1. Introduction
Hydrothermal synthesis of SiO
2 nanoparticles is the new technological method with a low production cost, high density of silanol groups, low toxicity, excluding the use of chemical reagents and other green chemistry methods of nanoparticle synthesis [
1,
2,
3], and allows to obtain different forms of nanosilica—sols, gels, powders, products of the sol-gel process applied in concrete, agricultural plants, medicines, and other materials [
4,
5,
6,
7]. The technology of hydrothermal synthesis includes the stages of OSA polymerization, ultrafiltration membrane concentration of nanoparticles, the sol-gel process, vacuum sublimation, and others [
4,
5,
6]. The temperature regime, the stage of polymerization, pH, initial OSA concentration C
s, and ionic strength influenced the rate of SiO
2 nanoparticle formation, the average nanoparticle size, and the index of polydispersity of nanoparticles in hydrothermal sols. Therefore, the density, morphology, agglomerate dimensions and fractal structures, volume, surface, diameters, and structure of pores of nanopowders produced from hydrothermal sols are also affected [
4,
5]. There is a need to regulate the parameters at the stage of OSA polymerization of silicic acid by means of numerical simulation to achieve the necessary characteristics of sols, gels, and nanopowders.
The kinetics of OSA polymerization with different mechanisms has been studied by many researchers under various conditions: pH, initial concentration of dissolved silica Cs, solution temperature, ionic strength, and sizes and molecular weight of SiO2 particles formed.
At sufficiently high concentrations of silica in 2–6 wt.% sodium silicate salt solutions with the ratio SiO
2:Na
2O = 3.25 and with acidification at pH = 1.7, when the increase in molecular weight was accompanied mainly by the aggregation of small particles the degree of polymerization increased proportionally to the square root of the polymerization time [
8].
At the monomer concentration of 0.24 % in the absence of salts [
9,
10], the average molecular weight at pH = 2.0 increased linearly depending on the square root of time; at pH = 3.2, 3.8, it increased proportionally with time; at pH = 4.36—linearly from the square of time. At pH = 2.2, temperature 90 °C, the average particle diameter was 1.3 nm. At a higher silica concentration of 8 g/L after about 150 h at pH = 3.0 or 75 h at pH = 2.0 the average molecular weight reached 300 SiO
2 with a smaller resulting particle size of 0.9 nm [
11]. The average molecular weight of the newly formed polymerized acid prepared by removing Na from a solution of sodium silicate at pH= 2–3 was about 660 SiO
2 atoms [
12]. At pH = 2.1, temperature 25 °C, in 0.24 wt.% SiO
2 solution after 60 days after the start of polymerization, the proportion of monomeric silica was 14% and the particle diameter was 1.4 nm [
13]. At pH = 8.8, growth of particles occurred during 1 min, that is 3∙10
4 times faster than at pH = 2.0. In studies performed at 25 °C, pH = 2.0, in the solution of monosilicic acid 2.4% SiO
2 for 24 days the particle diameter was 1.65 nm; after 8 days it was about 3.4 nm [
14,
15,
16]. In a wide range of SiO
2 contents at pH =3.0 and pH = 6.1, the initial average diameter of the nuclei was 1.5 nm and the final particle size was about 3 nm [
17]. In [
18], after 30 days of polymerization at pH = 7.0 and 25 °C with a total concentration of SiO
2 in a solution of 0.4 g/L the diameter of nanoparticles corresponding to the solubility was 3.7 nm.
In the pH = 7.0–10.0 region, when colloid particles are formed, but the aggregation process cannot proceed, the reaction order was 3 [
13,
19]. However, in [
20], at different initial concentrations of OSA the apparent “order” of the polymerization reaction was determined as equal to 2 at a pH above 2 and was equal to 3 at a pH below 2 [
20], whereas the second-order reaction rate constant decreased.
It was found that the rate of polymerization was the greatest at pH = 8.3 and close to the maximum in the pH = 7.0–9.0 region, and at pH < 7.0 and pH > 9.0 it noticeably decreased [
21].
