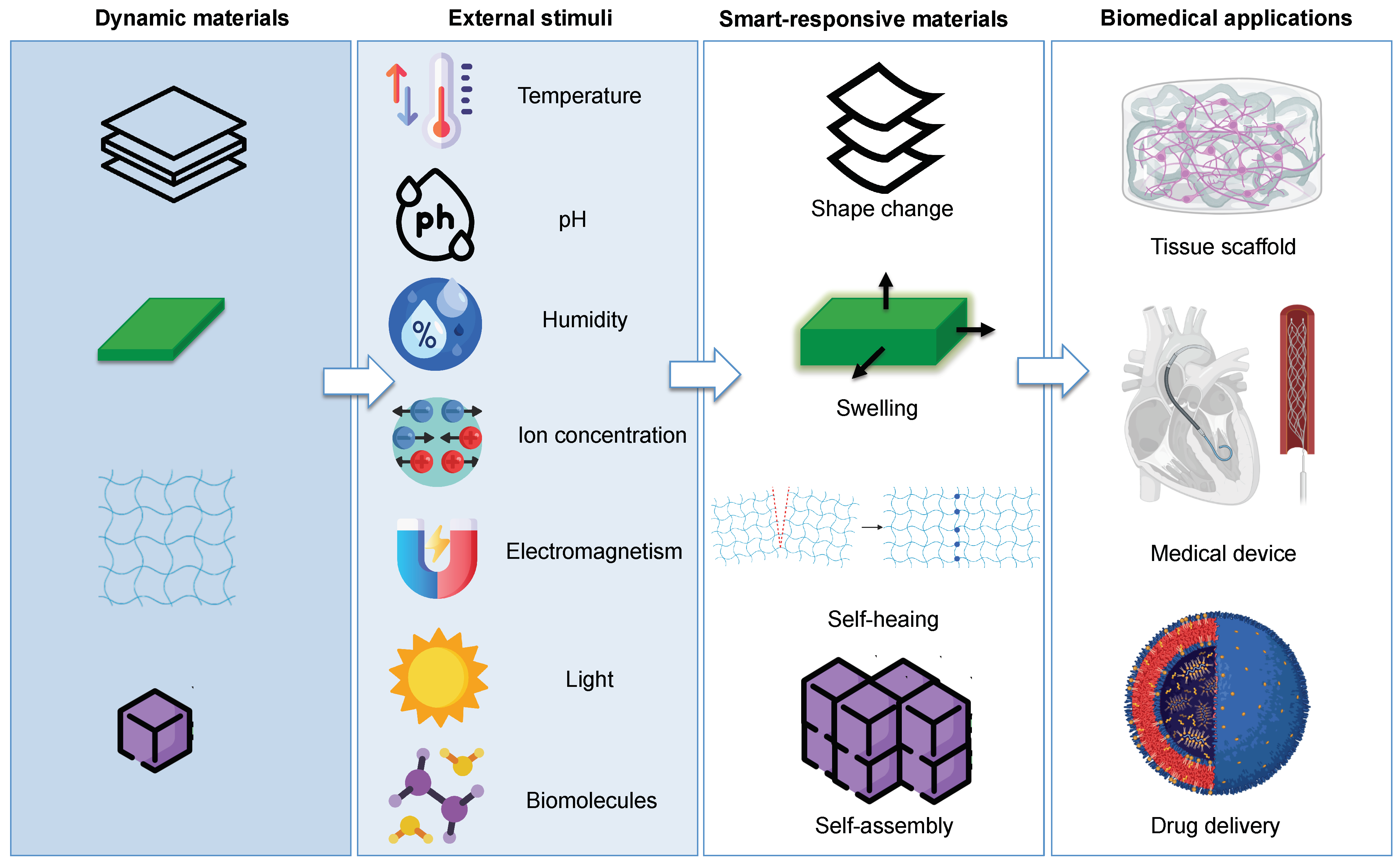
2. 4D Printing
2.1. Self-Adaptability
2.2. Self-Repair
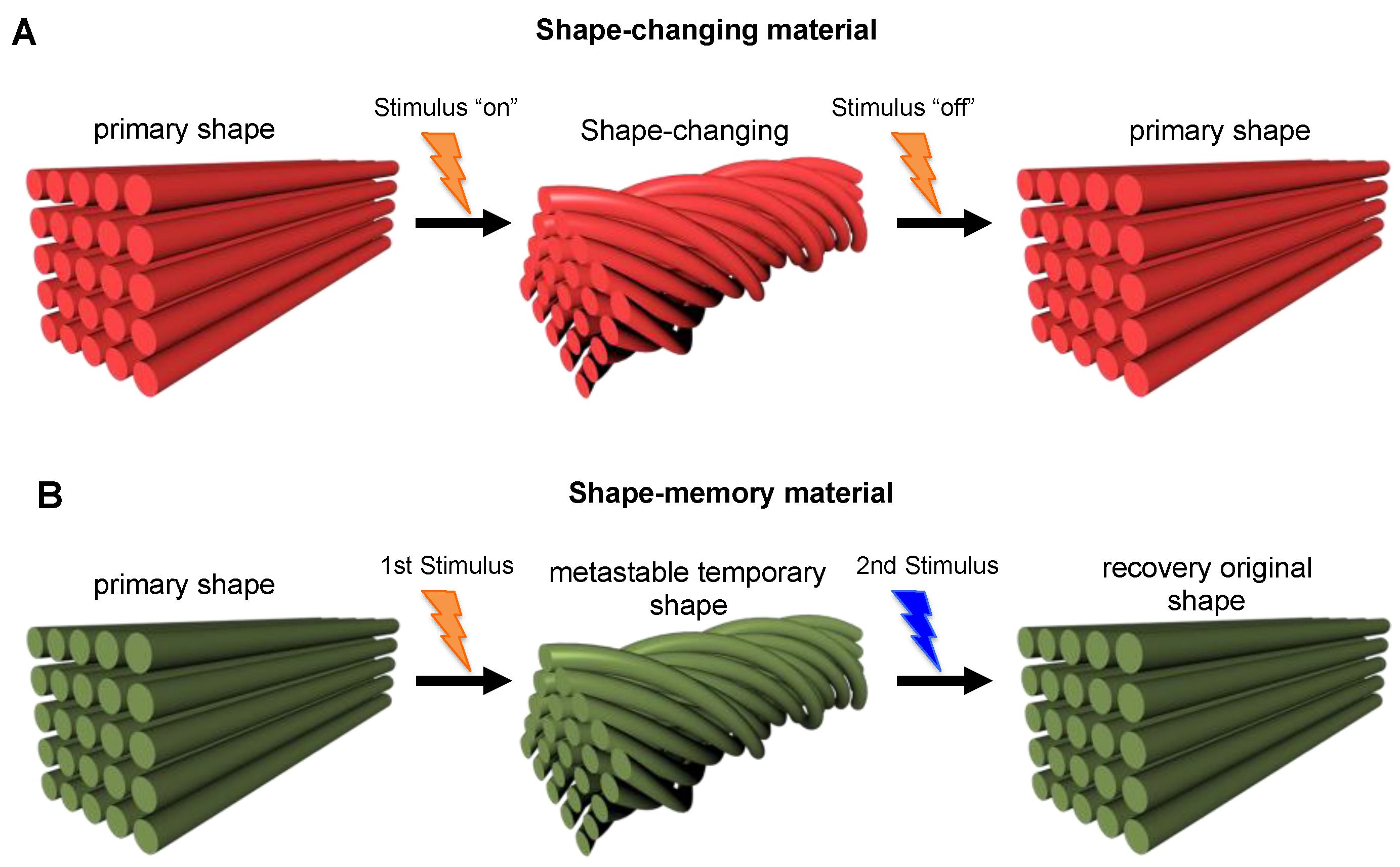
2.3. Shape-Shifting
2.4. Self-Assembly
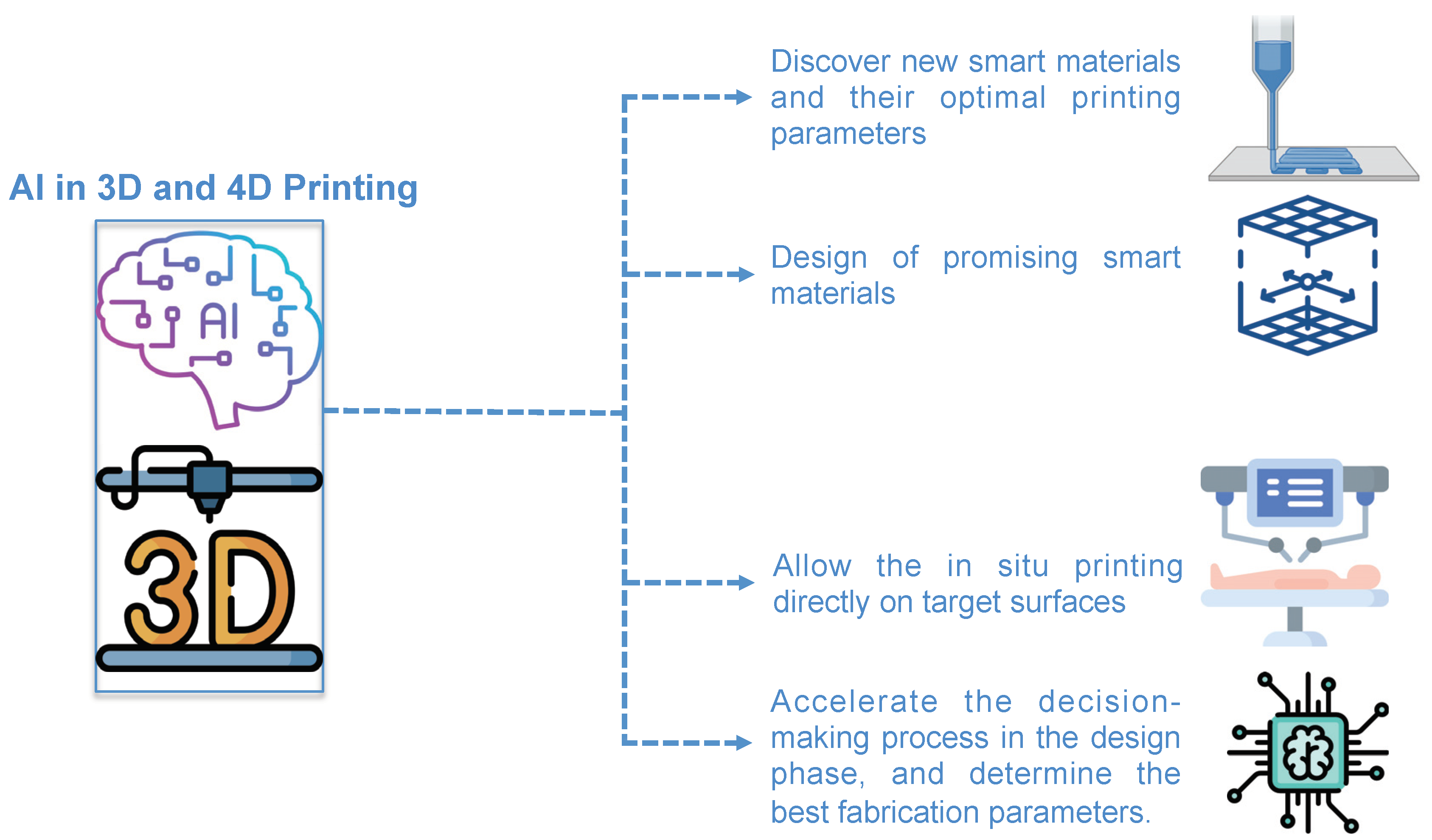
3. Open-Loop AI for 3D Printing
4. Closed-Loop AI for 3D Printing
| Common Terms Used in Additive Manufacturing and Artificial Intelligence | REFs |
|---|---|
| 3D printing: three-dimensional (3D) printing is an additive manufacturing process in which a physical object is created from a computer-aided design (CAD) model by printing the model on a pre-computed layer-by-layer toolpath. This process is fully deterministic and, therefore, is ideal for printing on planar surfaces that are stationary relative to the coordinate system of the printer (namely, ex situ 3D printing). To date, there are several 3D printing methods that include the following: fused deposition modeling (FDM), selective laser sintering (SLS), stereolithography (SLA), and direct ink writing (DIW). | [1,18,24,28,40] |
| 4D printing: four-dimensional (4D) printing uses the same techniques of 3D printing through computer-programmed deposition of material in successive layers to create a 3D object. However, in 4D printing, the resulting 3D object is able to change shape, structure, or function directly off the print bed in response to external stimulus, with the fourth dimension being the time-dependent shape change after the printing. It is therefore a type of programmable 3D printer, wherein after the fabrication process, the printed material reacts with parameters within the environment (humidity, temperature, mechanical force, pH, etc.) and changes its form accordingly. | [2,4,47,49,50,59,80] |
| Artificial Intelligence: artificial intelligence (AI) leverages computers and machines to mimic the problem-solving and decision-making capabilities of the human mind. Although a number of definitions of AI have surfaced over the last few decades, the most used is that of John McCarthy: “it is the science and engineering of making intelligent machines, especially intelligent computer programs. It is related to the similar task of using computers to understand human intelligence, but AI does not have to confine itself to methods that are biologically observable”. | [56,58,87] |
| Machine Learning: machine learning (ML) is a branch of AI and computer science, which focuses on the use of data and algorithms to imitate the way those humans learn, gradually improving its accuracy. ML involves the development and deployment of algorithms that, rather than being programmed to assign certain outputs in response to specific inputs from the environment, analyze data and their properties, and determine the action by using statistical tools. Usually, ML algorithms can be broadly classified into the following five categories: supervised learning, unsupervised learning, semi-supervised learning, reinforcement learning and federated learning. | [56,57,87] |
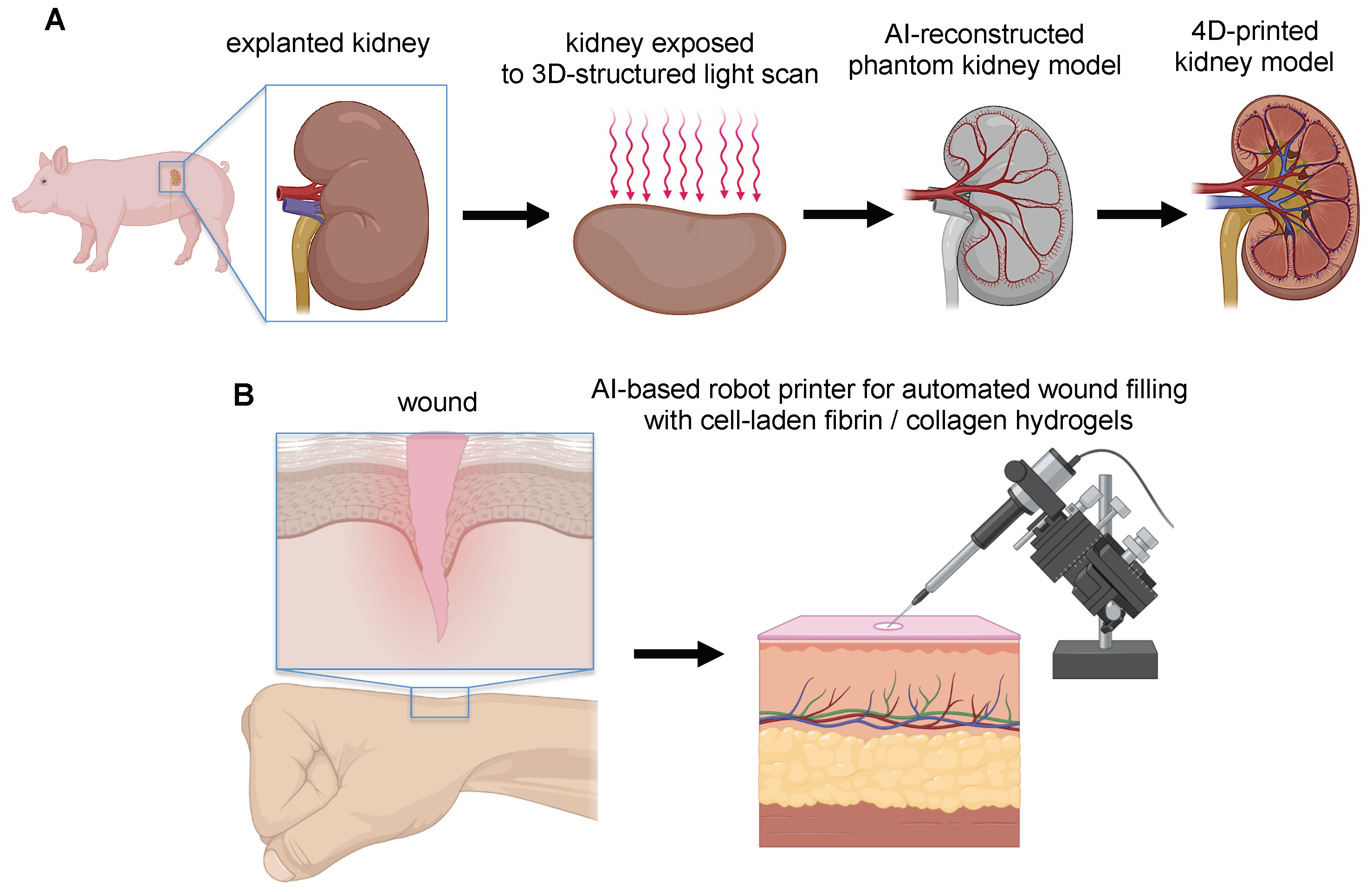
| Open-Loop AI printing: open-loop AI leverages pre-acquired sensory data (such as laser scanning and 3D tomography reconstructions) to obtain precise target geometry in various forms of 3D representations such as meshes and voxels. Then this geometry is calibrated with respect to the printing platform, thus enabling the generation of a toolpath on complex surfaces (i.e., organs or tissues). Based on this morphing path, open-loop AI can design the distribution of shape-morphing materials (whereby the morphing can be induced by mechanical load, change of temperature or pH, swelling) within the 3D-printed model to achieve improved compliance to a dynamically varying target surface. The AI-related computation occurs prior to the printing process. | [87,90] |
