4.3. Effect of pH and Surface Charges on Fluoride Sorption
The effect of initial solution pH (3–12) as it affects the surface charges of the CNF-AgMgOnHaP adsorbent in the removal of fluoride is shown in
Figure 8a,b. It is clear from the plot (
Figure 8a) that fluoride sorption capacity by the adsorbent was pH dependent and the maximum sorption percentage was observed at pH 5. Thereafter, the rate of removal dropped from 87% at pH 7 to 82% at pH 9, with significant adsorption reduction after pH 9, to pH 12 at 54%. This variation might be attributed to the changes within the solution pH environment which ultimately changes the surface charge and the protonation/deprotonation of the binding functional groups across the CNF-AgMgOnHaP adsorbent, as well as the solubility of fluoride ion species during the process [
58,
59]. The surface charges distributions of the CNF-AgMgOnHaP adsorbent in fluoride sorption at different initial solution pH values (3–12) were also evaluated by its point of zero charge (pHpzc). The pHpzc is defined as that pH value at which the net surface charge density of an adsorbent is zero [
60]. As shown in
Figure 8b, the pHpzc value of the CNF-AgMgOnHaP was at ~4.7, which is lower than the optimum pH value of 5. Consequently, the surface of the sorbent becomes negatively charged in the solutions at which the equilibrium pH was greater than pHpzc.
This means that there are abundant negative charges on the surface of CNF-AgMgOnHaP at pH > 4.7 which interact and replace the fluoride ions in solution. One of the main contributing surface groups for a high adsorption of F
− by the adsorbent is the availability of -OH groups in the cellulose and hydroxyapatite (recognized by FTIR in
Section 3.2.3). It is known that both F and -OH are structurally isoelectronic, having a comparable ionic radius, and can easily replace one another through ligand exchange, which ultimately favors F
− anions’ removal with consequent high sorption capacity and selectivity [
3,
61]. Besides, the hydrolyzing agent in the isolation of the stabilized cellulose nanofiber, sulfuric acid, reacts with the cellulose surface hydroxyl groups to form sulfate half-esters, which also contributed to the negatively charged surface of the adsorbent, thus, the high adsorption of fluoride by CNF-AgMgOnHaP [
62]. Therefore, the interaction between these surface groups and fluoride ions easily facilitates the high fluoride sorption capacity through an ion exchange mechanism, the formation of an ion pair, or through H bonding with the negatively charged surface. A similar pattern of surface exchange mechanisms of fluoride removal was reported elsewhere [
63,
64].
4.4. Effect of Coexisting Ions
The possible interference of coexisting ions (such as Cl
−, NO
3−, CO
32−, and SO
42−), along with fluoride for the active sorption sites in the sorbent material, was examined, and the results are presented in
Figure 9a,b.
Figure 9a illustrates the variation in F
− removal efficiency by the adsorbent with the existence of co anions in water. The result showed that the presence of anions such as Cl
−, NO
3−, and SO
42− had a slight effect on fluoride sorption by CNF-AgMgOnHaP, with NO
3− interfering the least when compared with the anion free water. However, the competitiveness of CO
32− appeared to have a significant effect in the F
− uptake for surface binding sites, leading to a reduction in % fluoride removal from ~93% to ~80%. In addition, the effect of co anion composite concentrations (10–50 mg/L), as a function of percentage fluoride removal by the CNF-AgMgOnHaP composite, is depicted in
Figure 9b. Evidently, from the plot (
Figure 9b), at higher composite anion concentrations, the potential affinity for fluoride by the sorbent reduces. This may be because of the strong hydrolysis contribution by stable CO
32− ions surface complex formation, which hinders fluoride ions being sorbed by the CNF-AgMgOnHaP composite.
4.5. Effect of Initial Concentration and Adsorption Isotherm
The rate of fluoride uptake by the CNF-AgMgOnHaP composite as a function of initial fluoride concentrations (5–100 mg/L) at different temperatures (303, 313, and 323 K) are shown in
Figure 10a,b. As depicted in
Figure 10a, the % F
− removal is found to decrease with increasing initial adsorbate concentration. This may be due to the reduction in active binding sites on the CNF-AgMgOnHaP adsorbent surface leading to its saturation by the sorbed fluoride ion. Furthermore, in contrast to
Figure 10a, the F
− sorption capacity increased gradually as the fluoride concentration increased from 5 to 100 mg/L (
Figure 10b). This suggests that, as the concentration increases, the driving force for overcoming the mass transfer resistance between the adsorbate in solution and adsorbent phases increases in the liquid–solid adsorption system [
64].
