Protein-Based Hydrogels: Promising Materials for Tissue Engineering
Abstract
:1. Introduction
2. Protein-Based Hydrogels
Protein Engineering for PBHs Formation
3. TE-Related Applications of PBHs
3.1. Collagen-Based Hydrogels
3.2. Gelatin-Based Hydrogels
3.3. Serum Albumin-Based Hydrogels
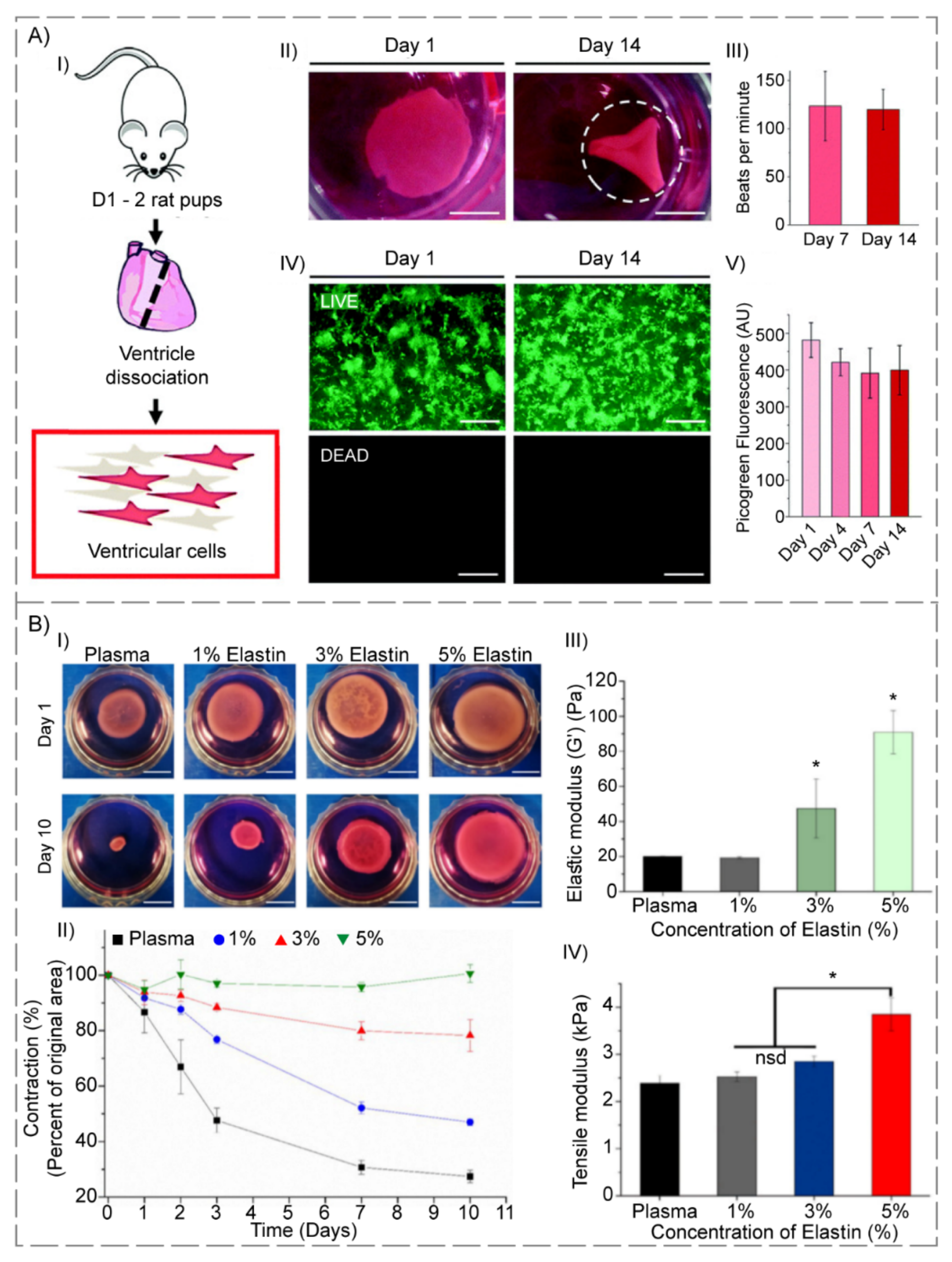
3.4. Elastin-Based Hydrogels
3.5. Keratin-Based Hydrogels
3.6. Resilin-Based Hydrogels
3.7. Silk-Based Hydrogels
4. Delivery Strategies
4.1. Infused GF Delivery
4.2. Scaffold Immobilization
4.3. Spatiotemporally Controlled Delivery
5. Critical Determinants of GF and PBHs
6. Summary, Challenges, and Outlook
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mamidi, N.; Zuniga, A.E.; Villela-Castrejon, J. Engineering and evaluation of forcespun functionalized carbon nano-onions reinforced poly (ε-caprolactone) composite nanofibers for pH-responsive drug release. Mater. Sci. Eng. C 2020, 112, 110928. [Google Scholar] [CrossRef] [PubMed]
- Frone, A.N.; Panaitescu, D.M.; Nicolae, C.A.; Gabor, A.R.; Trusca, R.; Casarica, A.; Stanescu, P.O.; Baciu, D.D.; Salageanu, A. Bacterial cellulose sponges obtained with green cross-linkers for tissue engineering. Mater. Sci. Eng. C 2020, 110, 110740. [Google Scholar] [CrossRef]
- Mozafari, M.; Ramedani, A.; Zhang, Y.N.; Mills, D.K. Thin films for tissue engineering applications. In Thin Film Coatings for Biomaterials and Biomedical Applications; Elsevier: Amsterdam, The Netherlands, 2016; pp. 167–195. [Google Scholar]
- Mamidi, N.; Delgadillo, R.M.V.; Barrera, E.V. Covalently functionalized carbon nano-onions integrated gelatin methacryloyl nanocomposite hydrogel containing γ-cyclodextrin as drug carrier for high-performance pH-triggered drug release. Pharmaceuticals 2021, 14, 291. [Google Scholar] [CrossRef] [PubMed]
- Mamidi, N.; Delgadillo, R.M.V. Design, fabrication and drug release potential of dual stimuli-responsive composite hydrogel nanoparticle interfaces. Colloids Surf. B Biointerfaces 2021, 204, 111819. [Google Scholar] [CrossRef] [PubMed]
- Sahranavard, M.; Zamanian, A.; Ghorbani, F.; Shahrezaee, M.H. A critical review on three dimensional-printed chitosan hydrogels for development of tissue engineering. Bioprinting 2020, 17, e00063. [Google Scholar] [CrossRef]
- Shin, H.; Jo, S.; Mikos, A.G. Biomimetic materials for tissue engineering. Biomaterials 2003, 24, 4353–4364. [Google Scholar] [CrossRef]
- Kohane, D.S.; Langer, R. Polymeric biomaterials in tissue engineering. Pediatr. Res. 2008, 63, 487–491. [Google Scholar] [CrossRef] [Green Version]
- Bakhtiary, N.; Liu, C.; Ghorbani, F. Bioactive inks development for osteochondral tissue engineering: A mini-review. Gels 2021, 7, 274. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Hydrogels for tissue engineering. Chem. Rev. 2001, 101, 1869–1880. [Google Scholar] [CrossRef]
- Sanchez, E.M.; Gomez-Blanco, J.C.; Nieto, E.L.; Casado, J.G.; Macias-Garcia, A.; Diez, M.A.D.; Carrasco-Amador, J.P.; Torrejon Martín, D.; Sanchez-Margallo, F.M.; Pagador, J.B. Hydrogels for bioprinting: A systematic review of hydrogels synthesis, bioprinting parameters, and bioprinted structures behavior. Front. Bioeng. Biotechnol. 2020, 8, 776. [Google Scholar] [CrossRef]
- Cha, G.D.; Lee, W.H.; Sunwoo, S.-H.; Kang, D.; Kang, T.; Cho, K.W.; Kim, M.; Park, O.K.; Jung, D.; Lee, J.; et al. Multifunctional Injectable hydrogel for in vivo diagnostic and therapeutic applications. ACS Nano 2022, 16, 554–567. [Google Scholar] [CrossRef] [PubMed]
- Jabbari, E.; Leijten, J.; Xu, Q.; Khademhosseini, A. The matrix reloaded: The evolution of regenerative hydrogels. Mater. Today 2016, 19, 190–196. [Google Scholar] [CrossRef]
- Pita-Lopez, M.L.; Fletes-Vargas, G.; Espinosa-Andrews, H.; Rodriguez-Rodriguez, R. Physically cross-linked chitosan-based hydrogels for tissue engineering applications: A state-of-the-art review. Eur. Polym. J. 2021, 145, 110176. [Google Scholar] [CrossRef]
- Augst, A.D.; Kong, H.J.; Mooney, D.J. Alginate hydrogels as biomaterials. Macromol. Biosci. 2006, 6, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Mamidi, N.; Delgadillo, R.M.V.; Castrejon, J.V. Unconventional and facile production of a stimuli-responsive multifunctional system for simultaneous drug delivery and environmental remediation. Environ. Sci. Nano 2021, 8, 2081–2097. [Google Scholar] [CrossRef]
- Silva, R.; Fabry, B.; Boccaccini, A.R. Fibrous protein-based hydrogels for cell encapsulation. Biomaterials 2014, 35, 6727–6738. [Google Scholar] [CrossRef]
- Sasiela, C. Proteins: Structure and function by david whitford (university of London). John Wiley and Sons, Ltd., Chichester. 2005. x + 528 pp. 19.5 × 24 cm. $65.00 (paper). J. Nat. Prod. 2006, 69, 1117. [Google Scholar] [CrossRef]
- Fisher, S.A.; Baker, A.E.G.; Shoichet, M.S. Designing peptide and protein modified hydrogels: Selecting the optimal conjugation strategy. J. Am. Chem. Soc. 2017, 139, 7416–7427. [Google Scholar] [CrossRef]
- Mirzaei, M.; Okoro, O.V.; Nie, L.; Petri, D.F.S.; Shavandi, A. Protein-Based 3D Biofabrication of Biomaterials. Bioengineering 2021, 8, 48. [Google Scholar] [CrossRef]
- Le Thi, P.; Lee, Y.; Nguyen, D.H.; Park, K.D. In situ forming gelatin hydrogels by dual-enzymatic cross-linking for enhanced tissue adhesiveness. J. Mater. Chem. B 2017, 5, 757–764. [Google Scholar] [CrossRef]
- Le, X.T.; Rioux, L.-E.; Turgeon, S.L. Formation and functional properties of protein–polysaccharide electrostatic hydrogels in comparison to protein or polysaccharide hydrogels. Adv. Colloid Interface Sci. 2017, 239, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Heaysman, C.L.; Willis, S.; Lewis, A.L. Physical hydrogels with self-assembled nanostructures as drug delivery systems. Expert Opin. Drug Deliv. 2011, 8, 1141–1159. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Aoyama, Y.; Yamanaka, J.; Toyotama, A.; Okuzono, T. Particle adsorption on hydrogel surfaces in aqueous media due to van der waals attraction. Sci. Rep. 2017, 7, 6099. [Google Scholar] [CrossRef] [Green Version]
- Tang, G.; Tan, Z.; Zeng, W.; Wang, X.; Shi, C.; Liu, Y.; He, H.; Chen, R.; Ye, X. Recent advances of chitosan-based injectable hydrogels for bone and dental tissue regeneration. Front. Bioeng. Biotechnol. 2020, 8, 1084. [Google Scholar] [CrossRef]
- Totosaus, A.; Montejano, J.G.; Salazar, J.A.; Guerrero, I. A review of physical and chemical protein-gel induction. Int. J. Food Sci. Technol. 2002, 37, 589–601. [Google Scholar] [CrossRef]
- Distler, T.; Kretzschmar, L.; Schneidereit, D.; Girardo, S.; Goswami, R.; Friedrich, O.; Detsch, R.; Guck, J.; Boccaccini, A.R.; Budday, S. Mechanical properties of cell- and microgel bead-laden oxidized alginate-gelatin hydrogels. Biomater. Sci. 2021, 9, 3051–3068. [Google Scholar] [CrossRef] [PubMed]
- Schloss, A.C.; Williams, D.M.; Regan, L.J. Protein-based hydrogels for tissue engineering. In Protein-Based Engineered Nanostructures; Cortajarena, A., Grove, T., Eds.; Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2016; Volume 940, pp. 167–177. [Google Scholar] [CrossRef] [Green Version]
- Shi, W.; Dumont, M.-J.; Ly, E.B. Synthesis and properties of canola protein-based superabsorbent hydrogels. Eur. Polym. J. 2014, 54, 172–180. [Google Scholar] [CrossRef]
- Kabiri, K.; Omidian, H.; Zohuriaan-Mehr, M.J.; Doroudiani, S. Superabsorbent hydrogel composites and nanocomposites: A review. Polym. Compos. 2011, 32, 277–289. [Google Scholar] [CrossRef]
- Mamidi, N.; Gonzalez-Ortiz, A.; Romo, I.L.; Barrera, E.V. Development of functionalized carbon nano-onions reinforced zein protein hydrogel interfaces for controlled drug release. Pharmaceutics 2019, 11, 621. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Scheiger, J.M.; Levkin, P.A. Design and applications of photoresponsive hydrogels. Adv. Mater. 2019, 31, 1807333. [Google Scholar] [CrossRef] [Green Version]
- Hardy, J.G.; Romer, L.M.; Scheibel, T.R. Polymeric materials based on silk proteins. Polymer 2008, 49, 4309–4327. [Google Scholar] [CrossRef]
- Li, Y.; Xue, B.; Cao, Y. 100th anniversary of macromolecular science viewpoint: Synthetic protein hydrogels. ACS Macro Lett. 2020, 9, 512–524. [Google Scholar] [CrossRef]
- Li, Z.; Xu, W.; Wang, X.; Jiang, W.; Ma, X.; Wang, F.; Zhang, C.; Ren, C. Fabrication of PVA/PAAm IPN hydrogel with high adhesion and enhanced mechanical properties for body sensors and antibacterial activity. Eur. Polym. J. 2021, 146, 110253. [Google Scholar] [CrossRef]
- Li, Z.; Meng, X.; Xu, W.; Zhang, S.; Ouyang, J.; Zhang, Z.; Liu, Y.; Niu, Y.; Ma, S.; Xue, Z.; et al. Single network double cross-linker (SNDCL) hydrogels with excellent stretchability, self-recovery, adhesion strength, and conductivity for human motion monitoring. Soft Matter 2020, 16, 7323–7331. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Kong, X.; Xiong, Y.; Zhang, X.; Chen, H.; Jiang, W.; Niu, Y.; Xu, W.; Ren, C. An overview of dynamic covalent bonds in polymer material and their applications. Eur. Polym. J. 2020, 141, 110094. [Google Scholar] [CrossRef]
