Spectroscopic Studies of Styrylquinoline Copolymers with Different Substituents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Measurement Methods and Equipments
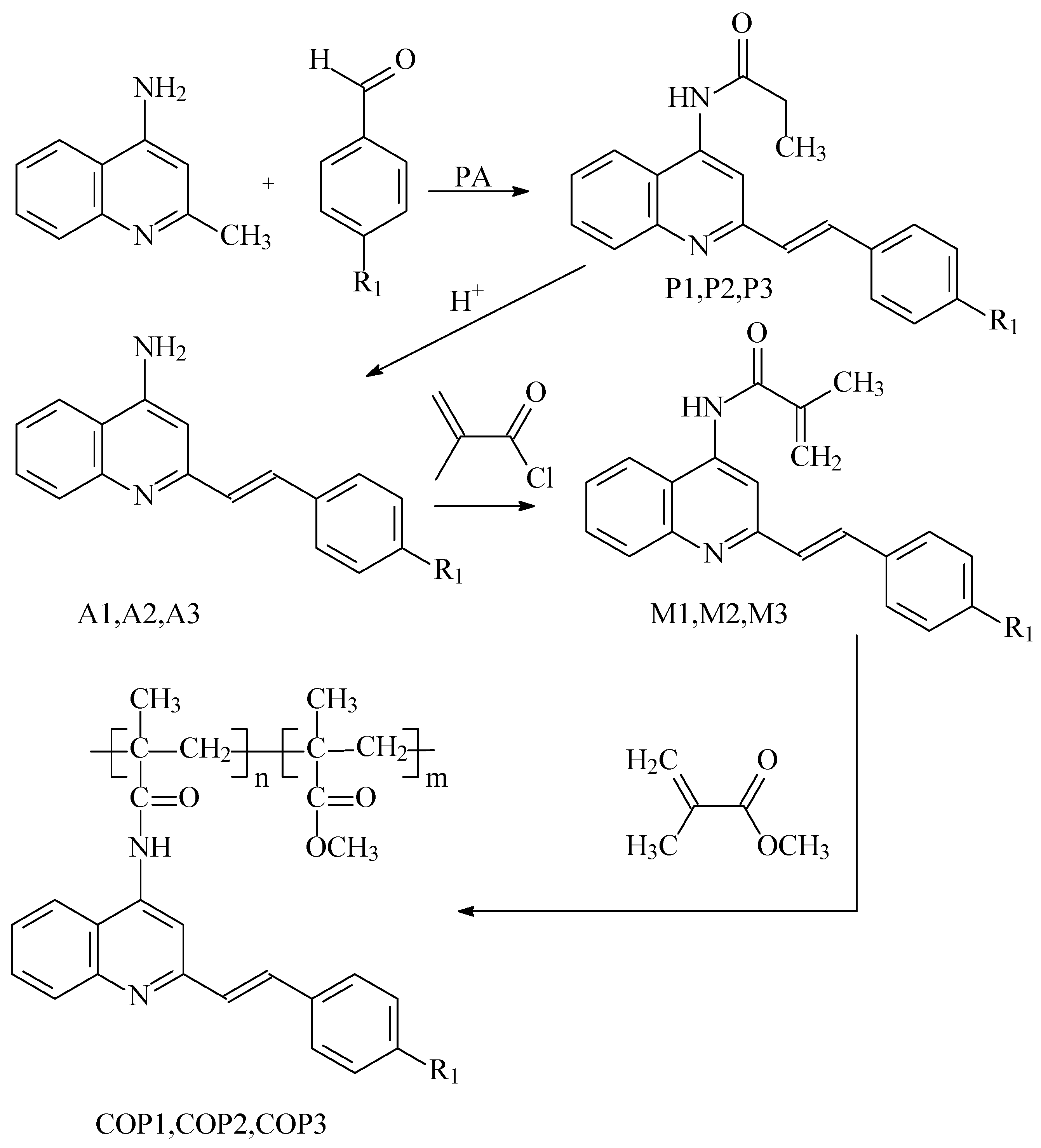
2.2. Synthesis
2.3. Polymerization
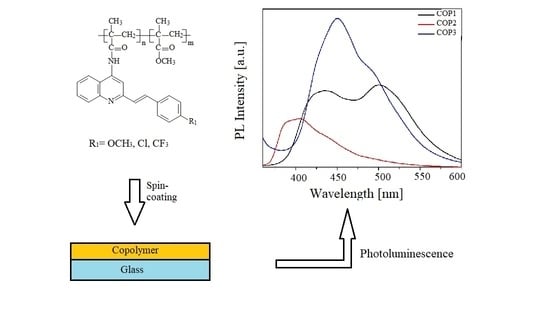
2.4. Thin Films Preparation
2.5. Theoretical Calculations
3. Results and Discussion
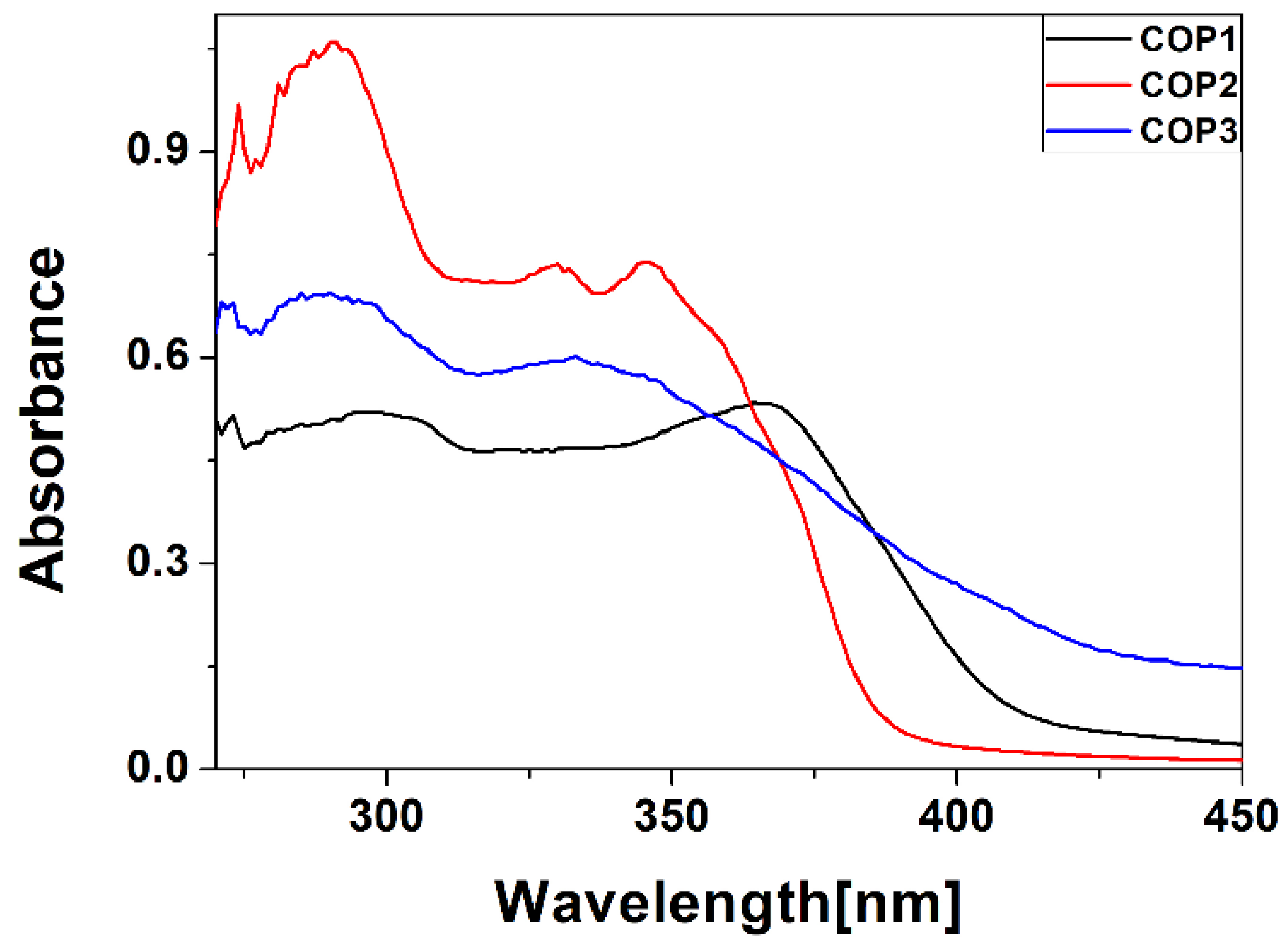
3.1. Experimental Results
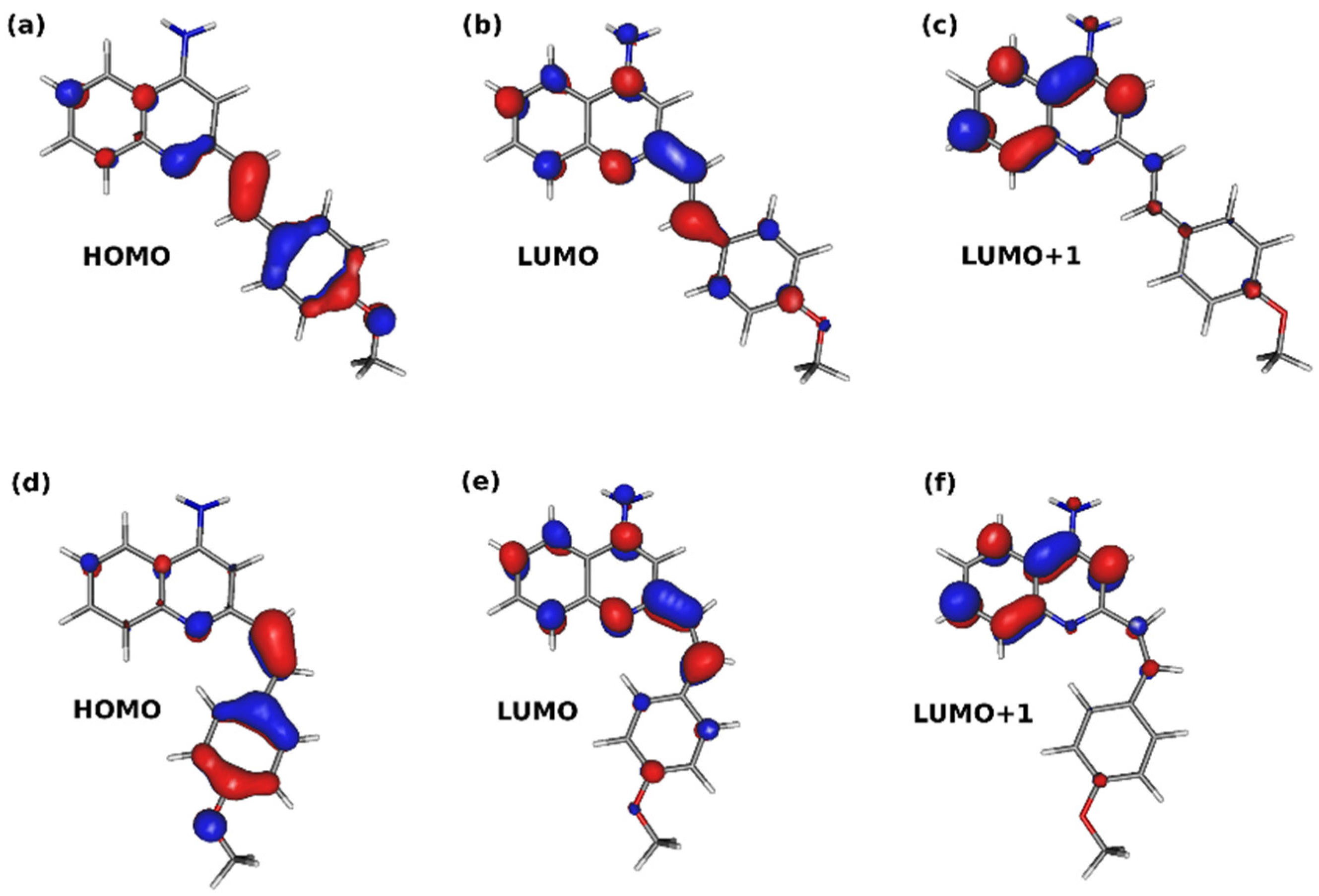
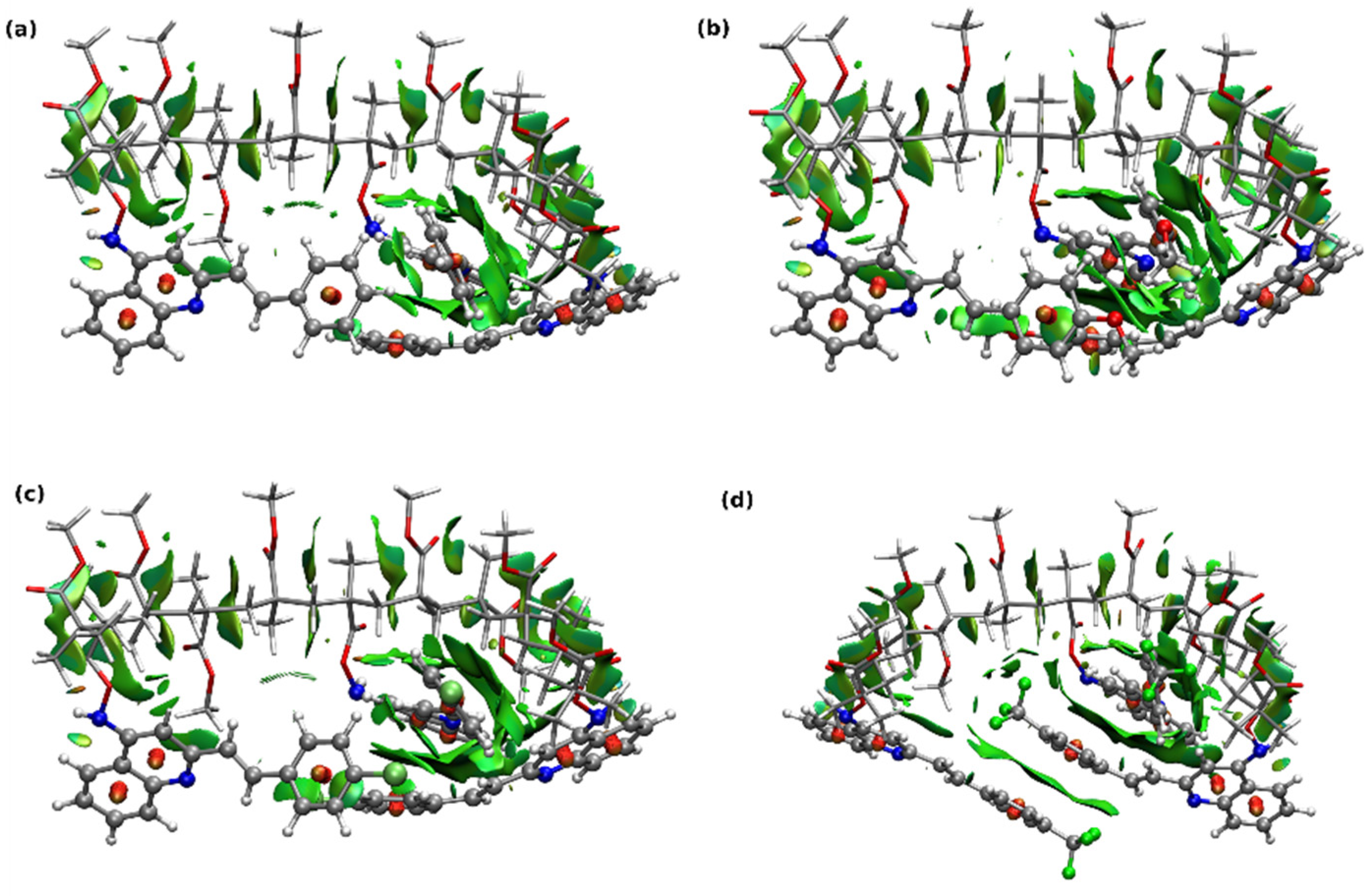
3.2. Theoretical Results
3.2.1. Chromophore Absorption
3.2.2. Copolymer Structure and Photophysics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaur, K.; Jain, M.; Reddy, R.P.; Jain, R. Quinolines and structurally related heterocycles as antimalarials. Eur. J. Med. Chem. 2010, 45, 3245–3264. [Google Scholar] [CrossRef] [PubMed]
- Baranoff, E.; Yum, J.H.; Graetzel, M.; Nazeeruddin, M.K. Cyclometallated iridium complexes for conversion of light into electricity and electricity into light. J. Organom. Chem. 2009, 694, 2661–2670. [Google Scholar] [CrossRef]
