A Study of the Phosphorylcholine Polymer Coating of a Polymethylpentene Hollow Fiber Membrane
Abstract
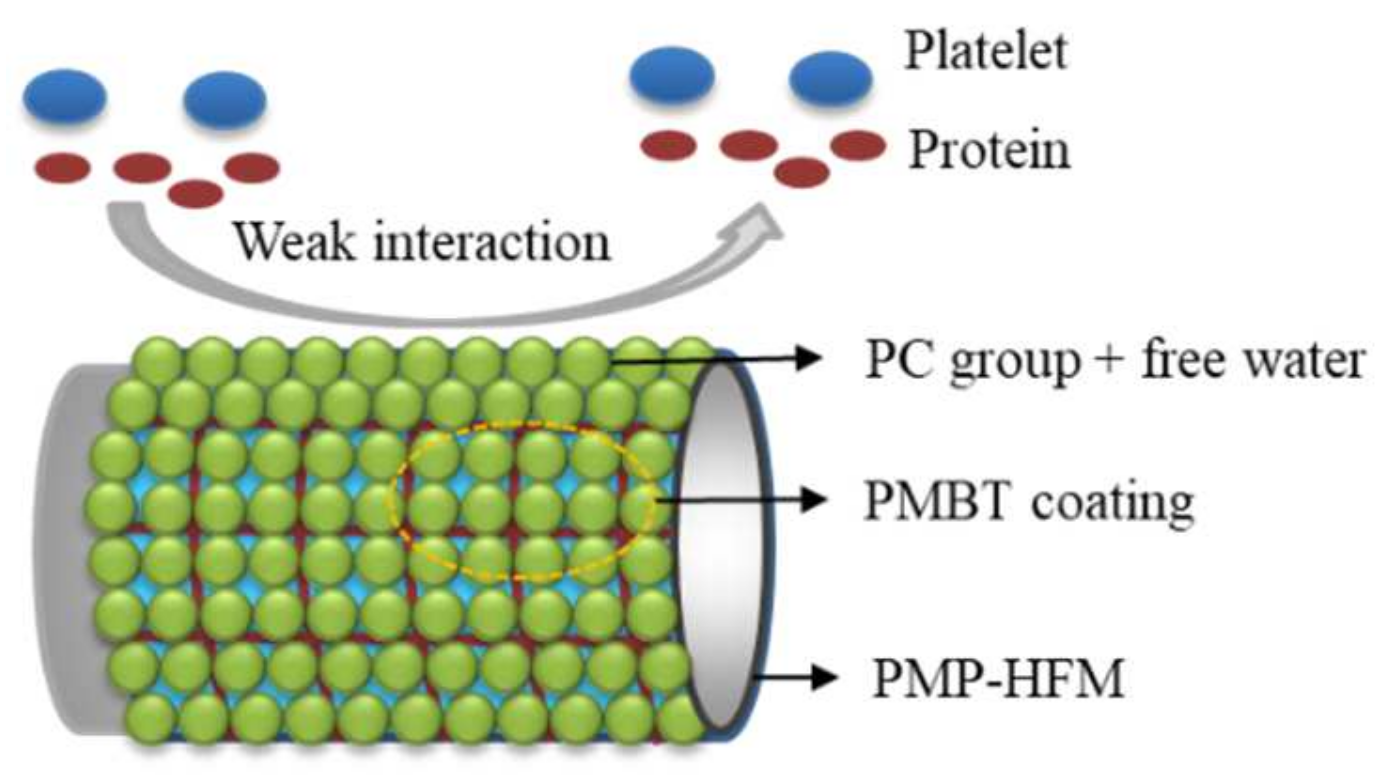
:1. Introduction
2. Experimental
2.1. Materials
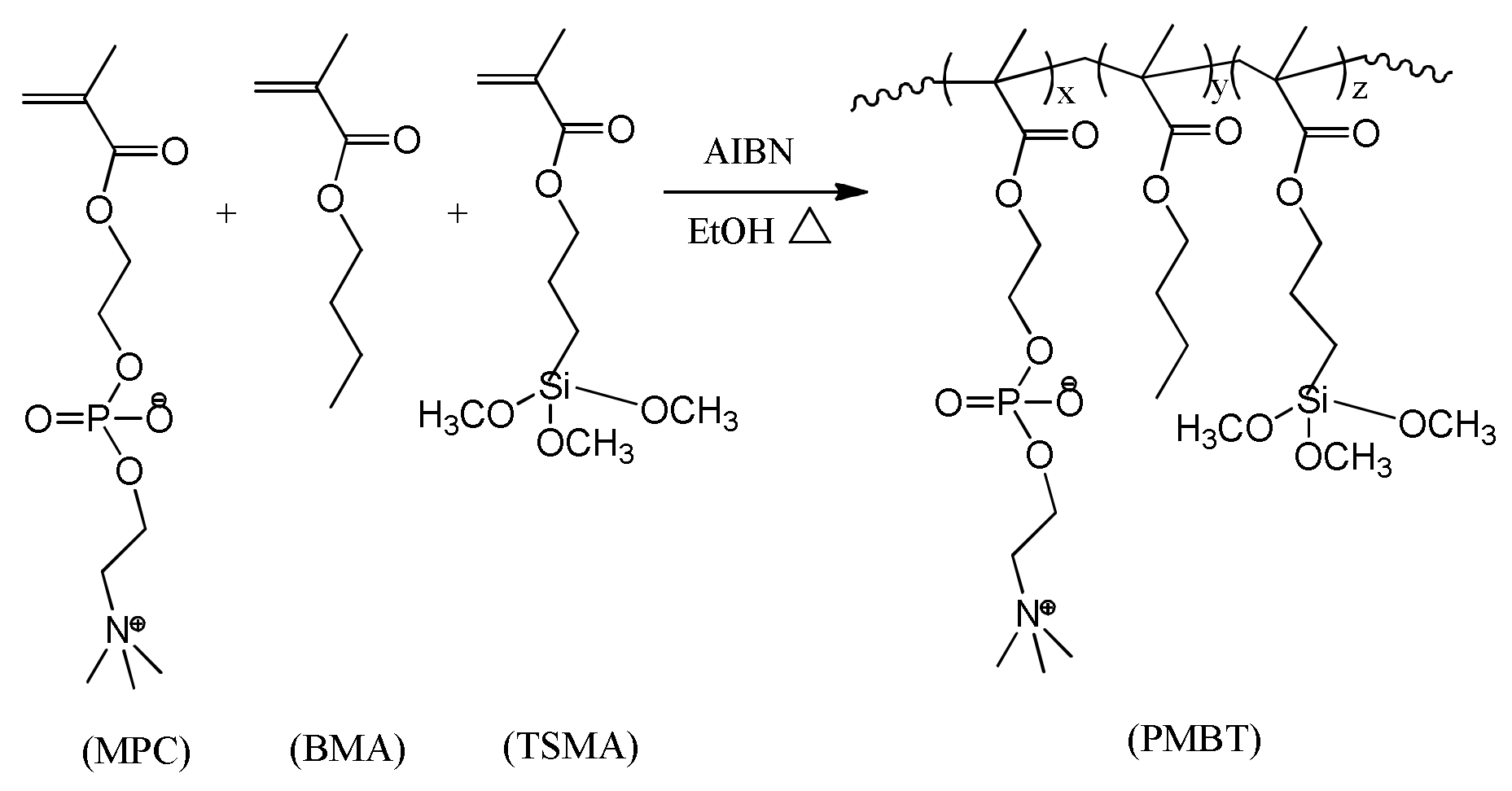
2.2. Synthesis of PMBT Polymer
2.3. Preparation of the PMBT Coating
2.4. Polymer Coating Analysis
2.5. Thermal Stability Test
2.6. Swelling Degree Test
2.7. Static Contact Angle Measurements
2.8. Fluorescence Properties of the Polymer Coating
2.9. Platelet Adhesion
2.10. Whole Blood Contact
3. Results and Discussion
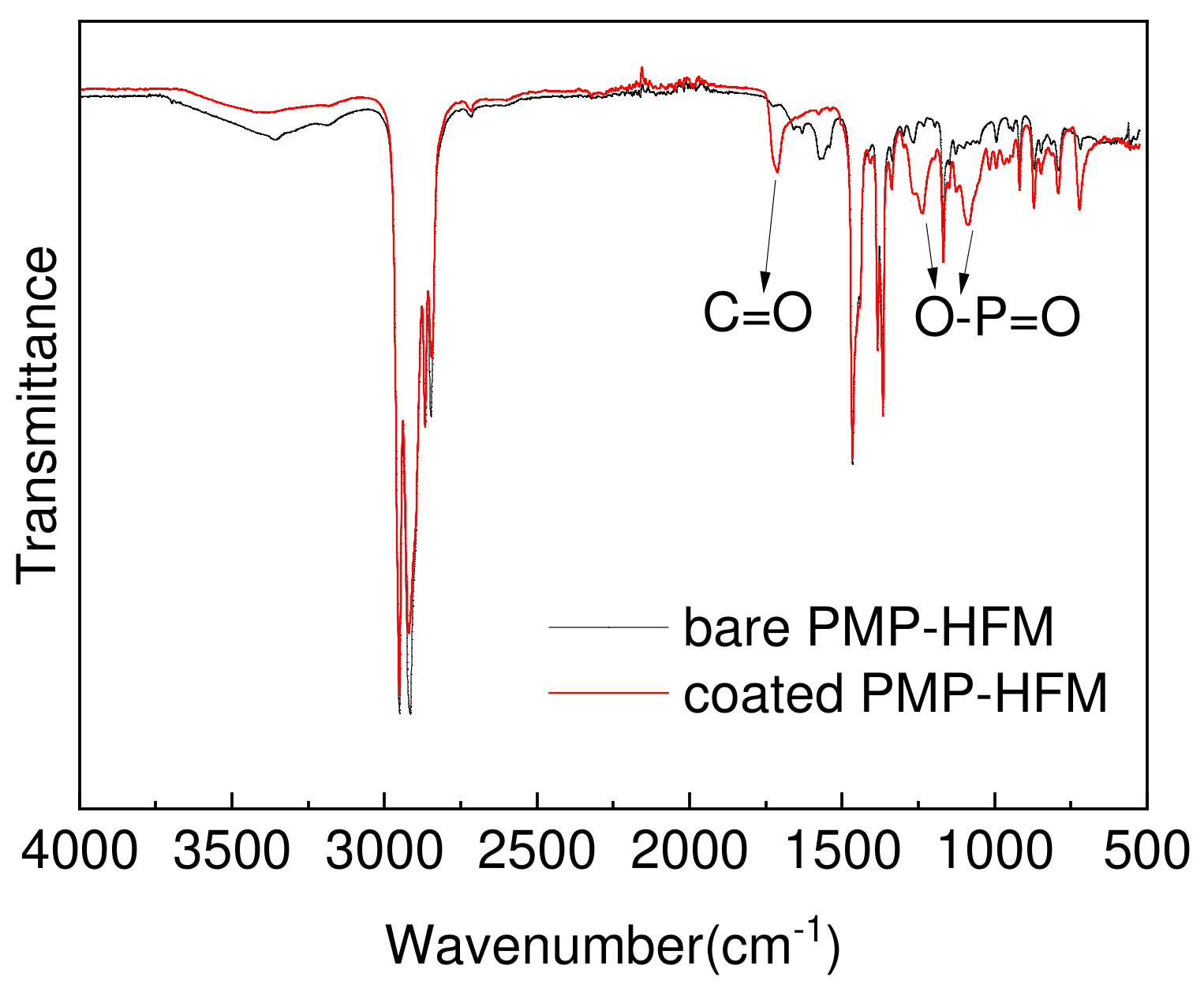
3.1. PMBT Coating Analysis
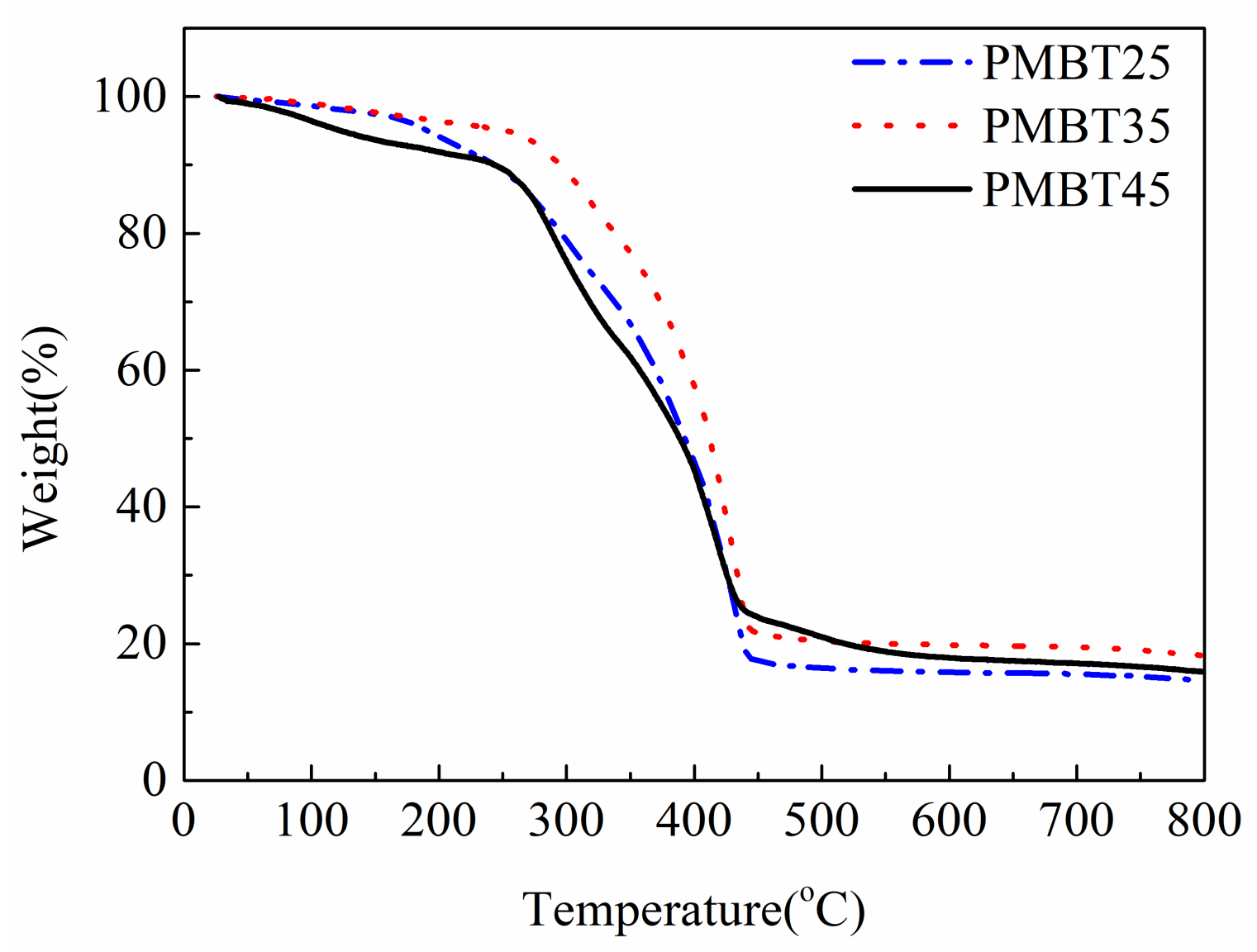
3.2. Thermal Stability Analysis of PMBT Polymer
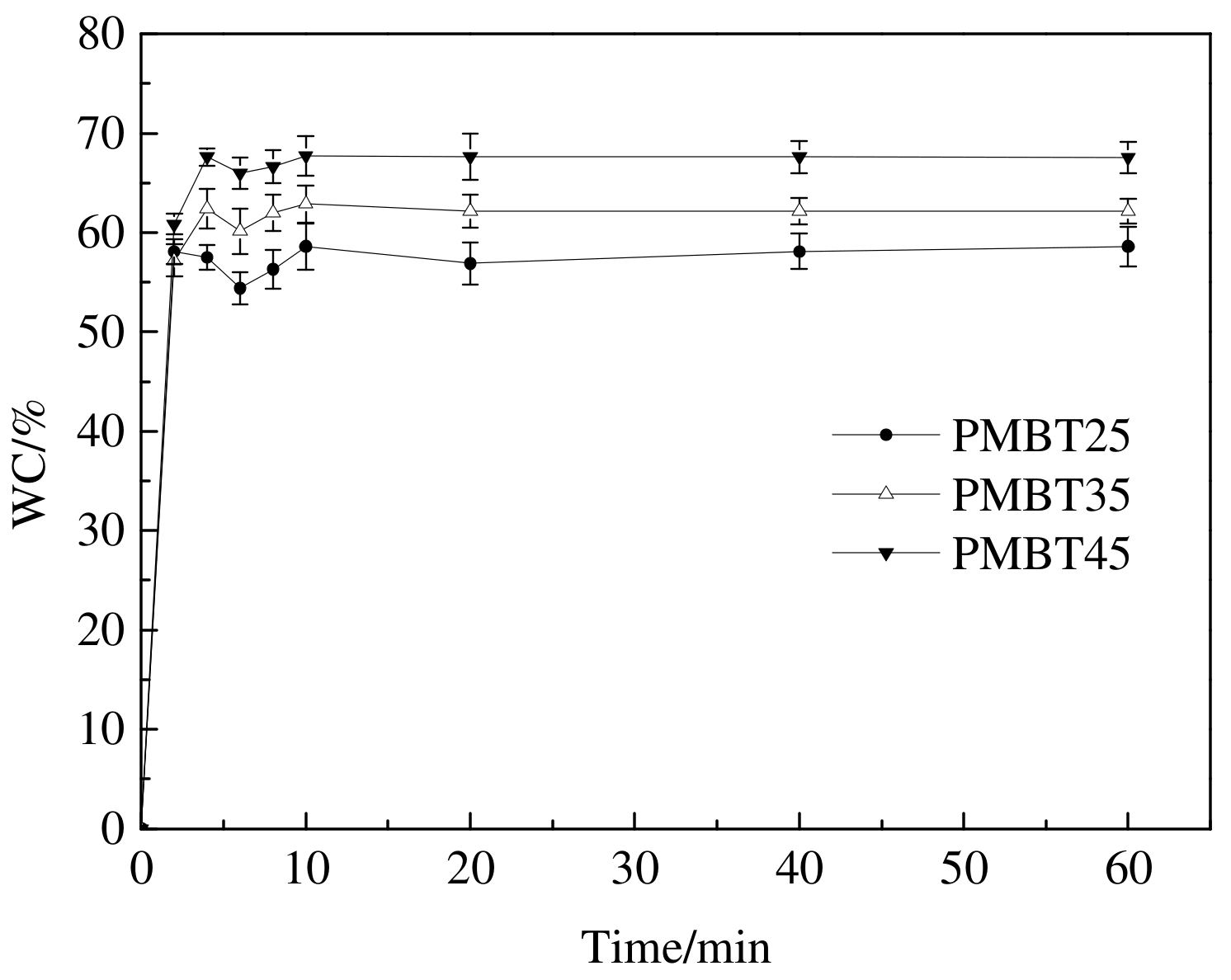
3.3. Swelling Degree Analysis of PMBT Polymer
3.4. Static Contact Angle Analysis
3.5. Analysis of the Fluorescence Properties of the Coatings
3.6. Biocompatibility Evaluation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fiore, A.; Piscitelli, M.; Adodo, D.K.; Thomas, C.; Dessap, A.M.; Bagate, F.; Folliguet, T. Successful Use of Extracorporeal Membrane Oxygenation Postpartum as Rescue Therapy in a Woman with COVID-19. J. Cardiothorac. Vasc. Anesth. 2021, 35, 2140–2143. [Google Scholar] [CrossRef] [PubMed]
