Thermosensitive Polymers and Thermo-Responsive Liposomal Drug Delivery Systems
Abstract
:1. Introduction
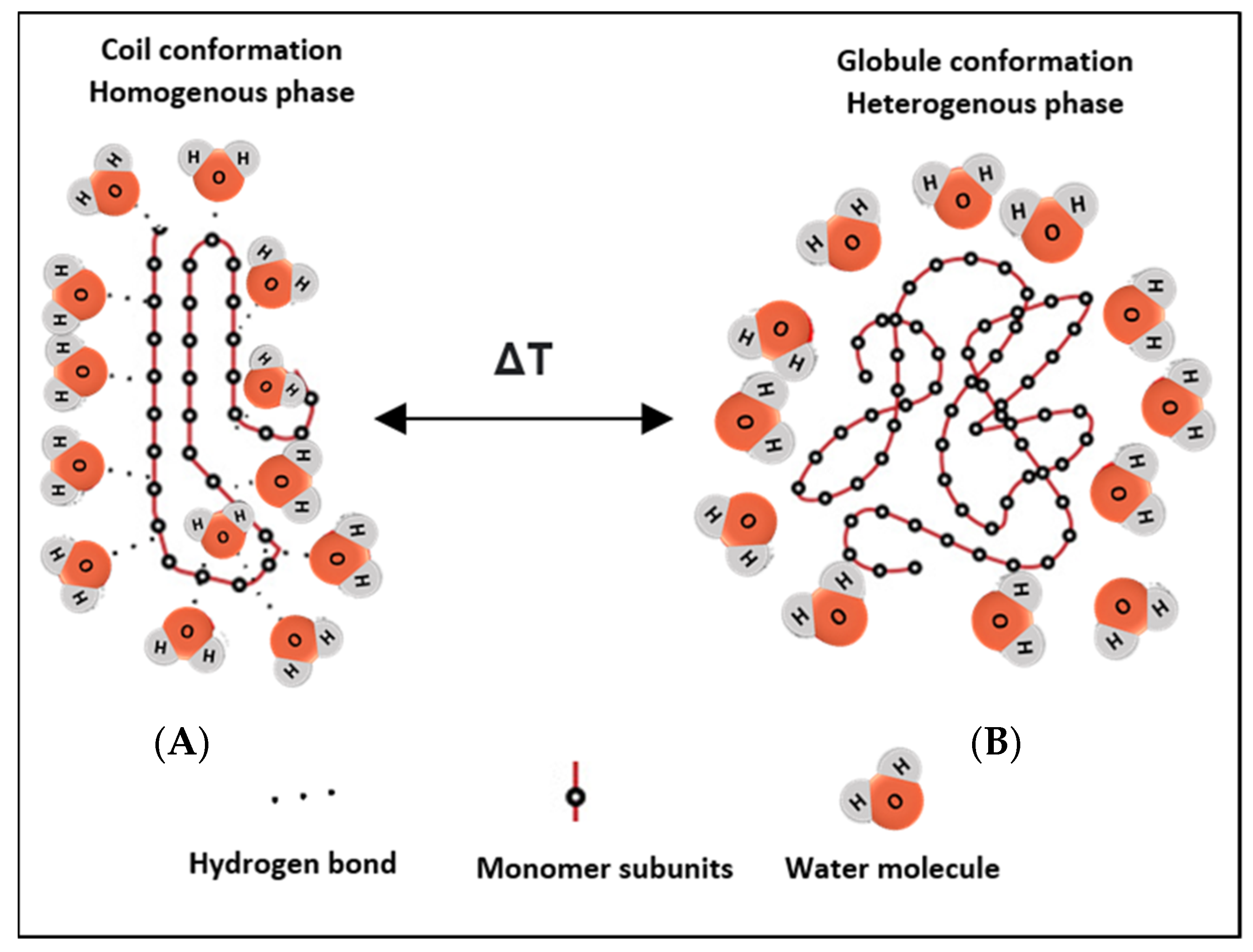
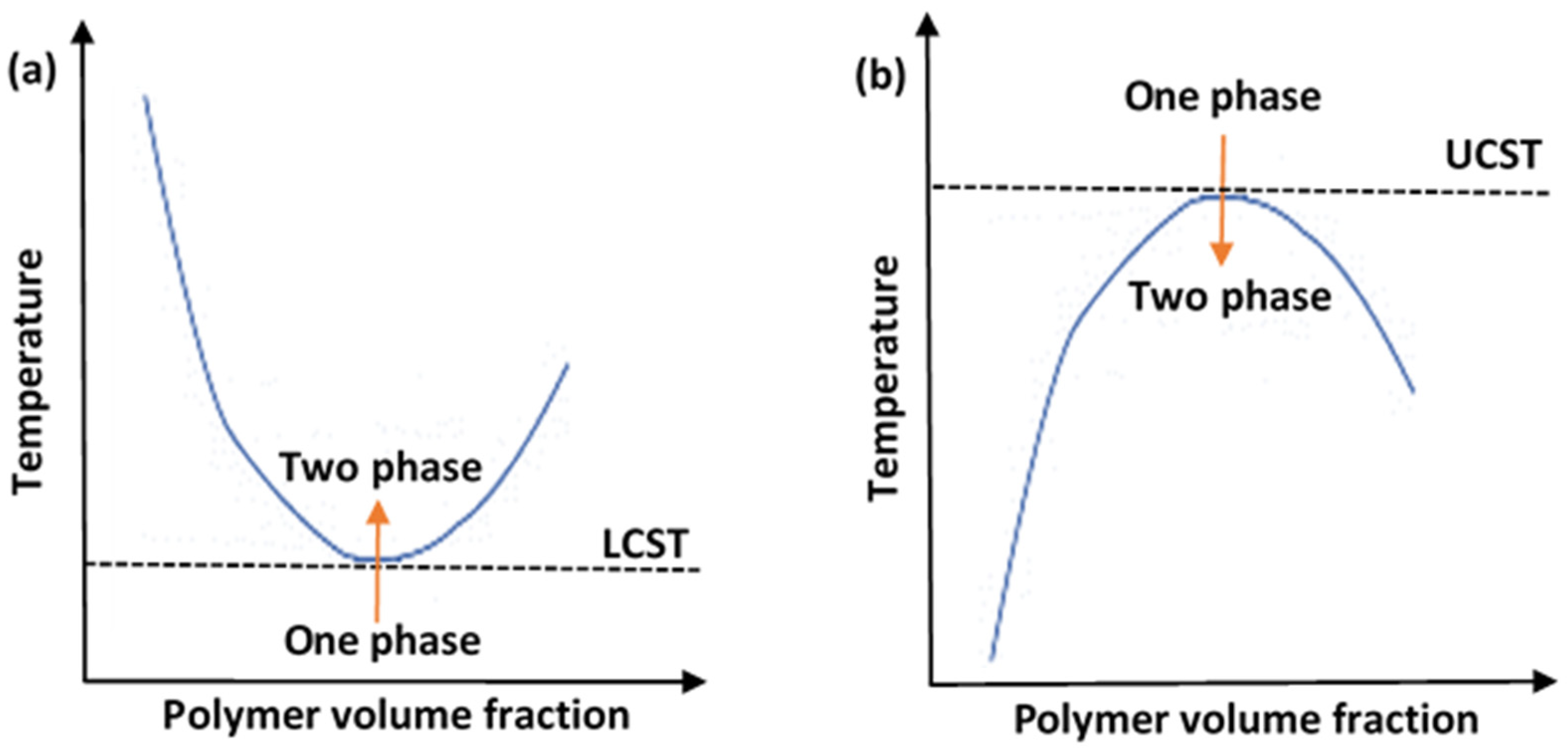
2. Thermosensitive Polymers
3. Thermoresponsive Liposomal-Based Drug Delivery Systems
3.1. Traditional Thermosensitive Liposomes (TTSL)
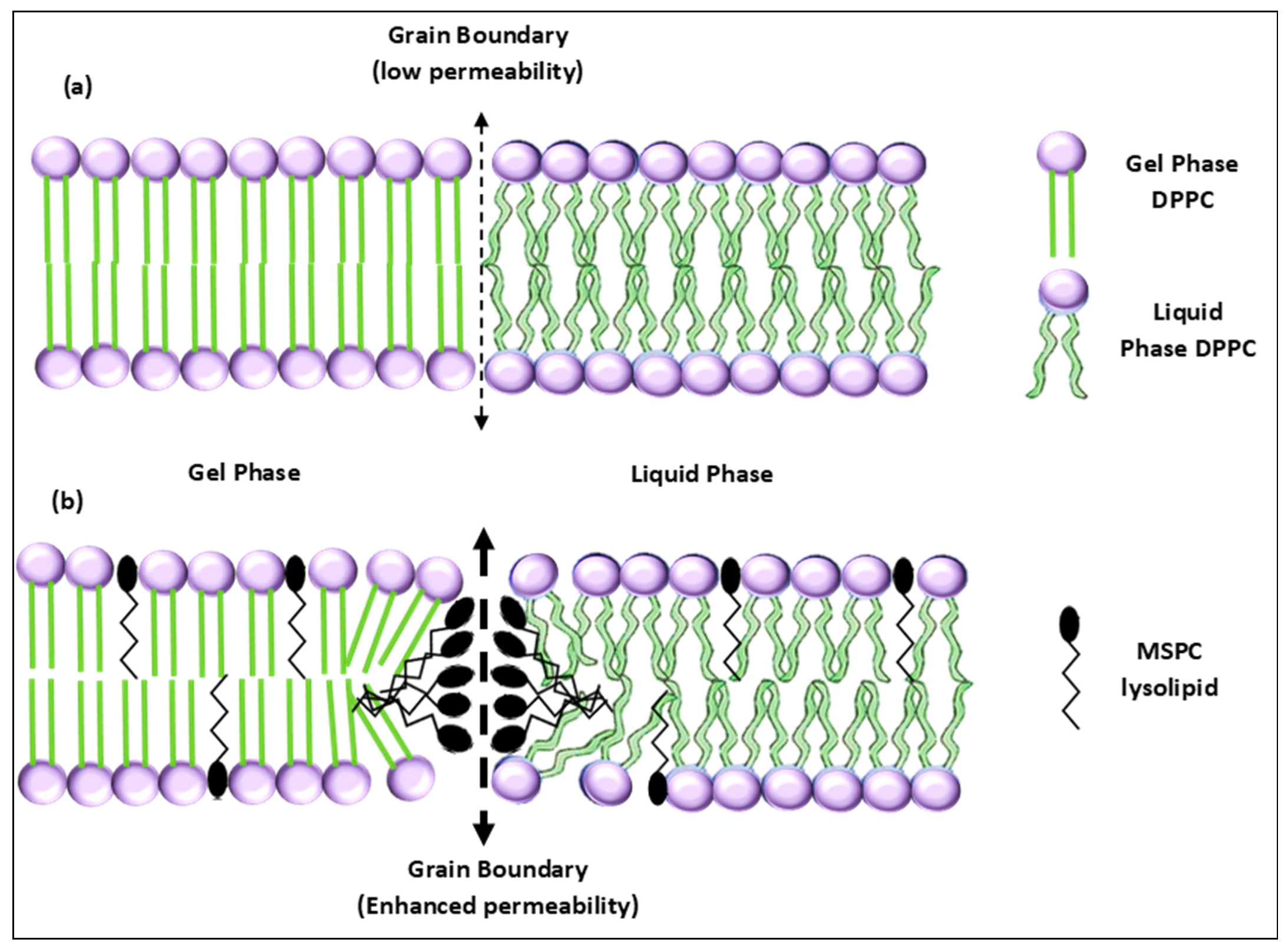
3.2. Lysolipid Thermosensitive Liposomes (LTSL)
| Study/ClinicalTrials ID | Status/Phase | Intervention | Indication | Remarks | Ref. |
|---|---|---|---|---|---|
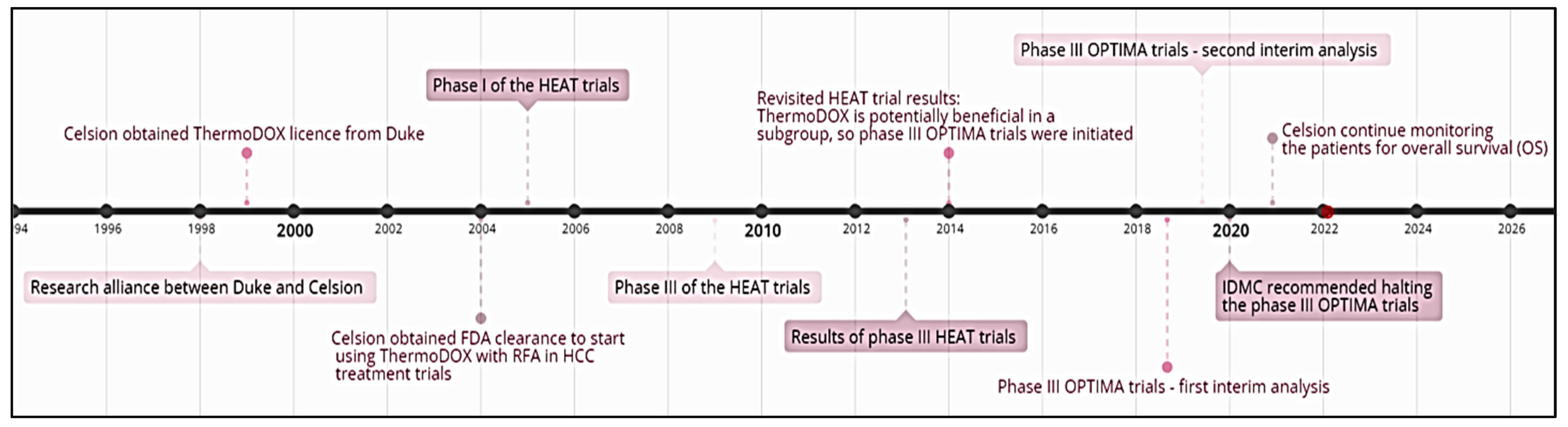
| HEAT/NCT02181075 | Completed/phase III | Lyso-thermosensitive liposomal doxorubicin (ThermoDOX) in conjunction with radiofrequency ablation (RFA) | Non-resectable hepatocellular carcinoma (HCC) | A total of 701 patients were divided into two experimental groups: 354 patients received a single ThermoDOX intravenous infusion (50 mg/m2) 15 min prior to RFA, while 347 were given a sham infusion of 5% Dextrose (placebo) 15 min before RFA. RFA was used to induce a thermal zone at the tumor site, where the entrapped doxorubicin was subsequently released from the liposome. Although the combination of ThermoDOX with RFA was safe, it did not increase the progression-free survival (PFS) and overall survival (OS) in the overall study subjects. | [80] |
| OPTIMA/NCT02112656 | Completed-phase III | ThermoDOX followed by standardized RFA | Non-resectable HCC | A total of 554 subjects enrolled in the trial; divided into an experimental group that received 50 mg/m2 doxorubicin, while the control group received a dummy infusion. RFA was initiated at least 15 min after drug administration and completed within a maximum of 3 h from administration time. RFA exposure was for a minimum of 45 min. CT scanning and MRI imaging were used to gauge the effectiveness of RFA. The second interim data analysis was unexpected, due to the consecutive death of 26 cases. The trial marginally crossed the futility preset boundary value of 0.900 by 0.003, which led to recommendations from the Independent Data Monitoring Committee (IDMC) to terminate the trial in 2020. However, the Celsion Corporation company announced that they will continue monitoring the patients for overall survival (OS). | [81,82] |
| TARDOX/NCT00617981 | Completed/phase I | ThermoDOX followed by focused ultrasound (FUS) exposure | Unresectable and non-ablatable primary or secondary liver tumors | The study was conducted in two parts, run in parallel: part I had 6 patients, while part II had 4. Optimized FUS parameters from part I were used in part II, determined based on real-time thermometry. Patients received ThermoDox® intravenously, at a dose of 50 mg/m2, followed by FUS exposure. Reported tumor biopsy results showed a 3.7-fold increase in intratumoral doxorubicin accumulation in patients treated with FUS, proving this combination treatment as safe, effective, and feasible for further clinical investigation. While no treatment-related deaths occurred, severe adverse events were registered in some patients (e.g., transient neutropenia, anemia). | [83] |
3.3. Polymer Thermosensitive Liposomes (PTSL)
4. Heat-Triggered Release Modalities
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deng, Y.; Zhang, X.; Shen, H.; He, Q.; Wu, Z.; Liao, W.; Yuan, M. Application of the Nano-Drug Delivery System in Treatment of Cardiovascular Diseases. Front. Bioeng. Biotechnol. 2020, 7, 489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mills, J.K.; Needham, D. Targeted Drug Delivery. Expert Opin. Ther. Patents 1999, 9, 1499–1513. [Google Scholar] [CrossRef]
- Sartaj, A.; Qamar, Z.; Qizilbash, F.F.; Annu; Shadab; Alhakamy, N.A.; Baboota, S.; Ali, J. Polymeric Nanoparticles: Exploring the Current Drug Development and Therapeutic Insight of Breast Cancer Treatment and Recommendations. Polymers 2021, 13, 4400. [Google Scholar] [CrossRef] [PubMed]
- Subjakova, V.; Oravczova, V.; Hianik, T. Polymer Nanoparticles and Nanomotors Modified by DNA/RNA Aptamers and Antibodies in Targeted Therapy of Cancer. Polymers 2021, 13, 341. [Google Scholar] [CrossRef]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced Targeted Therapies in Cancer: Drug Nanocarriers, the Future of Chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef] [Green Version]