- Lim, S.; Jung, G.A.; Muckom, R.J.; Glover, D.J.; Clark, D.S. Engineering bioorthogonal protein–polymer hybrid hydrogel as a functional protein immobilization platform. Chem. Commun. 2019, 55, 806–809. [Google Scholar] [CrossRef]
- Hu, X.; Xia, X.-X.; Huang, S.-C.; Qian, Z.-G. Development of adhesive and conductive resilin-based hydrogels for wearable sensors. Biomacromolecules 2019, 20, 3283–3293. [Google Scholar] [CrossRef]
- Ghosh, G.; Barman, R.; Sarkar, J.; Ghosh, S. pH-responsive biocompatible supramolecular peptide hydrogel. J. Phys. Chem. B 2019, 123, 5909–5915. [Google Scholar] [CrossRef]
- Dock-Bregeon, A.-C.; Lewis, K.A.; Conte, M.R. The la-related proteins: Structures and interactions of a versatile superfamily of RNA-binding proteins. RNA Biol. 2021, 18, 178–193. [Google Scholar] [CrossRef]
- Olechnovic, K.; Venclovas, C. VoroMQA web server for assessing three-dimensional structures of proteins and protein complexes. Nucleic Acids Res. 2019, 47, W437–W442. [Google Scholar] [CrossRef]
- Pickel, B.; Schaller, A. Dirigent proteins: Molecular characteristics and potential biotechnological applications. Appl. Microbiol. Biotechnol. 2013, 97, 8427–8438. [Google Scholar] [CrossRef] [PubMed]
- Satarker, S.; Nampoothiri, M. Structural proteins in severe acute respiratory syndrome coronavirus-2. Arch. Med. Res. 2020, 51, 482–491. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Qi, X.; Chen, Y.; Wu, Z. Thermo-sensitive hydrogels for delivering biotherapeutic molecules: A review. Saudi Pharm. J. 2019, 27, 990–999. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Jia, X.; Zhang, Q.; Chen, H.; Zhu, Q.; Yin, L. Interpenetrating polymer network hydrogels of soy protein isolate and sugar beet pectin as a potential carrier for probiotics. Food Hydrocoll. 2021, 113, 106453. [Google Scholar] [CrossRef]
- Joseph, C.A.; McCarthy, C.W.; Tyo, A.G.; Hubbard, K.R.; Fisher, H.C.; Altscheffel, J.A.; He, W.; Pinnaratip, R.; Liu, Y.; Lee, B.P.; et al. Development of an injectable nitric oxide releasing poly(ethylene) glycol-fibrin adhesive hydrogel. ACS Biomater. Sci. Eng. 2019, 5, 959–969. [Google Scholar] [CrossRef]
- Yan, W.; Zhang, B.; Yadav, M.P.; Feng, L.; Yan, J.; Jia, X.; Yin, L. Corn fiber gum-soybean protein isolate double network hydrogel as oral delivery vehicles for thermosensitive bioactive compounds. Food Hydrocoll. 2020, 107, 105865. [Google Scholar] [CrossRef]
- Chakraborty, P.; Das, S.; Nandi, A.K. Conducting gels: A chronicle of technological advances. Prog. Polym. Sci. 2019, 88, 189–219. [Google Scholar] [CrossRef]
- Rahmani, V.; Elshereef, R.; Sheardown, H. Optimizing electrostatic interactions for controlling the release of proteins from anionic and cationically modified alginate. Eur. J. Pharm. Biopharm. 2017, 117, 232–243. [Google Scholar] [CrossRef]
- Pacelli, S.; Basu, S.; Berkland, C.; Wang, J.; Paul, A. Design of a cytocompatible hydrogel coating to modulate properties of ceramic-based scaffolds for bone repair. Cell. Mol. Bioeng. 2018, 11, 211–217. [Google Scholar] [CrossRef]
- Dai, X.; Zhang, Y.; Gao, L.; Bai, T.; Wang, W.; Cui, Y.; Liu, W. A mechanically strong, highly stable, thermoplastic, and self-healable supramolecular polymer hydrogel. Adv. Mater. 2015, 27, 3566–3571. [Google Scholar] [CrossRef]
- Xu, R.; Ma, S.; Lin, P.; Yu, B.; Zhou, F.; Liu, W. High strength astringent hydrogels using protein as the building block for physically cross-linked multi-network. ACS Appl. Mater. Interfaces 2018, 10, 7593–7601. [Google Scholar] [CrossRef]
- Voorhaar, L.; Hoogenboom, R. Supramolecular polymer networks: Hydrogels and bulk materials. Chem. Soc. Rev. 2016, 45, 4013–4031. [Google Scholar] [CrossRef] [PubMed]
- Berger, J.; Reist, M.; Mayer, J.M.; Felt, O.; Peppas, N.A.; Gurny, R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur. J. Pharm. Biopharm. 2004, 57, 19–34. [Google Scholar] [CrossRef]
- Yan, S.; Han, G.; Wang, Q.; Zhang, S.; You, R.; Luo, Z.; Xu, A.; Li, X.; Li, M.; Zhang, Q.; et al. Directed assembly of robust and biocompatible silk fibroin/hyaluronic acid composite hydrogels. Compos. Part B Eng. 2019, 176, 107204. [Google Scholar] [CrossRef]
- Huang, S.-C.; Fan, R.-X.; Tian, K.-K.; Xia, X.-X.; Qian, Z.-G. Controllable fibrillization reinforces genetically engineered rubberlike protein hydrogels. Biomacromolecules 2021, 22, 961–970. [Google Scholar] [CrossRef]
- Hu, X.; Lu, Q.; Sun, L.; Cebe, P.; Wang, X.; Zhang, X.; Kaplan, D.L. Biomaterials from ultrasonication-induced silk fibroin−hyaluronic acid hydrogels. Biomacromolecules 2010, 11, 3178–3188. [Google Scholar] [CrossRef]
- Tang, Z.; Chen, Q.; Chen, F.; Zhu, L.; Lu, S.; Ren, B.; Zhang, Y.; Yang, J.; Zheng, J. General principle for fabricating natural globular protein-based double-network hydrogels with integrated highly mechanical properties and surface adhesion on solid surfaces. Chem. Mater. 2019, 31, 179–189. [Google Scholar] [CrossRef]
- Hoff, S.E.; Liu, J.; Heinz, H. Binding mechanism and binding free energy of amino acids and citrate to hydroxyapatite surfaces as a function of crystallographic facet, pH, and electrolytes. J. Colloid Interface Sci. 2022, 605, 685–700. [Google Scholar] [CrossRef]
- Cho, D.H.; Hahm, J. Protein–polymer interaction characteristics unique to nanoscale interfaces: A perspective on recent insights. J. Phys. Chem. B 2021, 125, 6040–6057. [Google Scholar] [CrossRef]
- Vila-Perello, M.; Liu, Z.; Shah, N.H.; Willis, J.A.; Idoyaga, J.; Muir, T.W. Streamlined expressed protein ligation using split inteins. J. Am. Chem. Soc. 2013, 135, 286–292. [Google Scholar] [CrossRef]
- Nguyen, G.K.T.; Wang, S.; Qiu, Y.; Hemu, X.; Lian, Y.; Tam, J.P. Butelase 1 is an Asx-specific ligase enabling peptide macrocyclization and synthesis. Nat. Chem. Biol. 2014, 10, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Wu, T.; Wang, M.; Shi, S.; Yuan, G.; Li, X.; Chong, H.; Wu, B.; Zheng, P. Enzymatic biosynthesis and immobilization of polyprotein verified at the single-molecule level. Nat. Commun. 2019, 10, 2775. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Gao, W. Precision conjugation: An emerging tool for generating protein–polymer conjugates. Angew. Chem. Int. Ed. 2021, 60, 11024–11035. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Zhang, W.-B.; Mahdavi, A.; Arnold, F.H.; Tirrell, D.A. Synthesis of bioactive protein hydrogels by genetically encoded spytag-spycatcher chemistry. Proc. Natl. Acad. Sci. USA 2014, 111, 11269–11274. [Google Scholar] [CrossRef] [Green Version]
- Lyu, S.; Fang, J.; Duan, T.; Fu, L.; Liu, J.; Li, H. Optically controlled reversible protein hydrogels based on photoswitchable fluorescent protein Dronpa. Chem. Commun. 2017, 53, 13375–13378. [Google Scholar] [CrossRef]
- Fierer, J.O.; Veggiani, G.; Howarth, M. SpyLigase peptide–peptide ligation polymerizes affibodies to enhance magnetic cancer cell capture. Proc. Natl. Acad. Sci. USA 2014, 111, E1176–E1181. [Google Scholar] [CrossRef] [Green Version]
- Hu, W.; Wang, Z.; Xiao, Y.; Zhang, S.; Wang, J. Advances in crosslinking strategies of biomedical hydrogels. Biomater. Sci. 2019, 7, 843–855. [Google Scholar] [CrossRef]
- Kadumudi, F.B.; Hasany, M.; Pierchala, M.K.; Jahanshahi, M.; Taebnia, N.; Mehrali, M.; Mitu, C.F.; Shahbazi, M.; Zsurzsan, T.; Knott, A.; et al. The manufacture of unbreakable bionics via multifunctional and self-healing silk–graphene hydrogels. Adv. Mater. 2021, 33, 2100047. [Google Scholar] [CrossRef]
- Wang, C.; Yang, L.; He, Y.; Xiao, H.; Lin, W. Microsphere-structured hydrogel crosslinked by polymerizable protein-based nanospheres. Polymer 2020, 211, 123114. [Google Scholar] [CrossRef]
- Su, J.; Lu, S.; Jiang, S.; Li, B.; Liu, B.; Sun, Q.; Li, J.; Wang, F.; Wei, Y. Engineered protein photo-thermal hydrogels for outstanding in situ tongue cancer therapy. Adv. Mater. 2021, 33, 2100619. [Google Scholar] [CrossRef]
- Chen, X.; Tan, B.; Wang, S.; Tang, R.; Bao, Z.; Chen, G.; Chen, S.; Tang, W.; Wang, Z.; Long, C.; et al. Rationally designed protein cross-linked hydrogel for bone regeneration via synergistic release of magnesium and zinc ions. Biomaterials 2021, 274, 120895. [Google Scholar] [CrossRef] [PubMed]
- Phan, V.H.G.; Le, T.M.D.; Janarthanan, G.; Ngo, P.-K.T.; Lee, D.S.; Thambi, T. Development of bioresorbable smart injectable hydrogels based on thermo-responsive copolymer integrated bovine serum albumin bioconjugates for accelerated healing of excisional wounds. J. Ind. Eng. Chem. 2021, 96, 345–355. [Google Scholar] [CrossRef]
- Wang, Y.; Xia, Y.; Xiang, P.; Dai, Y.; Gao, Y.; Xu, H.; Yu, J.; Gao, G.; Chen, K. Protein-assisted freeze-tolerant hydrogel with switchable performance toward customizable flexible sensor. Chem. Eng. J. 2022, 428, 131171. [Google Scholar] [CrossRef]
- Boonpavanitchakul, K.; Bast, L.K.; Bruns, N.; Magaraphan, R. Silk sericin-polylactide protein–polymer conjugates as biodegradable amphiphilic materials and their application in drug release systems. Bioconjug. Chem. 2020, 31, 2312–2324. [Google Scholar] [CrossRef]