| Closed-Loop AI printing: closed-loop AI printing integrates sensing as part of the printing process. The sensory data are processed in real time using AI tools to recognize the surface of the target. A feedback-control system adjusts the toolpath in real time to compensate the target motion, environmental disturbance, and calibration errors, thus ensuring the 3D printing procedures. | [87,90,92] |
5. 5D Printing: A New Route of AI and AM
6. Regulatory Standpoint for AI
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ji, Y.; Luan, C.; Yao, X.; Fu, J.; He, Y. Recent Progress in 3D Printing of Smart Structures: Classification, Challenges, and Trends. Adv. Intell. Syst. 2022, 3, 2000271. [Google Scholar] [CrossRef]
- Khoo, Z.X.; Teoh, J.E.M.; Liu, Y.; Chua, C.K.; Yang, S.; An, J.; Yeong, W.Y. 3D printing of smart materials: A review on recent progresses in 4D printing. Virtual Phys. Prototyp. 2015, 10, 103–122. [Google Scholar] [CrossRef]
- Breger, J.C.; Yoon, C.; Xiao, R.; Kwag, H.R.; Wang, M.O.; Fisher, J.P.; Nguyen, T.D.; Gracias, D.H. Self-folding thermo-magnetically responsive soft microgrippers. ACS Appl. Mater. Interfaces 2015, 7, 3398–3405. [Google Scholar] [CrossRef] [PubMed]
- Gladman, A.S.; Matsumoto, E.A.; Nuzzo, R.G.; Mahadevan, L.; Lewis, J.A. Biomimetic 4D printing. Nat. Mater. 2016, 15, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Guo, Q.; Cai, X.; Zhou, S.; Kobe, B.; Yang, J. Initiator-integrated 3D printing enables the formation of complex metallic architectures. ACS Appl. Mater. Interfaces 2014, 6, 2583–2587. [Google Scholar] [CrossRef]
- Kim, Y.; Yuk, H.; Zhao, R.; Chester, S.A.; Zhao, X. Printing ferromagnetic domains for untethered fast-transforming soft materials. Nature 2018, 558, 274–279. [Google Scholar] [CrossRef]
- Theato, P.; Sumerlin, B.S.; O’Reilly, R.K.; Epps, T.H., III. Stimuli responsive materials. Chem. Soc. Rev. 2013, 42, 7055–7056. [Google Scholar] [CrossRef]
- Roy, D.; Cambre, J.N.; Sumerlin, B.S. Future perspectives and recent advances in stimuli-responsive materials. Prog. Polym. Sci. 2010, 35, 278–301. [Google Scholar] [CrossRef]
- Shafranek, R.T.; Millik, S.C.; Smith, P.T.; Lee, C.U.; Boydston, A.J.; Nelson, A. Stimuli-responsive materials in additive manufacturing. Prog. Polym. Sci. 2019, 93, 36–67. [Google Scholar] [CrossRef]
- Moulin, E.; Faour, L.; Carmona-Vargas, C.C.; Giuseppone, N. From Molecular Machines to Stimuli-Responsive Materials. Adv. Mater. 2020, 32, 1906036. [Google Scholar] [CrossRef]
- Liu, X.; Yuk, H.; Lin, S.; Parada, G.A.; Tang, T.-C.; Tham, E.; de la Fuente-Nunez, C.; Lu, T.K.; Zhao, X. 3D Printing of Living Responsive Materials and Devices. Adv. Mater. 2018, 30, 1704821. [Google Scholar] [CrossRef]
- Duraj-Thatte, A.M.; Manjula-Basavanna, A.; Rutledge, J.; Xia, J.; Hassan, S.; Sourlis, A.; Rubio, A.G.; Lesha, A.; Zenkl, M.; Kan, A.; et al. Programmable microbial ink for 3D printing of living materials produced from genetically engineered protein nanofibers. Nat. Commun. 2021, 12, 6600. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Ou, Y.; Liu, J.; Huang, Q.; Tang, B.; Xin, F.; Yu, Z. 3D Printed Biocatalytic Living Materials with Dual-Network Reinforced Bioinks. Small 2021, 18, 2104820. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Aubin-Tam, M.E.; Meyer, A.S. 3D Printing for the Fabrication of Biofilm-Based Functional Living Materials. ACS Synth. Biol. 2019, 8, 1564–1567. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, F.; Yan, Z.; Ma, Q.; Li, X.; Huang, Y.; Rogers, J.A. Printing, folding and assembly methods for forming 3D mesostructures in advanced materials. Nat. Rev. Mater. 2017, 2, 17019. [Google Scholar] [CrossRef]