- Allahou, L.W.; Madani, S.Y.; Seifalian, A. Investigating the Application of Liposomes as Drug Delivery Systems for the Diagnosis and Treatment of Cancer. Int. J. Biomater. 2021, 2021, 3041969. [Google Scholar] [CrossRef]
- Kumari, P.; Ghosh, B.; Biswas, S. Nanocarriers for Cancer-Targeted Drug Delivery. J. Drug Target. 2016, 24, 179–191. [Google Scholar] [CrossRef]
- Dreher, M.R.; Liu, W.; Michelich, C.R.; Dewhirst, M.W.; Yuan, F.; Chilkoti, A. Tumor Vascular Permeability, Accumulation, and Penetration of Macromolecular Drug Carriers. J. Natl. Cancer Inst. 2006, 98, 335–344. [Google Scholar] [CrossRef] [Green Version]
- Husseini, G.A.; Pitt, W.G. The Use of Ultrasound and Micelles in Cancer Treatment. J. Nanosci. Nanotechnol. 2008, 8, 2205–2215. [Google Scholar] [CrossRef] [Green Version]
- Maeda, H.; Nakamura, H.; Fang, J. The EPR Effect for Macromolecular Drug Delivery to Solid Tumors: Improvement of Tumor Uptake, Lowering of Systemic Toxicity, and Distinct Tumor Imaging in Vivo. Adv. Drug Deliv. Rev. 2013, 65, 71–79. [Google Scholar] [CrossRef]
- Basha, S.A.; Salkho, N.; Dalibalta, S.; Husseini, G.A. Liposomes in Active, Passive and Acoustically-Triggered Drug Delivery. Mini-Rev. Med. Chem. 2019, 19, 961–969. [Google Scholar] [CrossRef] [PubMed]
- ben Daya, S.M.; Paul, V.; Awad, N.S.; al Sawaftah, N.M.; al Sayah, M.H.; Husseini, G.A. Targeting Breast Cancer Using Hyaluronic Acid-Conjugated Liposomes Triggered with Ultrasound. J. Biomed. Nanotechnol. 2021, 17, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Elamir, A.; Ajith, S.; Sawaftah, N.A.; Abuwatfa, W.; Mukhopadhyay, D.; Paul, V.; Al-Sayah, M.H.; Awad, N.; Husseini, G.A. Ultrasound-Triggered Herceptin Liposomes for Breast Cancer Therapy. Sci. Rep. 2021, 11, 7545. [Google Scholar] [CrossRef] [PubMed]
- Awad, N.S.; Paul, V.; Alsawaftah, N.M.; ter Haar, G.; Allen, T.M.; Pitt, W.G.; Husseini, G.A. Ultrasound-Responsive Nanocarriers in Cancer Treatment: A Review. ACS Pharmacol. Transl. Sci. 2021, 4, 589–612. [Google Scholar] [CrossRef]
- Mena-Giraldo, P.; Pérez-Buitrago, S.; Londoño-Berrío, M.; Ortiz-Trujillo, I.C.; Hoyos-Palacio, L.M.; Orozco, J. Photosensitive Nanocarriers for Specific Delivery of Cargo into Cells. Sci. Rep. 2020, 10, 2110. [Google Scholar] [CrossRef] [Green Version]
- Palanikumar, L.; Al-Hosani, S.; Kalmouni, M.; Nguyen, V.P.; Ali, L.; Pasricha, R.; Barrera, F.N.; Magzoub, M. PH-Responsive High Stability Polymeric Nanoparticles for Targeted Delivery of Anticancer Therapeutics. Commun. Biol. 2020, 3, 95. [Google Scholar] [CrossRef]
- Guo, X.; Cheng, Y.; Zhao, X.; Luo, Y.; Chen, J.; Yuan, W.E. Advances in Redox-Responsive Drug Delivery Systems of Tumor Microenvironment. J. Nanobiotechnol. 2018, 16, 74. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Zhao, G.; Su, W.K.; Shuai, Q. Enzyme-Responsive Nanoparticles for Anti-Tumor Drug Delivery. Front. Chem. 2020, 8, 647. [Google Scholar] [CrossRef]
- Sánchez-Moreno, P.; de Vicente, J.; Nardecchia, S.; Marchal, J.A.; Boulaiz, H. Thermo-Sensitive Nanomaterials: Recent Advance in Synthesis and Biomedical Applications. Nanomaterials 2018, 8, 935. [Google Scholar] [CrossRef] [Green Version]
- Roussakow, S. The History of Hyperthermia Rise and Decline. Conf. Pap. Med. 2013, 2013, 428027. [Google Scholar] [CrossRef]
- Jansen, W.; Haveman, J. Histopathological Changes in the Skin and Subcutaneous Tissues of Mouse Legs after Treatment with Hyperthermia. Pathol.-Res. Pract. 1990, 186, 247–253. [Google Scholar] [CrossRef]
- Borrelli, M.J.; Thompson, L.L.; Cain, C.A.; Dewey, W.C. Time-Temperature Analysis of Cell Killing of BHK Cells Heated at Temperatures in the Range of 43.5 °C to 57.0 °C. Int. J. Radiat. Oncol. Biol. Phys. 1990, 19, 389–399. [Google Scholar] [CrossRef]
- Meshorer, A.; Prionas, S.; Fajardo, L.; Meyer, J.; Hahn, G.; Martinez, A. The Effects of Hyperthermia on Normal Mesenchymal Tissues—Application of AaHistologic Grading System. Arch. Pathol. Lab. Med. 1983, 107, 328–334. [Google Scholar] [PubMed]
- Jung, H. A Generalized Concept for Cell Killing by Heat. Radiat. Res. 1986, 106, 56–72. [Google Scholar] [CrossRef]
- Jha, S.; Sharma, P.K.; Malviya, R. Hyperthermia: Role and Risk Factor for Cancer Treatment. Achiev. Life Sci. 2016, 10, 161–167. [Google Scholar] [CrossRef] [Green Version]
- Kousalová, J.; Etrych, T. Polymeric Nanogels as Drug Delivery Systems. Physiol. Res. 2018, 67, S305–S317. [Google Scholar] [CrossRef]
