Development and Preclinical Investigation of Physically Cross-Linked and pH-Sensitive Polymeric Gels as Potential Vaginal Contraceptives
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Formulation of Physically Cross-Linked Ci-PBA Gels
2.2.2. Formulation of Chemically Cross-Linked Ci-PVA-BA Gels
2.3. Rheological Measurements
2.3.1. Storage (G′) and Loss (G″) Modulus and Tan Δ Measurements
2.3.2. Complex Viscosity
2.4. Creep Recovery Analysis
2.5. Physical Observations
2.6. Osmolality Measurements
2.7. Fourier Transform Infrared Spectroscopy (FTIR)
2.8. Stability Analysis
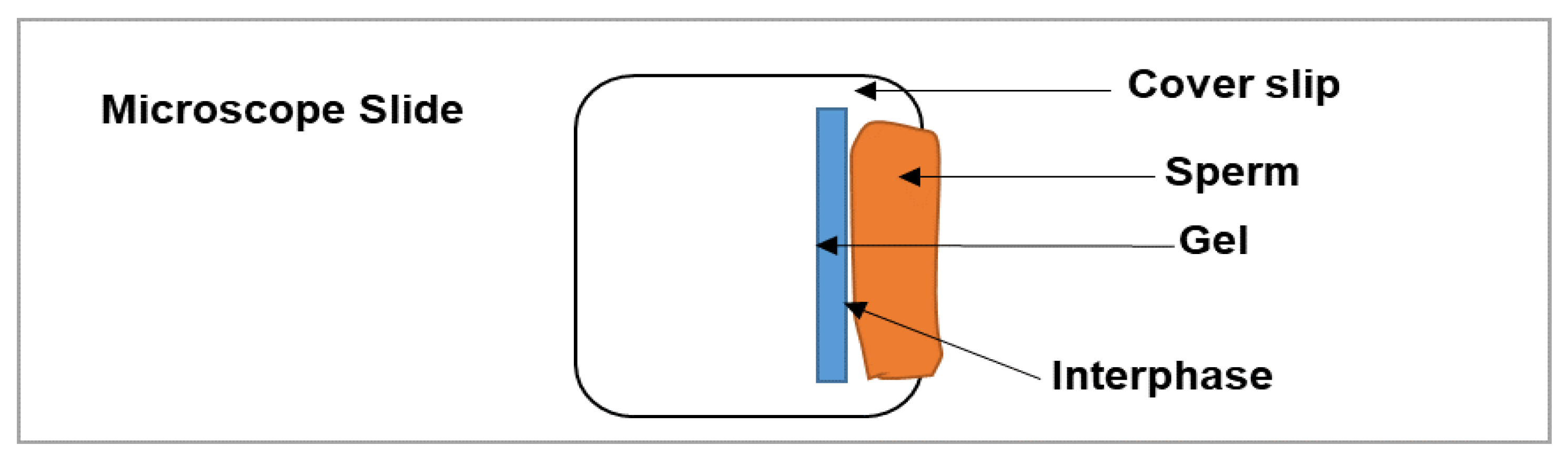
2.9. In Vitro Sperm Functionality Testing
2.10. Rabbit Contraceptive Efficacy Test (RCET)
3. Results and Discussion
3.1. Rheological Characterization of PBA-SHA (Control) Gel
3.2. Development of Physically Cross-Linked Ci-PBA Gels
3.2.1. Frequency Sweep Rheology and Tan Δ Measurements for Ci-PBA Gels
3.2.2. Effect of Excipients on Viscoelastic Behavior of Ci-PBA Gels
3.3. Development of pH-Dependent Chemically Cross-Linked Ci-PVA-BA Gels
Rheology of Ci-PVA-BA Gel (7P) in the Presence of Excipients
3.4. Creep Recovery Analysis of Ci-PBA and Ci-PVA-BA Gels
3.5. Osmolality Measurements of Ci-PBA and Ci-PVA-BA Gels
3.6. FTIR Analysis of Ci-PBA and Ci-PVA-BA Gels
3.7. Stability Testing
3.8. In Vitro Sperm Functionality Testing (Sperm Motility, Viability, and Penetration)
3.9. In Vivo Contraceptive Efficacy Testing of the Gels
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Contraception. Available online: https://apps.who.int/iris/bitstream/handle/10665/329884/WHO-RHR-19.18-eng.pdf?ua=1 (accessed on 7 February 2022).