To determine the adsorption interaction and mechanism of the F
− ions on the CNF-AgMgOnHaP composite surface, the nonlinear forms of Langmuir [
65,
66] (Equations (3) and (5)), as well as the linear form of Dubinin–Radushkevich [
67] (Equations (6)–(9)) isotherm models were used to evaluate the adsorption process.
These equations were used to simply calculate the adsorption parameters because of the usefulness of their model parameters, their clarity, and easy interpretability, as well as to reduce their respective error functions [
57,
68].
The nonlinear forms of the Langmuir adsorption isotherm model [
69] assume a monolayer of F
− sorption onto the surface of CNF-AgMgOnHaP containing a finite number of uniform adsorption binding sites without the migration of adsorbed molecules on the surface at a fixed temperature, and is given as:
where
Qm (mg/g) is the maximum adsorption capacity, which assumes the complete monolayer of an adsorbent,
Ce (mg/L) is the adsorbate equilibrium concentration,
(mg/g) is the amount of F
− ion adsorbed at equilibrium, and
KL (L/mg) is the Langmuir adsorption equilibrium constant.
The fundamental characteristics of the Langmuir isotherm model can also be calculated in terms of a dimensionless parameter called the separation factor,
RL [
68] (Equation (4)), denoted as:
where
Ci (mg/L) is the initial fluoride concentration and
KL is the Langmuir equilibrium constant. The
RL value is useful in determining if a sorption process is irreversible (
RL = 0), linear (
RL = l), favorable (0 <
RL< 1), or unfavorable (
RL > l).
The Freundlich adsorption isotherm [
66], which is an empirical model describing the surface heterogeneity of the CNF-AgMgOnHaP sorbent through a multilayer adsorption system, was also applied. It is expressed below as:
where
(mg/g) is the equilibrium adsorption capacity of the CNF-AgMgOnHaP adsorbent,
Ce (mg/L) is the equilibrium concentration, and
KF [(mg/g)/(mg/L)
n] and 1/
n are empirical Freundlich constants describing sorption capacity and intensity parameter, respectively.
KF characterizes the strength of adsorption, while
n indicates the magnitude of the adsorption driving force or the surface heterogeneity [
67]. The adsorption isotherm is favorable when 0 < 1/
n < 1, unfavorable when 1/
n > 1, and irreversible when 1/
n = 1.
The equilibrium adsorption results were also fitted into the linearized form of the Dubinin Radushkevich isotherm model [
68], which accounts for the effects of the porous nature of the CNF-AgMgOnHaP adsorbent and the mean free energy (E) of the sorption process. The D-R equations are shown below.
The energy
E (kJ/mol) of F
− sorption from Dubinin–Radushkevich is
The general linearized form of the D-R is in Equation (9):
where
(mg/g) is the CNF-AgMgOnHaP adsorption capacity;
(mol
2/kJ
2) is the activity coefficient constant related to mean sorption energy;
ε is the Polanyi potential; and
E (kJ/mol) is the mean adsorption energy. The slope of the plot of ln
against
ε2 gives
and the intercept gives the adsorption capacity
of CNF-AgMgOnHaP. If the magnitude of
E is between 8 and 16 kJ/mol, the adsorption process occurred via chemisorption, while for values where
E < 8 kJ/mol, the adsorption process is physisorption.
Figure 11 and
Table 3 show the adsorption isotherms plots with the corresponding calculated parameters of the sorbed F
− on CNF-AgMgOnHaP composite at different temperatures.
As shown in
Table 3, the respective values of
Qm and
KF from the nonlinear plots decrease as the temperature increases, an indication that the adsorption of F
− by the CNF AgMgOnHaP composite is unfavorable at higher temperatures. The Langmuir constant (
KL) values increase with temperature changes, showing the existence of an electrostatic attraction between the F
− and the CNF AgMgOnHaP surface [
69]. The obtained
RL values lying between 0 and 1 also confirm the favorability of the adsorption activity across all the operating temperatures. In addition, since
n lies between 1 and 10 as shown in
Table 3, the physical adsorption of F
− onto the adsorbent is demonstrated. It is important to note that the experimental maximum adsorption capacities were observed to be similar to the calculated Langmuir maximum adsorption capacities (
Qm) for all the temperatures. In addition, the adsorption energies,
E (kJ/mol) values, obtained by the linear D-R model generally increased as the temperature increased (
Table 3). These values suggested that the physical adsorption mechanism was involved in the F
− sorption process. Consequently, based on the higher correlation coefficients (
Adj. R2) and lower Chi-square (
χ2) values comparison (
Table 3), it was found that the fluoride sorption onto the CNF-AgMgOnHaP was best described by the Freundlich isotherm model across all the operating temperatures, compared to other models. This suggested that the dominant adsorption mechanism on the adsorbent surface was through multilayer binding sites.