- Nigro, V.; Angelini, R.; Bertoldo, M.; Buratti, E.; Franco, S.; Ruzicka, B. Chemical-Physical Behaviour of Microgels Made of Interpenetrating Polymer Networks of PNIPAM and Poly(Acrylic Acid). Polymers 2021, 13, 1353. [Google Scholar] [CrossRef]
- Iyer, S.; Das, A. Responsive Nanogels for Anti-Cancer Therapy. Mater. Today Proc. 2021, 44, 2330–2333. [Google Scholar] [CrossRef]
- Ghosh Dastidar, D.; Chakrabarti, G. Thermoresponsive Drug Delivery Systems, Characterization and Application. In Applications of Targeted Nano Drugs and Delivery Systems; Elsevier: Amsterdam, The Netherlands, 2019; pp. 133–155. [Google Scholar] [CrossRef]
- Jeong, B.; Kim, S.W.; Bae, Y.H. Thermosensitive Sol–Gel Reversible Hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 154–162. [Google Scholar] [CrossRef]
- Volpert, E.; Selb, J.; Candau, F. Associating Behaviour of Polyacrylamides Hydrophobically Modified with Dihexylacrylamide. Polymer 1998, 39, 1025–1033. [Google Scholar] [CrossRef]
- Lin, S.-Y.; Chen, K.-S.; Liang, R.-C. Thermal Micro ATR/FT-IR Spectroscopic System for Quantitative Study of the Molecular Structure of Poly(N-Isopropylacrylamide) in Water. Polymer 1999, 40, 2619–2624. [Google Scholar] [CrossRef]
- Aseyev, V.O.; Tenhu, H.; Winnik, F.M. Temperature Dependence of the Colloidal Stability of Neutral Amphiphilic Polymers in Water. Adv. Polym. Sci. 2006, 196, 1–85. [Google Scholar] [CrossRef]
- Zhang, Z. Switchable and Responsive Surfaces and Materials for Biomedical Applications; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar] [CrossRef]
- Hertle, Y.; Hellweg, T. Thermoresponsive Copolymer Microgels. J. Mater. Chem. B 2013, 1, 5874–5885. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Pei, Y.; Yang, L.M.; Shi, L.L.; Luo, H.J. Synthesis and Properties of Poly(N-Isopropylacrylamide-Co-Acrylamide) Hydrogels. Macromol. Symp. 2005, 225, 103–112. [Google Scholar] [CrossRef]
- Snowden, M.J.; Chowdhry, B.Z.; Vincent, B.; Morris, G.E. Colloidal Copolymer Microgels of N-Isopropylacrylamide and Acrylic Acid: PH, Ionic Strength and Temperature Effects. J. Chem. Soc. Faraday Trans. 1996, 92, 5013–5016. [Google Scholar] [CrossRef]
- Kratz, K.; Hellweg, T.; Eimer, W. Influence of Charge Density on the Swelling of Colloidal Poly(N-Isopropylacrylamide-Co-Acrylic Acid) Microgels. Colloids Surf. A Physicochem. Eng. Asp. 2000, 170, 137–149. [Google Scholar] [CrossRef]
- Xiong, W.; Gao, X.; Zhao, Y.; Xu, H.; Yang, X. The Dual Temperature/PH-Sensitive Multiphase Behavior of Poly(N-Isopropylacrylamide-Co-Acrylic Acid) Microgels for Potential Application in in Situ Gelling System. Colloids Surf. B Biointerfaces 2011, 84, 103–110. [Google Scholar] [CrossRef]
- Ruel-Gariépy, E.; Leroux, J.C. In Situ-Forming Hydrogels—Review of Temperature-Sensitive Systems. Eur. J. Pharm. Biopharm. 2004, 58, 409–426. [Google Scholar] [CrossRef]
- Diniz, I.M.A.; Chen, C.; Xu, X.; Ansari, S.; Zadeh, H.H.; Marques, M.M.; Shi, S.; Moshaverinia, A. Pluronic F-127 Hydrogel as a Promising Scaffold for Encapsulation of Dental-Derived Mesenchymal Stem Cells. J. Mater. Sci. Mater. Med. 2015, 26, 153. [Google Scholar] [CrossRef] [Green Version]
- Bae, K.H.; Lee, Y.; Park, T.G. Oil-Encapsulating PEO−PPO−PEO/PEG Shell Cross-Linked Nanocapsules for Target-Specific Delivery of Paclitaxel. Biomacromolecules 2007, 8, 650–656. [Google Scholar] [CrossRef]
- Choi, S.H.; Lee, S.H.; Park, T.G. Temperature-Sensitive Pluronic/Poly(Ethylenimine) Nanocapsules for Thermally Triggered Disruption of Intracellular Endosomal Compartment. Biomacromolecules 2006, 7, 1864–1870. [Google Scholar] [CrossRef]
- Bae, K.H.; Choi, S.H.; Park, S.Y.; Lee, Y.; Park, T.G. Thermosensitive Pluronic Micelles Stabilized by Shell Cross-Linking with Gold Nanoparticles. Langmuir 2006, 22, 6380–6384. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Lee, J.H.; Choi, S.M.; Park, T.G. Thermally Reversible Pluronic/Heparin Nanocapsules Exhibiting 1000-Fold Volume Transition. Langmuir 2006, 22, 1758–1762. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Sato, E.; Hirokawa, Y.; Hirotsu, S.; Peetermans, J. Critical Kinetics of Volume Phase Transition of Gels. Phys. Rev. Lett. 1985, 55, 2455–2458. [Google Scholar] [CrossRef]
- Kawaguchi, H. Functional Polymer Microspheres. Prog. Polym. Sci. 2000, 25, 1171–1210. [Google Scholar] [CrossRef]
- Pelton, R.; Hoare, T. Microgel Suspensions: Fundamentals and Applications; Fernandez-Nieves, A., Wyss, H., Mattsson, J., Weitz, D., Eds.; John Wiley & Sons: Singapore, 2011. [Google Scholar]
