Energy Release Characteristics and Reaction Mechanism of PTFE/Al/Bi2O3 Reactive Materials under Drop-Hammer Test
Abstract
:1. Introduction
2. Materials and Methods
2.1. Formulations and Preparation
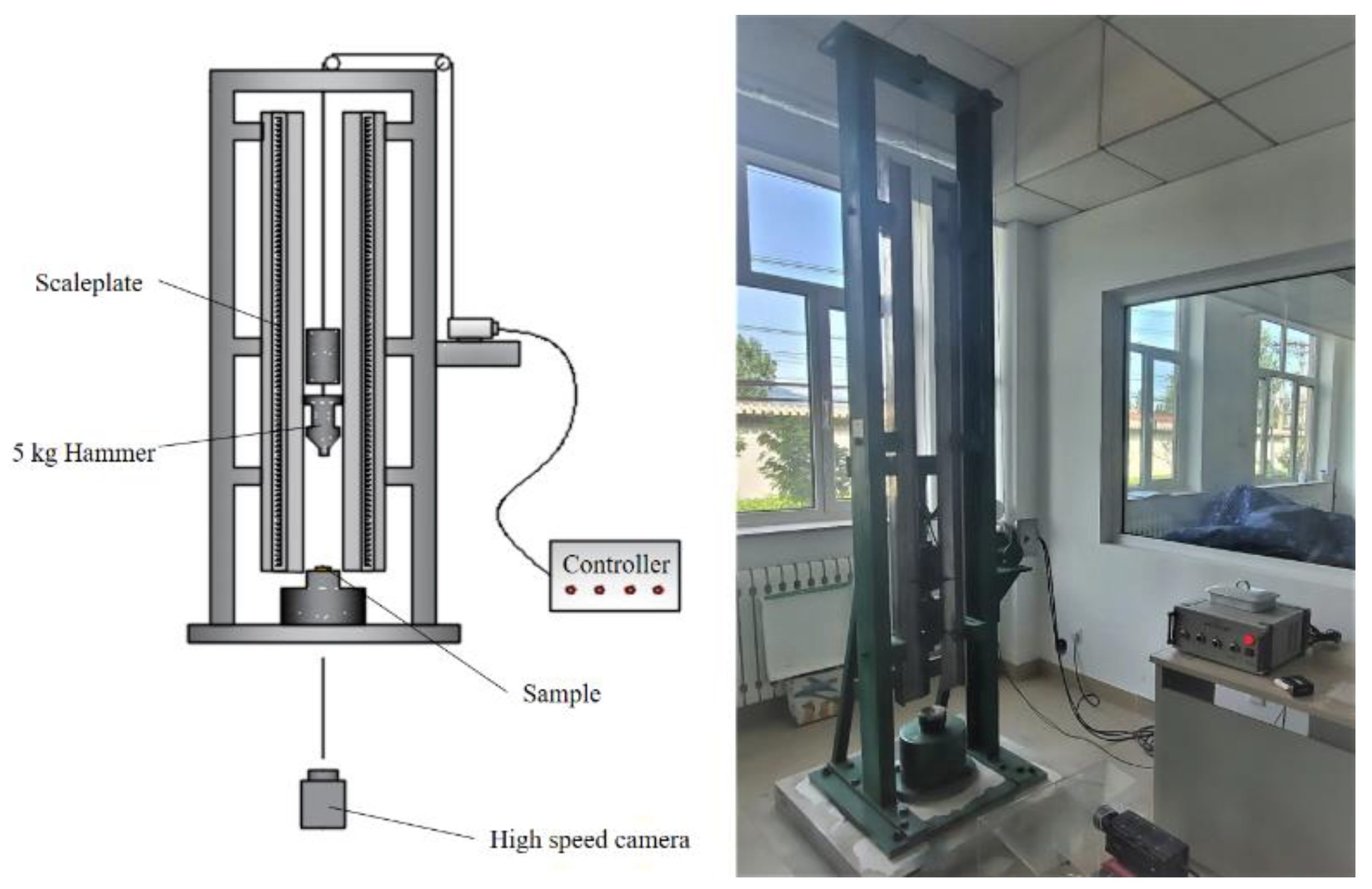
2.2. Drop-Hammer Test
2.3. Thermal Analysis
3. Results and Analysis
3.1. Energy Release Characteristics of PAB under Drop-Hammer Impact
3.1.1. Shock-Induced Chemical Reaction Phenomenon
3.1.2. Impact Sensitivity
3.1.3. Ignition Delay
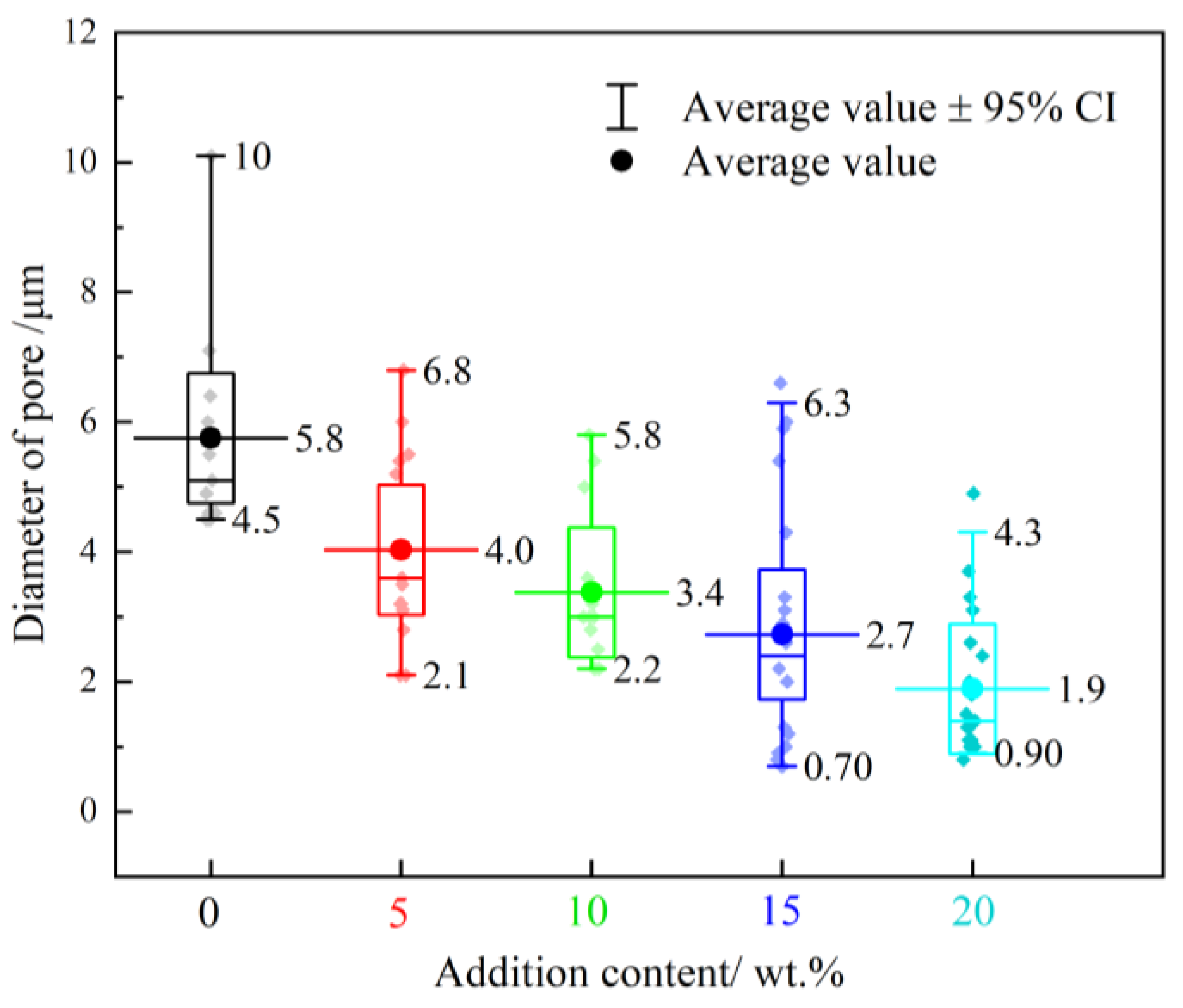
3.2. Shape of the Specimen before and after the Reaction
3.2.1. Macroscopic Appearance of Specimens
3.2.2. Microscopic Morphology of Specimens
3.3. Ignition and Reaction Mechanism of PAB under Drop-Hammer Impact
3.3.1. Hot-Spot Formation
3.3.2. Thermal Decomposition Process
3.3.3. Microscopic Morphology of Reaction Products
3.3.4. Ignition and Reaction Propagation Process
3.3.5. Ignition of Aluminum Particles
4. Conclusions
- (1)
- At a drop height of 150 cm, PABs can react vigorously under drop-hammer loading. The ratio influenced the reaction duration and intensity. As the Bi2O3 content increased to 9%, the reaction duration increased and the reactivity increased, and when the content continued to increase, the reaction duration decreased and the intensity of the reaction decreased. When the Bi2O3 content was 9%, the reaction was the most intense, and the duration was the longest at 1300 μs. With the increase in Bi2O3 content in the system, the sensitivity of the material first increased, and then fell. When the Bi2O3 content was 9%, the highest impact sensitivity was achieved with a characteristic drop height H50 of 130.5 cm.