- Lei, H.; Dong, L.; Li, Y.; Zhang, J.; Chen, H.; Wu, J.; Zhang, Y.; Fan, Q.; Xue, B.; Qin, M.; et al. Stretchable hydrogels with low hysteresis and anti-fatigue fracture based on polyprotein cross-linkers. Nat. Commun. 2020, 11, 4032. [Google Scholar] [CrossRef]
- Singh, S.; Topuz, F.; Hahn, K.; Albrecht, K.; Groll, J. Embedding of active proteins and living cells in redox-sensitive hydrogels and nanogels through enzymatic cross-linking. Angew. Chem. Int. Ed. 2013, 52, 3000–3003. [Google Scholar] [CrossRef]
- Chirila, T.V.; Suzuki, S.; Papolla, C. A comparative investigation of bombyx mori silk fibroin hydrogels generated by chemical and enzymatic cross-linking. Biotechnol. Appl. Biochem. 2017, 64, 771–781. [Google Scholar] [CrossRef]
- Hou, S.; Lake, R.; Park, S.; Edwards, S.; Jones, C.; Jeong, K.J. Injectable macroporous hydrogel formed by enzymatic cross-linking of gelatin microgels. ACS Appl. Bio Mater. 2018, 1, 1430–1439. [Google Scholar] [CrossRef]
- Williams, D.F. On the mechanisms of biocompatibility. Biomaterials 2008, 29, 2941–2953. [Google Scholar] [CrossRef]
- Rutz, A.L.; Shah, R.N. Protein-Based Hydrogels. In Polymeric Hydrogels as Smart Biomaterials; Kalia, S., Ed.; Springer Series on Polymer and Composite Materials; Springer: Berlin/Heidelberg, Germany, 2016; pp. 73–104. [Google Scholar] [CrossRef]
- Rafat, M.; Li, F.; Fagerholm, P.; Lagali, N.S.; Watsky, M.A.; Munger, R.; Matsuura, T.; Griffith, M. PEG-stabilized carbodiimide crosslinked collagen–chitosan hydrogels for corneal tissue engineering. Biomaterials 2008, 29, 3960–3972. [Google Scholar] [CrossRef]
- Cirino, P.C.; Qian, S. Protein Engineering as an Enabling Tool for Synthetic Biology. In Synthetic Biology; Elsevier: Amsterdam, The Netherlands, 2013; pp. 23–42. ISBN 9780123944306. [Google Scholar]
- Apostolovic, B.; Danial, M.; Klok, H.-A. Coiled coils: Attractive protein folding motifs for the fabrication of self-assembled, responsive and bioactive materials. Chem. Soc. Rev. 2010, 39, 3541. [Google Scholar] [CrossRef] [PubMed]
- Hill, L.K.; Meleties, M.; Katyal, P.; Xie, X.; Delgado-Fukushima, E.; Jihad, T.; Liu, C.F.; O’Neill, S.; Tu, R.S.; Renfrew, P.D.; et al. Thermoresponsive protein-engineered coiled-coil hydrogel for sustained small molecule release. Biomacromolecules 2019, 20, 3340–3351. [Google Scholar] [CrossRef] [PubMed]
- Hansel, T.T.; Kropshofer, H.; Singer, T.; Mitchell, J.A.; George, A.J.T. The safety and side effects of monoclonal antibodies. Nat. Rev. Drug Discov. 2010, 9, 325–338. [Google Scholar] [CrossRef]
- Bugelski, P.J.; Martin, P.L. Concordance of preclinical and clinical pharmacology and toxicology of therapeutic monoclonal antibodies and fusion proteins: Cell surface targets. Br. J. Pharmacol. 2012, 166, 823–846. [Google Scholar] [CrossRef]
- Pola, R.; Laga, R.; Ulbrich, K.; Sieglova, I.; Kral, V.; Fabry, M.; Kabesova, M.; Kovar, M.; Pechar, M. Polymer therapeutics with a coiled coil motif targeted against murine BCL1 leukemia. Biomacromolecules 2013, 14, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Thanasupawat, T.; Bergen, H.; Hombach-Klonisch, S.; Krcek, J.; Ghavami, S.; Del Bigio, M.R.; Krawitz, S.; Stelmack, G.; Halayko, A.; McDougall, M.; et al. Platinum (IV) coiled coil nanotubes selectively kill human glioblastoma cells. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 913–925. [Google Scholar] [CrossRef]
- Fischer, S.E.; Liu, X.; Mao, H.-Q.; Harden, J.L. Controlling cell adhesion to surfaces via associating bioactive triblock proteins. Biomaterials 2007, 28, 3325–3337. [Google Scholar] [CrossRef]
- Huang, C.-C.; Ravindran, S.; Yin, Z.; George, A. 3-D self-assembling leucine zipper hydrogel with tunable properties for tissue engineering. Biomaterials 2014, 35, 5316–5326. [Google Scholar] [CrossRef] [Green Version]
- Wheeldon, I.R.; Campbell, E.; Banta, S. A chimeric fusion protein engineered with disparate functionalities—Enzymatic activity and self–assembly. J. Mol. Biol. 2009, 392, 129–142. [Google Scholar] [CrossRef]
- Banta, S.; Wheeldon, I.R.; Blenner, M. Protein engineering in the development of functional hydrogels. Annu. Rev. Biomed. Eng. 2010, 12, 167–186. [Google Scholar] [CrossRef] [Green Version]
- Massodi, I.; Thomas, E.; Raucher, D. Application of thermally responsive elastin-like polypeptide fused to a lactoferrin-derived peptide for treatment of pancreatic cancer. Molecules 2009, 14, 1999–2015. [Google Scholar] [CrossRef] [PubMed]
- Bian, Q.; Fu, L.; Li, H. Engineering shape memory and morphing protein hydrogels based on protein unfolding and folding. Nat. Commun. 2022, 13, 137. [Google Scholar] [CrossRef]
- Zhang, X.; Chu, X.; Wang, L.; Wang, H.; Liang, G.; Zhang, J.; Long, J.; Yang, Z. Rational design of a tetrameric protein to enhance interactions between self-assembled fibers gives molecular hydrogels. Angew. Chem. Int. Ed. 2012, 51, 4388–4392. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Hudson, A.R.; Shiwarski, D.J.; Tashman, J.W.; Hinton, T.J.; Yerneni, S.; Bliley, J.M.; Campbell, P.G.; Feinberg, A.W. 3D bioprinting of collagen to rebuild components of the human heart. Science 2019, 365, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Jang, J.; Park, J.; Lee, K.-P.; Lee, S.; Lee, D.-M.; Kim, K.H.; Kim, H.K.; Cho, D.-W. Shear-induced alignment of collagen fibrils using 3D cell printing for corneal stroma tissue engineering. Biofabrication 2019, 11, 035017. [Google Scholar] [CrossRef]
- Yang, X.; Lu, Z.; Wu, H.; Li, W.; Zheng, L.; Zhao, J. Collagen-alginate as bioink for three-dimensional (3D) cell printing based cartilage tissue engineering. Mater. Sci. Eng. C 2018, 83, 195–201. [Google Scholar] [CrossRef]
- Simorgh, S.; Milan, P.B.; Saadatmand, M.; Bagher, Z.; Gholipourmalekabadi, M.; Alizadeh, R.; Hivechi, A.; Arabpour, Z.; Hamidi, M.; Delattre, C. Human olfactory mucosa stem cells delivery using a collagen hydrogel: As a potential candidate for bone tissue engineering. Materials 2021, 14, 3909. [Google Scholar] [CrossRef]
- Nichol, J.W.; Koshy, S.T.; Bae, H.; Hwang, C.M.; Yamanlar, S.; Khademhosseini, A. Cell-laden microengineered gelatin methacrylate hydrogels. Biomaterials 2010, 31, 5536–5544. [Google Scholar] [CrossRef] [Green Version]
- Balakrishnan, B.; Joshi, N.; Jayakrishnan, A.; Banerjee, R. Self-crosslinked oxidized alginate/gelatin hydrogel as injectable, adhesive biomimetic scaffolds for cartilage regeneration. Acta Biomater. 2014, 10, 3650–3663. [Google Scholar] [CrossRef]
- Satapathy, M.K.; Nyambat, B.; Chiang, C.W.; Chen, C.H.; Wong, P.C.; Ho, P.H.; Jheng, P.R.; Burnouf, T.; Tseng, C.L.; Chuang, E.Y. A gelatin hydrogel-containing nano-organic PEI–Ppy with a photothermal responsive effect for tissue engineering applications. Molecules 2018, 23, 1256. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Lang, Q.; Yildirimer, L.; Lin, Z.Y.; Cui, W.; Annabi, N.; Ng, K.W.; Dokmeci, M.R.; Ghaemmaghami, A.M.; Khademhosseini, A. Photocrosslinkable Gelatin Hydrogel for Epidermal Tissue Engineering. Adv. Healthc. Mater. 2016, 5, 108–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y.; Gao, L.; Peng, J.; Xing, M.; Han, Y.; Wang, X.; Xu, Y.; Chang, J. Bioglass activated albumin hydrogels for wound healing. Adv. Healthc. Mater. 2018, 7, e1800144. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Sun, J.; Sun, Y.; Xiang, Y.; Yan, Y.; Han, Z.; Bi, W.; Yang, F.; Zhou, Q.; Wang, L.; et al. Multifunctional injectable protein-based hydrogel for bone regeneration. Chem. Eng. J. 2020, 394, 124875. [Google Scholar] [CrossRef]
- Yuan, H.; Zheng, X.; Liu, W.; Zhang, H.; Shao, J.; Yao, J.; Mao, C.; Hui, J.; Fan, D. A novel bovine serum albumin and sodium alginate hydrogel scaffold doped with hydroxyapatite nanowires for cartilage defects repair. Colloids Surf. B Biointerfaces 2020, 192, 111041. [Google Scholar] [CrossRef]
- Amdursky, N.; Mazo, M.M.; Thomas, M.R.; Humphrey, E.J.; Puetzer, J.L.; St-Pierre, J.-P.; Skaalure, S.C.; Richardson, R.M.; Terracciano, C.M.; Stevens, M.M. Elastic serum-albumin based hydrogels: Mechanism of formation and application in cardiac tissue engineering. J. Mater. Chem. B 2018, 6, 5604–5612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Ali, M.S.; Lacerda, C.M.R. A three-dimensional collagen-elastin scaffold for heart valve tissue engineering. Bioengineering 2018, 5, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stojic, M.; Rodenas-Rochina, J.; Lopez-Donaire, M.L.; Gonzalez de Torre, I.; Gonzalez Perez, M.; Rodriguez-Cabello, J.C.; Vojtova, L.; Jorcano, J.L.; Velasco, D. Elastin-plasma hybrid hydrogels for skin tissue engineering. Polymers 2021, 13, 2114. [Google Scholar] [CrossRef]
- Pal, P.; Nguyen, Q.C.; Benton, A.H.; Marquart, M.E.; Janorkar, A.V. Drug-loaded elastin-like polypeptide–collagen hydrogels with high modulus for bone tissue engineering. Macromol. Biosci. 2019, 19, 1900142. [Google Scholar] [CrossRef]