- McPhee, W.; Tam, K.C.; Pelton, R. Poly(N-Isopropylacrylamide) Latices Prepared with Sodium Dodecyl Sulfate. J. Colloid Interface Sci. 1993, 156, 24–30. [Google Scholar] [CrossRef]
- Pelton, R.H.; Chibante, P. Preparation of Aqueous Latices with N-Isopropylacrylamide. Colloids Surf. 1986, 20, 247–256. [Google Scholar] [CrossRef]
- Landfester, K.; Willert, M.; Antonietti, M. Preparation of Polymer Particles in Nonaqueous Direct and Inverse Miniemulsions. Macromolecules 2000, 33, 2370–2376. [Google Scholar] [CrossRef]
- Chen, L.W.; Yang, B.Z.; Wu, M.L. Synthesis and Kinetics of Microgel in Inverse Emulsion Polymerization of Acrylamide. Prog. Org. Coat. 1997, 31, 393–399. [Google Scholar] [CrossRef]
- Feng, X.; Pelton, R.; Leduc, M.; Champ, S. Colloidal Complexes from Poly (Vinyl Amine) and Carboxymethyl Cellulose Mixtures. Langmuir 2007, 23, 2970–2976. [Google Scholar] [CrossRef]
- Ballauff, M.; Lu, Y. “Smart” Nanoparticles: Preparation, Characterization and Applications. Polymer 2007, 48, 1815–1823. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, A.M. Thermoresponsive Magnetic Colloids. Colloid Polym. Sci. 2007, 285, 953–966. [Google Scholar] [CrossRef]
- Kim, E.-M.; Jeong, H.-J. Liposomes: Biomedical Applications. Chonnam Med. J. 2021, 57, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Yatvin, M.B.; Weinstein, J.N.; Dennis, W.H.; Blumenthal, R. Design of Liposomes for Enhanced Local Release of Drugs by Hyperthermia. Science 1978, 202, 1290–1293. [Google Scholar] [CrossRef]
- Papahadjopoulos, D.; Jacobson, K.; Nir, S.; Isac, I. Phase Transitions in Phospholipid Vesicles. Fluorescence Polarization and Permeability Measurements Concerning the Effect of Temperature and Cholesterol. Biochim. Biophys. Acta-Biomembr. 1973, 311, 330–348. [Google Scholar] [CrossRef]
- Gaber, M.H.; Hong, K.; Huang, S.K.; Papahadjopoulos, D. Thermosensitive Sterically Stabilized Liposomes: Formulation and in Vitro Studies on Mechanism of Doxorubicin Release by Bovine Serum and Human Plasma. Pharm. Res. 1995, 12, 1407–1416. [Google Scholar] [CrossRef]
- Ta, T.; Porter, T.M. Thermosensitive Liposomes for Localized Delivery and Triggered Release of Chemotherapy. J. Control. Release 2013, 169, 112–125. [Google Scholar] [CrossRef] [Green Version]
- De Smet, M.; Heijman, E.; Langereis, S.; Hijnen, N.M.; Grüll, H. Magnetic Resonance Imaging of High Intensity Focused Ultrasound Mediated Drug Delivery from Temperature-Sensitive Liposomes: An in Vivo Proof-of-Concept Study. J. Control. Release 2011, 150, 102–110. [Google Scholar] [CrossRef]
- Hossann, M.; Wiggenhorn, M.; Schwerdt, A.; Wachholz, K.; Teichert, N.; Eibl, H.; Issels, R.D.; Lindner, L.H. In Vitro Stability and Content Release Properties of Phosphatidylglyceroglycerol Containing Thermosensitive Liposomes. Biochim. Biophys. Acta-Biomembr. 2007, 1768, 2491–2499. [Google Scholar] [CrossRef] [Green Version]
- Hosokawa, T.; Sami, M.; Kato, Y.; Hayakawa, E. Alteration in the Temperature-Dependent Content Release Property of Thermosensitive Liposomes in Plasma. Chem. Pharm. Bull. 2003, 51, 1227–1232. [Google Scholar] [CrossRef] [Green Version]
- Limmer, S.; Hahn, J.; Schmidt, R.; Wachholz, K.; Zengerle, A.; Lechner, K.; Eibl, H.; Issels, R.D.; Hossann, M.; Lindner, L.H. Gemcitabine Treatment of Rat Soft Tissue Sarcoma with Phosphatidyldiglycerol-Based Thermosensitive Liposomes. Pharm. Res. 2014, 31, 2276–2286. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Mei, X.G.; Goldberg, S.N.; Ahmed, M.; Lee, J.C.; Gong, W.; Han, H.B.; Yan, K.; Yang, W. Does Thermosensitive Liposomal Vinorelbine Improve Endpoint Survival after Percutaneous Radiofrequency Ablation of Liver Tumors in a Mouse Model? Radiology 2016, 279, 762–772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.; Li, Z.; Yang, Y.; Wang, Z.; Yang, Z.; Li, B.; Xie, X.; Song, J.; Zhang, H.; Li, Y.; et al. Thermo-Sensitive Liposome Co-Loaded of Vincristine and Doxorubicin Based on Their Similar Physicochemical Properties Had Synergism on Tumor Treatment. Pharm. Res. 2016, 33, 1881–1898. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Gong, W.; Wang, Z.-Y.; Yuan, S.-J.; Xie, X.-Y.; Yang, Y.-F.; Yang, Y.; Wang, S.-S.; Yang, D.-X.; Xuan, Z.-X.; et al. Preparation, Characterization, and Pharmacodynamics of Thermosensitive Liposomes Containing Docetaxel. J. Pharm. Sci. 2014, 103, 2177–2183. [Google Scholar] [CrossRef]
