Investigation of Cinnamic Acid Derivatives as Alternative Plasticizers for Improved Ductility of Polyvinyl Chloride Films
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Theoretical Solubility Parameters
2.3. Processing Plasticized PLA Formulations
2.4. Characterization of Plasticized PVC Formulations
2.4.1. Mechanical Properties
2.4.2. Thermal Properties
2.4.3. Thermomechanical Test
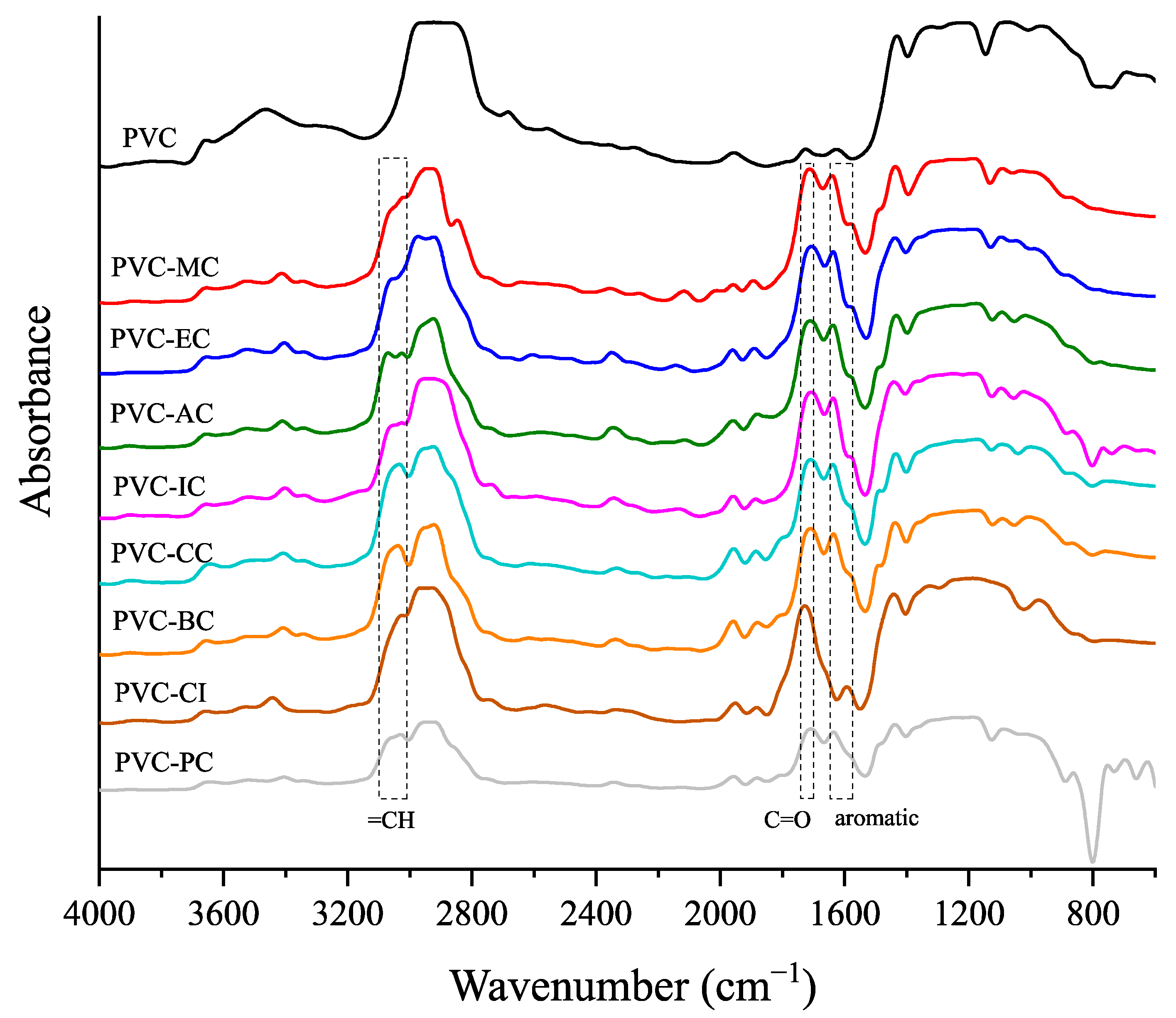
2.4.4. Fourier Transform Infrared Spectroscopy
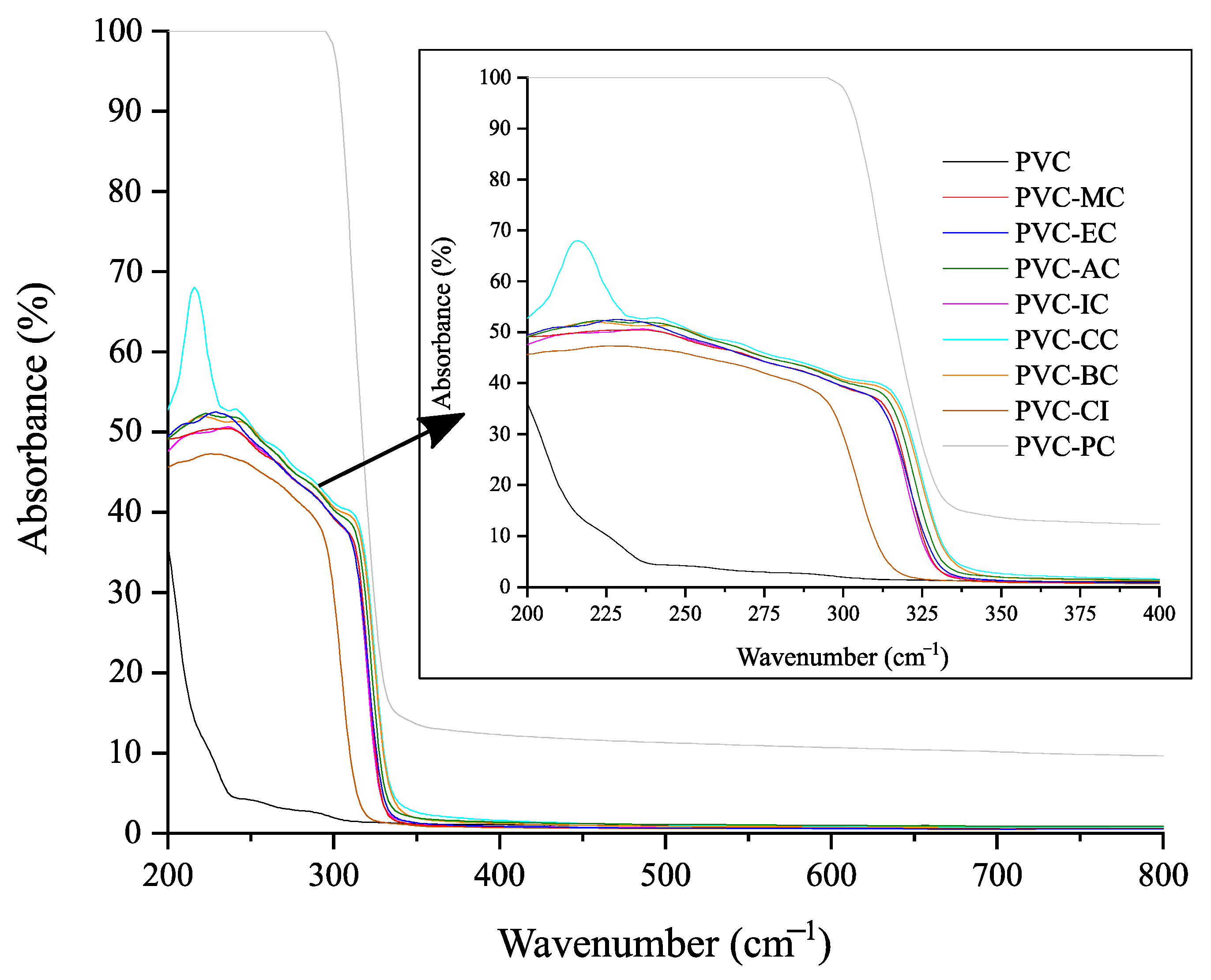
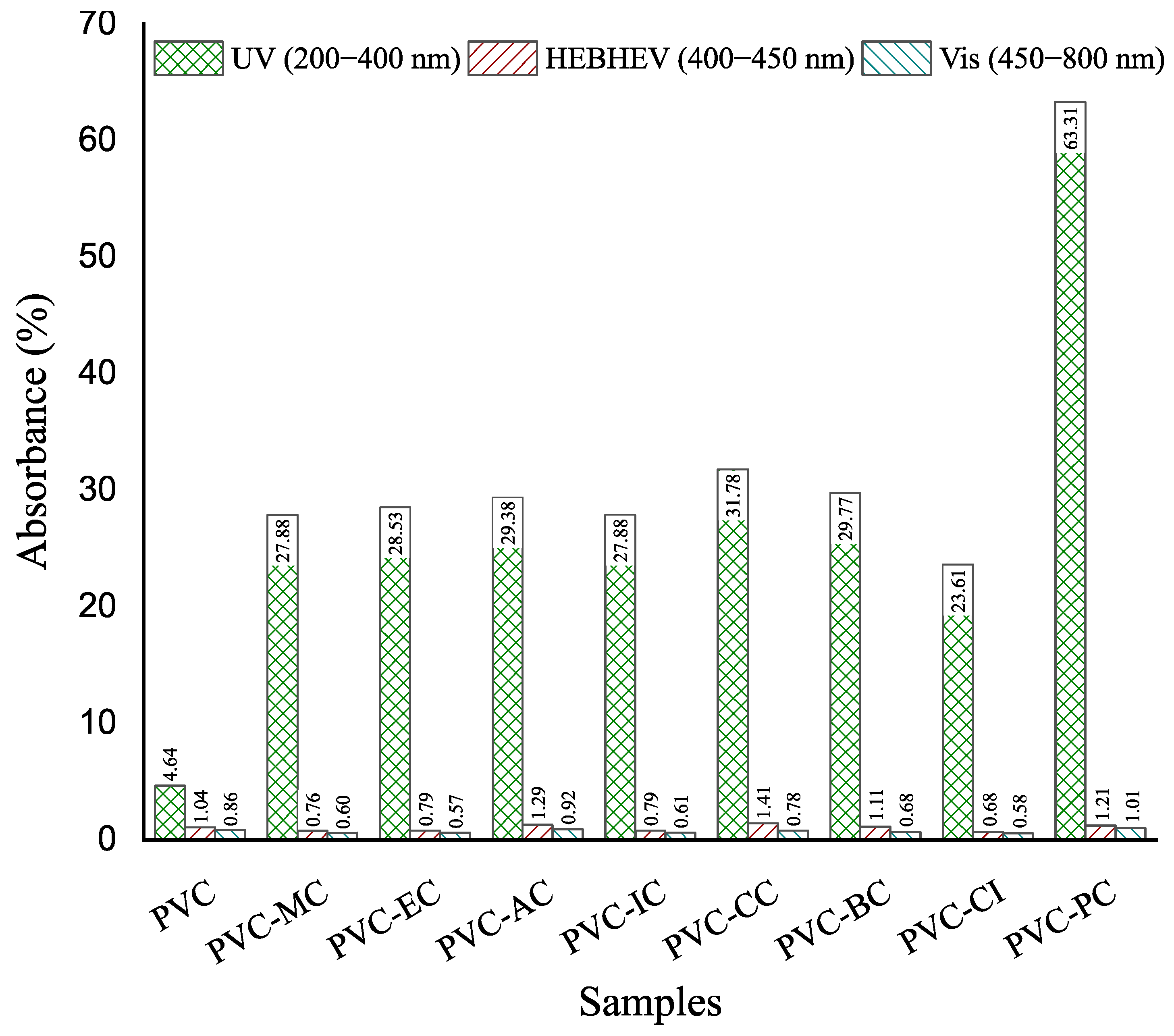
2.4.5. UV-Visible Spectrophotometry
2.4.6. Water Vapor Transmission Rate