- Zhu, Z.; Guo, S.Z.; Hirdler, T.; Eide, C.; Fan, X.; Tolar, J.; McAlpine, M.C. 3D Printed Functional and Biological Materials on Moving Freeform Surfaces. Adv. Mater. 2018, 30, 1707495. [Google Scholar] [CrossRef]
- Tofail, S.A.M.; Koumoulos, E.P.; Bandyopadhyay, A.B.S.; Odonoghue, L.; Charitidis, C.A. Additive manufacturing: Scientific and technological challenges, market uptake and opportunities. Mater. Today 2017, 21, 22–37. [Google Scholar] [CrossRef]
- Ngo, T.D.; Kashani, A.; Imbalzano, G.; Nguyen, K.T.Q.; Hui, D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos. Part B Eng. 2018, 143, 172–196. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- Dey, M.; Ozbolat, I.T. 3D bioprinting of cells, tissues and organs. Sci. Rep. 2020, 10, 14023. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Yan, W.C.; Lu, W.F.; Wang, C.H.; Fuh, J.Y.H. 3D bioprinting of tissues and organs for regenerative medicine. Adv. Drug Deliv. Rev. 2018, 132, 296–332. [Google Scholar] [CrossRef]
- Zhang, N.; Singh, S.; Liu, S.; Zbijewski, W.; Grayson, W.L. A robust, autonomous, volumetric quality assurance method for 3D printed porous scaffolds. 3D Print. Med. 2022, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Pang, S.S.; Fang, E.; Chen, K.W.; Leung, M.; Chow, V.L.; Fang, C. Patient-specific 3D-printed helmet for post-craniectomy defect-a case report. 3D Print. Med. 2022, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Zhan, S.; Guo, A.X.Y.; Cao, S.C.; Liu, N. 3D Printing Soft Matters and Applications: A Review. Int. J. Mol. Sci. 2022, 23, 3790. [Google Scholar] [CrossRef] [PubMed]
- Sayegh, M.A.; Daraghma, H.; Mekid, S.; Bashmal, S. Review of Recent Bio-Inspired Design and Manufacturing of Whisker Tactile Sensors. Sensors 2022, 22, 2705. [Google Scholar] [CrossRef]
- Bao, C.; Kim, T.H.; Hassanpoor Kalhori, A.; Kim, W.S. A 3D-printed neuromorphic humanoid hand for grasping unknown objects. iScience 2022, 25, 104119. [Google Scholar] [CrossRef]
- Sala, R.; Regondi, S.; Pugliese, R. Design Data and Finite Element Analysis of 3D Printed Poly(ε-Caprolactone)-Based Lattice Scaffolds: Influence of Type of Unit Cell, Porosity, and Nozzle Diameter on the Mechanical Behavior. Eng 2022, 3, 9–23. [Google Scholar] [CrossRef]
- Pugliese, R.; Beltrami, B.; Regondi, S.; Lunetta, C. Polymeric biomaterials for 3D printing in medicine: An overview. Ann. 3D Print. Med. 2021, 2, 100011. [Google Scholar] [CrossRef]
- Lin, H.; Shi, L.; Wang, D. A rapid and intelligent designing technique for patient-specific and 3D-printed orthopedic cast. 3D Print. Med. 2015, 2, 4. [Google Scholar] [CrossRef]
- Huang, L.Y.; Wang, T.H.; Chang, B.C.; Huang, C.I.; Chou, L.W.; Wang, S.J.; Chen, W.M. Printing a static progressive orthosis for hand rehabilitation. J. Chin. Med. Assoc. 2021, 84, 795–798. [Google Scholar] [CrossRef]