- Teber, O.O.; Altinay, A.D.; Mehrabani, S.A.N.; Tasdemir, R.S.; Zeytuncu, B.; Genceli, E.A.; Dulekgurgen, E.; Pekkan, K.; Koyuncu, İ. Polymeric hollow fiber membrane oxygenators as artificial lungs: A review. Biochem. Eng. J. 2022, 180, 108340. [Google Scholar] [CrossRef]
- Ogawa, T.; Uemura, T.; Matsuda, W.; Sato, M.; Ishizuka, K.; Fukaya, T.; Kinoshita, N.; Nakamoto, T.; Ohmagari, N.; Katano, H.; et al. SARS-CoV-2 Leakage from the Gas Outlet Port During Extracorporeal Membrane Oxygenation for COVID-19. Asaio J. 2021, 67, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.K.; Hei, F.L. lmprovement and new development of hollow fiber membrane materials for membrane artificiall lung. Chin. J. Tissue Eng. Res. 2022, 26, 2608–2612. [Google Scholar]
- Zhi, Z.; Wang, W.; Xin, H.; Fan, W.; Lei, L.; Zhi, Z.; Wang, W.; Xin, H.; Fan, W.; Lei, L. Surface modification of polysulfone hollow fiber membrane for extracorporeal membrane oxygenator using low-temperature plasma treatment. Plasma Process. Polym. 2017, 15, 1700122. [Google Scholar]