- (2)
- Under impact loading of the drop hammer, ignition occurs at the tips of open cracks. PAB undergoes several physicochemical processes such as PTFE melting, a PTFE/Bi2O3 reaction, an Al/ Bi2O3 reaction, molten PTFE pyrolysis, and a C2F4/Al reaction during the local temperature rise. The presence of Bi2O3 results in a lower reaction onset temperature of the reactive material, a lower excitation threshold of the reactive material, and a higher degree of reaction, improving the overall energy release of the reactive material.
- (3)
- The ignition process and propagation process in PAB can be roughly divided into three stages: local PTFE melting (mush zone); the reaction of the active component and thermal decomposition of the matrix to produce gas (multiphase zone); and violent combustion of Bi2O3 and Al particles in strong oxidizing C2F4 gas (flame zone). With a appropriate amount of Bi2O3, the reaction of molten PTFE and Bi2O3 and the thermit reaction between Al/Bi2O3 will occur more easily in the multiphasic zone; this will release a large amount of energy and promote diffusion in the multiphasic zone, thus promoting the propagation of the overall reaction and improving the reactivity of PAB.
Author Contributions
Funding
Conflicts of Interest
References
- Koch, E.-C. Metal-Fluorocarbon Based Energetic Materials; Wiley-VCH: Weinheim, Germany, 2012; ISBN 978-3-527-32920-5. [Google Scholar]
- Tao, J.; Wang, X. Research Progress in Metal-Fluoropolymer Mechanical Activation Energetic Composites. Chin. J. Explos. Propellants 2017, 40, 8–14. [Google Scholar]
- Nielson, D.B.; Truitt, R.M.; Ashcroft, B.N. Reactive Material Enhanced Projectiles and Related Methods. U.S. Patent US 2008/0035007 A1, 14 February 2008. [Google Scholar]
- Fischer, S.H.; Grubelich, M.C. A Survey of Combustible Metals, Thermites, and Intermetallics for Pyrotechnic Applications; American Institute of Aeronautics and Astronautics: Reston, VA, USA, 1996. [Google Scholar]
- White, B.W. Microstructure and Strain Rate Effects on the Mechanical Behavior of Particle Reinforced Epoxy-Based Reactive Materials. Ph.D. Thesis, Georgia Institute of Technology, Atlanta, GA, USA, 2011. [Google Scholar]
- Ferranti, L.; Thadhani, N.N. Dynamic Impact Characterization of Al+Fe2O3+30% Epoxy Composites Using Time Synchronized High-Speed Camera and VISAR Measurements. MRS Proc. 2005, 896, 0896-H06-05. [Google Scholar] [CrossRef]
- Ferranti, L. Dynamic Mechanical Behavior Characterization of Epoxy-Cast Al+Fe2O3 Mixtures. AIP Conf. Proc. 2006, 845, 805. [Google Scholar]
- Wu, J.; Feng, B.; Gao, Z.; Yin, Q.; Huang, J.; Wu, S.; Li, Y. Effect of Multi-Oxidants on Mechanical Response and Reactive Characteristics of PTFE/Al Reactive Material under Dynamic Impact. Mater. Lett. 2021, 283, 128886. [Google Scholar] [CrossRef]
- Zhou, J.; Ding, L.; Tang, W.; Ran, X. Experimental Study of Mechanical Properties and Impact-Induced Reaction Characteristics of PTFE/Al/CuO Reactive Materials. Materials 2019, 13, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J. Mechanical and Reaction Properties of PTFE/Al/MnO2 Reactive Materials at Different Strain Rates. Adv. Mater. 2018, 7, 50. [Google Scholar] [CrossRef]