- Staubli, S.M.; Cerino, G.; De Torre, I.G.; Alonso, M.; Oertli, D.; Eckstein, F.; Glatz, K.; Cabello, J.C.R.; Marsano, A. Control of angiogenesis and host response by modulating the cell adhesion properties of an elastin-like recombinamer-based hydrogel. Biomaterials 2017, 135, 30–41. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Cai, S.; Xu, L.; Yang, Y. Water-stable three-dimensional ultrafine fibrous scaffolds from keratin for cartilage tissue engineering. Langmuir 2014, 30, 8461–8470. [Google Scholar] [CrossRef] [Green Version]
- Veerasubramanian, P.K.; Thangavel, P.; Kannan, R.; Chakraborty, S.; Ramachandran, B.; Suguna, L.; Muthuvijayan, V. An investigation of konjac glucomannan-keratin hydrogel scaffold loaded with avena sativa extracts for diabetic wound healing. Colloids Surf. B Biointerfaces 2018, 165, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, Y.; Yang, X.; Cao, Z.; Nie, H.; Bian, Y.; Yang, G. Glucose-triggered in situ forming keratin hydrogel for the treatment of diabetic wounds. Acta Biomater. 2021, 125, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Yao, Y.; Li, Y.; Yang, X.; Cao, Z.; Yang, G. Tunable keratin hydrogel based on disulfide shuffling strategy for drug delivery and tissue engineering. J. Colloid Interface Sci. 2019, 544, 121–129. [Google Scholar] [CrossRef]
- Renner, J.N.; Cherry, K.M.; Su, R.S.-C.; Liu, J.C. Characterization of resilin-based materials for tissue engineering applications. Biomacromolecules 2012, 13, 3678–3685. [Google Scholar] [CrossRef] [PubMed]
- McGann, C.L.; Levenson, E.A.; Kiick, K.L. Resilin-based hybrid hydrogels for cardiovascular tissue engineering. Macromol. Chem. Phys. 2013, 214, 203–213. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Stiadle, J.M.; Levendoski, E.E.; Lau, H.K.; Thibeault, S.L.; Kiick, K.L. Biocompatibility of injectable resilin-based hydrogels. J. Biomed. Mater. Res. Part A 2018, 106, 2229–2242. [Google Scholar] [CrossRef]
- Su, R.S.C.; Galas, R.J.; Lin, C.; Liu, J.C. Redox-responsive resilin-like hydrogels for tissue engineering and drug delivery applications. Macromol. Biosci. 2019, 19, e1900122. [Google Scholar] [CrossRef] [PubMed]
- Patra, C.; Talukdar, S.; Novoyatleva, T.; Velagala, S.R.; Muhlfeld, C.; Kundu, B.; Kundu, S.C.; Engel, F.B. Silk protein fibroin from Antheraea mylitta for cardiac tissue engineering. Biomaterials 2012, 33, 2673–2680. [Google Scholar] [CrossRef] [Green Version]
- Moses, J.C.; Saha, T.; Mandal, B.B. Chondroprotective and osteogenic effects of silk-based bioinks in developing 3D bioprinted osteochondral interface. Bioprinting 2020, 17, e00067. [Google Scholar] [CrossRef]
- Hong, H.; Seo, Y.B.; Kim, D.Y.; Lee, J.S.; Lee, Y.J.; Lee, H.; Ajiteru, O.; Sultan, M.T.; Lee, O.J.; Kim, S.H.; et al. Digital light processing 3D printed silk fibroin hydrogel for cartilage tissue engineering. Biomaterials 2020, 232, 119679. [Google Scholar] [CrossRef]
- Pankongadisak, P.; Suwantong, O. Enhanced properties of injectable chitosan-based thermogelling hydrogels by silk fibroin and longan seed extract for bone tissue engineering. Int. J. Biol. Macromol. 2019, 138, 412–424. [Google Scholar] [CrossRef] [PubMed]
- Latorre, M.E.; Velazquez, D.E.; Purslow, P.P. Differences in the energetics of collagen denaturation in connective tissue from two muscles. Int. J. Biol. Macromol. 2018, 113, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Petcharat, T.; Benjakul, S.; Karnjanapratum, S.; Nalinanon, S. Ultrasound-assisted extraction of collagen from clown featherback (Chitala ornata) skin: Yield and molecular characteristics. J. Sci. Food Agric. 2021, 101, 648–658. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, V.D.M.; Assis, C.R.D.; Costa, B.D.A.M.; Neri, R.C.D.A.; Monte, F.T.D.; Freitas, H.M.S.D.C.V.; França, R.C.P.; Santos, J.F.; Bezerra, R.D.S.; Porto, A.L.F. Physical, biochemical, densitometric and spectroscopic techniques for characterization collagen from alternative sources: A review based on the sustainable valorization of aquatic by-products. J. Mol. Struct. 2021, 1224, 129023. [Google Scholar] [CrossRef]
- Song, Z.; Liu, H.; Chen, L.; Chen, L.; Zhou, C.; Hong, P.; Deng, C. Characterization and comparison of collagen extracted from the skin of the Nile tilapia by fermentation and chemical pretreatment. Food Chem. 2021, 340, 128139. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Gao, Y.; Shao, J.; Tumarbekova, A.; Zhang, D.; Zhu, J. Effects of ultrasound and thermal treatment on the ultrastructure of collagen fibers from bovine tendon using atomic force microscopy. Food Chem. 2021, 347, 128985. [Google Scholar] [CrossRef]
- McTiernan, C.D.; Simpson, F.C.; Haagdorens, M.; Samarawickrama, C.; Hunter, D.; Buznyk, O.; Fagerholm, P.; Ljunggren, M.K.; Lewis, P.; Pintelon, I.; et al. LiQD cornea: Pro-regeneration collagen mimetics as patches and alternatives to corneal transplantation. Sci. Adv. 2020, 6. [Google Scholar] [CrossRef]
- Silvipriya, K.; Kumar, K.; Bhat, A.; Kumar, B.; John, A.; Lakshmanan, P. Collagen: Animal sources and biomedical application. J. Appl. Pharm. Sci. 2015, 5, 123–127. [Google Scholar] [CrossRef] [Green Version]
- Hollmen, T.; Pikkarainen, J. Fibrous proteins. In Methods in Molecular Biology; Ling, S., Ed.; Springer: New York, NY, USA, 2021; Volume 2347, ISBN 978-1-0716-1573-7. [Google Scholar]
- Bhadra, B.; Sakpal, A.; Patil, S.; Patil, S.; Date, A.; Prasad, V.; Dasgupta, S. A Guide to Collagen Sources, Applications and Current Advancements. Syst. Biosci. Eng. 2021, 1, 67–87. [Google Scholar] [CrossRef]
- Maity, P.P.; Dutta, D.; Ganguly, S.; Kapat, K.; Dixit, K.; Chowdhury, A.R.; Samanta, R.; Das, N.C.; Datta, P.; Das, A.K.; et al. Isolation and mass spectrometry based hydroxyproline mapping of type II collagen derived from Capra hircus ear cartilage. Commun. Biol. 2019, 2, 1–11. [Google Scholar] [CrossRef]
- Ghorbani, F.; Zamanian, A.; Kermanian, F.; Shamoosi, A. A bioinspired 3D shape olibanum-collagen-gelatin scaffolds with tunable porous microstructure for efficient neural tissue regeneration. Biotechnol. Prog. 2020, 36, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Fu, Y.; Huang, S.; Liao, L.; Wu, Q.; Wang, Y.; Ge, F.; Fang, B. Identification and antioxidant activity of bovine bone collagen-derived novel peptides prepared by recombinant collagenase from Bacillus cereus. Food Chem. 2021, 349, 129143. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Fan, H.; Roy, B.C.; Wu, J. Amylase enhances production of low molecular weight collagen peptides from the skin of spent hen, bovine, porcine, and tilapia. Food Chem. 2021, 352, 129355. [Google Scholar] [CrossRef] [PubMed]
- Subhan, F.; Hussain, Z.; Tauseef, I.; Shehzad, A.; Wahid, F. A review on recent advances and applications of fish collagen. Crit. Rev. Food Sci. Nutr. 2021, 61, 1027–1037. [Google Scholar] [CrossRef]
- Pozzolini, M.; Tassara, E.; Dodero, A.; Castellano, M.; Vicini, S.; Ferrando, S.; Aicardi, S.; Cavallo, D.; Bertolino, M.; Petrenko, I.; et al. Potential biomedical applications of collagen filaments derived from the marine demosponges ircinia oros (Schmidt, 1864) and sarcotragus foetidus (Schmidt, 1862). Mar. Drugs 2021, 19, 563. [Google Scholar] [CrossRef] [PubMed]
- Kemp, D.C.; Kwon, J.Y. Fish and shellfish-derived anti-inflammatory protein products: Properties and mechanisms. Molecules 2021, 26, 3225. [Google Scholar] [CrossRef]
- Ahmed, Z.; Powell, L.C.; Matin, N.; Mearns-Spragg, A.; Thornton, C.A.; Khan, I.M.; Francis, L.W. Jellyfish collagen: A biocompatible collagen source for 3d scaffold fabrication and enhanced chondrogenicity. Mar. Drugs 2021, 19, 405. [Google Scholar] [CrossRef]
- Coppola, D.; Oliviero, M.; Vitale, G.A.; Lauritano, C.; D’Ambra, I.; Iannace, S.; de Pascale, D. Marine collagen from alternative and sustainable sources: Extraction, processing and applications. Mar. Drugs 2020, 18, 214. [Google Scholar] [CrossRef] [Green Version]
- Tian, M.L.; Zhou, J.F.; Qi, X.; Shen, R. Thermo-sensitive hydrogel and their biomedical applications. IOP Conf. Ser. Earth Environ. Sci. 2021, 714, 032062. [Google Scholar] [CrossRef]
- Sarrigiannidis, S.O.; Rey, J.M.; Dobre, O.; Gonzalez-Garcia, C.; Dalby, M.J.; Salmeron-Sanchez, M. A tough act to follow: Collagen hydrogel modifications to improve mechanical and growth factor loading capabilities. Mater. Today Bio 2021, 10, 100098. [Google Scholar] [CrossRef]
- Salvatore, L.; Gallo, N.; Natali, M.L.; Terzi, A.; Sannino, A.; Madaghiele, M. Mimicking the hierarchical organization of natural collagen: Toward the development of ideal scaffolding material for tissue regeneration. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ouyang, L.; Armstrong, J.P.K.; Stevens, M.M. Advances in the fabrication of biomaterials for gradient tissue engineering. Trends Biotechnol. 2021, 39, 150–164. [Google Scholar] [CrossRef] [PubMed]