- Ho, C.L.; Li, H.; Wong, W.Y. Red to near-infrared organometallic phosphorescent dyes for OLED applications. J. Organom. Chem. 2014, 751, 261–285. [Google Scholar] [CrossRef]
- Xie, L.H.; Yin, C.R.; Lai, W.Y.; Fan, Q.L.; Huang, W. Polyfluorene-based semiconductors combined with various periodic table elements for organic electronics. Prog. Polym. Sci. 2012, 37, 1192–1264. [Google Scholar] [CrossRef]
- Guo, X.; Baumgarten, M.; Müllen, K. Designing π-conjugated polymers for organic electronics. Prog. Polym. Sci. 2013, 38, 1832–1908. [Google Scholar] [CrossRef]
- Jenekhe, S.A.; Lu, L.; Alam, M.M. New Conjugated Polymers with Donor−Acceptor Architectures: Synthesis and Photophysics of Carbazole−Quinoline and Phenothiazine−Quinoline Copolymers and Oligomers Exhibiting Large Intramolecular Charge Transfer. Macromolecules 2001, 34, 7315–7324. [Google Scholar] [CrossRef]
- Khalid, M.; Ali, A.; Jawaria, R.; Asghar, M.A.; Asim, S.; Khan, M.U.; Hussain, R.; Fayyaz ur Rehman, M.; Ennise, C.J.; Akram, M.S. First Principles Study of Electronic and Nonlinear Optical Properties of A-D−π−A and D-A-D−π−A Configured Compounds Containing Novel Quinoline-Carbazole Derivatives. RSC Adv. 2020, 10, 22273–22283. [Google Scholar] [CrossRef]
- Danel, A.; Gondek, E.; Kityk, I. 1H-pyrazolo[3,4-b] quinoline and 1H-pyrazolo [3, 4-b] quinoxaline derivatives as promising materials for optoelectronic applications. Opt. Mater. 2009, 32, 267–273. [Google Scholar] [CrossRef]
- Gulakova, E.N.; Berdnikova, D.V.; Aliyeu, T.M.; Fedorov, Y.V.; Godovikov, I.A.; Fedorova, O.A. Regiospecific C-N photocyclization of 2-styrylquinolines. J. Org. Chem. 2014, 79, 5533–5537. [Google Scholar] [CrossRef]
- Budyka, M.F.; Potashova, N.I.; Gavrishova, T.N.; Lee, V.M. The effect of substituents in the styryl moiety on the photocyclization of 4-styrylquinoline derivatives. High Energy Chem. 2010, 44, 404–411. [Google Scholar] [CrossRef]