- Yildiz, K.; Medetalibeyoglu, F.; Kaymaz, I.; Ulusoy, G.R. Triad of foot deformities and its conservative treatment: With a 3D customized insole. Proc. Inst. Mech. Eng. H 2021, 235, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Portnoy, S.; Barmin, N.; Elimelech, M.; Assaly, B.; Oren, S.; Shanan, R.; Levanon, Y. Automated 3D-printed finger orthosis versus manual orthosis preparation by occupational therapy students: Preparation time, product weight, and user satisfaction. J. Hand Ther. 2020, 33, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhang, P.; Yan, M.; Xie, Y.M.; Huang, G.Z. Additive manufacturing of specific ankle-foot orthoses for persons after stroke: A preliminary study based on gait analysis data. Math. Biosci. Eng. 2019, 16, 8134–8143. [Google Scholar] [CrossRef]
- Ielapi, A.; Lammens, N.; Van Paepegem, W.; Forward, M.; Deckers, J.P.; Vermandel, M.; De Beule, M. A validated computational framework to evaluate the stiffness of 3D printed ankle foot orthoses. Comput. Methods Biomech. Biomed. Eng. 2019, 22, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Wojciechowski, E.; Chang, A.Y.; Balassone, D.; Ford, J.; Cheng, T.L.; Little, D.; Menezes, M.P.; Hogan, S.; Burns, J. Feasibility of designing, manufacturing and delivering 3D printed ankle-foot orthoses: A systematic review. J. Foot Ankle Res. 2019, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Lal, H.; Patralekh, M.K. 3D printing and its applications in orthopaedic trauma: A technological marvel. J. Clin. Orthop. Trauma 2018, 9, 260–268. [Google Scholar] [CrossRef]
- Pietrabissa, A.; Marconi, S.; Negrello, E.; Mauri, V.; Peri, A.; Pugliese, L.; Marone, E.M.; Auricchio, F. An overview on 3D printing for abdominal surgery. Surg. Endosc. 2020, 34, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Marone, E.M.; Rinaldi, L.F.; Conti, M.; Marconi, S.; Auricchio, F.; Pietrabissa, A.; Basile, G. Three-Dimensional Printed Models Can Help Settle Malpractice Litigation Over Surgical Interventions. Ann. Vasc. Surg. 2020, 65, e292–e294. [Google Scholar] [CrossRef]
- Pugliese, L.; Marconi, S.; Negrello, E.; Mauri, V.; Peri, A.; Gallo, V.; Auricchio, F.; Pietrabissa, A. The clinical use of 3D printing in surgery. Updates Surg. 2018, 70, 381–388. [Google Scholar] [CrossRef]
- Guvendiren, M.; Molde, J.; Soares, R.M.; Kohn, J. Designing Biomaterials for 3D Printing. ACS Biomater. Sci. Eng. 2016, 2, 1679–1693. [Google Scholar] [CrossRef]
- Ligon, S.C.; Liska, R.; Stampfl, J.; Gurr, M.; Mulhaupt, R. Polymers for 3D Printing and Customized Additive Manufacturing. Chem. Rev. 2017, 117, 10212–10290. [Google Scholar] [CrossRef] [PubMed]
- Ratnamani, M.P.C.; Zhang, X.; Wang, H. A Comprehensive Assessment on the Pivotal Role of Hydrogels in Scaffold-Based Bioprinting. Gels 2022, 8, 239. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.Q.; Ng, F.L.; Kai, J.T.Y.; Feih, S.; Nai, M.L.S. Tensile Strength Enhancement of Fused Filament Fabrication Printed Parts: A Review of Process Improvement Approaches and Respective Impact. Addit. Manuf. 2022, 54, 102724. [Google Scholar] [CrossRef]
- Tran, T.Q.; Canturri, C.; Deng, X.; Tham, C.L.; Ng, F.L. Enhanced tensile strength of acrylonitrile butadiene styrene composite specimens fabricated by overheat fused filament fabrication printing. Compos. Part B Eng. 2022, 235, 109783. [Google Scholar] [CrossRef]
- Shaikh, M.Q.; Graziosi, S.; Atre, S.V. Supportless printing of lattice structures by metal fused filament fabrication (MF3) of Ti-6Al-4V: Design and analysis. Rapid Prototyp. J. 2021, 27, 1408–1422. [Google Scholar] [CrossRef]
- Thiam, B.G.; El Magri, A.; Vanaei, H.R.; Vaudreuil, S. 3D Printed and Conventional Membranes-A Review. Polymers 2022, 14, 1023. [Google Scholar] [CrossRef]
- Tibbits, S. 4D Printing: Multi-Material Shape Change. Arch. Des. 2014, 84, 116–121. [Google Scholar] [CrossRef]