- Xin, H.; Wang, W.; Zhi, Z.; Fan, W.; Mao, C.; Shi, J.; Lei, L. Surface monofunctionalized polymethyl pentene hollow fiber membranes by plasma treatment and hemocompatibility modification for membrane oxygenators. Appl. Surf. Sci. 2016, 362, 355–363. [Google Scholar]
- Garimella, N.; Wu, Z.; Griffith, B. Biocompatible Coating Composition. Patent US20150352265, 10 December 2015. [Google Scholar]
- Ishihara, K. Phospholipid Polymers; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012. [Google Scholar]
- Iwasaki, Y.; Ishihara, K. Cell membrane-inspired phospholipid polymers for developing medical devices with excellent biointerfaces. Sci. Technol. Adv. Mater. 2012, 13, 064101. [Google Scholar] [CrossRef]
- Santin, M.; Phillips, G. History of Biomimetic, Bioactive and Bioresponsive Biomaterials. In Biomimetic, Bioresponsive, and Bioactive Materials; Wiley: Hoboken, NJ, USA, 2012; pp. 1–34. [Google Scholar]
- Goda, T.; Ishihara, K.; Miyahara, Y. Review: Critical Update on 2-Methacryloyloxyethyl Phosphorylcholine (MPC) Polymer Science. J. Appl. Polym. Sci. 2015, 132, 41766. [Google Scholar] [CrossRef]