- Huang, J.; Fang, X.; Wu, S.; Yang, L.; Yu, Z.; Li, Y. Mechanical Response and Shear-Induced Initiation Properties of PTFE/Al/MoO3 Reactive Composites. Materials 2018, 11, 1200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martirosyan, K.S.; Wang, L.; Vicent, A.; Luss, D. Synthesis and Performance of Bismuth Trioxide Nanoparticles for High Energy Gas Generator Use. Nanotechnology 2009, 20, 405609. [Google Scholar] [CrossRef]
- Wang, L.; Luss, D.; Martirosyan, K.S. The Behavior of Nanothermite Reaction Based on Bi2O3/Al. J. Appl. Phys. 2011, 110, 074311. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Jiang, Z.; Feng, C. Metastable Intermolecular Composite Al/Bi2O3 and Its Applications. Prog. Chem. 2016, 28, 391–400. [Google Scholar] [CrossRef]
- Glavier, L.; Taton, G.; Ducéré, J.-M.; Baijot, V.; Pinon, S.; Calais, T.; Estève, A.; Djafari Rouhani, M.; Rossi, C. Nanoenergetics as Pressure Generator for Nontoxic Impact Primers: Comparison of Al/Bi2O3, Al/CuO, Al/MoO3 Nanothermites and Al/PTFE. Combust. Flame 2015, 162, 1813–1820. [Google Scholar] [CrossRef]
- Jiang, Z. Preparation and Properties of Metastable Intermolecular Composite Al/Bi2O3. Master’s Thesis, Beijing Institute of Technology, Beijing, China, 2016. [Google Scholar]
- Lan, J.; Liu, J.; Zhang, S.; Xue, X.; He, C.; Wu, Z.; Yang, M.; Li, S. Influence of Multi-Oxidants on Reaction Characteristics of PTFE-Al-XmOY Reactive Material. Mater. Des. 2020, 186, 108325. [Google Scholar] [CrossRef]
- Yuan, Y.; Geng, B.; Sun, T.; Yu, Q.; Wang, H. Impact-Induced Reaction Characteristic and the Enhanced Sensitivity of PTFE/Al/Bi2O3 Composites. Polymers 2019, 11, 2049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, Y.; Liu, Z.; He, S.; Ge, C.; Yu, Q.; Zheng, Y.; Wang, H. Shock-Induced Reaction Behaviors of Functionally Graded Reactive Material. Def. Technol. 2021, 17, 1687–1698. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, Q.; Huang, F. Detonation Physics, 1st ed.; The Publishing House of Ordnance Industry: Beijing, China, 2009. [Google Scholar]
- Sano, Y.; Nagaishi, T.; Fukui, H. Thermal reaction or sensitivity of Bi2O3 and metal powder. Sci. Technol. Energ. Mater. 2013, 74, 93–99. [Google Scholar]
- Dolgoborodov, A.Y.; Streletskii, A.N.; Makhov, M.N.; Kolbanev, I.V.; Fortov, V.E. Explosive Compositions Based on the Mechanoactivated Metal-Oxidizer Mixtures. Russ. J. Phys. Chem. B 2007, 1, 606–611. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, C.; Wang, Z.; Luo, P. Experimental Study on Reaction Characteristics of PTFE/Ti/W Energetic Materials under Explosive Loading. Materials 2016, 9, 936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, L.; Wei, C.; Hu, R.; Hu, W.; Luo, P.; Qi, Y.; Jiang, C. Effects of Al Particle Size on the Impact Energy Release of Al-Rich PTFE/Al Composites under Different Strain Rates. Materials 2021, 14, 1911. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.-P. Study on the Modification of PTFE and the Properties of Its Composites. Master’s Thesis, Norethwestern Polytechnical University, Xi’an, China, 2006. [Google Scholar]