- Council, N.R. Contraception and Reproduction: Health Consequences for Women and Children in the Developing World; The National Academies Press: Washington, DC, USA, 1989; p. 120. [Google Scholar]
- Sitruk-Ware, R.; Nath, A.; Mishell, D.R., Jr. Contraception technology: Past, present and future. Contraception 2013, 87, 319–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schreiber, C.A.; Meyn, L.A.; Creinin, M.D.; Barnhart, K.T.; Hillier, S.L. Effects of long-term use of nonoxynol-9 on vaginal flora. Obs. Gynecol. 2006, 107, 136–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ndesendo, V.M.K.; Pillay, V.; Choonara, Y.E.; Buchmann, E.; Bayever, D.N.; Meyer, L.C.R. A review of current intravaginal drug delivery approaches employed for the prophylaxis of HIV/AIDS and prevention of sexually transmitted infections. AAPS PharmSciTech 2008, 9, 505–520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahalingam, A.; Jay, J.I.; Langheinrich, K.; Shukair, S.; McRaven, M.D.; Rohan, L.C.; Herold, B.C.; Hope, T.J.; Kiser, P.F. Inhibition of the transport of HIV in vitro using a pH-responsive synthetic mucin-like polymer system. Biomaterials 2011, 32, 8343–8355. [Google Scholar] [CrossRef] [Green Version]
- Jay, J.I.; Shukair, S.; Langheinrich, K.; Hanson, M.C.; Cianci, G.C.; Johnson, T.J.; Clark, M.R.; Hope, T.J.; Kiser, P.F. Modulation of Viscoelasticity and HIV Transport as a Function of pH in a Reversibly Crosslinked Hydrogel. Adv. Funct. Mater. 2009, 19, 2969–2977. [Google Scholar] [CrossRef] [Green Version]
- Roberts, M.C.; Mahalingam, A.; Hanson, M.C.; Kiser, P.F. Chemorheology of phenylboronate-salicylhydroxamate crosslinked hydrogel networks with a sulfonated polymer backbone. Macromolecules 2008, 41, 8832–8840. [Google Scholar] [CrossRef] [Green Version]
- Roberts, M.C.; Hanson, M.C.; Massey, A.P.; Karren, E.A.; Kiser, P.F. Dynamically Restructuring Hydrogel Networks Formed with Reversible Covalent Crosslinks. Adv. Mater. 2007, 19, 2503–2507. [Google Scholar] [CrossRef]
- Kiser, P. GCE Phase II: Synthetic Mucin Polymers for Reproductive Health. Available online: https://www.scholars.northwestern.edu/en/projects/gce-phase-ii-synthetic-mucin-polymers-for-reproductive-health-6 (accessed on 6 February 2022).
- Elshaarani, T.; Yu, H.; Wang, L.; Zain ul, A.; Ullah, R.S.; Haroon, M.; Khan, R.U.; Fahad, S.; Khan, A.; Nazir, A.; et al. Synthesis of hydrogel-bearing phenylboronic acid moieties and their applications in glucose sensing and insulin delivery. J. Mater. Chem. B 2018, 6, 3831–3854. [Google Scholar] [CrossRef]
- Lacina, K.; Skladal, P.; James, T.D. Boronic acids for sensing and other applications—a mini-review of papers published in 2013. Chem. Cent. J. 2014, 8, 60. [Google Scholar] [CrossRef] [Green Version]
- Tarus, D.; Hachet, E.; Messager, L.; Catargi, B.; Ravaine, V.; Auzely-Velty, R. Readily prepared dynamic hydrogels by combining phenyl boronic acid- and maltose-modified anionic polysaccharides at neutral pH. Macromol. Rapid Commun. 2014, 35, 2089–2095. [Google Scholar] [CrossRef]
- Ma, S.; Wang, S.; Li, Q.; Leng, Y.; Wang, L.; Hu, G.-H. A Novel Method for Preparing Poly(vinyl alcohol) Hydrogels: Preparation, Characterization, and Application. Ind. Eng. Chem. Res. 2017, 56, 7971–7976. [Google Scholar] [CrossRef]
- Park, J.-C.; Ito, T.; Kim, K.-O.; Kim, K.-W.; Kim, B.-S.; Khil, M.-S.; Kim, H.-Y.; Kim, I.-S. Electrospun poly(vinyl alcohol) nanofibers: Effects of degree of hydrolysis and enhanced water stability. Polym. J. 2010, 42, 273–276. [Google Scholar] [CrossRef] [Green Version]
- Madlova, M.; Jones, S.A.; Zwerschke, I.; Ma, Y.; Hider, R.C.; Forbes, B. Poly(vinyl alcohol) nanoparticle stability in biological media and uptake in respiratory epithelial cell layers in vitro. Eur. J. Pharm. Biopharm. 2009, 72, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Zied, S.; Elie, A.-J.; Luc, P.; Didier Le, C. Anionic Polysaccharide Hydrogels with Charges Provided by the Polysaccharide or the Crosslinking Agent. Drug Deliv. Lett. 2012, 2, 240–250. [Google Scholar] [CrossRef]