- Wei, Y.; Jing-xun, Z.; Ying, J.; Jian, J. REDV/Rapamycin-loaded polymer combinations as a coordinated strategy to enhance endothelial cells selectivity for a stent system. Colloids Surf. B. 2015, 136, 1166–1173. [Google Scholar] [CrossRef]
- Xie, L.; Gong, L.; Zhang, J.; Han, L.; Xiang, L.; Chen, J.; Liu, J.; Yan, B.; Zeng, H. Wet Adhesion Strategy Via Synergistic Cation–π and Hydrogen Bonding Interactions of Antifouling Zwitterions and Mussel-Inspired Binding Moiety. J. Mater. Chem. A 2019, 7, 21944–21952. [Google Scholar] [CrossRef]
- Sang, H.Y.; Watanabe, J.; Takai, M.; Iwasaki, Y.; Ishihara, K. High functional hollow fiber membrane modified with phospholipid polymers for a liver assist bioreactor. Biomaterials 2006, 27, 1955–1962. [Google Scholar]
- Sang, H.Y.; Watanabe, J.; Takai, M.; Iwasaki, Y.; Ishihara, K. Design of functional hollow fiber membranes modified with phospholipid polymers for application in total hemopurification system. Biomaterials 2005, 26, 5032–5041. [Google Scholar]
- Sang-Ho, Y.; Arazawa, D.T.; Yang, Z.; Venkat, S.; Malkin, A.D.; Kimmel, J.D.; Gamble, L.J.; Kazuhiko, I.; Federspiel, W.J.; Wagner, W.R. Hollow fiber membrane modification with functional zwitterionic macromolecules for improved thromboresistance in artificial lungs. Langmuir 2015, 31, 2463–2471. [Google Scholar]