- González-Henríquez, C.M.; Sarabia-Vallejos, M.A.; Sanz-Horta, R.; Rodriguez-Hernandez, J. Additive Manufacturing of Polymers: 3D and 4D Printing, Methodologies, Type of Polymeric Materials, and Applications. Macromol. Eng. Precise Synth. Macrosc. Mater. Appl. 2022, 1, 57–116. [Google Scholar]
- Momeni, F.; Liu, X.; Ni, J. A review of 4D printing. Mater. Des. 2017, 122, 42–79. [Google Scholar] [CrossRef]
- Mallakpour, S.; Tabesh, F.; Hussain, C.M. 3D and 4D printing: From innovation to evolution. Adv. Colloid Interface Sci. 2021, 294, 102482. [Google Scholar] [CrossRef]
- Zhou, W.; Qiao, Z.; Nazarzadeh Zare, E.; Huang, J.; Zheng, X.; Sun, X.; Shao, M.; Wang, H.; Wang, X.; Chen, D.; et al. 4D-Printed Dynamic Materials in Biomedical Applications: Chemistry, Challenges, and Their Future Perspectives in the Clinical Sector. J. Med. Chem. 2020, 63, 8003–8024. [Google Scholar] [CrossRef] [PubMed]
- Hendrikson, W.J.; Rouwkema, J.; Clementi, F.; van Blitterswijk, C.A.; Fare, S.; Moroni, L. Towards 4D printed scaffolds for tissue engineering: Exploiting 3D shape memory polymers to deliver time-controlled stimulus on cultured cells. Biofabrication 2017, 9, 031001. [Google Scholar] [CrossRef] [PubMed]
- Malachowski, K.; Breger, J.; Kwag, H.R.; Wang, M.O.; Fisher, J.P.; Selaru, F.M.; Gracias, D. Stimuli-responsive theragrippers for chemomechanical controlled release. Angew. Chem. Int. Ed. 2014, 53, 8045–8049. [Google Scholar] [CrossRef]
- Zarek, M.; Mansour, N.; Shapira, S.; Cohn, D. 4D Printing of Shape Memory-Based Personalized Endoluminal Medical Devices. Macromol. Rapid Commun. 2017, 38, 1600628. [Google Scholar] [CrossRef] [PubMed]
- Riedl, M.O. Human-centered artificial intelligence and machine learning. Hum. Behav. Emerg. Technol. 2019, 1, 33–36. [Google Scholar] [CrossRef]
- Pugliese, R.; Regondi, S.R.M. Machine learning-based approach: Global trends, research directions, and regulatory standpoints. Data Sci. Manag. 2021, 4, 19–29. [Google Scholar] [CrossRef]
- Goh, G.D.; Sing, S.L.; Yeong, W.Y. A review on machine learning in 3D printing: Applications, potential, and challenges. Artif. Intell. Rev. 2021, 54, 63–94. [Google Scholar] [CrossRef]
- Milazzo, M.; Libonati, F. The Synergistic Role of Additive Manufacturing and Artificial Intelligence for the Design of New Advanced Intelligent Systems. Adv. Intell. Syst. 2022, 4, 2100278. [Google Scholar] [CrossRef]
- Ahmed, A.; Arya, S.; Gupta, V.; Furukawa, H.; Khosla, A. 4D printing: Fundamentals, materials, applications and challenges. Polymer 2021, 228, 123926. [Google Scholar] [CrossRef]
- Spiegel, C.A.; Hippler, M.; Münchinger, A.; Bastmeyer, M.; Barner-Kowollik, C.; Wegener, M. 4D Printing at the Microscale. Adv. Funct. Mater. 2019, 30, 1907615. [Google Scholar] [CrossRef]
- Zhang, Z.; Demir, K.G.; Gu, G.X. Developments in 4D-printing: A review on current smart materials, technologies, and applications. Int. J. Smart Nano Mater. 2019, 10, 205–224. [Google Scholar] [CrossRef]
- Gao, B.; Yang, Q.; Zhao, X.; Jin, G.; Ma, Y.; Xu, F. 4D Bioprinting for Biomedical Applications. Trends Biotechnol. 2016, 34, 746–756. [Google Scholar] [CrossRef] [PubMed]
- Kumari, G.; Abhishek, K.; Singh, S.; Hussain, A.; Altamimi, M.A.; Madhyastha, H.; Webster, T.J.; Dev, A. A voyage from 3D to 4D printing in nanomedicine and healthcare: Part II. Nanomedicine 2022, 17, 255–270. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Han, S.; Wu, W.; Wu, Z.; Yuan, Y.; Wu, J.; Liu, C. Harnessing 4D Printing Bioscaffolds for Advanced Orthopedics. Small 2022, 18, e2106824. [Google Scholar] [CrossRef]