4.6. Adsorption Kinetic
To evaluate the mechanism and time dependence kinetic parameters of F
− sorption by the CNF-AgMgOnHaP surface, the contact time experimental data (
Figure 6) were simulated and fitted to both reaction and diffusion based kinetic models.
The Lagergren [
70] pseudo-first-order model can be used for a simple sorption process and its linear equation is stated in Equation (10):
The linearized pseudo-second-order (Equation (11)) model, which is normally used to describe cation exchange and chemisorption reaction mechanisms [
71], was also employed and is represented by Equation (11):
The linear form of the intraparticle diffusion model [
72] given in Equation (12) was also used to describe the transfer of the solute in the solid/liquid system.
where
qe and
qt are the amounts of adsorbate uptake per mass of CNF-AgMgOnHaP adsorbent (mg/g) at equilibrium and at time
t (min), respectively; with
, and
Ki (min
−1) representing the rate constant of the pseudo-first-order, pseudo-second-order, and intraparticle diffusion rate constant, respectively.
The normalized standard deviation (S.D. (%)) usually used to describe the applicability of the kinetic model in the fluoride sorption process on the adsorbent was determined using Equation (13).
where
N is the number of data points, and
qe, exp, and
qe, cal (mg/g) are the experimental and the calculated equilibrium sorption capacity, respectively.
The calculated corresponding adsorption kinetic parameters from the three different model plots for fluoride sorption are summarized in
Table 4. Based on the higher correlation coefficient (
R2) and the lowest S.D. and root mean square error (RMSE) values obtained regarding reaction based pathways, the pseudo-second-order model was the most suitable model and best describes the fluoride sorption behavior on the adsorbent surface. This confirmed that the reaction rate was rapid, as observed in
Figure 6. This is an indication that the reaction mechanism may have been controlled by chemisorption, which involved the sharing or exchange of ions between the CNF-AgMgOnHaP adsorbent and the sorbed F
−. This was supported by the XPS (
Section 4.8) and pH at various sorbate sorbent pH values (
Section 4.3). Equally, the intraparticle diffusion models parameters (
Table 4) further showed that the overall kinetic models might not only depend upon the chemical adsorption process but were also governed by an equilibrium diffusion mechanism, through a mass transfer of the adsorbate molecules from the external surface through the pores of the adsorbent [
73].
4.7. Thermodynamics
The thermodynamic parameters (
Table 5), such as standard enthalpy change (∆
H°), standard free energy (∆
G°), and standard entropy change (∆
S°), were evaluated from the experimental sorption data (303–323 K). These parameters were obtained from the plot of 490 1/T vs. ln
Kc (
Figure 12) using the following equations (Equations (14)–(17));
where
R (8.314 J mol
−1 K
−1) is the gas constant, and
T is the absolute temperature (K). The equilibrium constant values,
Kc, were obtained by plotting
qe/Ce against
Ce and extrapolating to zero [
74,
75].
The calculated thermodynamic parameters are listed in
Table 5. The positive value of Δ
H° is an indication that the fluoride sorption process stimulation occurred at a reasonably higher temperature and is, thus, endothermic [
76]. This was further displayed by the decrease in the values of
Qm and
KF, respectively, as well as the increased
Ka values from the nonlinear isotherm parameters (
Table 3) as the temperature increases. The values of ∆
G° obtained are all negative across the temperature range, which implies that the fluoride adsorption process occurred favorably and spontaneously with minimal requirements of the adsorption and activation energies [
68]. The positive ∆S° values suggest that the F
− sorption phenomenon was governed by the increasing randomness of the F
− ions at the CNF-AgMgOnHaP adsorbent–solution interface.