The detailed study of polymerization in the wide area of concentrations of 0.2–1.8 g/L of silica and pH = 4.0–10.0 at 25 °C confirmed that there is an induction period during which the polymerization of the monomer is only slight or nonexistent. A decrease in the monomer content as it polymerizes at pH = 7.0 and 25 °C at an initial content of 0.05 wt. % occurred according to the law 0.37∙(t − 0.35)
−0.333 [
22]. The induction period was 0.35 h. After 1 h after the decrease in the monomer content, silica particles began to grow rapidly without the formation of any intermediate product. The presence of a long induction period at pH = 4.5–5.5 and a temperature of 95 °C is shown in [
23]. The induction period characterizes the metastability of the system and for aqueous silicic acid at pH = 7.75, t = 20 °C, C
s = 1450 mg/kg under mixing conditions it decreased from 3.25 to 2.75 h [
20,
21]. In the strongly acidic region, the rate of the process is proportional to [H+]
1.2 at pH < 1.8, and in the weakly acidic region at pH > 3.4 –[OH-]
0.9 [
24,
25]. The rate increases initially with an increase in temperature from 20 to 90 °C and decreases with a decrease in the initial OSA concentration [
24,
25]. The results obtained in [
26] for OSA solutions with a SiO
2 content of 300–1200 mg/kg and low salinity in the temperature range from 5 to 180 °C confirmed that the temperature changes at a constant silica concentration do not significantly affect the increase in induction time at temperatures from 90 to 180 °C.
In a dimer–monomer mixture at pH = 2.0 [
27] the polymerization mechanism and particle sizes differed from the results of monosilicic acid polymerization obtained for polycyclic polymer formation in [
14,
15,
16]: at the beginning the discrete particles of less than 4 nm in size formed, then such particles aggregated [
27].
Thus, the experimental data obtained by the authors showed significant differences in the values of the polymerization reaction order, the rate constant, the induction period, the sizes of the formed nanoparticles, and the different effects of temperature, pH, and ionic strength on these parameters. Taking this into account, it is justified to use the method of numerical simulation of polymerization based on a certain mathematical model.
One of the mathematical models of orthosilicic acid polymerization is Fleming’s model [
28]. Fleming suggested that the process under study is characterized by two main areas of flow. In the first region, the initial concentration of silicic acid exceeds the pseudo-equilibrium concentration of Cx, and the polycondensation reaction is a first-order reaction both with respect to the difference (C
s-C
x) and with respect to the surface concentration of C
SiO-ionized groups of SiO
−, that is, the surface charge of colloid particles. The second area is defined by the condition C
s < C
x.
The decrease in the rate of transition of monomeric silica to colloid is explained using the Weres-Yi-Tsao model [
29]. The authors studied two processes: the formation of colloid particles at the homogeneous stage of nucleation and the subsequent growth of particles due to OSA molecular deposition. From this model, developed on the basis of classical concepts of nucleation by Lose-Pound, it follows that the supersaturation of S
N (T), equal to C
s/C
e, C
e (solubility of amorphous silica in water at temperature T), and pH are the main factors determining the rate of nucleation I
N of silicic acid in an aqueous solution.
The authors of [
30] used two methods to study the OSA polymerization, transfer in nanocolloids, and nanosilica precipitation in synthetic solutions based on the Na
2SiO
3 precursor: (1) the concentration model, when the rate of monomeric silica concentration C
s decrease is proportional to the 4-th order of k
1∙C
s4 and the rate of nanosilica transfer to precipitated silica is proportional to the 1-st order of k
2∙C
nano; and (2) the supersaturated model, when the rate of monomeric silica concentration C
s decrease is proportional to the 4-th order of the difference k
1∙(C
s–C
e)
4, C
e—solubility of amorphous silica in the water at 25 °C. In the wide range of initial C
s 240–1200 mg/kg, ionic strength 0.01–0.24 mol/kg, and pH = 3–7 it was obtained that constants k
1 and k
2 did not differ for concentration and supersaturation models. The models cannot predict the sizes and distribution of SiO
2 particles.
The method of molecular dynamic simulation for OSA polymerization was developed in [
31,
32] at the temperature up 2700 K and can provide the structure of polymerized clusters which was in accordance with the
29Si NMR method.
The models with different orders of polymerization reaction [
8], taking into account the concentration of OH
− groups [
33] and ionic strength [
28] have been used for analysis of experimental results of orthosilicic acid polymerization in hydrothermal solutions [
34].