- Khademhosseini, A.; Langer, R. A decade of progress in tissue engineering. Nat. Protoc. 2016, 11, 1775–1781. [Google Scholar] [CrossRef]
- Taylor, D.L.; Panhuis, M.i.h. Self-Healing Hydrogels. Adv. Mater. 2016, 28, 9060–9093. [Google Scholar] [CrossRef]
- Pugliese, R.; Gelain, F. Characterization of elastic, thermo-responsive, self-healable supramolecular hydrogel made of self-assembly peptides and guar gum. Mater. Des. 2019, 186, 108370. [Google Scholar] [CrossRef]
- Imam, S.S.; Hussain, A.; Altamimi, M.A.; Alshehri, S. Four-Dimensional Printing for Hydrogel: Theoretical Concept, 4D Materials, Shape-Morphing Way, and Future Perspectives. Polymers 2021, 13, 3858. [Google Scholar] [CrossRef]
- Hou, S.; Wang, X.; Park, S.; Jin, X.; Ma, P.X. Rapid Self-Integrating, Injectable Hydrogel for Tissue Complex Regeneration. Adv. Healthc Mater. 2015, 4, 1491–1495. [Google Scholar] [CrossRef]
- Malekmohammadi, S.; Aminabad, N.S.; Sabzi, A.; Zarebkohan, A.; Razavi, M.; Vosough, M.; Bodaghi, M.; Maleki, H. Smart and Biomimetic 3D and 4D Printed Composite Hydrogels: Opportunities for Different Biomedical Applications. Biomedicines 2021, 9, 1537. [Google Scholar] [CrossRef]
- Wan, X.; Wei, H.; Zhang, F.; Liu, Y.; Leng, J. 3D printing of shape memory poly(d,l-lactide-co-trimethylene carbonate) by direct ink writing for shape-changing structures. J. Appl. Polym. Sci. 2019, 136, 48177. [Google Scholar] [CrossRef]
- Kim, D.; Kim, T.; Lee, Y.-G. 4D Printed Bifurcated Stents with Kirigami-Inspired Structures. J. Vis. Exp. 2019, 1, e59746. [Google Scholar] [CrossRef] [PubMed]
- Whitesides, G.M.; Grzybowski, B. Self-assembly at all scales. Science 2002, 295, 2418–2421. [Google Scholar] [CrossRef] [PubMed]
- Whitesides, G.M.; Boncheva, M. Beyond molecules: Self-assembly of mesoscopic and macroscopic components. Proc. Natl. Acad. Sci. USA 2002, 99, 4769–4774. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S. Fabrication of novel biomaterials through molecular self-assembly. Nat. Biotechnol. 2003, 21, 1171–1178. [Google Scholar] [CrossRef]
- Pugliese, R.; Gelain, F. Peptidic Biomaterials: From Self-Assembling to Regenerative Medicine. Trends Biotechnol. 2017, 35, 145–158. [Google Scholar] [CrossRef]
- Zhou, Y.; Huang, W.M.; Kang, S.F.; Wu, X.L.; Lu, H.B.; Fu, J.; Cui, H. From 3D to 4Dprinting: Approaches and typical applications. J. Mech. Sci. Technol. 2015, 29, 4281–4288. [Google Scholar] [CrossRef]
- Tibbits, S.; McKnelly, C.; Olguin, C.; Dikovsky, D.; Hirsch, S. 4D Printing and Universal Transformation. ACADIA 14 2014, 1, 539. [Google Scholar]
- Campbell, T.A.; Tibbits, S.; Garrett, B. Can We Program the Material World? Sci. Am. 2014, 311, 60–65. [Google Scholar] [CrossRef]
- Hawkes, E.; An, B.; Benbernou, N.M.; Tanaka, H.; Kim, S.; Demaine, E.D.; Wood, R.J. Programmable Matter by Folding. Proc. Natl. Acad. Sci. USA 2010, 107, 12441–12445. [Google Scholar] [CrossRef]
- Cheung, K.C.; Demaine, E.D.; Bachrach, J.R.; Griffith, S. Programmable Assembly With Universally Foldable Strings. IEEE Trans. Robot. 2011, 27, 718–729. [Google Scholar] [CrossRef]
- Li, L.; Yu, F.; Shi, J.; Shen, S.; Teng, H.; Yang, J.; Wang, X.; Jiang, Q. In situ repair of bone and cartilage defects using 3D scanning and 3D printing. Sci. Rep. 2017, 7, 9416. [Google Scholar] [CrossRef] [PubMed]