- Antoine, E.E.; Vlachos, P.P.; Rylander, M.N. Review of collagen I hydrogels for bioengineered tissue microenvironments: Characterization of mechanics, structure, and transport. Tissue Eng. Part B Rev. 2014, 20, 683–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sorushanova, A.; Delgado, L.M.; Wu, Z.; Shologu, N.; Kshirsagar, A.; Raghunath, R.; Mullen, A.M.; Bayon, Y.; Pandit, A.; Raghunath, M.; et al. The collagen suprafamily: From biosynthesis to advanced biomaterial development. Adv. Mater. 2019, 31, e1801651. [Google Scholar] [CrossRef] [Green Version]
- Ashokkumar, M.; Ajayan, P.M. Materials science perspective of multifunctional materials derived from collagen. Int. Mater. Rev. 2021, 66, 160–187. [Google Scholar] [CrossRef]
- Zhang, T.; Yu, Z.; Ma, Y.; Chiou, B.-S.; Liu, F.; Zhong, F. Modulating physicochemical properties of collagen films by cross-linking with glutaraldehyde at varied pH values. Food Hydrocoll. 2022, 124, 107270. [Google Scholar] [CrossRef]
- Cho, W.T.; Kim, S.Y.; Jung, S.I.; Kang, S.S.; Kim, S.E.; Hwang, S.H.; Jeong, C.M.; Huh, J.B. Effects of gamma radiation-induced crosslinking of collagen type i coated dental titanium implants on osseointegration and bone regeneration. Materials 2021, 14, 3268. [Google Scholar] [CrossRef]
- Grabska-Zielinska, S.; Sionkowska, A.; Carvalho, A.; Monteiro, F.J. Biomaterials with potential use in bone tissue regeneration-collagen/chitosan/silk fibroin scaffolds cross-linked by EDC/NHS. Materials 2021, 14, 1105. [Google Scholar] [CrossRef]
- Bax, D.V.; Davidenko, N.; Gullberg, D.; Hamaia, S.W.; Farndale, R.W.; Best, S.M.; Cameron, R.E. Fundamental insight into the effect of carbodiimide crosslinking on cellular recognition of collagen-based scaffolds. Acta Biomater. 2017, 49, 218–234. [Google Scholar] [CrossRef]
- Wang, Y.; Ruan, Y.; Du, B.; Li, J.; Ebendorff-Heidepriem, H.; Wang, X. Real-time raman analysis of the hydrolysis of formaldehyde oligomers for enhanced collagen fixation. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 264, 120285. [Google Scholar] [CrossRef]
- Leon-Campos, M.I.; Claudio-Rizo, J.A.; Rodriguez-Fuentes, N.; Cabrera-Munguia, D.A.; Becerra-Rodriguez, J.J.; Herrera-Guerrero, A.; Soriano-Corral, F. Biocompatible interpenetrating polymeric networks in hydrogel state comprised from jellyfish collagen and polyurethane. J. Polym. Res. 2021, 28, 1–15. [Google Scholar] [CrossRef]
- Li, Z.; Du, T.; Ruan, C.; Niu, X. Bioinspired mineralized collagen scaffolds for bone tissue engineering. Bioact. Mater. 2021, 6, 1491–1511. [Google Scholar] [CrossRef]
- Yu, Y.; Xu, S.; Li, S.; Pan, H. Genipin-cross-linked hydrogels based on biomaterials for drug delivery: A review. Biomater. Sci. 2021, 9, 1583–1597. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Wang, W.; Li, Y.; Gao, G.; Zhang, K.; Zhou, J.; Wu, Z. Cross-linking and film-forming properties of transglutaminase-modified collagen fibers tailored by denaturation temperature. Food Chem. 2019, 271, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Grabska-Zielinska, S.; Sionkowska, A.; Reczynska, K.; Pamula, E. Physico-chemical characterization and biological tests of collagen/silk fibroin/chitosan scaffolds cross-linked by dialdehyde starch. Polymers 2020, 12, 372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radenkovic, M.; Alkildani, S.; Stoewe, I.; Bielenstein, J.; Sundag, B.; Bellmann, O.; Jung, O.; Najman, S.; Stojanovic, S.; Barbeck, M. Comparative in vivo analysis of the integration behavior and immune response of collagen-based dental barrier membranes for guided bone regeneration (GBR). Membranes 2021, 11, 712. [Google Scholar] [CrossRef]
- Vazquez-Portalatin, N.; Alfonso-Garcia, A.; Liu, J.C.; Marcu, L.; Panitch, A. Physical, biomechanical, and optical characterization of collagen and elastin blend hydrogels. Ann. Biomed. Eng. 2020, 48, 2924–2935. [Google Scholar] [CrossRef]
- Dille, M.J.; Haug, I.J.; Draget, K.I. Gelatin and collagen. In Handbook of Hydrocolloids; Elsevier: Amsterdam, The Netherlands, 2021; pp. 1073–1097. [Google Scholar]
- Ghorbani, F.; Zamanian, A.; Shams, A.; Shamoosi, A.; Aidun, A. Fabrication and characterisation of super-paramagnetic responsive PLGA-gelatine-magnetite scaffolds with the unidirectional porous structure: A physicochemical, mechanical, and in vitro evaluation. IET Nanobiotechnol. 2019, 13, 860–867. [Google Scholar] [CrossRef]
- Jaipan, P.; Nguyen, A.; Narayan, R.J. Gelatin-based hydrogels for biomedical applications. MRS Commun. 2017, 7, 416–426. [Google Scholar] [CrossRef]
- Kang, J.I.; Park, K.M. Advances in gelatin-based hydrogels for wound management. J. Mater. Chem. B 2021, 9, 1503–1520. [Google Scholar] [CrossRef]
- Rigueto, C.V.T.; Nazari, M.T.; Massuda, L.A.; Ostwald, B.E.P.; Piccin, J.S.; Dettmer, A. Production and environmental applications of gelatin-based composite adsorbents for contaminants removal: A review. Environ. Chem. Lett. 2021, 19, 2465–2486. [Google Scholar] [CrossRef]
- Samadian, H.; Maleki, H.; Allahyari, Z.; Jaymand, M. Natural polymers-based light-induced hydrogels: Promising biomaterials for biomedical applications. Coord. Chem. Rev. 2020, 420, 213432. [Google Scholar] [CrossRef]
- Mamidi, N.; Villela Castrejon, J.; Gonzalez-Ortiz, A. Rational design and engineering of carbon nano-onions reinforced natural protein nanocomposite hydrogels for biomedical applications. J. Mech. Behav. Biomed. Mater. 2020, 104, 103696. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Duan, Q.; Yu, L.; Xie, F. Thermomechanically processed chitosan:gelatin films being transparent, mechanically robust and less hygroscopic. Carbohydr. Polym. 2021, 272, 118522. [Google Scholar] [CrossRef]
- Ghorbani, F.; Zamanian, A.; Nojehdehian, H. Effects of pore orientation on in-vitro properties of retinoic acid-loaded PLGA/gelatin scaffolds for artificial peripheral nerve application. Mater. Sci. Eng. C 2017, 77, 159–172. [Google Scholar] [CrossRef]
- Kruger-Genge, A.; Hauser, S.; Neffe, A.T.; Liu, Y.; Lendlein, A.; Pietzsch, J.; Jung, F. Response of endothelial cells to gelatin-based hydrogels. ACS Biomater. Sci. Eng. 2021, 7, 527–540. [Google Scholar] [CrossRef]
- Ghanbari, M.; Salavati-Niasari, M.; Mohandes, F.; Firouzi, Z. Modified silicon carbide NPs reinforced nanocomposite hydrogels based on alginate-gelatin by with high mechanical properties for tissue engineering. Arab. J. Chem. 2022, 15, 103520. [Google Scholar] [CrossRef]
- Teimouri, S.; Kasapis, S. Mechanistic interpretation of vitamin B6 transport from swelling matrices of genipin-crosslinked gelatin, BSA and WPI. Food Hydrocoll. 2022, 123, 107195. [Google Scholar] [CrossRef]
- Jahanban-Esfahlan, A.; Ostadrahimi, A.; Jahanban-Esfahlan, R.; Roufegarinejad, L.; Tabibiazar, M.; Amarowicz, R. Recent developments in the detection of bovine serum albumin. Int. J. Biol. Macromol. 2019, 138, 602–617. [Google Scholar] [CrossRef]
- Ghalandari, B.; Asadollahi, K.; Ghorbani, F.; Ghalehbaghi, S.; Rafiee, S.; Komeili, A.; Kamrava, S.K. Determinants of gold nanoparticle interactions with Proteins: Off-Target effect study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 269, 120736. [Google Scholar] [CrossRef]
- Nourizadeh, S.; Divsalar, A.; Fekri, M.; Ghalandari, B.; Eslami-Moghadam, M.; Akbar Saboury, A.; Abbasi, S. Multiple Spectroscopic, docking and cytotoxic study of a synthesized 2,2′ bipyridin phenyl isopentylglycin pt(ii) nitrate complex: Human Serum albumin and breast cancer cell line of MDA-MB231 as targets. J. Fluoresc. 2018, 28, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Ong, J.; Zhao, J.; Justin, A.W.; Markaki, A.E. Albumin-based hydrogels for regenerative engineering and cell transplantation. Biotechnol. Bioeng. 2019, 116, 3457–3468. [Google Scholar] [CrossRef] [PubMed]
- Mamidi, N.; Delgadillo, R.M.V.; Gonzalez-Ortiz, A. Engineering of carbon nano-onion bioconjugates for biomedical applications. Mater. Sci. Eng. C 2021, 120, 111698. [Google Scholar] [CrossRef] [PubMed]
- Lantigua, D.; Nguyen, M.A.; Wu, X.; Suvarnapathaki, S.; Kwon, S.; Gavin, W.; Camci-Unal, G. Synthesis and characterization of photocrosslinkable albumin-based hydrogels for biomedical applications. Soft Matter 2020, 16, 9242–9252. [Google Scholar] [CrossRef] [PubMed]
- Fiorica, C.; Palumbo, F.S.; Pitarresi, G.; Allegra, M.; Puleio, R.; Giammona, G. Hyaluronic acid and α-elastin based hydrogel for three dimensional culture of vascular endothelial cells. J. Drug Deliv. Sci. Technol. 2018, 46, 28–33. [Google Scholar] [CrossRef]
- Zhu, D.; Wang, H.; Trinh, P.; Heilshorn, S.C.; Yang, F. Elastin-like protein-hyaluronic acid (ELP-HA) hydrogels with decoupled mechanical and biochemical cues for cartilage regeneration. Biomaterials 2017, 127, 132–140. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.; Sani, E.S.; Spencer, A.R.; Guan, Y.; Weiss, A.S.; Annabi, N. Human-recombinant-elastin-based bioinks for 3d bioprinting of vascularized soft tissues. Adv. Mater. 2020, 32, e2003915. [Google Scholar] [CrossRef]
- Dunphy, S.E.; Bratt, J.A.J.; Akram, K.M.; Forsyth, N.R.; El Haj, A.J. Hydrogels for lung tissue engineering: Biomechanical properties of thin collagen-elastin constructs. J. Mech. Behav. Biomed. Mater. 2014, 38, 251–259. [Google Scholar] [CrossRef]