- Myers, G.; Johnstone, D.; Swyer, W.; McTeer, S.; Maxwell, S.; Squires, C.; Ditmore, S.; Power, C.; Mitchell, L.; Ditmore, J.; et al. Evaluation of Mimesys® phosphorylcholine (PC)–coated oxygenators during cardiopulmonary bypass in adults. J. Extra Corpor. Technol. 2003, 35, 6–12. [Google Scholar] [PubMed]
- Teotia, R.S.; Dahe, G.J.; Bellare, J. In-Situ Coating of 2-Methacryloyloxyethyl Phosphorylcholine Polymer on Polysulfone Hollow Fiber Membranes for Hemodialysis. J. Comput. Theor. Nanosci. 2014, 20, A105. [Google Scholar] [CrossRef]
- Nishigochi, S.; Ishigami, T.; Maruyama, T.; Hao, Y.; Ohmukai, Y.; Iwasaki, Y.; Matsuyama, H. Improvement of Antifouling Properties of Polyvinylidene Fluoride Hollow Fiber Membranes by Simple Dip Coating of Phosphorylcholine Copolymer via Hydrophobic Interactions. Ind. Eng. Chem. Res. 2014, 53, 2491–2497. [Google Scholar] [CrossRef]
- Wang, Y.B.; Gong, M.; Yang, S.; Nakashima, K.; Gong, Y.K. Hemocompatibility and film stability improvement of crosslinkable MPC copolymer coated polypropylene hollow fiber membrane. J. Membr. Sci. 2014, 452, 29–36. [Google Scholar] [CrossRef]