- Oshkin, I.V.; Budyka, M.F. Quantum-chemical study of the photoisomerization and photocyclization reactions of styrylquinolines: Potential energy surfaces. High Energy Chem. 2010, 44, 472–481. [Google Scholar] [CrossRef]
- Deligeorgiev, T.; Vasilev, A.; Kaloyanova, S.; Vaquero, J.J. Styryl dyes—Synthesis and applications during the last 15 years. Soc. Dyers Col. 2010, 126, 55–80. [Google Scholar] [CrossRef]
- Rams-Baron, M.; Dulski, M.; Mrozek-Wilczkiewicz, A.; Korzec, M.; Cieslik, W.; Spaczyńska, E.; Bartczak, P.; Ratuszna, A.; Polanski, J.; Musiol, R. Synthesis of New Styrylquinoline Cellular Dyes, Fluorescent Properties, Cellular Localization and Cytotoxic Behavior. PLoS ONE 2015, 10, e0131210. [Google Scholar] [CrossRef] [PubMed]
- Budyka, M.F.; Potashova, N.I.; Gavrishova, T.N.; Li, V.M. Design of fully photonic molecular logic gates based on the supramolecular bis-styrylquinoline dyad. Nanotechnol. Russ. 2012, 7, 280–287. [Google Scholar] [CrossRef]
- Podeszwa, B.; Niedbala, H.; Polanski, J.; Musiol, R.; Tabak, D.; Finster, J.; Serafin, K.; Milczarek, M.; Wietrzyk, J.; Boryczka, S.; et al. Investigating