- Chen, P.; Qu, K.; Chen, R.; Yuan, B. Reactive Properties of Reactive Material under the Load of Drop-weight. Chin. J. Explos. Propellants 2019, 42, 197–201+206. [Google Scholar]
- Ge, C.; Wubuliaisan, M.; Tian, C.; Dong, Y.; Song, Q. Impact-induced initiation thresholds of polytetrafluoroethylene/Al composite by gas gun. Explos. Shock Waves 2018, 37, 1–8. [Google Scholar]
- Feng, B.; Li, Y.; Wu, S.; Wang, H.; Tao, Z.; Fang, X. A Crack-Induced Initiation Mechanism of Al-PTFE under Quasi-Static Compression and the Investigation of Influencing Factors. Mater. Des. 2016, 108, 411–417. [Google Scholar] [CrossRef]
- Dienes, J.K. Frictional Hot-Spots and Statistical Crack Mechanics. [Solid Propellant Cylinders]; Los Alamos National Laboratory (LANL): Los Alamos, NM, USA, 1983.
- Fuller, K.N.G.; Fox, P.G.; Field, J.E. The Temperature Rise at the Tip of Fast-Moving Cracks in Glassy Polymers. Proc. R. Soc. Lond. Math. Phys. Sci. 1975, 341, 537–557. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, L.; Mao, Y.; Gong, F. An Effective Way to Enhance Energy Output and Combustion Characteristics of Al/PTFE. Combust. Flame 2020, 214, 419–425. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, J.; Fang, X.; Li, Y.; Yu, Z.; Gao, Z.; Wu, S.; Yang, L.; Wu, J.; Kui, J. Thermal Decomposition and Thermal Reaction Process of PTFE/Al/MnO2 Fluorinated Thermite. Materials 2018, 11, 2451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y. Study on Energy Release Characteristics of Aluminum Particles and Aluminum-Based Solid Propellants. Ph.D. Thesis, Zhejiang University, Hangzhou, China, 2019. [Google Scholar]
- Wang, J.; Qu, Y.; Gong, F.; Shen, J.; Zhang, L. A Promising Strategy to Obtain High Energy Output and Combustion Properties by Self-Activation of Nano-Al. Combust. Flame 2019, 204, 220–226. [Google Scholar] [CrossRef]
- Xin, W. Studies on Preparation, Modification and Electrochemical Performance of BiF3 as a Positive Electrode for Rechargeable Lithium Battery. Master’s Thesis, Xiangtan University, Xiangtan, China, 2009. [Google Scholar]
- Hunt, E.M.; Malcolm, S.; Pantoya, M.L.; Davis, F. Impact Ignition of Nano and Micron Composite Energetic Materials. Int. J. Impact Eng. 2009, 36, 842–846. [Google Scholar] [CrossRef]
- Levitas, V.I.; Pantoya, M.L.; Watson, K.W. Melt-Dispersion Mechanism for Fast Reaction of Aluminum Particles: Extension for Micron Scale Particles and Fluorination. Appl. Phys. Lett. 2008, 92, 201917. [Google Scholar] [CrossRef]
- Rai, A.; Lee, D.; Park, K.; Zachariah, M.R. Importance of Phase Change of Aluminum in Oxidation of Aluminum Nanoparticles. J. Phys. Chem. B 2004, 108, 14793–14795. [Google Scholar] [CrossRef]