- Ye, F.H.; Yi, G.B. Cross-linkable phosphonylcholine polymer modified polymethylpentene hollow fiber membrane. Acta Mater. Compos. Sinica. 2021, 38, 479–486. [Google Scholar]
- Wang, Y.B.; Shi, K.H.; Jiang, H.L.; Gong, Y.K. Significantly reduced adsorption and activation of blood components in a membrane oxygenator system coated with crosslinkable zwitterionic copolymer. Acta Biomater. 2016, 40, 153–161. [Google Scholar] [CrossRef]
- Hasegawa, T.; Iwasaki, Y.; Ishihara, K. Preparation of blood-compatible hollow fibers from a polymer alloy composed of polysulfone and 2-methacryloyloxyethyl phosphorylcholine polymer. J. Biomed. Mater. Res. 2002, 63, 333–341. [Google Scholar] [CrossRef]
- Asanuma, Y.; Inoue, Y.; Yusa, S.; Ishihara, K. Hybridization of poly(2-methacryloyloxyethyl phosphorylcholine-block-2-ethylhexyl methacrylate) with segmented polyurethane for reducing thrombogenicity. Colloids Surf. B. 2013, 108, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Sugihara, S.; Blanazs, A.; Armes, S.P.; Ryan, A.J.; Lewis, A.L. Aqueous Dispersion Polymerization: A New Paradigm for in Situ Block Copolymer Self-Assembly in Concentrated Solution. J. Am. Chem. Soc. 2011, 133, 15707–15713. [Google Scholar] [CrossRef]