the antiproliferative activity of quinoline-5,8-diones and styrylquinolinecarboxylic acids on tumor cell lines. Bioorg. Med. Chem. Lett. 2007, 17, 6138–6141. [Google Scholar] [CrossRef] [PubMed]
- Mao, F.; Yan, J.; Li, J.; Jia, X.; Miao, H.; Sun, Y.; Huang, L.; Li, X. New multi-target-directed small molecules against Alzheimer’s disease: A combination of resveratrol and clioquinol. Org. Biomol. Chem. 2014, 12, 5936–5944. [Google Scholar] [CrossRef]
- Mekouar, K.; Mouscadet, J.F.; Desmaële, D.; Subra, F.; Leh, H.; Savouré, D.; Auclair, C.; d’Angelo, J. Styrylquinoline derivatives: A new class of potent HIV-1 integrase inhibitors that block HIV-1 replication in CEM cells. J. Med. Chem. 1998, 41, 2846–2857. [Google Scholar] [CrossRef]
- Budyka, M.F.; Potashova, N.I.; Gavrishova, T.N.; Li, V.M. Photoisomerization of 2-styrylquinoline in neutral and protonated forms. High Energy Chem. 2008, 42, 446–453. [Google Scholar] [CrossRef]
- Nosova, E.V.; Trashakhova, T.V.; Ustyugov, V.S.; Mochul’skaya, N.N.; Valova, M.S.; Lipunova, G.N.; Charushin, V.N. Fluorine containing quinoline and quinoxaline styryl derivatives: Synthesis and photophysical properties. Russ. Chem. Bullet. 2011, 60, 942–947. [Google Scholar] [CrossRef]
- Dorranian, D.; Abedini, Z.; Hojabri, A.; Ghoranneviss, M. Structural and optical characterization of PMMA surface treated in low power nitrogen and oxygen RF plasmas. J. Non-Oxide Glasses 2009, 1, 217–229. [Google Scholar]
- Derkowska-Zielinska, B.; Kaczmarek-Kedziera, A.; Sypniewska, M.; Chomicki, D.; Szczesny, R.; Skowronski, L.; Figà, V.; Krupka, O. Distance Effects of Phenylpiperazine-Containing Methacrylic Polymers on Optical and Structural Properties. J. Phys. Chem. B 2021, 125, 10629–10638. [Google Scholar] [CrossRef] [PubMed]
- Boto, R.A.; Peccati, F.; Laplaza, R.; Quan, C.; Carbone, A.; Piquemal, J.-P.; Maday, Y.; Contreras-Garcia, J. NCIPLOT4: A New Step towards a Fast Quantification of Noncovalent Interactions. Available online: https://github.com/juliacontrerasgarcia/nciplot (accessed on 16 August 2022).