- Al Sabbagh, C.; Tsapis, N.; Novell, A.; Calleja-Gonzalez, P.; Escoffre, J.-M.; Bouakaz, A.; Chacun, H.; Denis, S.; Vergnaud, J.; Gueutin, C.; et al. Formulation and Pharmacokinetics of Thermosensitive Stealth® Liposomes Encapsulating 5-Fluorouracil. Pharm. Res. 2015, 32, 1585–1603. [Google Scholar] [CrossRef]
- Landon, C.D.; Park, J.-Y.; Needham, D.; Dewhirst, M.W. Nanoscale Drug Delivery and Hyperthermia: The Materials Design and Preclinical and Clinical Testing of Low Temperature-Sensitive Liposomes Used in Combination with Mild Hyperthermia in the Treatment of Local Cancer. Open Nanomed. J. 2011, 3, 38–64. [Google Scholar] [CrossRef]
- Mills, J.K.; Needham, D. Lysolipid Incorporation in Dipalmitoylphosphatidylcholine Bilayer Membranes Enhances the Ion Permeability and Drug Release Rates at the Membrane Phase Transition. Biochim. Biophys. Acta-Biomembr. 2005, 1716, 77–96. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Krol, A.; Wright, A.; Needham, D.; Dewhirst, M.W.; Yuan, F. Tumor Microvascular Permeability Is a Key Determinant for Antivascular Effects of Doxorubicin Encapsulated in a Temperature Sensitive Liposome. Int. J. Hyperth. 2008, 24, 475–482. [Google Scholar] [CrossRef] [Green Version]
- Dewhirst, M.W.; Tso, C.Y.; Oliver, R.; Gustafson, C.S.; Secomb, T.W.; Gross, J.F. Morphologic and Hemodynamic Comparison of Tumor and Healing Normal Tissue Microvasculature. Int. J. Radiat. Oncol. Biol. Phys. 1989, 17, 91–99. [Google Scholar] [CrossRef]
- Needham, D.; Anyarambhatla, G.; Kong, G.; Dewhirst, M.W. A New Temperature-Sensitive Liposome for Use with Mild Hyperthermia: Characterization and Testing in a Human Tumor Xenograft Model. Cancer Res. 2000, 60, 1197–1201. [Google Scholar]
- Kong, G.; Anyarambhatla, G.; Petros, W.P.; Braun, R.D.; Colvin, O.M.; Needham, D.; Dewhirst, M.W. Efficacy of Liposomes and Hyperthermia in a Human Tumor Xenograft Model: Importance of Triggered Drug Release. Cancer Res. 2000, 60, 6950–6957. [Google Scholar] [PubMed]
- Li, L.; Hagen, T.L.T.; Haeri, A.; Soullié, T.; Scholten, C.; Seynhaeve, A.L.; Eggermont, A.M.; Koning, G.A. A Novel Two-Step Mild Hyperthermia for Advanced Liposomal Chemotherapy. J. Control. Release 2014, 174, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Regenold, M.; Bannigan, P.; Evans, J.C.; Waspe, A.; Temple, M.J.; Allen, C. Turning down the Heat: The Case for Mild Hyperthermia and Thermosensitive Liposomes. Nanomed. Nanotechnol. Biol. Med. 2022, 40, 102484. [Google Scholar] [CrossRef]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal Formulations in Clinical Use: An Updated Review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- PanDox: Targeted Doxorubicin in Pancreatic Tumours—Tabular View—Clinical Trials. Available online: https://clinicaltrials.gov/ct2/show/record/NCT04852367?term=thermosensitive&recrs=ab&cond=cancer&draw=2&rank=3 (accessed on 6 February 2022).
- de Maar, J.S.; Suelmann, B.B.M.; Braat, M.N.G.J.A.; van Diest, P.J.; Vaessen, H.H.B.; Witkamp, A.J.; Linn, S.C.; Moonen, C.T.W.; van der Wall, E.; Deckers, R. Phase i Feasibility Study of Magnetic Resonance Guided High Intensity Focused Ultrasound-Induced Hyperthermia, Lyso-Thermosensitive Liposomal Doxorubicin and Cyclophosphamide in de Novo Stage IV Breast Cancer Patients: Study Protocol of the i-GO Study. BMJ Open 2020, 10, e040162. [Google Scholar] [CrossRef] [PubMed]
- Tak, W.Y.; Lin, S.-M.; Wang, Y.; Zheng, J.; Vecchione, A.; Park, S.Y.; Chen, M.H.; Wong, S.; Xu, R.; Peng, C.-Y.; et al. Phase III HEAT Study Adding Lyso-Thermosensitive Liposomal Doxorubicin to Radiofrequency Ablation in Patients with Unresectable Hepatocellular Carcinoma Lesions. Clin. Cancer Res. 2018, 24, 73–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Celsion Corporation to Continue Following Patients in Phase III OPTIMA Study for Overall Survival Celsion Corporation. Available online: https://investor.celsion.com/news-releases/news-release-details/celsion-corporation-continue-following-patients-phase-iii-optima (accessed on 6 February 2022).
- Study of ThermoDox with Standardized Radiofrequency Ablation (RFA) for Treatment of Hepatocellular Carcinoma (HCC)—Tabular View—Clinica lTrials. Available online: https://clinicaltrials.gov/ct2/show/record/NCT02112656?view=record (accessed on 6 February 2022).