- Rai, A.; Park, K.; Zhou, L.; Zachariah, M.R. Understanding the Mechanism of Aluminium Nanoparticle Oxidation. Combust. Theory Model. 2006, 10, 843–859. [Google Scholar] [CrossRef]
- Trunov, M.A.; Schoenitz, M.; Zhu, X.; Dreizin, E.L. Effect of Polymorphic Phase Transformations in Al2O3 Film on Oxidation Kinetics of Aluminum Powders. Combust. Flame 2005, 140, 310–318. [Google Scholar] [CrossRef]















| Formulation Codes | Al/Bi2O3 Additive Content/(wt.%) | Content/(wt.%) | Theoretical Density /(g∙cm−3) | Theoretical Calorific Value/(kJ∙g−1) | ||
|---|---|---|---|---|---|---|
| PTFE | Al | Bi2O3 | ||||
| 1 | 0 | 74 | 26 | - | 2.32 | 8.42 |
| 2 | 5 | 70 | 25 | 5 | 2.41 | 8.11 |
| 3 | 10 | 67 | 24 | 9 | 2.48 | 7.79 |
| 4 | 15 | 63 | 24 | 13 | 2.57 | 7.48 |
| 5 | 20 | 59 | 23 | 18 | 2.68 | 7.16 |
| Formulation Codes | Bi2O3 Content/(%) | Theoretical Density /(g∙cm−3) | Actual Density /(g∙cm−3) | Relative Density /(%) | Porosity Ratio /(%) |
|---|---|---|---|---|---|
| 1 | 0 | 2.32 | 2.19 | 94.92 | 5.08 |
| 2 | 5 | 2.41 | 2.27 | 93.59 | 6.41 |
| 3 | 9 | 2.48 | 2.32 | 92.77 | 7.23 |
| 4 | 13 | 2.57 | 2.33 | 90.66 | 9.34 |
| 5 | 18 | 2.68 | 2.38 | 88.43 | 11.67 |
| Bi2O3 Content/(%) | Characteristic Drop Height H50/(cm) |
|---|---|
| 0 | 138.4 |
| 5 | 135.8 |
| 9 | 130.5 |
| 13 | 137.5 |
| 18 | 143.6 |
| Formulation | Peak | Start Temperature /(°C) | Peak Temperature /(°C) | End Temperature /(°C) | Energy Release /(J/g) | Physicochemical Changes |
|---|---|---|---|---|---|---|
| PTFE-Al | A | 325 | 343 | 360 | −36.0 | Melting of PTFE |
| B | 522 | 561 | 576 | −243.8 | Decomposition of PTFE | |
| C | 564 | 590 | 605 | 344.4 | PTFE/Al reaction | |
| D | 645 | 658 | 668 | −35.4 | Melting of Al | |
| E | 755 | 825 | 877 | −380.4 | Sublimation of AlF3 | |
| PTFE-Al-Bi2O3 | F | 329 | 342 | 354 | −23.6 | Melting of PTFE |
| G | 385 | 422 | 446 | 61.4 | PTFE/Bi2O3 reaction | |
| H | 472 | 574 | 602 | 1042.2 | Al/Bi2O3 reaction, Decomposition of PTFE, and PTFE/Al reaction | |
| I | 642 | 656 | 661 | −7.5 | Melting of Al |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, C.; Hu, R.; Mao, L.; Wang, Z.; Xu, W.; Hu, W. Energy Release Characteristics and Reaction Mechanism of PTFE/Al/Bi2O3 Reactive Materials under Drop-Hammer Test. Polymers 2022, 14, 1415. https://doi.org/10.3390/polym14071415
Jiang C, Hu R, Mao L, Wang Z, Xu W, Hu W. Energy Release Characteristics and Reaction Mechanism of PTFE/Al/Bi2O3 Reactive Materials under Drop-Hammer Test. Polymers. 2022; 14(7):1415. https://doi.org/10.3390/polym14071415
Chicago/Turabian StyleJiang, Chunlan, Rong Hu, Liang Mao, Zaicheng Wang, Wenyu Xu, and Wanxiang Hu. 2022. "Energy Release Characteristics and Reaction Mechanism of PTFE/Al/Bi2O3 Reactive Materials under Drop-Hammer Test" Polymers 14, no. 7: 1415. https://doi.org/10.3390/polym14071415
APA StyleJiang, C., Hu, R., Mao, L., Wang, Z., Xu, W., & Hu, W. (2022). Energy Release Characteristics and Reaction Mechanism of PTFE/Al/Bi2O3 Reactive Materials under Drop-Hammer Test. Polymers, 14(7), 1415. https://doi.org/10.3390/polym14071415