- Farahani, N.; Braun, A.; Jutt, D.; Huffman, T.; Reder, N.; Liu, Z.; Yagi, Y.; Pantanowitz, L. Three-dimensional Imaging and Scanning: Current and Future Applications for Pathology. J. Pathol. Inform. 2017, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Aboali, M.; Manap, N.A.; Darsono, A.M.; Yusof, Z.M. Review on Three-Dimensional (3-D) Acquisition and Range Imaging Techniques. Int. J. Appl. Eng. Res. 2017, 12, 2409–2421. [Google Scholar]
- Zhu, Z.; Ng, D.W.H.; Park, H.S.; McAlpine, M.C. 3D-printed multifunctional materials enabled by artificial-intelligence-assisted fabrication technologies. Nat. Rev. Mater. 2020, 6, 27–47. [Google Scholar] [CrossRef]
- Singh, M.; Tong, Y.; Webster, K.; Cesewski, E.; Haring, A.P.; Laheri, S.; Carswell, B.; O’Brien, T.J.; Aardema, C.H.; Senger, R.S.; et al. 3D printed conformal microfluidics for isolation and profiling of biomarkers from whole organs. Lab Chip 2017, 17, 2561–2571. [Google Scholar] [CrossRef]
- Albanna, M.; Binder, K.W.; Murphy, S.V.; Kim, J.; Qasem, S.A.; Zhao, W.; Tan, J.; El-Amin, I.B.; Dice, D.D.; Marco, J.; et al. In Situ Bioprinting of Autologous Skin Cells Accelerates Wound Healing of Extensive Excisional Full-Thickness Wounds. Sci. Rep. 2019, 9, 1856. [Google Scholar] [CrossRef]
- Ferguson, A.L. Machine learning and data science in soft materials engineering. J. Phys. Conde. Matter. 2018, 30, 043002. [Google Scholar] [CrossRef]
- Hamel, C.M.; Roach, D.J.; Long, K.N.; Demoly, F.; Dunn, M.L.; Qi, H.J. Machine-learning based design of active composite structures for 4D printing. Smart Mater. Struct. 2019, 28, 065005. [Google Scholar] [CrossRef]
- Mercado Rivera, F.J.; Rojas Arciniegas, A.J. Additive manufacturing methods: Techniques, materials, and closed-loop control applications. Int. J. Adv. Manuf. Technol. 2020, 109, 17–31. [Google Scholar] [CrossRef]
- Ashammakhi, N.; Ahadian, S.; Pountos, I.; Hu, S.K.; Tellisi, N.; Bandaru, P.; Ostrovidov, S.; Dokmeci, M.R.; Khademhosseini, A. In situ three-dimensional printing for reparative and regenerative therapy. Biomed. Microdevices 2019, 21, 42. [Google Scholar] [CrossRef] [PubMed]
- Zolfagharian, A.; Kaynak, A.; Kouzani, A. Closed-loop 4D-printed soft robots. Mater. Des. 2020, 188, 108411. [Google Scholar] [CrossRef]
- Zhu, Z.; Park, H.S.; McAlpine, M.C. 3D printed deformable sensors. Sci. Adv. 2020, 6, eaba5575. [Google Scholar] [CrossRef] [PubMed]
- Losi, P.; Burchielli, S.; Spiller, D.; Finotti, V.; Kull, S.; Briganti, E.; Soldani, G. Cyanoacrylate surgical glue as an alternative to suture threads for mesh fixation in hernia repair. J. Surg. Res. 2010, 163, e53–e58. [Google Scholar] [CrossRef]
- Hakimi, N.; Cheng, R.; Leng, L.; Sotoudehfar, M.; Ba, P.Q.; Bakhtyar, N.; Amini-Nik, S.; Jeschke, M.G.; Gunther, A. Handheld skin printer: In situ formation of planar biomaterials and tissues. Lab Chip 2018, 18, 1440–1451. [Google Scholar] [CrossRef]
- Di Bella, C.; Duchi, S.; O’Connell, C.D.; Blanchard, R.; Augustine, C.; Yue, Z.; Thompson, F.; Richards, C.; Beirne, S.; Onofrillo, C.; et al. In situ handheld three-dimensional bioprinting for cartilage regeneration. J. Tissue Eng. Regen. Med. 2018, 12, 611–621. [Google Scholar] [CrossRef]
- O’Connell, C.D.; Di Bella, C.; Thompson, F.; Augustine, C.; Beirne, S.; Cornock, R.; Richards, C.J.; Chung, J.; Gambhir, S.; Yue, Z.; et al. Development of the Biopen: A handheld device for surgical printing of adipose stem cells at a chondral wound site. Biofabrication 2016, 8, 015019. [Google Scholar] [CrossRef]
- Nio, D.; Diks, J.; Bemelman, W.A.; Wisselink, W.; Legemate, D.A. Laparoscopic vascular surgery: A systematic review. Eur. J. Vasc. Endovasc. Surg. 2007, 33, 263–271. [Google Scholar] [CrossRef]