2.4.7. Atomic Force Microscopy
2.4.8. Statistical Analysis
3. Results and Discussion
3.1. Theoretical Approach to Miscibility of PLA and Cinnamates
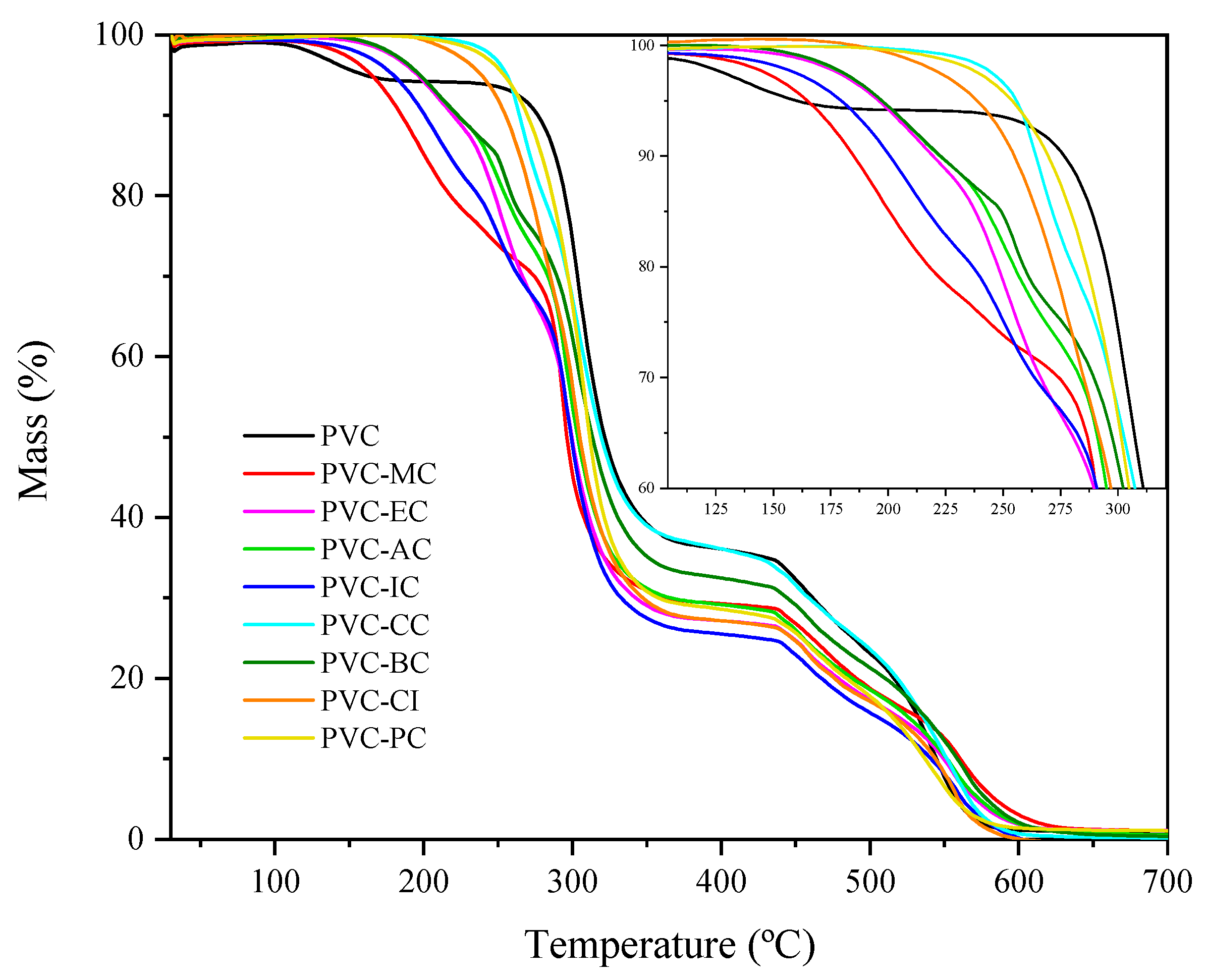
3.2. Analysis of Thermal Stability and Residual Plasticizer after Processing
3.3. Mechanical and Thermomechanical Properties of the Plasticized PVC with the Cinnamic Acid-Based Plasticizers
3.4. Optical Properties and Spectroscopic Characterization
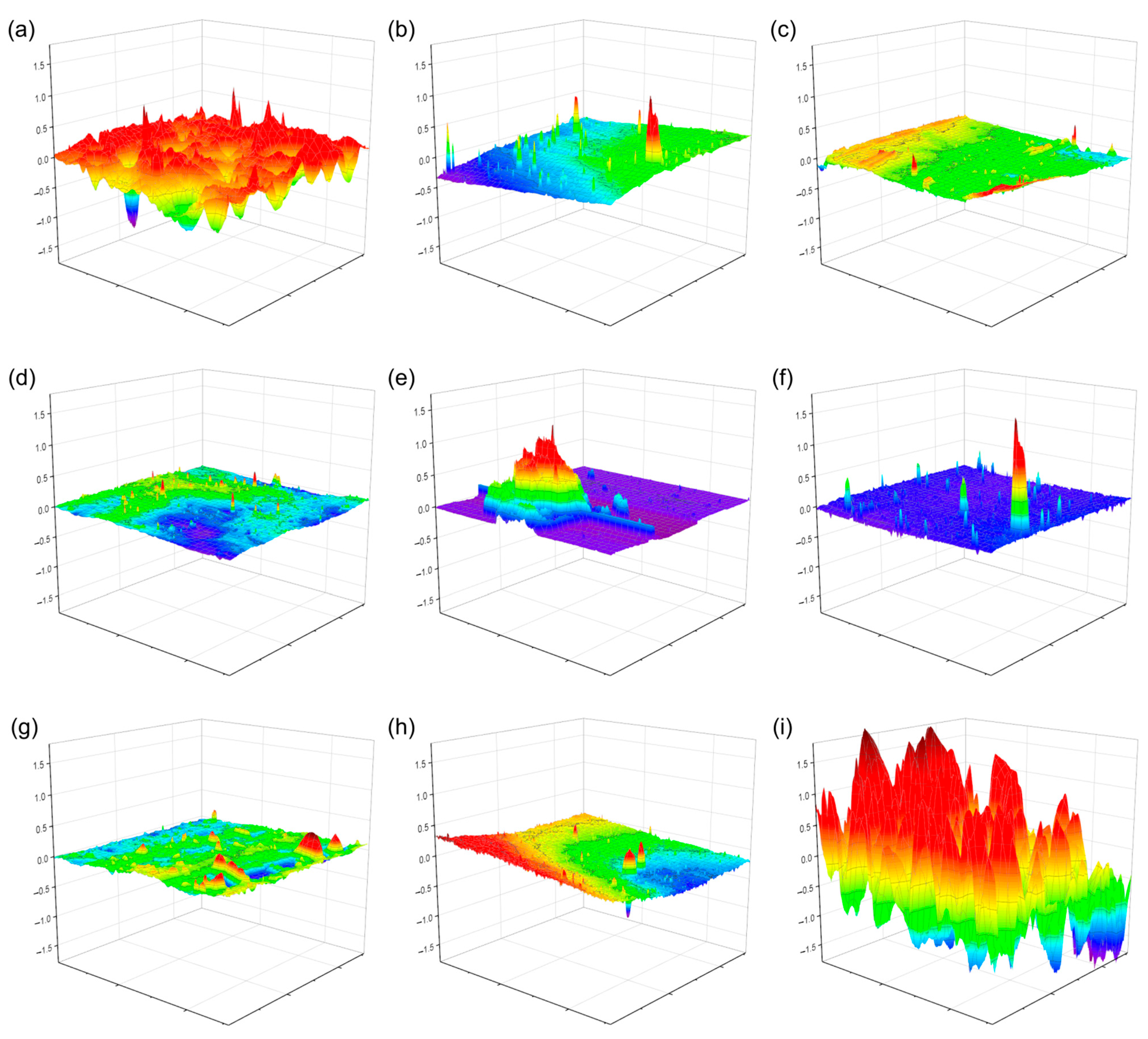
3.5. Water Vapor Transmission Rate (WVTR) and Surface Morphology
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Verified Market Research. Global Polyvinyl Chloride (PVC) Market Size by Type (Rigid, Flexible), by Application (Pipes & Fittings, Profiles, Films & Sheets, Cables), by End-User (Construction, Electrical Cables, Packaging), by Geographic Scope and Forecast. Available online: https://www.precedenceresearch.com/polyvinyl-chloride-market (accessed on 29 September 2023).
- Lewandowski, K.; Skórczewska, K. A Brief Review of Poly (Vinyl Chloride) (PVC) Recycling. Polymers 2022, 14, 3035. [Google Scholar] [CrossRef] [PubMed]
- Elgharbawy, A. Poly Vinyl Chloride Additives and Applications-A Review. J. Risk Anal. Crisis Response 2022, 12, 143–151. [Google Scholar] [CrossRef]
- Kumaresan, S.; Rokade, D.S.; Marathe, Y.N.; Ingole, P.G.; Pawar, R.R.; Pol, H.V.; Bajaj, H.C. Synthesis and characterization of nylon 6 polymer nanocomposite using organically modified Indian bentonite. SN Appl. Sci. 2020, 2, 1412. [Google Scholar] [CrossRef]
- Czogała, J.; Pankalla, E.; Turczyn, R. Recent attempts in the design of efficient PVC plasticizers with reduced migration. Materials 2021, 14, 844. [Google Scholar] [CrossRef] [PubMed]
- European Commission. The Use of PVC (Poly Vinyl Chloride) in the Context of a Non-Toxic Environment; Directorate-General for Environment: Final Report; Publications Office of the European Union: Luxembourg, 2022.