- Goda, T.; Kjall, P.; Ishihara, K.; Richter-Dahlfors, A.; Miyahara, Y. Biomimetic Interfaces Reveal Activation Dynamics of C-Reactive Protein in Local Microenvironments. Adv. Healthc. Mater. 2014, 3, 1733–1738. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.L.; Tolhurst, L.A.; Stratford, P.W. Analysis of a phosphorylcholine-based polymer coating on a coronary stent pre- and post-implantation. Biomaterials 2002, 23, 1697–1706. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Bartlett, J.D.; Dunn, A.C.; Small, S.; Willis, S.L.; Driver, M.J.; Lewis, A.L. The use of rhodamine 6G and fluorescence microscopy in the evaluation of phospholipid-based polymeric biomaterials. J. Microsc. 2005, 217, 216–224. [Google Scholar] [CrossRef] [PubMed]





| Sample | MPC (mol%) | BMA (mol%) | TSMA (mol%) |
|---|---|---|---|
| PMBT25 | 25 | 65 | 10 |
| PMBT35 | 35 | 55 | 10 |
| PMBT45 | 45 | 45 | 10 |
| Samples | T(5%)/°C | T(10%)/°C | T(50%)/°C |
|---|---|---|---|
| PMBT25 | 191 | 245 | 393 |
| PMBT35 | 252 | 295 | 412 |