- Agarwal, D.K.; Sharma, V.; Toussi, A.; Viers, B.R.; Tollefson, M.K.; Gettman, M.T.; Frank, I. Initial Experience with da Vinci Single-port Robot-assisted Radical Prostatectomies. Eur. Urol. 2020, 77, 373–379. [Google Scholar] [CrossRef]
- Morelli, L.; Guadagni, S.; Di Franco, G.; Palmeri, M.; Di Candio, G.; Mosca, F. Da Vinci single site(c) surgical platform in clinical practice: A systematic review. Int. J. Med. Robot. 2016, 12, 724–734. [Google Scholar] [CrossRef]
- Reshmy, R.; Philip, E.; Madhavan, A.; Arun, K.B.; Binod, P.; Pugazhendhi, A.; Sindhu, R. Promising eco-friendly biomaterials for future biomedicine: Cleaner production and applications of Nanocellulose. Environ. Technol. Innov. 2021, 21, 101855. [Google Scholar]
- Zhao, X.; Kim, J.; Cezar, C.A.; Huebsch, N.; Lee, K.; Bouhadir, K.; Mooney, D.J. Active scaffolds for on-demand drug and cell delivery. Proc. Natl. Acad. Sci. USA 2011, 108, 67–72. [Google Scholar] [CrossRef] [PubMed]
- van Manen, T.; Janbaz, S.; Jansen, K.M.B.; Zadpoor, A.A. 4D printing of reconfigurable metamaterials and devices. Commun. Mater. 2021, 2, 1–8. [Google Scholar] [CrossRef]
- Zhou, N.; Bekenstein, Y.; Eisler, C.N.; Zhang, D.; Schwartzberg, A.M.; Yang, P.; Alivisatos, A.P.; Lewis, J.A. Perovskite nanowire-block copolymer composites with digitally programmable polarization anisotropy. Sci. Adv. 2019, 5, eaav8141. [Google Scholar] [CrossRef] [PubMed]
- Jaafreh, R.; Abuhmed, T.; Kim, J.-G.; Hamad, K. Crystal structure guided machine learning for the discovery and design of intrinsically hard materials. J. Materiomics 2021, 8, 678–684. [Google Scholar] [CrossRef]
- Veale, M.; Borgesius, F.Z. Demystifying the Draft EU Artificial Intelligence Act—Analysing the good, the bad, and the unclear elements of the proposed approach. Comput. Law Rev. Int. 2021, 22, 97–112. [Google Scholar] [CrossRef]
- Service, E.P.R. Regulatory Divergences in the Draft AI Act Differences in Public and Private Sector Obligations, Scientific Foresight Unit (STOA) PE 729.507; European Parliament: Strasbourg, France, 2022. [Google Scholar]
- Ebers, M.; Hoch, V.R.S.; Rosenkranz, F.; Ruschemeier, H.; Steinrötter, B. The European Commission’s Proposal for an Artificial Intelligence Act—A Critical Assessment by Members of the Robotics and AI Law Society (RAILS). J. Multidiscip. Sci. J. 2021, 4, 589–603. [Google Scholar] [CrossRef]
- Justo-Hanani, R. The politics of Artificial Intelligence regulation and governance reform in the European Union. Policy Sci. 2022, 55, 137–159. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pugliese, R.; Regondi, S. Artificial Intelligence-Empowered 3D and 4D Printing Technologies toward Smarter Biomedical Materials and Approaches. Polymers 2022, 14, 2794. https://doi.org/10.3390/polym14142794
Pugliese R, Regondi S. Artificial Intelligence-Empowered 3D and 4D Printing Technologies toward Smarter Biomedical Materials and Approaches. Polymers. 2022; 14(14):2794. https://doi.org/10.3390/polym14142794
Chicago/Turabian StylePugliese, Raffaele, and Stefano Regondi. 2022. "Artificial Intelligence-Empowered 3D and 4D Printing Technologies toward Smarter Biomedical Materials and Approaches" Polymers 14, no. 14: 2794. https://doi.org/10.3390/polym14142794
APA StylePugliese, R., & Regondi, S. (2022). Artificial Intelligence-Empowered 3D and 4D Printing Technologies toward Smarter Biomedical Materials and Approaches. Polymers, 14(14), 2794. https://doi.org/10.3390/polym14142794