- Zhang, Z.; Jiang, P.; Liu, D.; Feng, S.; Zhang, P.; Wang, Y.; Fu, J.; Agus, H. Research progress of novel bio-based plasticizers and their applications in poly(vinyl chloride). J. Mater. Sci. 2021, 56, 10155–10182. [Google Scholar] [CrossRef]
- He, Z.; Lu, Y.; Lin, C.; Jia, H.; Wu, H.; Cao, F.; Ouyang, P. Designing anti-migration furan-based plasticizers and their plasticization properties in poly(vinyl chloride) blends. Polym. Test. 2020, 91, 106793. [Google Scholar] [CrossRef]
- Ji, S.; Gao, C.; Wang, H.; Liu, Y.; Zhang, D.; Zhang, S.; Lu, X.; Wu, Y.; Hu, Z. Application of a bio-based polyester plasticizer modified by hydrosilicon-hydrogenation reaction in soft PVC films. Polym. Adv. Technol. 2019, 30, 1126–1134. [Google Scholar] [CrossRef]
- Jia, P.; Zheng, M.; Ma, Y.; Feng, G.; Xia, H.; Hu, L.; Zhang, M.; Zhou, Y. Clean synthesis of epoxy plasticizer with quaternary ammonium phosphotungstate as catalyst from a byproduct of cashew nut processing. J. Clean. Prod. 2019, 206, 838–849. [Google Scholar] [CrossRef]
- Tan, J.; Liu, B.; Fu, Q.; Wang, L.; Xin, J.; Zhu, X. Role of the Oxethyl Unit in the Structure of Vegetable Oil-Based Plasticizer for PVC: An Efficient Strategy to Enhance Compatibility and Plasticization. Polymers 2019, 11, 779. [Google Scholar] [CrossRef]
- Diario Oficial de la Unión Europea. Reglamento (CE) No 1223/2009 del Parlamento Europeo Y del Consejo de 30 de Noviembre de 2009 Sobre los Productos Cosméticos; Visto el dictamen del Comité Económico y Social Europeo; Diario Oficial de la Unión Europea: Luxembourg, 2013. [Google Scholar]
- Muobom, S.S.; Umar, A.; Soongseok, Y.; Brolin, A.-P. A review on plasticizers and eco-friendly bioplasticizers: Biomass sources and market. Int. J. Eng. Res 2020, 9, 1138–1144. [Google Scholar]
- Han, J.; Zhang, M.; Zhang, H.; Liu, H.; Xu, S. Effects of modified tributyl citrate as a novel environmentally friendly plasticizer on the mechanical property and migration stability of soft polyvinyl chloride. J. Vinyl Addit. Technol. 2022, 28, 751–761. [Google Scholar] [CrossRef]
- Global, S.P. Chemical Economics Handbook—Plasticizers. 2021. Available online: https://www.spglobal.com/commodityinsights/en/ci/products/plasticizers-chemical-economics-handbook.html (accessed on 29 September 2023).
- Ceresena. Report details growth and change for global plasticizers market. Addit. Polym. 2019, 2012. Available online: https://ceresana.com/en/produkt/plasticizers-market-report (accessed on 29 September 2023).
- Jia, P.; Xia, H.; Tang, K.; Zhou, Y. Plasticizers derived from biomass resources: A short review. Polymers 2018, 10, 1303. [Google Scholar] [CrossRef] [PubMed]
- Abdelghany, A.; Meikhail, M.; Asker, N. Synthesis and structural-biological correlation of PVC\PVAc polymer blends. J. Mater. Res. Technol. 2019, 8, 3908–3916. [Google Scholar] [CrossRef]
- Chen, J.; Nie, X.; Jiang, J. Synthesis of a Novel Bio-Oil-Based Hyperbranched Ester Plasticizer and Its Effects on Poly(vinyl chloride) Soft Films. ACS Omega 2020, 5, 5480–5486. [Google Scholar] [CrossRef] [PubMed]
- Ledniowska, K.; Nosal-Kovalenko, H.; Janik, W.; Krasuska, A.; Stańczyk, D.; Sabura, E.; Bartoszewicz, M.; Rybak, A. Effective, environmentally friendly PVC plasticizers based on succinic acid. Polymers 2022, 14, 1295. [Google Scholar] [CrossRef] [PubMed]
- Bocqué, M.; Voirin, C.; Lapinte, V.; Caillol, S.; Robin, J.J. Petro-based and bio-based plasticizers: Chemical structures to plasticizing properties. J. Polym. Sci. Part A Polym. Chem. 2016, 54, 11–33. [Google Scholar] [CrossRef]
- Barandiaran, A.; Gomez-Caturla, J.; Ivorra-Martinez, J.; Lascano, D.; Selles, M.A.; Moreno, V.; Fenollar, O. Esters of Cinnamic Acid as Green Plasticizers for Polylactide Formulations with Improved Ductility. Macromol. Mater. Eng. 2023, 308, 2300022. [Google Scholar] [CrossRef]
- Bonifacio, A.; Bonetti, L.; Piantanida, E.; De Nardo, L. Plasticizer design strategies enabling advanced applications of cellulose acetate. Eur. Polym. J. 2023, 197, 112360. [Google Scholar] [CrossRef]
- Eslami, Z.; Elkoun, S.; Robert, M.; Adjalle, K. A Review of the Effect of Plasticizers on the Physical and Mechanical Properties of Alginate-Based Films. Molecules 2023, 28, 6637. [Google Scholar] [CrossRef]
- Venkatram, S.; Kim, C.; Chandrasekaran, A.; Ramprasad, R. Critical assessment of the Hildebrand and Hansen solubility parameters for polymers. J. Chem. Inf. Model. 2019, 59, 4188–4194. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.S.; Jena, H.M. Performance assessment of polyvinyl chloride films plasticized with Citrullus lanatus seed oil based novel plasticizer. Polym. Test. 2021, 101, 107271. [Google Scholar] [CrossRef]
- Moya, M.S.A.; Asensio, R.C.; González, M.L.G. Factores responsables de la degradación química de los polímeros: Efectos provocados por la radiación lumínica sobre algunos materiales utilizados en conservación: Primeros resultados. In Proceedings of the Conservación de Arte Contemporáneo: 11ª jornada, Madrid, Spain, 11 February 2010; pp. 283–308. [Google Scholar]
- UNE EN ISO 527; Determination of Tensile Properties Part 1: General Principles. ISO: Geneva, Switzerland, 2019; pp. 1–26.