- Li, B.; Zaveri, T.; Ziegler, G.R.; Hayes, J.E. User preferences in a carrageenan-based vaginal drug delivery system. PLoS One 2013, 8, e54975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elias, C.J.; Coggins, C.; Alvarez, F.; Brache, V.; Fraser, I.S.; Lacarra, M.; Lahteenmaki, P.; Massai, R.; Mishell, D.R., Jr.; Phillips, D.M.; et al. Colposcopic evaluation of a vaginal gel formulation of iota-carrageenan. Contraception 1997, 56, 387–389. [Google Scholar] [CrossRef]
- Maguire, R.A.; Zacharopoulos, V.R.; Phillips, D.M. Carrageenan-based nonoxynol-9 spermicides for prevention of sexually transmitted infections. Sex. Transm. Dis. 1998, 25, 494–500. [Google Scholar] [CrossRef]
- Flores, S.L.; Descallar, F.B.A.; Matsukawa, S.; Bacabac, R.G. Dynamic rheological properties of mixed carrageenan gels under large strains. J. Biorheol. 2017, 31, 35–39. [Google Scholar] [CrossRef] [Green Version]
- Yati, K.; Srifiana, Y.; Lestari, A.I. Optimization of SRC (Semi Refined Carrageenan) and Glucomannan Concentration as Gelling Agent to the Physical Stability Sunscreen Gel of Dry Corncob Extract (Zea mays L.). Open Access Maced J. Med. Sci. 2019, 7, 3833–3836. [Google Scholar] [CrossRef] [Green Version]
- Godha, K.; Tucker, K.M.; Biehl, C.; Archer, D.F.; Mirkin, S. Human vaginal pH and microbiota: An update. Gynecol. Endocrinol. 2018, 34, 451–455. [Google Scholar] [CrossRef]
- Tevi-Benissan, C.; Belec, L.; Levy, M.; Schneider-Fauveau, V.; Si Mohamed, A.; Hallouin, M.C.; Matta, M.; Gresenguet, G. In vivo semen-associated pH neutralization of cervicovaginal secretions. Clin. Diagn Lab. Immunol. 1997, 4, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Kleine-Borger, L.; Meyer, R.; Kalies, A.; Kerscher, M. Approach to differentiate between hyaluronic acid skin quality boosters and fillers based on their physicochemical properties. J. Cosmet. Derm. 2022, 21, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, S.; Nishinari, K. “Weak gel”-type rheological properties of aqueous dispersions of nonaggregated kappa-carrageenan helices. J. Agric. Food Chem. 2001, 49, 4436–4441. [Google Scholar] [CrossRef] [PubMed]
- Moreno, R. Rheology. In Encyclopedia of Materials: Science and Technology; Buschow, K.H.J., Cahn, R.W., Flemings, M.C., Ilschner, B., Kramer, E.J., Mahajan, S., Veyssière, P., Eds.; Elsevier: Oxford, UK, 2001; pp. 8192–8196. [Google Scholar]
- Tien, D.; Schnaare, R.L.; Kang, F.; Cohl, G.; McCormick, T.J.; Moench, T.R.; Doncel, G.; Watson, K.; Buckheit, R.W.; Lewis, M.G.; et al. In vitro and in vivo characterization of a potential universal placebo designed for use in vaginal microbicide clinical trials. AIDS Res. Hum. Retrovir. 2005, 21, 845–853. [Google Scholar] [CrossRef]
- Douglas, J.F. Weak and Strong Gels and the Emergence of the Amorphous Solid State. Gels 2018, 4, 19. [Google Scholar] [CrossRef] [Green Version]
- Soto, F.; Chrostowski, R. Frontiers of Medical Micro/Nanorobotics: In vivo Applications and Commercialization Perspectives Toward Clinical Uses. Front. Bioeng. Biotechnol. 2018, 6, 170. [Google Scholar] [CrossRef]
- Feola, A.; Duerr, R.; Moalli, P.; Abramowitch, S. Changes in the rheological behavior of the vagina in women with pelvic organ prolapse. Int. Urogynecol. J. 2013, 24, 1221–1227. [Google Scholar] [CrossRef] [Green Version]
- Bastholm, S.K.; Becher, N.; Stubbe, P.R.; Chronakis, I.S.; Uldbjerg, N. The viscoelastic properties of the cervical mucus plug. Acta Obs. Gynecol. Scand. 2014, 93, 201–208. [Google Scholar] [CrossRef]
- BeMiller, J.N. 13—Carrageenans. In Carbohydrate Chemistry for Food Scientists, 3rd ed.; BeMiller, J.N., Ed.; AACC International Press: St. Paul, MN, USA, 2019; pp. 279–291. [Google Scholar]
- Trios. AR-G2 Rheometer Analysis Manual. Available online: http://www.ifug.ugto.mx/~labblanda/manuales/Content/Rheology%20Common%20Topics/Analysis%20Models/Williamson.htm (accessed on 7 February 2022).