In [
35], the model of Weres-Yi-Tsao [
29] has been applied to experimental results in hydrothermal solutions because it can predict not only time dependence of the OSA concentration but also evaluate how the average size of SiO
2 nanoparticles depends on time. The main purpose of this work was to investigate nanoparticle size distribution dependence on the temperature, ionic strength, and other parameters of the polymerization process needed to regulate the characteristics of nanosilica sol, gel, and powders produced using the technology of hydrothermal synthesis [
4,
5,
6,
7]. For this purpose, it is necessary: (1) to study the dependence of surface tension σ
sw, rate of molecular deposition R
md, and Zeldovich factor Z on the temperature and pH to explain the mechanisms of their influence on the results of the polymerization process; (2) to examine the dependence of the average nanoparticle size and induction period on temperature, pH, and ionic strength; (3) to study the nanoparticle size distribution under conditions of time variable temperatures; (4) to verify the results of the numerical simulation with the experimental data in wide range of T, pH, and I
s; and (5) to compare the results of the numerical simulation under time-variable temperature conditions at different technological stages of the hydrothermal sol production with the results of determining the average size of SiO
2 nanoparticles and their size distribution using the DLS method.
2. Method of Numerical Simulation of OSA Polymerization
The initial concentration of molecules of orthosilicic acid formed due to dissolution of minerals of rocks [
4,
5,
6,
7] is determined using quartz solubility C
cr (mol/kg) of α-quartz in water at the temperature T(K) [
36]:
After the hydrothermal solution reaches the surface pressure and the temperature decreases, the supersaturation of the solution depends on solubility C
e of amorphous silica [
37]:
Polymerization of orthosilicic molecules in oversaturated solution is provided by the formation of siloxane bonds and dehydration:
The total silica concentration C
t in the solution is equal to the sum of the concentrations of colloid silica C
col, soluble silicic acid C
s, and ions of silicic acids C
in:
The fraction of dimers does not exceed 1.0% from C
e, the fraction of trimers is no more than 0.1% [
26], the fraction of tetramers and low-molecular cyclic polymers is less than 0.1%, and the fraction of C
in does not exceed 14.0%.
The physical and chemical characteristics of the hydrothermal solution from the Mutnovsky geothermal electric plant are presented in the
Table 1.
We developed the mathematical model [
29] which is based on the calculation of the rate of nucleation I
N (nucl/(kg∙s)) dependent on oversaturation S
N (T) = C
s/C
e:
where ΔF
cr is the change in free energy due to the formation of a nucleus of critical radius R
cr with a surface area of A
cr = 4·π·R
cr2 and the number of SiO
2 molecules n
cr, σ
sw is the surface tension at the silica–water interface, R
md is the rate of molecular deposition of SiO
2 on a solid surface, kg/(m
2∙s), k
B = 1.38∙10
−23 J/K, M
Si = 0.060 kg/mol, N
A =6.02∙10
23 mol
−1, Q
LP is the Lothe–Pound factor, Q
LP = 3.34∙10
25 kg
−1, and Z is the Zeldovich factor, calculated as follows:
with ρ = 2200 kg/m
3—the density of amorphous silica.
The function R
md, which is the rate of molecular deposition of SiO
2 as a function of temperature and pH of the solution, is expressed as follows in the model [
29]:
The coefficient of surface tension σ
sw depends on temperature and pH via the function I(pH, pH
nom):
where S
a = (1 − α
i)·S
N, α
i is a fraction of silicic acid in ionic form, pH
nom = pH + lg([Na
+]/0.069), [Na
+] is an ion activity [Na
+], mol/kg, pK
i = 6.4, f(7.0) = 0.119, h
f = 0.45, H
σ and S
σ are specific enthalpy and entropy of silica surface in water, H
σ = 63.68·10
−3 J/m
2, S
σ = 0.049·10
−3 J/m
2·K, and n
o = 6.84∙10
18 m
−2 [
29].
Time dependence I
N (t) was calculated considering the induction time τ
in that is necessary to grow and form a stable population of the particles with sizes close to critical [
29]:
Initial values were entered: the temperature T, pH, concentration of ions and ionic strength of the solution, total silica Ct content, time step DTp, initial radii R(I), and particle quantity MP(I) of every class “I”. The Runge–Cutt method was used for the numerical simulation.
The total content of colloid, monomeric silica, and the current value of supersaturation were estimated:
Solution degree of supersaturation S
N was:
Then, the calculation of values of σsw, Rmd, Rcr, Z, τin, and IN(t) corresponding to the current values of SN, pH, and T using the equations was undertaken.