- Almeida, H.; Domingues, R.M.A.; Mithieux, S.M.; Pires, R.A.; Gonçalves, A.I.; Gomez-Florit, M.; Reis, R.L.; Weiss, A.S.; Gomes, M.E. Tropoelastin-coated tendon biomimetic scaffolds promote stem cell tenogenic commitment and deposition of elastin-rich matrix. ACS Appl. Mater. Interfaces 2019, 11, 19830–19840. [Google Scholar] [CrossRef]
- Costa, F.; Silva, R.; Boccaccini, A.R. Fibrous protein-based biomaterials (silk, keratin, elastin, and resilin proteins) for tissue regeneration and repair. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Elsevier: Amsterdam, The Netherlands, 2018; pp. 175–204. [Google Scholar]
- Keleshteri, A.R.; Moztarzadeh, F.; Farokhi, M.; Mehrizi, A.A.; Basiri, H.; Mohseni, S.S. Preparation of microfluidic-based pectin microparticles loaded carbon dots conjugated with BMP-2 embedded in gelatin-elastin-hyaluronic acid hydrogel scaffold for bone tissue engineering application. Int. J. Biol. Macromol. 2021, 184, 29–41. [Google Scholar] [CrossRef]
- McKittrick, J.; Chen, P.-Y.; Bodde, S.G.; Yang, W.; Novitskaya, E.E.; Meyers, M.A. The structure, functions, and mechanical properties of keratin. JOM 2012, 64, 449–468. [Google Scholar] [CrossRef]
- Park, M.; Shin, H.K.; Kim, B.-S.; Kim, M.J.; Kim, I.-S.; Park, B.-Y.; Kim, H.-Y. Effect of discarded keratin-based biocomposite hydrogels on the wound healing process in vivo. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 55, 88–94. [Google Scholar] [CrossRef]
- Calo, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef] [Green Version]
- Cao, G.; Rong, M.Z.; Zhang, M.Q. Continuous high-content keratin fibers with balanced properties derived from wool waste. ACS Sustain. Chem. Eng. 2020, 8, 18148–18156. [Google Scholar] [CrossRef]
- Ye, J.-P.; Gong, J.-S.; Su, C.; Liu, Y.-G.; Jiang, M.; Pan, H.; Li, R.-Y.; Geng, Y.; Xu, Z.-H.; Shi, J.-S. Fabrication and characterization of high molecular keratin based nanofibrous membranes for wound healing. Colloids Surf. B Biointerfaces 2020, 194, 111158. [Google Scholar] [CrossRef] [PubMed]
- Dou, Y.; Zhang, L.; Zhang, B.; He, M.; Shi, W.; Yang, S.; Cui, Y.; Yin, G. Preparation and characterization of edible dialdehyde carboxymethyl cellulose crosslinked feather keratin films for food packaging. Polymers 2020, 12, 158. [Google Scholar] [CrossRef] [Green Version]
- Nuutinen, E.M.; Virtanen, T.; Lantto, R.; Vaha-Nissi, M.; Jaaskelainen, A.S. Ductile keratin films from deep eutectic solvent-fractionated feathers. RSC Adv. 2021, 11, 27512–27522. [Google Scholar] [CrossRef]
- Patil, N.V.; Netravali, A.N. Enhancing strength of wool fiber using a soy flour sugar-based “green” cross-linker. ACS Omega 2019, 4, 5392–5401. [Google Scholar] [CrossRef]
- Tinoco, A.; Rodrigues, R.M.; Machado, R.; Pereira, R.N.; Cavaco-Paulo, A.; Ribeiro, A. Ohmic heating as an innovative approach for the production of keratin films. Int. J. Biol. Macromol. 2020, 150, 671–680. [Google Scholar] [CrossRef] [Green Version]
- Feroz, S.; Muhammad, N.; Ratnayake, J.; Dias, G. Keratin—Based materials for biomedical applications. Bioact. Mater. 2020, 5, 496–509. [Google Scholar] [CrossRef]
- Li, L.; Mahara, A.; Tong, Z.; Levenson, E.A.; Mcgann, C.L.; Jia, X.; Yamaoka, T.; Kiick, K.L. Recombinant resilin-based bioelastomers for regenerative medicine applications. Adv. Healthc. Mater. 2016, 5, 266–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abascal, N.C.; Regan, L. The past, present and future of protein-based materials. Open Biol. 2018, 8, 180113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, G.; Hu, X.; Cebe, P.; Kaplan, D.L. Mechanism of resilin elasticity. Nat. Commun. 2012, 3, 1003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Kiick, K.L. Resilin-based materials for biomedical applications. ACS Macro Lett. 2013, 2, 635–640. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, W.; Gregory, D.A.; Tomeh, M.A.; Zhao, X. Silk fibroin as a functional biomaterial for tissue engineering. Int. J. Mol. Sci. 2021, 22, 1499. [Google Scholar] [CrossRef]
- Zuluaga-Velez, A.; Combita-Merchan, D.F.; Buitrago-Sierra, R.; Santa, J.F.; Aguilar-Fernandez, E.; Sepulveda-Arias, J.C. Silk fibroin hydrogels from the Colombian silkworm bombyx mori l: Evaluation of physicochemical properties. PLoS ONE 2019, 14, e0213303. [Google Scholar] [CrossRef]
- Chawla, S.; Midha, S.; Sharma, A.; Ghosh, S. Silk-based bioinks for 3D bioprinting. Adv. Healthc. Mater. 2018, 7, 1701204. [Google Scholar] [CrossRef]
- Cheng, B.; Yan, Y.; Qi, J.; Deng, L.; Shao, Z.-W.; Zhang, K.-Q.; Li, B.; Sun, Z.; Li, X. Cooperative assembly of a peptide gelator and silk fibroin afford an injectable hydrogel for tissue engineering. ACS Appl. Mater. Interfaces 2018, 10, 12474–12484. [Google Scholar] [CrossRef]
- Kapoor, S.; Kundu, S.C. Silk protein-based hydrogels: Promising advanced materials for biomedical applications. Acta Biomater. 2016, 31, 17–32. [Google Scholar] [CrossRef]
- Lv, L.; Han, X.; Zong, L.; Li, M.; You, J.; Wu, X.; Li, C. Biomimetic hybridization of kevlar into silk fibroin: Nanofibrous strategy for improved mechanic properties of flexible composites and filtration membranes. ACS Nano 2017, 11, 8178–8184. [Google Scholar] [CrossRef]
- Rodriguez, M.J.; Brown, J.; Giordano, J.; Lin, S.J.; Omenetto, F.G.; Kaplan, D.L. Silk based bioinks for soft tissue reconstruction using 3-dimensional (3D) printing with in vitro and in vivo assessments. Biomaterials 2017, 117, 105–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Yu, X.; Li, Y.; Su, Z.; Jandt, K.D.; Wei, G. Protein-mimetic peptide nanofibers: Motif design, self-assembly synthesis, and sequence-specific biomedical applications. Prog. Polym. Sci. 2018, 80, 94–124. [Google Scholar] [CrossRef]
- Zhu, Y.; Song, K.; Jiang, S.; Chen, J.; Tang, L.; Li, S.; Fan, J.; Wang, Y.; Zhao, J.; Liu, T. Numerical simulation of mass transfer and three-dimensional fabrication of tissue-engineered cartilages based on chitosan/gelatin hybrid hydrogel scaffold in a rotating bioreactor. Appl. Biochem. Biotechnol. 2017, 181, 250–266. [Google Scholar] [CrossRef] [PubMed]
- Kurzyk, A.; Ostrowska, B.; Swiȩszkowski, W.; Pojda, Z. Characterization and optimization of the seeding process of adipose stem cells on the polycaprolactone scaffolds. Stem Cells Int. 2019, 2019, 1201927. [Google Scholar] [CrossRef]
- Aguilar, L.M.C.; Silva, S.M.; Moulton, S.E. Growth factor delivery: Defining the next generation platforms for tissue engineering. J. Control. Release 2019, 306, 40–58. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zhang, L.; Bao, Y.; Yan, X.; Yin, Y.; Li, Y.; Wang, X.; Huang, Z.; Xu, P. Preparation and characterization of injectable chitosan–hyaluronic acid hydrogels for nerve growth factor sustained release. J. Bioact. Compat. Polym. 2017, 32, 146–162. [Google Scholar] [CrossRef]
- Baker, H.B.; Passipieri, J.A.; Siriwardane, M.; Ellenburg, M.D.; Vadhavkar, M.; Bergman, C.R.; Saul, J.M.; Tomblyn, S.; Burnett, L.; Christ, G.J. Cell and growth factor-loaded keratin hydrogels for treatment of volumetric muscle loss in a mouse model. Tissue Eng. Part A 2017, 23, 572–584. [Google Scholar] [CrossRef]
- Jaatinen, L.; Voros, J.; Hyttinen, J. Controlling cell migration and adhesion into a scaffold by external electric currents. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; IEEE: New York, NY, USA, 2015; pp. 3549–3552. [Google Scholar]
- Chen, C.-Y.; Ke, C.-J.; Yen, K.-C.; Hsieh, H.-C.; Sun, J.-S.; Lin, F.-H. 3D porous calcium-alginate scaffolds cell culture system improved human osteoblast cell clusters for cell therapy. Theranostics 2015, 5, 643–655. [Google Scholar] [CrossRef] [Green Version]
- Villalona, G.A.; Udelsman, B.; Duncan, D.R.; McGillicuddy, E.; Sawh-Martinez, R.F.; Hibino, N.; Painter, C.; Mirensky, T.; Erickson, B.; Shinoka, T.; et al. Cell-seeding techniques in vascular tissue engineering. Tissue Eng. Part B Rev. 2010, 16, 341–350. [Google Scholar] [CrossRef]
- Yeatts, A.B.; Fisher, J.P. Bone tissue engineering bioreactors: Dynamic culture and the influence of shear stress. Bone 2011, 48, 171–181. [Google Scholar] [CrossRef]
- Gaspar, D.A.; Gomide, V.; Monteiro, F.J. The role of perfusion bioreactors in bone tissue engineering. Biomatter 2012, 2, 167–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neiman, J.A.S.; Raman, R.; Chan, V.; Rhoads, M.G.; Raredon, M.S.B.; Velazquez, J.J.; Dyer, R.L.; Bashir, R.; Hammond, P.T.; Griffith, L.G. Photopatterning of hydrogel scaffolds coupled to filter materials using stereolithography for perfused 3D culture of hepatocytes. Biotechnol. Bioeng. 2015, 112, 777–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pirosa, A.; Gottardi, R.; Alexander, P.G.; Tuan, R.S. Engineering in-vitro stem cell-based vascularized bone models for drug screening and predictive toxicology. Stem Cell Res. Ther. 2018, 9, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mulyasasmita, W.; Cai, L.; Hori, Y.; Heilshorn, S.C. Avidity-controlled delivery of angiogenic peptides from injectable molecular-recognition hydrogels. Tissue Eng. Part A 2014, 20, 2102–2114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Enriquez-Ochoa, D.; Robles-Ovalle, P.; Mayolo-Deloisa, K.; Brunck, M.E.G. Immobilization of growth factors for cell therapy manufacturing. Front. Bioeng. Biotechnol. 2020, 8, 620. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, Z.; Lu, W.W.; Zhen, W.; Yang, D.; Peng, S. Novel biomaterial strategies for controlled growth factor delivery for biomedical applications. NPG Asia Mater. 2017, 9, e435. [Google Scholar] [CrossRef]