- Li, X.; Xu, J. Synthesis of CdS QDs with single cubic and hexagonal lattice for blue-light-blocking nanocomposite films with a narrow absorbing transitional band. Mater. Today Commun. 2020, 24, 101108. [Google Scholar] [CrossRef]
- ASTM E96/E96M-16; Standard Test Methods for Water Vapor Transmission of Materials. Annual Book of American Standard Testing Methods. American Society for Testing and Materials: West Conshohocken, PA, USA, 2016; pp. 719–725.
- Shi, Y.; Yao, Y.; Lu, S.; Chen, L.; Chen, S.; He, H.; Ma, M.; Wang, X. Synergistic Effect of Two Plasticizers on Thermal Stability, Transparency, and Migration Resistance of Zinc Arginine Stabilized PVC. Polymers 2022, 14, 4560. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Xie, W.; Wu, H.; Wang, L.; Liang, S.; Chang, H.; Liu, B. Effect of volatile solvent and evaporation time on formation and performance of PVC/PVC-g-PEGMA blended membranes. RSC Adv. 2019, 9, 34486–34495. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yu, E.; Li, Y.; Wei, Z. Novel branched poly(ɛ-caprolactone) as a nonmigrating plasticizer in flexible PVC: Synthesis and characterization. J. Appl. Polym. Sci. 2018, 135, 46542. [Google Scholar] [CrossRef]
- Chen, J.; Liu, Z.; Nie, X.; Zhou, Y.; Jiang, J.; Murray, R.E. Plasticizers derived from cardanol: Synthesis and plasticization properties for polyvinyl chloride(PVC). J. Polym. Res. 2018, 25, 128. [Google Scholar] [CrossRef]
- Ma, Y.; Liao, S.; Li, Q.; Guan, Q.; Jia, P.; Zhou, Y. Physical and chemical modifications of poly(vinyl chloride) materials to prevent plasticizer migration—Still on the run. React. Funct. Polym. 2020, 147, 104458. [Google Scholar] [CrossRef]
- Cruz, P.P.R.; da Silva, L.C.; Fiuza-Jr, R.A.; Polli, H. Thermal dehydrochlorination of pure PVC polymer: Part I—Thermal degradation kinetics by thermogravimetric analysis. J. Appl. Polym. Sci. 2021, 138, 50598. [Google Scholar] [CrossRef]
- Feng, G.; Ma, Y.; Zhang, M.; Jia, P.; Liu, C.; Zhou, Y. Synthesis of bio-base plasticizer using waste cooking oil and its performance testing in soft poly (vinyl chloride) films. J. Bioresour. Bioprod. 2019, 4, 99–110. [Google Scholar] [CrossRef]
- Ivorra-Martinez, J.; Peydro, M.A.; Gomez-Caturla, J.; Boronat, T.; Balart, R. The Potential of an Itaconic Acid Diester as Environmentally Friendly Plasticizer for Injection-Molded Polylactide Parts. Macromol. Mater. Eng. 2022, 307, 2200360. [Google Scholar] [CrossRef]
- Hwang, T.; Frank, Z.; Neubauer, J.; Kim, K.J. High-performance polyvinyl chloride gel artificial muscle actuator with graphene oxide and plasticizer. Sci. Rep. 2019, 9, 9658. [Google Scholar] [CrossRef] [PubMed]
- Najafi, V.; Abdollahi, H. Internally Plasticized PVC by Four Different Green Plasticizer Compounds. Eur. Polym. J. 2020, 128, 109620. [Google Scholar] [CrossRef]
- Zhu, H.; Yang, J.; Wu, M.; Wu, Q.; Liu, J.; Zhang, J. Biobased Plasticizers from Tartaric Acid: Synthesis and Effect of Alkyl Chain Length on the Properties of Poly(vinyl chloride). ACS Omega 2021, 6, 13161–13169. [Google Scholar] [CrossRef] [PubMed]
- Su, K.; Tao, Y.; Zhang, J. Highly transparent plasticized PVC composite film with ideal ultraviolet/high-energy short-wavelength blue light shielding. J. Mater. Sci. 2021, 56, 17353–17367. [Google Scholar] [CrossRef]
- Hasan, A.A.; Al-Mashhadani, M.H.; Al-Dahhan, W.H.; Hashim, H.; Yousif, E. Synthesized and Designed New Modified Poly(vinyl chloride) Structures to Enhance Their Photo-Resistance Characteristics. Chemistry 2022, 4, 1101–1122. [Google Scholar] [CrossRef]