The quantity of new particles appearing during the time DT
p in accordance with the current value of nucleation rate I
N on a given program step (N+1) was calculated and it was equal to the quantity of particles in the new class N+1:
The summary of the concentration of particles CONP was calculated in a program cycle:
The increment in mass DPM of every class “I” during the time DT
p was calculated:
The calculation of particle radius R(I) corresponding to a new value of particle mass in every class I was:
The mean values of R
a, R
2a, and R
3a in all classes of particles were estimated using the program cycle:
4. Simulation the Particle Size Distribution in Hydrothermal Sol Production Technology
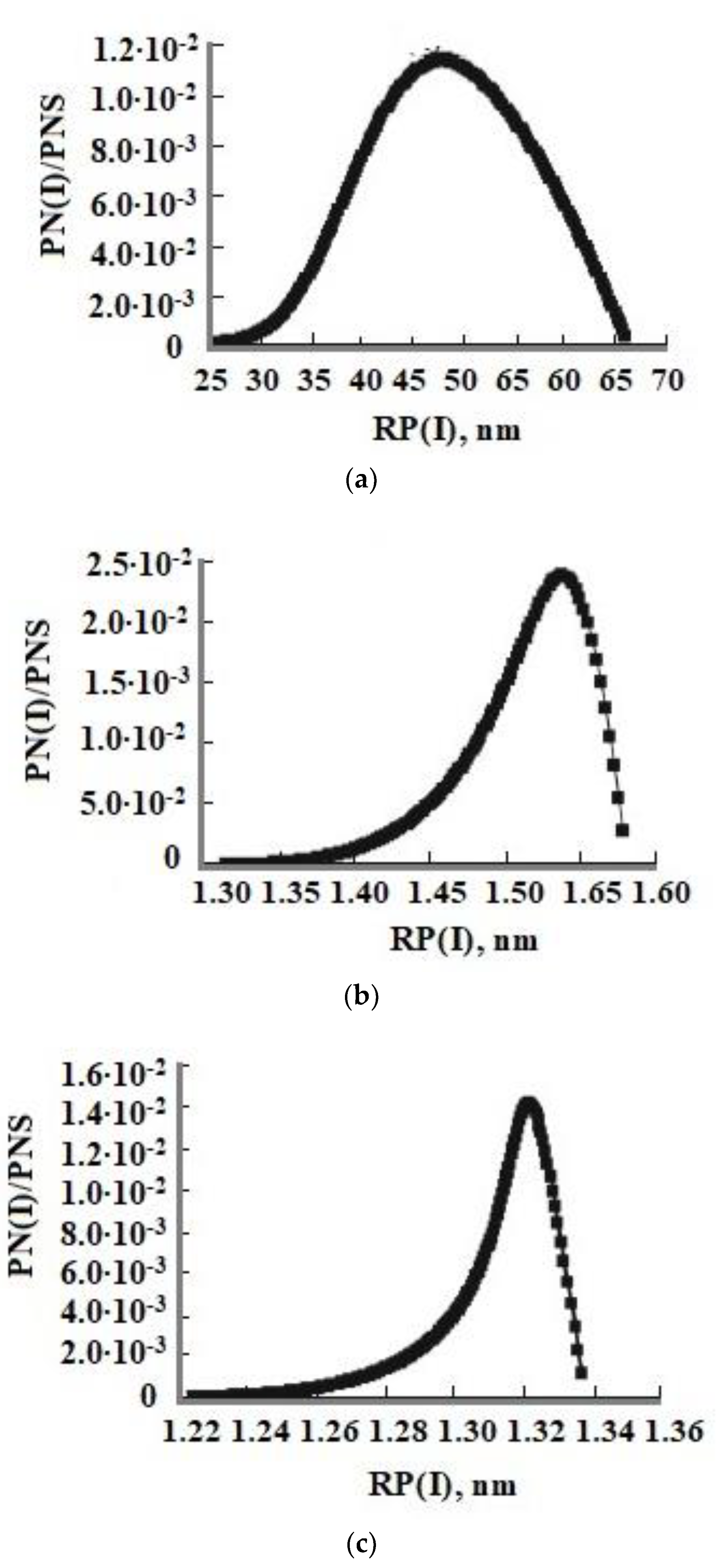
The technological flow sheet for the solution of the Mutnovsky geothermal electric power plant GeoPP includes the well, separator, pipe silencer, tank for the solution cooling and aging to develop OSA polymerization, ultrafiltration membrane module, and volumes for concentrated sol,
Figure 12a. The temperature decreases with the linear dependence in every element from the well to silencer from 300 to 96 °C at pH = 8.0 and with the dependence (1.33) in the aging tank in which the temperature decreases from 96 to 70 °C. When the duration of aging was 20 h and taun = 11 h, the average SiO
2 nanoparticle radius calculated using the numerical simulation was 12.5 nm (
Figure 12b) and it was 12.9 nm measured using the DLS method (
Figure 13). When the duration of aging was 100 h and taun = 15 h, the final average diameters were 68.6 and 66 nm using the DLS method. If pH = 5.0, the temperature decrease during aging of 24 h was from 96 to 23 °C, taun = 12 h, and the calculated diameter was 160 nm and the diameter measured using the DLS method was 154 nm.