- Kobayashi, T.; Mizuta, M.; Hiwatashi, N.; Kishimoto, Y.; Nakamura, T.; Kanemaru, S.; Hirano, S. Drug delivery system of basic fibroblast growth factor using gelatin hydrogel for restoration of acute vocal fold scar. Auris Nasus Larynx 2017, 44, 86–92. [Google Scholar] [CrossRef]
- Chen, G.; Yu, Y.; Wu, X.; Wang, G.; Ren, J.; Zhao, Y. Bioinspired multifunctional hybrid hydrogel promotes wound healing. Adv. Funct. Mater. 2018, 28, 1801386. [Google Scholar] [CrossRef]
- Tallawi, M.; Rosellini, E.; Barbani, N.; Cascone, M.G.; Rai, R.; Saint-Pierre, G.; Boccaccini, A.R. Strategies for the chemical and biological functionalization of scaffolds for cardiac tissue engineering: A review. J. R. Soc. Interface 2015, 12, 20150254. [Google Scholar] [CrossRef]
- Kastritis, P.L.; Bonvin, A.M.J.J. On the binding affinity of macromolecular interactions: Daring to ask why proteins interact. J. R. Soc. Interface 2013, 10, 20120835. [Google Scholar] [CrossRef]
- Spicer, C.D.; Pashuck, E.T.; Stevens, M.M. Achieving controlled biomolecule–Biomaterial conjugation. Chem. Rev. 2018, 118, 7702–7743. [Google Scholar] [CrossRef] [PubMed]
- Richbourg, N.R.; Peppas, N.A.; Sikavitsas, V.I. Tuning the biomimetic behavior of scaffolds for regenerative medicine through surface modifications. J. Tissue Eng. Regen. Med. 2019, 13, 1275–1293. [Google Scholar] [CrossRef] [PubMed]
- Chiu, L.L.Y.; Weisel, R.D.; Li, R.-K.; Radisic, M. Defining conditions for covalent immobilization of angiogenic growth factors onto scaffolds for tissue engineering. J. Tissue Eng. Regen. Med. 2011, 5, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Deng, D.; Xing, Y.; Li, S.; Yuan, B.; Chen, J.; Xia, N. Activity analysis of the carbodiimide-mediated amine coupling reaction on self-assembled monolayers by cyclic voltammetry. Electrochim. Acta 2013, 89, 616–622. [Google Scholar] [CrossRef]
- Hajimiri, M.; Shahverdi, S.; Esfandiari, M.A.; Larijani, B.; Atyabi, F.; Rajabiani, A.; Dehpour, A.R.; Amini, M.; Dinarvand, R. Preparation of hydrogel embedded polymer-growth factor conjugated nanoparticles as a diabetic wound dressing. Drug Dev. Ind. Pharm. 2016, 42, 707–719. [Google Scholar] [CrossRef]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-inspired surface chemistry for multifunctional coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef] [Green Version]
- Harrington, M.J.; Masic, A.; Holten-Andersen, N.; Waite, J.H.; Fratzl, P. Iron-clad fibers: A metal-based biological strategy for hard flexible coatings. Science 2010, 328, 216–220. [Google Scholar] [CrossRef] [Green Version]
- Ghalandari, B.; Yu, Y.; Ghorbani, F.; Warden, A.R.; Ahmad, K.Z.; Sang, X.; Huang, S.; Zhang, Y.; Su, W.; Divsalar, A.; et al. Polydopamine nanospheres coated with bovine serum albumin permit enhanced cell differentiation: Fundamental mechanism and practical application for protein coating formation. Nanoscale 2021, 13, 20098–20110. [Google Scholar] [CrossRef]
- Ghorbani, F.; Ghalandari, B.; Liu, C. A facile method to synthesize 3D pomegranate-like polydopamine microspheres. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef]
- Gan, D.; Wang, Z.; Xie, C.; Wang, X.; Xing, W.; Ge, X.; Yuan, H.; Wang, K.; Tan, H.; Lu, X. Mussel-inspired tough hydrogel with in situ nanohydroxyapatite mineralization for osteochondral defect repair. Adv. Healthc. Mater. 2019, 8, 1901103. [Google Scholar] [CrossRef]
- Qu, M.; Jiang, X.; Zhou, X.; Wang, C.; Wu, Q.; Ren, L.; Zhu, J.; Zhu, S.; Tebon, P.; Sun, W.; et al. Stimuli-responsive delivery of growth factors for tissue engineering. Adv. Healthc. Mater. 2020, 9, 1901714. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Jin, Y.; Qi, R.; Fan, W.; Li, H.; Sun, X.; Lai, S. Dual pH and oxidation-responsive nanogels crosslinked by diselenide bonds for controlled drug delivery. Polymer 2016, 101, 370–378. [Google Scholar] [CrossRef]
- Garbern, J.C.; Minami, E.; Stayton, P.S.; Murry, C.E. Delivery of basic fibroblast growth factor with a pH-responsive, injectable hydrogel to improve angiogenesis in infarcted myocardium. Biomaterials 2011, 32, 2407–2416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Li, Y.; Yang, R.; Gao, X.; Ying, G. pH-responsive polyethylene glycol monomethyl ether-ε-polylysine-g-poly (Lactic acid)-based nanoparticles as protein delivery systems. PLoS ONE 2016, 11, e0159296. [Google Scholar] [CrossRef] [PubMed]
- Salimath, A.S.; Phelps, E.A.; Boopathy, A.V.; Che, P.; Brown, M.; Garcia, A.J.; Davis, M.E. Dual delivery of hepatocyte and vascular endothelial growth factors via a protease-degradable hydrogel improves cardiac function in rats. PLoS ONE 2012, 7, e50980. [Google Scholar] [CrossRef] [Green Version]
- Yao, X.; Niu, X.; Ma, K.; Huang, P.; Grothe, J.; Kaskel, S.; Zhu, Y. Graphene quantum dots-capped magnetic mesoporous silica nanoparticles as a multifunctional platform for controlled drug delivery, magnetic hyperthermia, and photothermal therapy. Small 2017, 13, 1602225. [Google Scholar] [CrossRef]
- Zamora-Mora, V.; Fernandez-Gutierrez, M.; Gonzalez-Gomez, A.; Sanz, B.; Roman, J.S.; Goya, G.F.; Hernandez, R.; Mijangos, C. Chitosan nanoparticles for combined drug delivery and magnetic hyperthermia: From preparation to in vitro studies. Carbohydr. Polym. 2017, 157, 361–370. [Google Scholar] [CrossRef] [Green Version]
- Rastegari, B.; Karbalaei-Heidari, H.R.; Zeinali, S.; Sheardown, H. The enzyme-sensitive release of prodigiosin grafted β-cyclodextrin and chitosan magnetic nanoparticles as an anticancer drug delivery system: Synthesis, characterization and cytotoxicity studies. Colloids Surf. B Biointerfaces 2017, 158, 589–601. [Google Scholar] [CrossRef]
- Kim, H.; Park, H.; Lee, J.W.; Lee, K.Y. Magnetic field-responsive release of transforming growth factor beta 1 from heparin-modified alginate ferrogels. Carbohydr. Polym. 2016, 151, 467–473. [Google Scholar] [CrossRef]
- Feng, Q.; Liu, Y.; Huang, J.; Chen, K.; Huang, J.; Xiao, K. Uptake, distribution, clearance, and toxicity of iron oxide nanoparticles with different sizes and coatings. Sci. Rep. 2018, 8, 2082. [Google Scholar] [CrossRef]
- Azagarsamy, M.A.; Anseth, K.S. Wavelength-controlled photocleavage for the orthogonal and sequential release of multiple proteins. Angew. Chem. 2013, 125, 14048–14052. [Google Scholar] [CrossRef] [Green Version]
- Fan, H.; Vitharana, S.N.; Chen, T.; O’Keefe, D.; Middaugh, C.R. Effects of pH and polyanions on the thermal stability of fibroblast growth factor 20. Mol. Pharm. 2007, 4, 232–240. [Google Scholar] [CrossRef]
- Subbiah, R.; Guldberg, R.E. Materials science and design principles of growth factor delivery systems in tissue engineering and regenerative medicine. Adv. Healthc. Mater. 2019, 8, 1801000. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, X. Protein isoelectric point. In Bioinformatics and the Cell; Springer: Boston, MA, USA, 2007; pp. 207–219. [Google Scholar]
- Mohanta, T.K.; Khan, A.; Hashem, A.; Abd_Allah, E.F.; Al-Harrasi, A. The molecular mass and isoelectric point of plant proteomes. BMC Genom. 2019, 20, 631. [Google Scholar] [CrossRef] [Green Version]
- Grayson, A.C.R.; Choi, I.S.; Tyler, B.M.; Wang, P.P.; Brem, H.; Cima, M.J.; Langer, R. Multi-pulse drug delivery from a resorbable polymeric microchip device. Nat. Mater. 2003, 2, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Tayalia, P.; Mooney, D.J. Controlled growth factor delivery for tissue engineering. Adv. Mater. 2009, 21, 3269–3285. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.W.; Zhang, J.X.; Zeng, L.; Xu, J.J.; Du, F.T.; Luo, W.; Luo, Z.J.; Jiang, J.H. Vascular endothelial growth factor gene therapy with intramuscular injections of plasmid DNA enhances the survival of random pattern flaps in a rat model. Br. J. Plast. Surg. 2005, 58, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Kim, N.A.; Lim, D.G.; Kim, K.H.; Jeong, S.H. Effects of thermal and mechanical stress on the physical stability of human growth hormone and epidermal growth factor. Arch. Pharm. Res. 2015, 38, 1488–1498. [Google Scholar] [CrossRef] [PubMed]
- La Corte, A.L.C.; Philippou, H.; Ariens, R.A.S. Role of Fibrin Structure in Thrombosis and Vascular Disease; Elsevier: Amsterdam, The Netherlands, 2011; pp. 75–127. [Google Scholar]
- Fagundes, M.B.; Vendruscolo, R.G.; Wagner, R. Sterols from microalgae. In Handbook of Microalgae-Based Processes and Products; Elsevier: Amsterdam, The Netherlands, 2020; pp. 573–596. [Google Scholar]