- Ghani, H.; Yousif, E.; Ahmed, D.S.; Kariuki, B.M.; El-Hiti, G.A. Tin Complexes of 4-(Benzylideneamino)benzenesulfonamide: Synthesis, Structure Elucidation and Their Efficiency as PVC Photostabilizers. Polymers 2021, 13, 2434. [Google Scholar] [CrossRef]
- Janik, R.; Kohutiar, M.; Dubec, A.; Eckert, M.; Moricova, K.; Pajtasova, M.; Ondrusova, D.; Krbata, M. DMA Analysis of Plasma Modified PVC Films and the Nature of Initiated Surface Changes. Materials 2022, 15, 4658. [Google Scholar] [CrossRef]
- Howell, B.A.; Sun, W. Biobased Plasticizers from Tartaric Acid, an Abundantly Available, Renewable Material. Ind. Eng. Chem. Res. 2018, 57, 15234–15242. [Google Scholar] [CrossRef]
- Jia, P.; Ma, Y.; Zhang, M.; Hu, L.; Zhou, Y. Designing Rosin-Based Plasticizers: Effect of Differently Branched Chains on Plasticization Performance and Solvent Resistance of Flexible Poly(vinyl chloride) Films. ACS Omega 2019, 4, 3178–3187. [Google Scholar] [CrossRef]



| Cinnamic Acid-Derivative | Melting Point (°C) | Boiling Point (°C) | Molecular Weight (g mol−1) | Density (g cm−3) | Molar Volume (cm3 mol−1) |
|---|---|---|---|---|---|
| MC | 34–38 a | 260–262 a | 162.188 | 1.092 | 148.524 |
| IC | - | 287 a | 204.269 | 1.005 | 203.658 |
| AC | - | 289 a | 188.226 | 1.053 | 178.752 |
| EC | 6–7 a | 271 a | 176.215 | 1.050 | 168.305 |
| CC | 42–45 a | 370 a | 264.324 | 1.121 | 235.793 |
| CI | - | 295–297 a | 204.269 | 1.008 | 202.648 |
| BC | 37–39 a | 195–200 b | 238.286 | 1.11 | 214.672 |
| PC | 54–56 a | 300–301 a | 252.313 | 1.108 | 227.719 |
| Code | (MPa1/2) | (MPa1/2) | (MPa1/2) | (MPa1/2) | (MPa1/2) |
|---|---|---|---|---|---|
| PVC [31,32] | 18.54 | 8.56 | 3.31 | 20.68 | - |
| MC | 15.83 | 4.66 | 11.81 | 20.29 | 0.39 |
| EC | 16.06 | 6.63 | 10.50 | 20.94 | 0.26 |
| AC | 15.06 | 6.72 | 10.09 | 20.65 | 0.03 |
| IC | 16.47 | 5.92 | 8.43 | 19.58 | 1.10 |
| CC | 15.11 | 5.47 | 8.75 | 19.13 | 1.55 |
| BC | 16.76 | 7.15 | 11.53 | 22.68 | 2.00 |
| CI | 15.85 | 5.72 | 9.05 | 19.89 | 0.79 |
| PC | 16.91 | 6.59 | 10.63 | 21.95 | 1.27 |
| Code | T5 (°C) | T15 (°C) | T50 (°C) |
|---|---|---|---|
| PVC | 283.4 ± 2.7 | 287.4 ± 3.2 | 321.7 ± 3.6 |
| PVC-MC | 165.5 ± 2.1 | 200.3 ± 2.7 | 296.9 ± 2.7 |
| PVC-EC | 195.7 ± 1.9 | 238.2 ± 2.4 | 299.5 ± 2.0 |
| PVC-AC | 197.5 ± 1.7 | 242.7 ± 1.9 | 303.7 ± 2.4 |
| PVC-IC | 180.1 ± 1.9 | 217.3 ± 2.0 | 299.5 ± 2.2 |
| PVC-CC | 256.2 ± 3.7 | 271.4 ± 2.9 | 319.3 ± 1.7 |
| PVC-BC | 197.5 ± 2.4 | 249.2 ± 1.7 | 314.5 ± 1.9 |
| PVC-CI | 238.9 ± 3.3 | 264.2 ± 2.2 | 304.8 ± 2.5 |
| PVC-PC | 254.0 ± 0.7 | 279.3 ± 3.3 | 311.6 ± 1.8 |
| Tensile Strength (MPa) | Young’s Modulus (MPa) | Elongation at Break (%) | |
|---|---|---|---|
| PVC | 37.7 ± 2.0 | 1023.6 ± 56.0 | 8.8 ± 2.0 |
| PVC-MC | 11.6 ± 0.9 | 7.9 ± 1.5 | 343.0 ± 27.3 |
| PVC-EC | 9.6 ± 0.6 | 5.8 ± 1.0 | 312.1 ± 23.4 |
| PVC-AC | 11.6 ± 1.0 | 7.5 ± 0.3 | 308.3 ± 18.0 |
| PVC-IC | 10.4 ± 0.6 | 8.1 ± 1.3 | 308.8 ± 13.5 |
| PVC-CC | 22.2 ± 1.0 | 119.9 ± 16.5 | 279.3 ± 10.2 |
| PVC-BC | 16.3 ± 1.3 | 17.0 ± 1.8 | 261.4 ± 15.9 |
| PVC-CI | 13.5 ± 0.8 | 6.2 ± 0.5 | 371.6 ± 12.8 |
| PVC-PC | 16.8 ± 0.7 | 47.8 ± 4.5 | 298.2 ± 14.4 |
| Code | Storage Modulus, E’ (MPa) | Tg (°C) | |
|---|---|---|---|
| at 30 °C | at 70 °C | ||
| PVC | 2008.0 ± 39.0 | 1077.1 ± 26.8 | 83.8 ± 2.4 |
| PVC-MC | 14.1 ± 1.4 | 3.6 ± 0.2 | 14.7 ± 0.5 |
| PVC-EC | 10.5 ± 0.7 | 3.2 ± 0.2 | 12.9 ± 0.4 |
| PVC-AC | 16.4 ± 1.1 | 3.9 ± 0.3 | 17.2 ± 0.3 |
| PVC-IC | 7.8 ± 0.7 | 2.4 ± 0.3 | 11.7 ± 0.4 |
| PVC-CC | 75.9 ± 2.2 | 2.6 ± 0.2 | 33.4 ± 0.8 |
| PVC-BC | 10.3 ± 0.4 | 2.4 ± 0.1 | 17.5 ± 0.4 |