- Pettinelli, N.; Rodriguez-Llamazares, S.; Bouza, R.; Barral, L.; Feijoo-Bandin, S.; Lago, F. Carrageenan-based physically crosslinked injectable hydrogel for wound healing and tissue repairing applications. Int. J. Pharm. 2020, 589, 119828. [Google Scholar] [CrossRef]
- Cheng, A.T.Y.; Rodriguez, F. Mechanical properties of borate crosslinked poly(vinyl alcohol) gels. J. Appl. Polym. Sci. 1981, 26, 3895–3908. [Google Scholar] [CrossRef]
- Riedo, C.; Caldera, F.; Poli, T.; Chiantore, O. Poly(vinylalcohol)-borate hydrogels with improved features for the cleaning of cultural heritage surfaces. Herit. Sci. 2015, 3, 23. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Ye, L.; Cui, M.; Yang, B.; Li, J.; Sun, H.; Yao, F. Physically crosslinked poly(vinyl alcohol)–carrageenan composite hydrogels: Pore structure stability and cell adhesive ability. RSC Adv. 2015, 5, 78180–78191. [Google Scholar] [CrossRef]
- El-Fawal, G.F.; Yassin, A.M.; El-Deeb, N.M. The Novelty in Fabrication of Poly Vinyl Alcohol/kappa-Carrageenan Hydrogel with Lactobacillus bulgaricus Extract as Anti-inflammatory Wound Dressing Agent. AAPS PharmSciTech 2017, 18, 1605–1616. [Google Scholar] [CrossRef]
- Ochiai, H.; Shimizu, S.; Tadokoro, Y.; Murakami, I. Complex formation between poly(vinyl alcohol) and borate ion. Polymer 1981, 22, 1456–1458. [Google Scholar] [CrossRef]
- Itou, T.; Kitai, H.; Shimazu, A.; Miyazaki, T.; Tashiro, K. Clarification of cross-linkage structure in boric acid doped poly(vinyl alcohol) and its model compound as studied by an organized combination of X-ray single-crystal structure analysis, Raman spectroscopy, and density functional theoretical calculation. J. Phys. Chem. B 2014, 118, 6032–6037. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, Y.; Gu, Y.; Xue, P.; Xu, X. Self-Healing and Highly Stretchable Hydrogel for Interfacial Compatible Flexible Paper-Based Micro-Supercapacitor. Materials (Basel) 2021, 14, 1852. [Google Scholar] [CrossRef]
- Zhang, Y.; Hui, B.; Ye, L. Reactive toughening of polyvinyl alcohol hydrogel and its wastewater treatment performance by immobilization of microorganisms. RSC Adv. 2015, 5, 91414–91422. [Google Scholar] [CrossRef]
- Chiappe, C.; Signori, F.; Valentini, G.; Marchetti, L.; Pomelli, C.S.; Bellina, F. Novel (Glycerol)borate-Based Ionic Liquids: An Experimental and Theoretical Study. J. Phys. Chem. B 2010, 114, 5082–5088. [Google Scholar] [CrossRef]
- Fink, J.K. (Ed.) Chapter III—Fracturing Fluids. In Water-Based Chemicals and Technology for Drilling, Completion, and Workover Fluids; Gulf Professional Publishing: Boston, MA, USA, 2015; pp. 115–178. [Google Scholar]
- Ham, A.S.; Nugent, S.T.; Peters, J.J.; Katz, D.F.; Shelter, C.M.; Dezzutti, C.S.; Boczar, A.D.; Buckheit, K.W.; Buckheit, R.W., Jr. The rational design and development of a dual chamber vaginal/rectal microbicide gel formulation for HIV prevention. Antivir. Res. 2015, 120, 153–164. [Google Scholar] [CrossRef] [Green Version]