- Lyon, P.C.; Gray, M.D.; Mannaris, C.; Folkes, L.K.; Stratford, M.; Campo, L.; Chung, D.Y.F.; Scott, S.; Anderson, M.; Goldin, R.; et al. Safety and Feasibility of Ultrasound-Triggered Targeted Drug Delivery of Doxorubicin from Thermosensitive Liposomes in Liver Tumours (TARDOX): A Single-Centre, Open-Label, Phase 1 Trial. Lancet Oncol. 2018, 19, 1027–1039. [Google Scholar] [CrossRef] [Green Version]
- Bi, H.; Xue, J.; Jiang, H.; Gao, S.; Yang, D.; Fang, Y.; Shi, K. Current Developments in Drug Delivery with Thermosensitive Liposomes. Asian J. Pharm. Sci. 2019, 14, 365–379. [Google Scholar] [CrossRef]
- de Smet, M.; Langereis, S.; van den Bosch, S.V.D.; Grüll, H. Temperature-Sensitive Liposomes for Doxorubicin Delivery under MRI Guidance. J. Control. Release 2010, 143, 120–127. [Google Scholar] [CrossRef]
- Ta, T.; Convertine, A.J.; Reyes, C.R.; Stayton, P.S.; Porter, T.M. Thermosensitive Liposomes Modified with Poly(N-Isopropylacrylamide-Co-Propylacrylic Acid) Copolymers for Triggered Release of Doxorubicin. Biomacromolecules 2010, 11, 1915–1920. [Google Scholar] [CrossRef] [Green Version]
- Mazzotta, E.; Tavano, L.; Muzzalupo, R. Thermo-Sensitive Vesicles in Controlled Drug Delivery for Chemotherapy. Pharmaceutics 2018, 10, 150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ringsdorf, H.; Venzmer, J.; Winnik, F.M. Interaction of Hydrophobically-Modified Poly-N-Isopropylacrylamides with Model Membranes—or Playing a Molecular Accordion. Angew. Chem. Int. Ed. Engl. 1991, 30, 315–318. [Google Scholar] [CrossRef]
- De Leo, V.; Milano, F.; Agostiano, A.; Catucci, L. Recent Advancements in Polymer/Liposome Assembly for Drug Delivery: From Surface Modifications to Hybrid Vesicles. Polymers 2021, 13, 1027. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.C.; Bae, S.K.; Kim, J.D. Temperature-Sensitivity of Liposomal Lipid Bilayers Mixed with Poly(N-Isopropylacrylamide-Co-Acrylic Acid). J. Biochem. 1997, 121, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Han, H.D.; Shin, B.C.; Choi, H.S. Doxorubicin-Encapsulated Thermosensitive Liposomes Modified with Poly(N-Isopropylacrylamide-Co-Acrylamide): Drug Release Behavior and Stability in the Presence of Serum. Eur. J. Pharm. Biopharm. 2006, 62, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Kono, K.; Hayashi, H.; Takagishi, T. Temperature-Sensitive Liposomes: Liposomes Bearing Poly(N-Isopropylacrylamide). J. Control. Release 1994, 30, 69–75. [Google Scholar] [CrossRef]
- Hayashi, H.; Kono, K.; Takfagishi, T. Temperature Sensitization of Liposomes Using Copolymers of N-Isopropylacrylamide. Bioconjugate Chem. 1999, 10, 412–418. [Google Scholar] [CrossRef]
- Yoshino, K.; Kadowaki, A.; Takagishi, T.; Kono, K. Temperature Sensitization of Liposomes by Use of N-Isopropylacrylamide Copolymers with Varying Transition Endotherms. Bioconjugate Chem. 2004, 15, 1102–1109. [Google Scholar] [CrossRef]
- Gomes, I.P.; Duarte, J.A.; Maia, A.L.C.; Rubello, D.; Townsend, D.M.; de Barros, A.L.B.; Leite, E.A. Thermosensitive Nanosystems Associated with Hyperthermia for Cancer Treatment. Pharmaceuticals 2019, 12, 171. [Google Scholar] [CrossRef] [Green Version]
- Song, C.W. Effect of Local Hyperthermia on Blood Flow and Microenvironment: A Review. Cancer Res. 1984, 44 (Suppl. 10), 4721s–4730s. [Google Scholar]
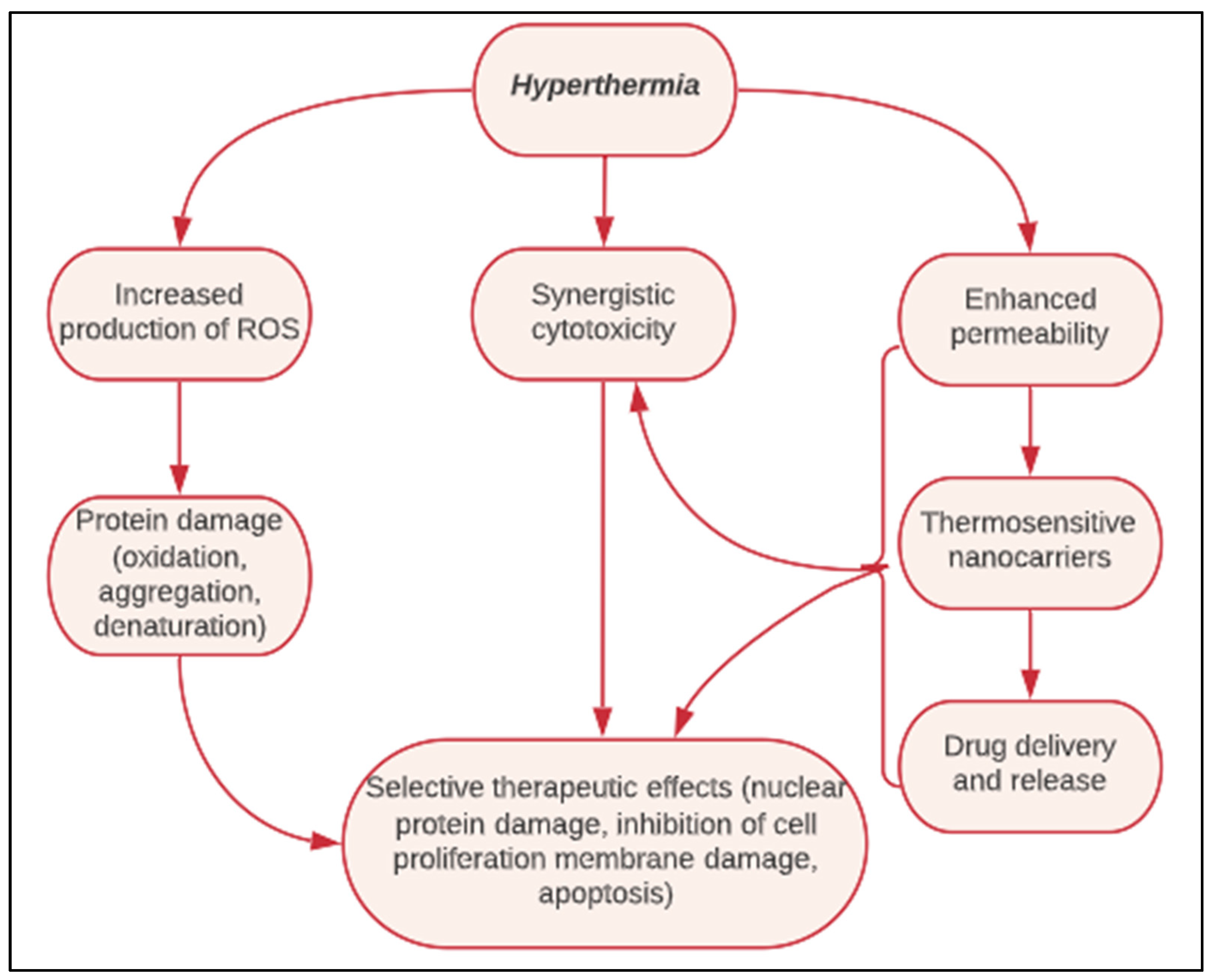
- Bettaieb, A.; Wrzal, P.K.; Averill-Bates, D.A. Hyperthermia: Cancer Treatment and Beyond. Cancer Treat. 2013, 257–283. [Google Scholar] [CrossRef] [Green Version]
- Yoshikawa, T.; Kokura, S.; Tainaka, K.; Itani, K.; Oyamada, H.; Kaneko, T.; Naito, Y.; Kondo, M. The Role of Active Oxygen Species and Lipid Peroxidation in the Antitumor Effect of Hyperthermia. Cancer Res. 1993, 53, 2326–2329. [Google Scholar] [PubMed]
- Wells, J.; Sen, A.; Hui, S.W. Localized Delivery to CT-26 Tumors in Mice Using Thermosensitive Liposomes. Int. J. Pharm. 2003, 261, 105–114. [Google Scholar] [CrossRef]
- Gaber, M.H.; Wu, N.Z.; Hong, K.; Huang, S.K.; Dewhirst, M.W.; Papahadjopoulos, D. Thermosensitive Liposomes: Extravasation and Release of Contents in Tumor Microvascular Networks. Int. J. Radiat. Oncol. Biol. Phys. 1996, 36, 1177–1187. [Google Scholar] [CrossRef]
- Nishimura, Y.; Ono, K.; Hiraoka, M.; Masunaga, S.I.; Jo, S.; Shibamoto, Y.; Sasai, K.; Abe, M.; Iga, K.; Ogawa, Y. Treatment of Murine SCC VII Tumors with Localized Hyperthermia and Temperature-Sensitive Liposomes Containing Cisplatin. Radiat. Res. 1990, 122, 161–167. [Google Scholar] [CrossRef]
- Maekawa, S.; Sugmachi, K.; Kitamura, M. Selective Treatment of Metastatic Lymph Nodes with Combination of Local Hyperthermia and Temperature-Sensitive Liposomes Containing Bleomycin. Cancer Treat. Rep. 1987, 71, 1053–1059. [Google Scholar]
- Kong, G.; Braun, R.; Dewhirst, M. Characterization of the Effect of Hyperthermia on Nanoparticle Extravasation from Tumor Vasculature. Cancer Res. 2001, 61, 3027–3032. [Google Scholar]
- Willerding, L.; Limmer, S.; Hossann, M.; Zengerle, A.; Wachholz, K.; ten Hagen, T.L.M.; Koning, G.A.; Sroka, R.; Lindner, L.H.; Peller, M. Method of Hyperthermia and Tumor Size Influence Effectiveness of Doxorubicin Release from Thermosensitive Liposomes in Experimental Tumors. J. Control. Release 2016, 222, 47–55. [Google Scholar] [CrossRef]
- You, J.; Zhang, P.; Hu, F.; Du, Y.; Yuan, H.; Zhu, J.; Wang, Z.; Zhou, J.; Li, C. Near-Infrared Light-Sensitive Liposomes for the Enhanced Photothermal Tumor Treatment by the Combination with Chemotherapy. Pharm. Res. 2014, 31, 554–565. [Google Scholar] [CrossRef]