| PMBT45 | 123 | 244 | 389 |
| Samples | Bare PMP-HFM | PMBT35 Coated PMP-HFM | Treated with Ethanol Solution | Treated with SDS Solution |
|---|---|---|---|---|
| θ (deg) | 106 ± 0.6 * | 79 ± 0.8 * | 77 ± 1.3 * | 76 ± 1.1 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, F.; Chen, Z.; Li, C.; Chen, J.; Yi, G. A Study of the Phosphorylcholine Polymer Coating of a Polymethylpentene Hollow Fiber Membrane. Polymers 2023, 15, 2881. https://doi.org/10.3390/polym15132881
Ye F, Chen Z, Li C, Chen J, Yi G. A Study of the Phosphorylcholine Polymer Coating of a Polymethylpentene Hollow Fiber Membrane. Polymers. 2023; 15(13):2881. https://doi.org/10.3390/polym15132881
Chicago/Turabian StyleYe, Feihua, Zhisheng Chen, Chunsheng Li, Junhua Chen, and Guobin Yi. 2023. "A Study of the Phosphorylcholine Polymer Coating of a Polymethylpentene Hollow Fiber Membrane" Polymers 15, no. 13: 2881. https://doi.org/10.3390/polym15132881
APA StyleYe, F., Chen, Z., Li, C., Chen, J., & Yi, G. (2023). A Study of the Phosphorylcholine Polymer Coating of a Polymethylpentene Hollow Fiber Membrane. Polymers, 15(13), 2881. https://doi.org/10.3390/polym15132881