- Ayehunie, S.; Wang, Y.-Y.; Landry, T.; Bogojevic, S.; Cone, R.A. Hyperosmolal vaginal lubricants markedly reduce epithelial barrier properties in a three-dimensional vaginal epithelium model. Toxicol. Rep. 2018, 5, 134–140. [Google Scholar] [CrossRef]
- WHO/UNFPA/FHI. Use and Procurement of Additional Lubricants for Male and Female Condoms; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Ghani, N.A.A.; Othaman, R.; Ahmad, A.; Anuar, F.H.; Hassan, N.H. Impact of purification on iota carrageenan as solid polymer electrolyte. Arab. J. Chem. 2019, 12, 370–376. [Google Scholar] [CrossRef]
- Croitoru, C.; Pop, M.A.; Bedo, T.; Cosnita, M.; Roata, I.C.; Hulka, I. Physically Crosslinked Poly (Vinyl Alcohol)/Kappa-Carrageenan Hydrogels: Structure and Applications. Polymers 2020, 12, 560. [Google Scholar] [CrossRef] [Green Version]
- Alhosseini, S.N.; Moztarzadeh, F.; Mozafari, M.; Asgari, S.; Dodel, M.; Samadikuchaksaraei, A.; Kargozar, S.; Jalali, N. Synthesis and characterization of electrospun polyvinyl alcohol nanofibrous scaffolds modified by blending with chitosan for neural tissue engineering. Int. J. Nanomed. 2012, 7, 25–34. [Google Scholar] [CrossRef] [Green Version]
- Kharazmi, A.; Faraji, N.; Mat Hussin, R.; Saion, E.; Yunus, W.M.; Behzad, K. Structural, optical, opto-thermal and thermal properties of ZnS-PVA nanofluids synthesized through a radiolytic approach. Beilstein J. Nanotechnol. 2015, 6, 529–536. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.R. Laboratory development of a constitutive model for visco-elasto-plastic materials. KSCE J. Civ. Eng. 1999, 3, 261–271. [Google Scholar] [CrossRef]
- Available online: https://www.tainstruments.com/wp-content/uploads/Boston-Rheology-Training-2019.pdf (accessed on 1 March 2022).








| Gel | Ci (mg/mL) | PBA (mg/mL) | Williamson Model (r2) | Zero Shear Rate Viscosity (Pa. S) | Physical Properties | G′max (Pa) | G″max (Pa) |
|---|---|---|---|---|---|---|---|
| 1 | 20 | 33.33 | N.D. * | N.D. | Crumbly, hard, non-syringeable | 173.56 | 30.73 |
| 2 | 10 | 33.33 | N.D. | N.D. | Soft, syringeable with self-healing property | 43.48 | 5.70 |
| 3 | 5 | 30 | N.D. | N.D. | Fluidic, no gel behavior | 3.33 | 1.54 |
| 4 | 10 | 30 | 0.98 | 125,107 | Soft, syringeable | 34.40 | 8.65 |
| 5 | 20 | 30 | 0.99 | 163,155 | Hard but syringeable with self-healing property | 42.84 | 7.17 |
| 6 | 10 | 50 | 0.99 | 354,482 | Relatively hard with some syringeability | 72.22 | 7.63 |
| 7 | 8 | 20 | 0.99 | 103,782 | Extremely soft, syringeable | 28.67 | 8.81 |
| Gel | Ci (mg/mL) | PVA (mg/mL) | BA (mg/mL) | KCl (mg/mL) | Cross-linking | G′max (Pa) | G″max (Pa) | ωc (rad/s) | Modulus at Crossover (Pa) |
|---|---|---|---|---|---|---|---|---|---|