- Zagar, T.M.; Vujaskovic, Z.; Formenti, S.; Rugo, H.; Muggia, F.; O’Connor, B.; Myerson, R.; Stauffer, P.; Hsu, I.-C.; Diederich, C.; et al. Two Phase I Dose-Escalation/Pharmacokinetics Studies of Low Temperature Liposomal Doxorubicin (LTLD) and Mild Local Hyperthermia in Heavily Pretreated Patients with Local Regionally Recurrent Breast Cancer. Int. J. Hyperth. 2014, 30, 285–294. [Google Scholar] [CrossRef]
- O’Neill, B.E.; Li, K.C.P. Augmentation of Targeted Delivery with Pulsed High Intensity Focused Ultrasound. Int. J. Hyperth. 2009, 24, 506–520. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.R.; Luk, A.; Durrani, A.; Dromi, S.; Cuesta, J.; Angstadt, M.; Dreher, M.R.; Wood, B.J.; Frenkel, V. In Vitro and in Vivo Evaluations of Increased Effective Beam Width for Heat Deposition Using a Split Focus High Intensity Ultrasound (HIFU) Transducer. Int. J. Hyperth. 2008, 24, 537–549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- VanOsdol, J.; Ektate, K.; Ramasamy, S.; Maples, D.; Collins, W.; Malayer, J.; Ranjan, A. Sequential HIFU Heating and Nanobubble Encapsulation Provide Efficient Drug Penetration from Stealth and Temperature Sensitive Liposomes in Colon Cancer. J. Control. Release 2017, 247, 55–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dromi, S.; Frenkel, V.; Luk, A.; Traughber, B.; Angstadt, M.; Bur, M.; Poff, J.; Xie, J.; Libutti, S.K.; Li, K.; et al. Pulsed-High Intensity Focused Ultrasound and Low Temperature-Sensitive Liposomes for Enhanced Targeted Drug Delivery and Antitumor Effect. Clin. Cancer Res. 2007, 13, 2722–2727. [Google Scholar] [CrossRef] [Green Version]
- Negussie, A.H.; Yarmolenko, P.S.; Partanen, A.; Ranjan, A.; Jacobs, G.; Woods, D.; Bryant, H.; Thomasson, D.; Dewhirst, M.W.; Wood, B.; et al. Formulation and Characterisation of Magnetic Resonance Imageable Thermally Sensitive Liposomes for Use with Magnetic Resonance-Guided High Intensity Focused Ultrasound. Int. J. Hyperth. 2011, 27, 140–155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lokerse, W.J.M.; Bolkestein, M.; Dalm, S.U.; Eggermont, A.M.M.; de Jong, M.; Grüll, H.; Koning, G.A. Comparing the Therapeutic Potential of Thermosensitive Liposomes and Hyperthermia in Two Distinct Subtypes of Breast Cancer. J. Control. Release 2017, 258, 34–42. [Google Scholar] [CrossRef]
- Peller, M.; Willerding, L.; Limmer, S.; Hossann, M.; Dietrich, O.; Ingrisch, M.; Sroka, R.; Lindner, L.H. Surrogate MRI Markers for Hyperthermia-Induced Release of Doxorubicin from Thermosensitive Liposomes in Tumors. J. Control. Release 2016, 237, 138–146. [Google Scholar] [CrossRef]
- Ranjan, A.; Jacobs, G.C.; Woods, D.L.; Negussie, A.H.; Partanen, A.; Yarmolenko, P.S.; Gacchina, C.E.; Sharma, K.V.; Frenkel, V.; Wood, B.J.; et al. Image-Guided Drug Delivery with Magnetic Resonance Guided High Intensity Focused Ultrasound and Temperature Sensitive Liposomes in a Rabbit Vx2 Tumor Model. J. Control. Release 2012, 158, 487–494. [Google Scholar] [CrossRef] [Green Version]



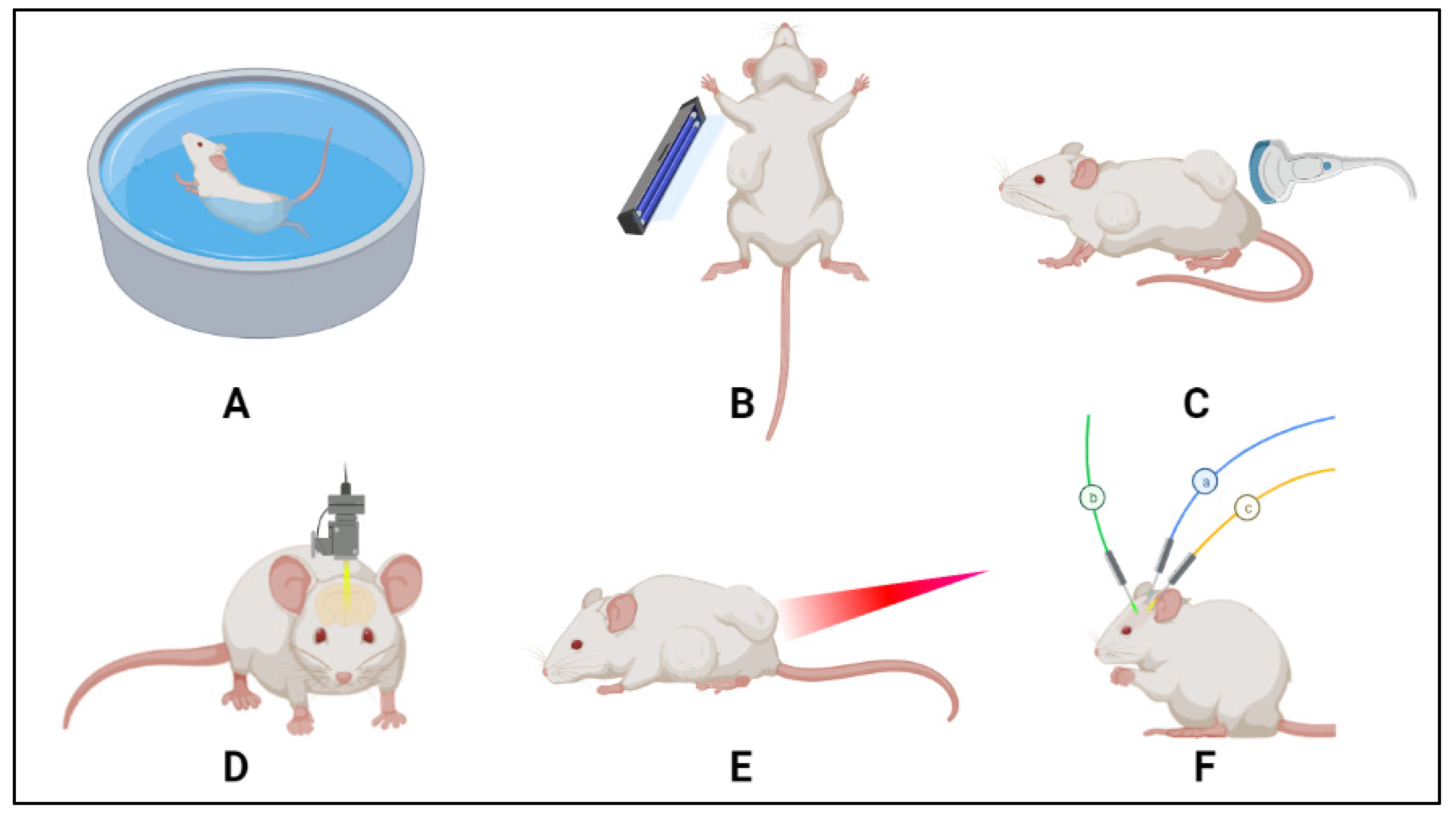
| Material | Preparation Method | Volume Change (%) | Transition Onset (T °C) | Ref. |
|---|---|---|---|---|
| Pluronic® F127/heparin | Modified emulsification/solvent evaporation method | ~99 | ~25 | [45] |
| Pluronic® F127/poly(ethylenimine) | Modified emulsification/solvent evaporation method | 92–97 | ~21 | [43] |
| Au/Pluronic® F127 | Self-assembly then conjugation | ~96 | ~18 | [44] |
| Pluronic® F127/PEG | Modified emulsification/solvent evaporation method | ~89 | ~23 | [42] |
| Encapsulated Drug | Composition Molar Ratio | Experimental/Release Conditions | Findings | Ref. |
|---|---|---|---|---|
| Doxorubicin and MRI contrast agent [Gd(HPDO3A)(H2O)] | DPPC/HSPC/CH/DPPE-PEG2000/DOTA-DSPE 50:25:15:3:1 | Hyperthermia was induced by exposing TSLs samples homogeneously distributed in a gel, by heating from 37 °C to 42 °C inside a clinical (magnetic resonance high intensity focused ultrasound) MR-HIFU setup. | Simultaneous and quantitative release of the drug and the MRI contrast agent was observed from the TSLs at 42 °C, while none was observed 37 °C after exposure for 1 h. | [61] |
| Doxorubicin | DPPC/DSPC/DPPGOG 50:20:30 | TSLs were added to preheated fetal calm serum (FCS) or HN buffer where the temperature was varied from 37 to 45 °C over a time period of up to 180 min, and doxorubicin release was measured using fluorescence spectrometry. | 89.1 ± 4.0% of doxorubicin over was retained in the TSLs for 3 h at 37 °C in the presence of serum. The release rate was significantly increased by incorporating DPPGOG. | [62] |
| Mitomycin C (MMC) | DPPC/DPPG 7:3 | Diluted TSL samples were incubated in 30% (volume/volume) rat plasma for 60 min at the desired temperature (37 °C and 44 °C), then the released MMC was removed with cation exchange resin and the concentration of the retained drug in the liposomes was measured by high-performance liquid chromatography (HPLC). | The temperature-dependent content release efficiency % increased to 96% at the higher temperature. MMC leakage from the TSLs was suppressed in the presence of rat plasma and reached a plateau of 15%. | [63] |
| Pyrimidine Analogue Gemcitabine (dFdC) | DPPC/DSPC/DPPG2 50:20:30 | Diluted TSL samples were incubated in a preheated thermoshaker in FCS or HN for 5 min at 43 °C. After incubation, the samples were centrifuged where the filtrate containing the released drug was analyzed by HPLC. | The TSLs were stable at 37 °C in serum after 6 h of incubation and showed less than 20% release, while at 43 °C, 81.8 ± 15.0% of dFdC was released. | [64] |
| Vinorelbine | DPPC/MPPC/DSPE-PEG2000 86:5:4 | In vitro WST-1 proliferation assay was used to evaluate the TSL dose-dependent effect and temperature on the viability of H22 cells. Cells were incubated for 72 h with the treatment at 37 °C and 42 °C. | Cells incubated at the higher temperature exhibited less cell viability%. | [65] |