4.8. Surface Chemistry
Figure 13 shows the wide XPS spectral elemental analysis, whose peaks fitted according to the surface composition and chemistry of the fluoride sorbed CNF-AgMgOnHaP adsorbent. The fundamental elements of the adsorbent, as displayed by the XPS spectrum, include carbon, oxygen, calcium, phosphorus, magnesium, fluorine, and traces of silver content. As shown in
Figure 13, the C1s (287.1 eV) spectra are resolved into four gaussian peaks assigned to C-C (284.9 eV), the C-O contribution of -OH groups (286.3 eV), C=O (287.6 eV), and O-C=O (289.2 eV) bonding states [
77,
78], associated with the cellulose nanofiber layer within the composite formation. The peaks of O 1s and Mg 1s, which appeared between 531 and 535 eV (deconvoluted into three major peaks) and 1307.2 eV (single peak), respectively, are a representation of lattice O atoms bonded with carbonyl, Mg atoms, hydroxyl groups, and adsorbed water [
78]. The presence of 2p (134.0 eV) and Ca 2p (347.7 eV) were attributed to organic P and Ca-C=O interaction for calcium phosphates in the hydroxyapatite. The fewer related peaks at ~367.8 eV (two lines) corresponded to the characteristic peak of Ag NPs, confirming its existence within the composite. The appearance of an F 1s peak at 685 eV, which was resolved into two peaks at 685.5 and 689.9 eV, attributed to inorganic and organic fluoride, respectively, indicates F
− ions being bound to the CNF-AgMgOnHaP adsorbent. This was further supported by the possible Ca-F bond, as shown by the Ca 2p (347.7 eV). Thus, the XPS, EDS, FTIR, and solution pH results clearly show that the surface mechanisms of fluoride removal by the CNF-AgMgOnHaP composite could have occurred via ligand exchange and electrostatic attraction between F
− and the OH
−, Mg
2+ or Mg-OH
+ and Ca-OH
+ species at the sorbent–sorbate solution interface.
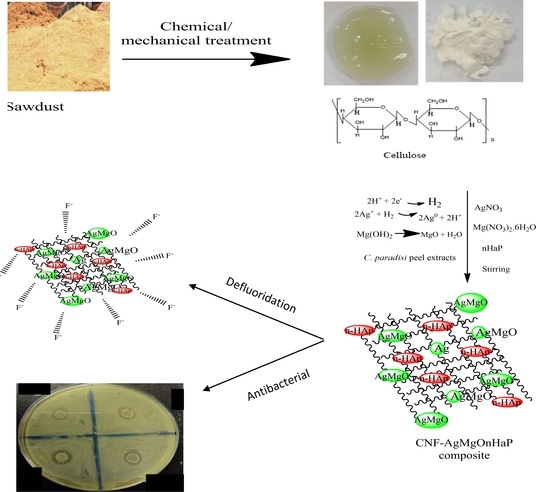
4.10. Antibacterial Activity of the CNF-AgMgOnHaP Adsorbent
Figure 15a,b displayed the observed antibacterial property of the CNF-AgMgOnHaP adsorbent through the values exhibited by the zone of inhibition against Gram-negative and Gram-positive bacterial strains. The diameter of the zone of inhibition was found to increase with an increase in the concentration of the adsorbent (Label: a, b, c, d depicting 1, 5, 7, and 10 mg/L, respectively) (
Figure 15b). The observed zone of inhibition showed that the CNF-AgMgOnHaP adsorbent possesses antibacterial activity against all the bacterial strains. However, this antibacterial property varies, with higher antimicrobial potency observed towards
E. Coli compared to
S. aureus and
K. pneumonia.
The antibacterial potency observed by this material depends on several factors, including the synergistic effects of Ag-MgO within the synthesized CNF-AgMgOnHaP composite. It has been established that MgO and Ag based materials have excellent broad spectrum, potent antimicrobial properties and are easily impregnated into cellulosic materials as a stabilizing agent [
81,
82,
83]. The antibacterial property of the CNF-AgMgOnHaP adsorbent may be due to the diffusion of Ag-MgO nanoparticles within the adsorbent through the release of metal ions disrupting the cell wall structure of the bacteria genome in producing intracellular reactive oxygen species (ROS), resulting in microbial cell death [
18,
84,
85]; the Ag-MgO nanocomposite antibacterial activity may also depend on the reducing and capping agents used in the synthesis route, as well as the size and surface properties of the composite [
38,
86,
87]. It is important to emphasize that the Ag ions release, though in low concentration, was affected by the time and rate of dissolution; metal ions release increases as time progresses. Therefore, the adsorbent presents specific antibacterial activity against all the bacteria strains.