| 1P | 5 | 10 | 5 | 2.5 | No | 18.73 | 3.93 | NA | NA |
| 2P | 5 | 20 | 10 | 2.5 | Weak | 75.77 | 46.32 | 39.03 | 46.17 |
| 3P | 5 | 30 | 15 | 2.5 | Yes | 752.13 | 431.05 | 2.44 | 330.59 |
| 4P | 6 | 30 | 15 | 2.5 | Yes | 5020.03 | 2294.13 | 0.90 | 1570.58 |
| 5P | 5 | 10 | 5 | - | No | 10.14 | 4.39 | NA | NA |
| 6P | 9 | 30 | 10 | - | Yes | 175.97 | 125.05 | 9.49 | 122.59 |
| 7P | 9.5 | 30 | 10 | - | Yes | 321.93 | 444.94 | 13.42 | 388.11 |
| Gel | Acidic pH | Basic pH | ||
|---|---|---|---|---|
| Zero-Shear Rate Viscosity (Pa·S) | Williamson r2 Value | Zero-Shear Rate Viscosity (Pa·S) | Williamson r2 Value | |
| 2P | 2.99 | 0.93 | 44.39 | 0.99 |
| 3P | 8.46 | 0.93 | 448.12 | 0.99 |
| 4P | 50.67 | 0.99 | 4964.79 | 0.99 |
| 6P | 2.23 | 0.94 | 41.59 | 0.99 |
| 7P | 19.16 | 0.98 | 161.85 | 0.98 |
| Samples * | Polymer Composition and Gel pH | Approximate Sperm Penetration Distance (µm) ** | |
|---|---|---|---|
| 0 min | 20 min | ||
| Ci-PBA (Gel 2) | 33.33 mg/mL of PBA, 10 mg/mL of Ci, pH~4.4 | 0 ± 0 | 0 ± 0 |
| Ci-PBA (Gel 4) | 30 mg/mL of PBA, 10 mg/mL of Ci, pH~4.4 | 140 ± 3.5 | 390 ± 29.2 |
| Ci-PVA-BA (Gel 7P) | 30 mg/mL of PVA, 9.5 mg/mL of Ci, 10 mg/mL of BA, pH~4.8 | 150 ± 11.2 | 350 ± 35.4 |
| HEC gel | 2.7% w/w, pH~4.4 | >500 µm | Spread all over |
| N-9 gel | 4% w/w | 0 ± 0 *** | Not applicable |
| Treatment Arms | RCET # 1 | RCET # 2 | Summary of Two RCET Studies |
|---|---|---|---|
| # Pregnant/Total (% Contraceptive Efficacy) | # Pregnant/Total (% Contraceptive Efficacy) | # Pregnant/Total (% Contraceptive Efficacy) | |
| Sham control | 3/3 (0%) | 3/3 (0%) | 6/6 (0%) |
| HEC gel (2.7% w/w) | 4/5 (20%) | 4/4 (0%) | 8/9 (~11%) |
| N-9 gel (4% w/w) | 0/2 (100%) | 0/2 (100%) | 0/4 (100%) |
| Ci-PBA (Gel 2) | 2/5 (60%) | Not applicable | 2/5 (60%) |
| Ci-PBA (Gel 7) | Not applicable | 3/5 (40%) | 3/5 (40%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rochani, A.; Agrahari, V.; Chandra, N.; Singh, O.N.; McCormick, T.J.; Doncel, G.F.; Clark, M.R.; Kaushal, G. Development and Preclinical Investigation of Physically Cross-Linked and pH-Sensitive Polymeric Gels as Potential Vaginal Contraceptives. Polymers 2022, 14, 1728. https://doi.org/10.3390/polym14091728
Rochani A, Agrahari V, Chandra N, Singh ON, McCormick TJ, Doncel GF, Clark MR, Kaushal G. Development and Preclinical Investigation of Physically Cross-Linked and pH-Sensitive Polymeric Gels as Potential Vaginal Contraceptives. Polymers. 2022; 14(9):1728. https://doi.org/10.3390/polym14091728
Chicago/Turabian StyleRochani, Ankit, Vivek Agrahari, Neelima Chandra, Onkar N. Singh, Timothy J. McCormick, Gustavo F. Doncel, Meredith R. Clark, and Gagan Kaushal. 2022. "Development and Preclinical Investigation of Physically Cross-Linked and pH-Sensitive Polymeric Gels as Potential Vaginal Contraceptives" Polymers 14, no. 9: 1728. https://doi.org/10.3390/polym14091728