| Doxorubicin and Vincristine | DPPC/DSPE-PEG2000/MSPC 75:17:8 | Drug release from the TSLs was determined at 37 °C, 39 °C, 41 °C, and 42 °C over a period of 60 min. The time-dependent drug release profiles at 37 and 42 °C were assessed by HPLC. | Released amount of both payloads was about 85% within the first 5 min of heating at 42 °C from the TSLs, while less than 10% of the total drugs amount was released at 37 °C after heating for 30 min. | [66] |
| Docetaxel | DPPC/DSPE/PEG2000/EPC/MSPC 82:11:4:3:4 | The TSLs were suspended in phosphate-buffered saline (PBS) 37 °C and 42 °C, then an analysis made done by dialysis. | In vitro drug release showed less drug released at 37 °C than at 42 °C, as after 2 h of incubation the TSLs released 15% and 40% of their load, respectively. | [67] |
| 5-Fluorouracil (5-FU) | DPPC/CHO/DSPE-PEG 90:5:5 | TSLs were diluted in PBS and exposed to temperatures varying from 25 to 49 °C over a time period of 30 min, in a heated water bath to determine time-dependent release. Temperature-dependent release was measured at each temperature in the range, by heating the samples for 10 min in Eppendorf tubes in heated water bath. | Drug release approached 70% as temperature increased from 37 °C to 49 °C. | [68] |
| Comonomer | Liposome Composition | Encapsulated Drug | Modulation to Thermo-Responsiveness | Ref. |
|---|---|---|---|---|
| Free radical copolymerization of PNIPAAm with ODA | EPC | Calcein/carboxyfluorescein | The copolymer containing 1 mol% ODA had a LCST of 27 °C, compared to 32 °C for pure NIPAAm. ODA chains served as fixation sites of NIPAAm onto the core of the liposomes. Liposomes incorporating the copolymer exhibited enhanced thermosensitivity and showed more sustainable release profiles. | [92] |
| Free radical copolymerization of PNIPAAm with AAm | DOPE/EPC (6:4, w/w) | Calcein | Incorporating 10%, 20%, and 30% of AAm with NIPAAm increased the LCST to 39, 47, and 53 °C, respectively. Tuning the polymer LCST directly affects the liposomes Tc. At T > 50 °C, the liposomal formulation incorporating 10% AAm showed 80% drug release. | [93] |
| Free radical copolymerization of PNIPAAm with AAm | DPPC | Doxorubicin | Increasing the AAm% in the copolymer from 17 to 24% resulted in increasing the LCST from 40 to 47 °C. The respective modified liposomal formulations exhibited a Tc similar to the copolymers’ LCST. | [91] |
| Free radical copolymerization with 3 structurally different comonomers: Apr, DMAM, NIPMAM | EPC | Calcein | The three copolymers’ LCST was ~40 °C. The transition enthalpy (ΔH) of the copolymers: NIPMAM > DMAM > Apr. Drug release % from the different modified liposomes increased as ΔH increased. The copolymer containing NIPMAM formed the highest hydrophobic domains above the LCST, which resulted in stronger interactions between the copolymer and the lipid bilayer; thus, augmenting perturbation upon heating, which caused the highest release. | [94] |
| Reversible-deactivation radical copolymerization with PAA | DPPC | Doxorubicin | Incorporating 5% PAA increased the copolymer LCST to 42 °C as its hydrophobicity increased. The modified liposomes were stable at physiological conditions, but released 70% and 100% after 5 min of heating at 40 °C and 42 °C, respectively. | [86] |
| Liposomal Formulation | Heating Modality | Encapsulated Drug/Targeted Cancer | In Vivo Model | Response | Ref. |
|---|---|---|---|---|---|
| DPPC/DSPC/DSPE–PEG2000/70:25:5 | Water bath | Doxorubicin/breast cancer | Orthotopic mice bearing breast tumors (MDA-MB-231 and T-47D) | The potency of neoadjuvant hyperthermia with TSLs was demonstrated, where heated tumors showed increased vascularization and permeability | [112] |
| DPPC/DSPC/DPPG2/50:20:30 | Laser | Doxorubicin/soft tissue sarcoma | Brown Norway rats bearing syngeneic soft tissue sarcomas (BN175) | Heated tumors treated with TSLs showed more selective Doxorubicin uptake and accumulation | [113] |
| DPPC/DSPC/DPPG2/50:20:30 | Laser | Doxorubicin/soft tissue sarcoma | Brown Norway rats bearing syngeneic soft tissue sarcomas (BN175) | Hyperthermal-triggered drug release from TSLs resulted in a 13-fold increase in Doxorubicin accumulation inside tumors. | [104] |
| DPPC/DSPC/DPPG2/50:20:30 | HIFUS | Gemcitabine/soft tissue sarcoma | Brown Norway rats bearing syngeneic soft tissue sarcomas (BN175) | Combining HIFUS and TSLs showed distinguished tumor growth suppression | [64] |
| DPPC/MSPC/DSPE–PEG2000/DSPG/83:3:10:4 | Water bath | Paclitaxel/lung cancer | Male Kunming mice bearing Lewis lung carcinoma (LLC) | Tumors treated with TSLs and exposed to heating experienced an arrest in growth | [67] |
| lyso-lecithin containing LTSLs | MR-HIFUS | Doxorubicin/squamous cell carcinoma | Rabbits bearing Vx2 carcinoma | LTSLs combined with MR-HIFUS enhanced tumor specificity and increased Dox uptake. | [114] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abuwatfa, W.H.; Awad, N.S.; Pitt, W.G.; Husseini, G.A. Thermosensitive Polymers and Thermo-Responsive Liposomal Drug Delivery Systems. Polymers 2022, 14, 925. https://doi.org/10.3390/polym14050925
Abuwatfa WH, Awad NS, Pitt WG, Husseini GA. Thermosensitive Polymers and Thermo-Responsive Liposomal Drug Delivery Systems. Polymers. 2022; 14(5):925. https://doi.org/10.3390/polym14050925
Chicago/Turabian StyleAbuwatfa, Waad H., Nahid S. Awad, William G. Pitt, and Ghaleb A. Husseini. 2022. "Thermosensitive Polymers and Thermo-Responsive Liposomal Drug Delivery Systems" Polymers 14, no. 5: 925. https://doi.org/10.3390/polym14050925