| PVC-CI | 10.0 ± 0.6 | 2.9 ± 0.2 | 5.0 ± 0.3 |
| PVC-PC | 84.6 ± 2.0 | 2.3 ± 0.2 | 21.2 ± 0.7 |
| Code | WVTR (×10−12 g µm m2 h−1 Pa−1) |
|---|---|
| PVC | 1.04 ± 0.05 a |
| PVC-MC | 1.58 ± 0.09 b |
| PVC-EC | 2.00 ± 0.12 b |
| PVC-AC | 2.92 ± 0.10 b |
| PVC-IC | 2.26 ± 0.09 b |
| PVC-CC | 1.31 ± 0.06 b |
| PVC-BC | 1.35 ± 0.05 b |
| PVC-CI | 4.18 ± 0.15 b |
| PVC-PC | 1.11 ± 0.03 a |
| Plastification of PVC Films with Different Plasticizers and Involved Methods | |||
|---|---|---|---|
| Plasticizer(s) | Processing | Comments | Reference |
| 2,5-furandicarboxylic acid and butyl oligoglycol ethers | Esterification synthesis | Reduction of the melt-point. Increased plasticization efficiency and drop in Tg | [8] |
| Cardanol and quaternary ammonium phosphotungstate | Casting method | Improved thermal stability and drop in Tg | [10] |
| Esters/ethers derived from tartaric acid | Casting method | Good plasticizing effect, displays low migration potential, and does not negatively impact the thermal stability of polymers | [46] |
| Hyperbranched poly(ε-caprolactone | Copolymerization | High plasticization efficiency and minimal plasticizers migration, even under very harsh conditions | [33] |
| Polyethylene glycol methyl ether and dimer acid | Hydrolysis of fatty acids from soybean oil at 70 °C and subsequent Diels-Alder reaction at 250 °C | Tensile properties, transparency and thermal stability of plasticized PVC increased significantly with more oxyethyl units | [11] |
| Hyperbranched ester plasticizer (SOHE) | Synthesis with a large number of steps | Greater thermal stability and flexibility. The migration stability of PVC samples enhances when increasing the amount of SOHE | [19] |
| Castor oil methyl ester, cardanol, and triethyl citrate with rosin-based plasticizers | Synthesis with a large number of steps according to free volume theory and lubricity theory | Good plasticizing efficiency and miscibility | [47] |
| Cardanol, cardanol acetate and epoxidized cardanol acetate | Synthesis with several steps | Overall superior flexibility, compatibility, thermal stability and workability than commercial phthalate plasticizers | [34] |
| Cinnamic acid derivatives | Solvent casting method | Good compatibility with PVC. Improved ductility, thermal and mechanical properties. Increased permeability | This paper |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barandiaran, A.; Montanes, N.; Sanchez-Nacher, L.; Balart, R.; Selles, M.A.; Moreno, V. Investigation of Cinnamic Acid Derivatives as Alternative Plasticizers for Improved Ductility of Polyvinyl Chloride Films. Polymers 2023, 15, 4265. https://doi.org/10.3390/polym15214265
Barandiaran A, Montanes N, Sanchez-Nacher L, Balart R, Selles MA, Moreno V. Investigation of Cinnamic Acid Derivatives as Alternative Plasticizers for Improved Ductility of Polyvinyl Chloride Films. Polymers. 2023; 15(21):4265. https://doi.org/10.3390/polym15214265
Chicago/Turabian StyleBarandiaran, Alejandro, Nestor Montanes, Lourdes Sanchez-Nacher, Rafael Balart, Miguel Angel Selles, and Virginia Moreno. 2023. "Investigation of Cinnamic Acid Derivatives as Alternative Plasticizers for Improved Ductility of Polyvinyl Chloride Films" Polymers 15, no. 21: 4265. https://doi.org/10.3390/polym15214265
APA StyleBarandiaran, A., Montanes, N., Sanchez-Nacher, L., Balart, R., Selles, M. A., & Moreno, V. (2023). Investigation of Cinnamic Acid Derivatives as Alternative Plasticizers for Improved Ductility of Polyvinyl Chloride Films. Polymers, 15(21), 4265. https://doi.org/10.3390/polym15214265