The temperature and aging duration can be varied to regulate the average particle diameter from 5 to 160 nm.
For the solution from wells at Cerro Prieto GeoPP with the values C
s = 950 mg/kg, pH = 7.3, I
s = 0.4227 mol/kg, aging duration 65 min, taun = 60 min, and temperature decrease from 100 to 30 °C [
23,
24], the final average radius calculated in the present work was R
A = 1.32 nm and it was 1.35 nm as a result of the calculation in [
29].
For the solutions of the Wairakei GeoPP, New Zealand, at aging temperatures of 70 and 20 °C during 50 h, Cs = 490 mg/kg, and Is = 0.05 mol/kg, the calculated average radii were 60.7 and 9.5 nm, and the radii measured using DLS were 60 and 10 nm.
5. Conclusions
The mathematical model we adopted allowed us to investigate the influence of various physical and chemical factors in a wide range of values on the rate of polymerization of OSA in a hydrothermal solution and on the SiO2 nanoparticle size distribution. The results of the numerical simulation were verified using a comparison with experimental data. The temperature, pH, initial OSA concentration, and ionic strength are the main parameters that determine the kinetics of colloid phase formation, the final average size of SiO2 nanoparticles, and the form of the particle size distribution.
The final average radius decreases when the temperature decreases, and the degree of oversaturation becomes higher taking into account the surface tension, rate of molecular deposition, and Z-factor. The average radius increases when the pH decreases due to the increase in the surface tension and the critical size of the nucleus. The average radius decreases when the initial OSA concentration and degree of oversaturation increases.
The function of oversaturation SN depending on the time of polymerization Tp showed the long period of induction τin and homogeneous nucleation which becomes much longer at low values of pH. The period of homogeneous nucleation increases when pH decreases and the initial degree of oversaturation SN decreases at higher temperatures and low initial Cs concentrations. The relation of homogeneous and heterogenous durations (thom/thetg) increases when the temperature increases and pH decreases.
The dependence of the curve Cs vs. Tp and final average particle size Ra on the ionic strength Is is non-monotonic. With an increase in Is from 0.014 to 0.8 mol/kg, the kinetics of OSA polymerization accelerate, but Ra increases. Starting from the value of Is = 0.8 mol/kg, the final average particle size decreases. This non-monotonic dependence is explained by the fact that with an increase in Is, the rate of molecular deposition of Rmd increases due to an increase in the degree of ionization of orthosilicic acid αSIL. Therefore, the rate IN of OSA nucleation increases. At the same time, the growth rate of the formed particles increases. Thus, the final average size of silica particles is influenced by two competing factors—the increases in IN and Rmd which determine the non-monotonic behavior.
At variable temperatures, the supersaturation functions of time have a non-uniform behavior. The supersaturation of the solution SN = Cs/Ce depends on the solubility of Ce, which decreases, and faster than the value of the concentration of Cs. Both branches of the silica particle size distribution are concave in contrast to the case of constant temperatures. The variation in temperatures during the stage of OSA polymerization can influence the index of polydispersity of particles, its distribution regarding sizes, and the homogeneity and symmetrical form of the distribution. The temperature regime at the stage of polymerization highly influenced the index of polydispersity of hydrothermal sols and, thus, the density, morphology, agglomerate dimensions and fractal structures, volume, surface, diameters, and structures of pores of nanopowders produced from hydrothermal sols.
The results obtained can be used for regulating the kinetics of SiO2 nanoparticle formations, sizes, the polydispersity of the distribution of sizes, and the concentrations of nanoparticles in the technology of the hydrothermal synthesis of sol, gel, and powder.
The results can be applied to the technology of the production of synthetic water sols based on Na2SiO3 precursors and the technology of precipitated silica.