- Johnson, E.R.; Keinan, S.; Mori-Sánchez, P.; Contreras-García, J.; Cohen, A.J.; Yang, W. Revealing noncovalent interactions. J. Am. Chem. Soc. 2010, 132, 6498–6506. [Google Scholar] [CrossRef] [PubMed]
- Contreras-García, J.; Johnson, E.R.; Keinan, S.; Chaudret, R.; Piquemal, J.P.; Beratan, D.N.; Yang, W. NCIPLOT: A program for plotting noncovalent interaction regions. J. Chem. Theory Comput. 2011, 7, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Le Bahers, T.; Adamo, C.; Ciofini, I. A Qualitative Index of Spatial Extent in Charge-Transfer Excitations. J. Chem. Theory Comput. 2011, 7, 2498–2506. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision B.01; Gaussian, Inc.: Wallingford, CT, USA, 2016.
- Sayyah, S.M.; Khaliel, A.B.; Abd El-Salam, H.M.; Younis, M.A. Infrared Spectroscopic Studies on Some Thermally Degraded Poly(methyl methacrylate) Doped with N,N,N′,N′- tetraoxaloyl Para Sulphanilamide. Egypt. J. Chem. 2012, 55, 603–623. [Google Scholar]
- Saavedra, L.N.; Penido, R.G.; Santos, L.A.; Ramalho, T.C.; Lobo Baeta, B.E.; Pereira, M.C.; da Silva, A.C. Molecularly imprinted polymers for selective adsorption of quinoline: Theoretical and experimental studies. RSC Adv. 2018, 8, 28775–28786. [Google Scholar] [CrossRef]
- Hemalatha, C. FT-Raman, FTIR Spectra and Normal Co-Ordinate Analysis of Isoquinoline. Int. J. Phys. Appl. 2011, 3, 83–90. [Google Scholar]
- Molard, Y.; Dorson, F.; Brylev, K.A.; Shestopalov, M.A.; Gal, Y.L.; Cordier, S.; Mironov, Y.V.; Kitamura, N.; Perrin, C. Red-NIR Luminescent Hybrid Poly(methyl methacrylate) Containing Covalently Linked Octahedral Rhenium Metallic Clusters. Chem. Eur. J. 2010, 16, 5613–5619. [Google Scholar] [CrossRef]
- Lipunova, G.N.; Nosova, E.V.; Trashakhova, T.V.; Charushin, V.N. Azinylarylethenes: Synthesis and photophysical and photochemical properties. Russ. Chem. Rev. 2011, 80, 1115. [Google Scholar] [CrossRef]
- Li, L. Tunable Memristic Characteristics Based on Graphene Oxide Charge-Trap Memory. Micromachines 2019, 10, 151. [Google Scholar] [CrossRef]
- Singh, V.D.; Dwivedi, B.K.; Paitandi, R.P.; Kumar, Y.; Pandey, D.S. Effect of substituents on photophysical and aggregation behaviour in quinoline based bis-terpyridine Zn(II) complexes. Inorg. Chim. Acta 2019, 487, 24–30. [Google Scholar] [CrossRef]
- Shiraishi, Y.; Ichimura, C.; Sumiya, S.; Hirai, T. Multicolor Fluorescence of a Styrylquinoline Dye Tuned by Metal Cations. Chem. Eur. J. 2011, 17, 8324–8332. [Google Scholar] [CrossRef] [PubMed]