- Zhao, H.; Wu, J.; Zhu, J.; Xiao, Z.Z.; He, C.; Shi, H.; Li, X.; Ya, S.; Zheng, X.; An, Y.; et al. Insulin-like growth factor (IGF)-I and IGF binding proteins axis in diabetes mellitus. J. Control. Release 2020, 25, 581–611. [Google Scholar] [CrossRef]
- Xiao, Z.; Zheng, X.; An, Y.; Wang, K.; Zhang, J.; He, H.; Wu, J. Zwitterionic hydrogel for sustained release of growth factors to enhance wound healing. Biomater. Sci. 2020. [Google Scholar] [CrossRef]
- Bhosle, V.K.; Altit, G.; Autmizguine, J.; Chemtob, S. Basic pharmacologic principles. In Fetal and Neonatal Physiology; Elsevier: Amsterdam, The Netherlands, 2017; pp. 187–201.e3. [Google Scholar]
- Toutain, P.L.; Bousquet-Melou, A. Bioavailability and its assessment. J. Vet. Pharmacol. Ther. 2004, 27, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Elmasry, S.; Asfour, S.; de Rivero Vaccari, J.P.; Travascio, F. A computational model for investigating the effects of changes in bioavailability of insulin-like growth factor-1 on the homeostasis of the intervertebral disc. Comput. Biol. Med. 2016, 78, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Yang, L.; Li, X.; Sun, X.; Zhao, J.; Hou, X.; Li, Z.; Yuan, X.; Cui, Z.; Yang, X. Systemic administration of enzyme-responsive growth factor nanocapsules for promoting bone repair. Biomater. Sci. 2019, 7, 1675–1685. [Google Scholar] [CrossRef] [PubMed]
- Utterstrom, J.; Naeimipour, S.; Selegard, R.; Aili, D. Coiled coil-based therapeutics and drug delivery systems. Adv. Drug Deliv. Rev. 2021, 170, 26–43. [Google Scholar] [CrossRef]
- Epstein-Barash, H.; Stefanescu, C.F.; Kohane, D.S. An in situ cross-linking hybrid hydrogel for controlled release of proteins. Acta Biomater. 2012, 8, 1703–1709. [Google Scholar] [CrossRef] [Green Version]
- Shin, G.R.; Kim, H.E.; Kim, J.H.; Choi, S.; Kim, M.S. Advances in injectable in situ-forming hydrogels for intratumoral treatment. Pharmaceutics 2021, 13, 1953. [Google Scholar] [CrossRef]
- Bernhard, S.; Tibbitt, M.W. Supramolecular engineering of hydrogels for drug delivery. Adv. Drug Deliv. Rev. 2021, 171, 240–256. [Google Scholar] [CrossRef]
- Cirillo, G.; Curcio, M.; Nicoletta, F.P.; Iemma, F. Injectable Hydrogels for Cancer Therapy over the Last Decade. Pharmaceutics 2019, 11, 486. [Google Scholar] [CrossRef] [Green Version]
- Shevach, M.; Fleischer, S.; Shapira, A.; Dvir, T. Gold nanoparticle-decellularized matrix hybrids for cardiac tissue engineering. Nano Lett. 2014, 14, 5792–5796. [Google Scholar] [CrossRef]
- Zhang, Q.; Fan, A.; Fu, J.; Zhao, Q. Precise engineering of iron oxide nanoparticle-encapsulated protein hydrogel: Implications for cardiac toxicity and ultrasound contrast agents. Process Biochem. 2021, 102, 296–303. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, G.; Xu, K.; Wang, L.; Yu, L.; Xing, M.M.Q.; Qiu, X. Mussel-inspired dual-functional PEG hydrogel inducing mineralization and inhibiting infection in maxillary bone reconstruction. Mater. Sci. Eng. C 2018, 90, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Krishnan, N.; Heo, J.; Fang, R.H.; Zhang, L. Nanoparticle–hydrogel superstructures for biomedical applications. J. Control. Release 2020, 324, 505–521. [Google Scholar] [CrossRef] [PubMed]






| Proteins | Cross-Linking | Hydrogel Components | Effects of Protein Component | Target Tissue | Ref. |
|---|---|---|---|---|---|
| Collagen | Physical cross-linking Adjustment of pH (50 mM HEPES), employment of CaCl2 and thrombin, and UV exposure | - | Showed rapid mouse myoblast cells’ infiltration and micro-vascularization | Heart | [98] |
| Thermally cross-linking Incubation for 30 min | - | Formed a lattice pattern for cornea structure | Cornea | [99] | |
| - | Alginate | Increased chondrocyte cell viability (up to 90%) | Cartilage | [100] | |
| Thermally cross-linking Incubation at 37 °C | - | Displayed significant osteogenic differentiation | Bone | [101] | |
| Gelatin | Physical cross-linking UV exposure, Irgacure 2959 (0.5% w/v) | GelMA | Produced endothelial cell-responsive tissues | Blood vessel | [102] |
| Chemical cross-linking Borax (0.1 M, 30 s) | Alginate | Promoted mouse chondrocytes’ adhesion, viability, and proliferation | Cartilage | [103] | |
| - | PEI-Ppy | Developed antibacterial properties | Skin | [104] | |
| Physical cross-linking UV exposure, Irgacure 2959 (0.5% w/v) | GelMA | Aided keratinocytes’ proliferation and differentiation | Skin | [105] | |
| Serum albumin | Chemical cross-linking Adjustment of pH (NaH2PO4 and Na2HPO4) | PEG-SS2-bioglass | Accelerated the wound healing process | Skin | [106] |
| Ionic cross-linking (Ag+) | - | Significantly increased osteogenesis differentiation | Bone | [107] | |
| Ionic cross-linking (CaCl2) | Sodium alginate-Hydroxyapatite | Affected the differentiation and proliferation of human bone marrow-derived mesenchymal stem cells | Cartilage | [108] | |
| Thermally cross-linking Incubation at 75–80 °C | Fibroin | Created contractile heart tissue | Heart | [109] | |
| Elastin | Thermally cross-linking incubation at 37 °C | Collagen | Accelerated the heart valve endothelial cells’ growth | Heart | [110] |
| Modification of SKS concentration | Plasma | Improved mechanical characteristics and biological capabilities | Skin | [111] | |
| Chemical cross-linking N-hydroxysuccinimide (NHS)-1-Ethyl-3-(3-dimethylaminopropyl(EDC) | Collagen | Tackled bacterial infection | Bone | [112] | |
| - | - | Controlled angiogenesis | Blood vessel | [113] | |
| Keratin | Disulfide cross-linking | - | Showed rapid penetration, propagation, and differentiation of MSCs | Cartilage | [114] |
| Chemical cross-linking Sodium trimetaphosphate | Konjac glucomannan, Oat | Aided collagen formation | Skin | [115] | |
| Disulfide cross-linking | Glucose-triggered | Decreased gel formation time | Skin | [116] | |
| Disulfide cross-linking | - | Developed hydrogel biocompatibility | Bone and Skin | [117] | |
| Resilin | Chemical cross-linking Tris(hydroxymethyl phosphine) | Fibronectin | Increased human MSCs’ proliferation | Cartilage | [118] |
| Chemical cross-linking PEG macromers | PEG-vinyl sulfone | Increased aortic cell viability | Cardiovascular | [119] | |
| Chemical cross-linking Tris(hydroxymethyl phosphine) | - | Increased hydrogel flexibility and bioactivity | Vocal fold | [120] | |
| Chemical cross-linking 3,3′-dithiobis(sulfosuccinimidyl propionate) | - | Displayed remarkable NIH/3T3 fibroblasts’ growth in a day (>95%) | - | [121] | |
| Silk | - | Fibroin | Improved rat cardiomyocytes cells’ attachment and activities | Heart | [122] |
| Enzyme-mediated cross-linking | - | Provided the repair of osteochondral tissue | Bone and Cartilage | [123] | |
| Physical cross-linking UV exposure, LAP (0.6% w/v) | Glycidyl methacrylate | Displayed proliferation and viability of chondrocyte cell after four week | Cartilage | [124] | |
| Thermally cross-linking at physiological temperature | Chitosan | Positively impacted MC3T3-E1 cells’ adhesion and proliferation | Bone | [125] |
| Hydrogels Composition | Growth Factor | Delivery Method | Tissue | Ref. |
|---|---|---|---|---|
| Chitosan-Hyaluronic acid | NGF | Static scaffold seeding | Nerve | [211] |
| Keratin-Keratose | IGF-1 and bFGF | Static scaffold seeding | Skeletal muscle | [212] |
| PEGDA | EGF | Bioreactor utilization | Liver | [218] |
| Chitosan–Gelatin | TGF-β2 | Bioreactor utilization | Cartilage | [219] |
| Gelatin | bFGF | Physical immobilization | Vocal fold | [225] |
| Aldehyde chitosan-amino-end PEG | VEGF | Physical immobilization | Skin | [226] |
| Sodium carboxymethyl chitosan | rhEGF | Chemical immobilization | Skin | [233] |
| HA-GelMA | TGF-β3 and BMP-2 | Chemical immobilization | Osteochondral | [238] |
| Poly (N-isopropylacrylamide-co-propyl acrylic acid-co-butyl acrylate) | bFGF | Spatiotemporally controlled delivery | Heart | [241] |
| PEG | HGF and VEGF | Spatiotemporally controlled delivery | Heart | [243] |
| Heparin-modified alginate-iron oxide nanoparticles | TGF-β1 | Spatiotemporally controlled delivery | Cartilage | [247] |
| PEG | BMP-2 and BMP-7 | Spatiotemporally controlled delivery | Bone | [249] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davari, N.; Bakhtiary, N.; Khajehmohammadi, M.; Sarkari, S.; Tolabi, H.; Ghorbani, F.; Ghalandari, B. Protein-Based Hydrogels: Promising Materials for Tissue Engineering. Polymers 2022, 14, 986. https://doi.org/10.3390/polym14050986
Davari N, Bakhtiary N, Khajehmohammadi M, Sarkari S, Tolabi H, Ghorbani F, Ghalandari B. Protein-Based Hydrogels: Promising Materials for Tissue Engineering. Polymers. 2022; 14(5):986. https://doi.org/10.3390/polym14050986
Chicago/Turabian StyleDavari, Niyousha, Negar Bakhtiary, Mehran Khajehmohammadi, Soulmaz Sarkari, Hamidreza Tolabi, Farnaz Ghorbani, and Behafarid Ghalandari. 2022. "Protein-Based Hydrogels: Promising Materials for Tissue Engineering" Polymers 14, no. 5: 986. https://doi.org/10.3390/polym14050986