- Correia, F.C.; Santos, T.C.F.; Garcia, J.R.; Peres, L.O.; Wang, S.H. Synthesis and Characterization of a New Semiconductor Oligomer Having Quinoline and Fluorene Units. J. Braz. Chem. Soc. 2015, 26, 84–91. [Google Scholar] [CrossRef] [Green Version]








| Polymer | R1 | Hammett σ Constant | Monomers Mole Ratio | n/m a | Yield % | Mw b g/mol | Mn b g/mol | Mw/Mn b | Tg c |
|---|---|---|---|---|---|---|---|---|---|
| COP1 | OCH3 | −0.268 donor | 1:3 | 1:2.9 | 80 | 10,500 | 6000 | 1.75 | 136.82 |
| COP2 | Cl | 0.227 acceptor | 1:3 | 1:2.7 | 65 | 6000 | 4100 | 1.45 | 120.08 |
| COP3 | CF3 | 0.540 acceptor | 1:3 | 1:2.8 | 73 | 9800 | 5500 | 1.78 | 139.68 |
| Ground State S0 | Excited State S1 | Excited State 4 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Dipole Moment μ | Dipole Moment μ | Δμ (S1-S0) | D [Å] | Q [a.u.] | Dipole Moment μ | Δμ (S4-S0) | D [Å] | Q [a.u.] | ||
| A0 | E | 3.4702 | 3.1978 | −0.2724 | 0.262 | 0.347 | 3.9490 | 0.4788 | 0.844 | 0.360 |
| Z | 3.8096 | 1.9713 | −1.8383 | 0.660 | 0.581 | 3.2343 | −0.5753 | 0.863 | 0.383 | |
| A1 | E | 1.8144 | 3.0786 | 1.2642 | 1.684 | 0.412 | 2.7227 | 0.9083 | 1.102 | 0.374 |
| Z | 2.6324 | 3.4264 | 0.7940 | 1.985 | 0.500 | 2.6414 | 0.0090 | 0.870 | 0.373 | |
| A2 | E | 4.8433 | 4.5960 | −0.2473 | 0.148 | 0.352 | 4.6476 | −0.1957 | 0.843 | 0.365 |
| Z | 5.5503 | 3.6939 | −1.8564 | 0.691 | 0.563 | 4.8970 | −0.6533 | 0.777 | 0.380 | |
| A3 | E | 6.0855 | 8.1472 | 2.0617 | 1.315 | 0.361 | 6.2343 | 0.1488 | 0.619 | 0.361 |
| Z | 6.6407 | 5.8906 | −0.7501 | 0.315 | 0.580 | 7.1717 | 0.5310 | 0.672 | 0.359 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sypniewska, M.; Kaczmarek-Kędziera, A.; Apostoluk, A.; Smokal, V.; Krupka, A.; Szczesny, R.; Derkowska-Zielinska, B. Spectroscopic Studies of Styrylquinoline Copolymers with Different Substituents. Polymers 2022, 14, 4040. https://doi.org/10.3390/polym14194040
Sypniewska M, Kaczmarek-Kędziera A, Apostoluk A, Smokal V, Krupka A, Szczesny R, Derkowska-Zielinska B. Spectroscopic Studies of Styrylquinoline Copolymers with Different Substituents. Polymers. 2022; 14(19):4040. https://doi.org/10.3390/polym14194040
Chicago/Turabian StyleSypniewska, Malgorzata, Anna Kaczmarek-Kędziera, Alexandra Apostoluk, Vitaliy Smokal, Anastasiia Krupka, Robert Szczesny, and Beata Derkowska-Zielinska. 2022. "Spectroscopic Studies of Styrylquinoline Copolymers with Different Substituents" Polymers 14, no. 19: 4040. https://doi.org/10.3390/polym14194040