1. Introduction
As a series of new functional materials, intelligent materials have greatly promoted the development of modern technology in the 21st century. Intelligent systems composed of these materials can sense and respond to external stimuli, mimicking the operation of living systems [
1,
2,
3]. The emergence of information technology, high-end equipment manufacturing and the innovation of material research and technology drive the rapid development of advanced smart materials. Recently, in view of the unique properties of smart materials, such as the shape memory effect and damage–repair characteristics, related research has attracted extensive attention.
Shape memory materials are intelligent materials that can reversibly transform between their initial permanent shape and their temporary shape under specific environmental conditions (such as specified temperature, humidity, light, pH and magnetic and electric fields [
4,
5,
6,
7,
8,
9]). Compared to alloys and ceramics, the other two shape memory materials, polymers have the advantages of adjustable mechanical properties, good biocompatibility, light weights and low prices. Based on the above advantages, shape memory polymers (SMPs) can be widely used as important components in aerospace [
10], biomedical devices [
11,
12,
13,
14] and sensing actuators [
15,
16].
Self-healing polymers (SHPs) are also responsive, intelligent materials that can repair damage and prolong the service life of products. Therefore, SHPs are of great significance in saving energy and reducing environmental pollution. According to the healing mechanism, SHPs can be divided into two categories: exogenous and endogenous. Exogenous SHPs usually need to incorporate external healing agents, the main forms of which include embedded microcapsules, filled hollow fibers and biomimetic, three-dimensional, microvascular networks [
17,
18,
19]. Endogenous SHPs mostly rely on chemically reversible bonds to achieve a self-healing process. Some bonds, such as reversible ionic bonds [
20,
21], covalent bonds [
22,
23,
24] and supramolecular chemical bonds, play an effective role in endogenous SHPs [
25,
26,
27]. In addition, the effect of physical diffusion cannot be underestimated in SHPs [
28,
29].
In recent years, the excellent physical properties and fast responsiveness make intelligent, self-healing rubber materials promising candidates for flexible devices and long-life structural materials. To date, research on self-healing rubber materials mainly focuses on natural rubber (NR), butyl rubber (IIR), nitrile rubber (NBR) and other materials. For instance, Landro et al. investigated the self-healing behavior of epoxidized natural rubber (ENR) and cis-1, 4-polyisoprene (PISP) blends [
30]. Heinrich et al. modified the bromine functional group of bromobutyl rubber (BIIR) into ionic imidazolium bromide groups to prepare highly elastic composites with extraordinary self-healing properties [
31]. Carboxyl nitrile rubber (XNBR) was reported to be ionically cross-linked by zinc oxide (ZnO) to form ionic clusters, which exhibited a certain self-repairing ability and prolonged the life cycle of the rubber material [
32].
However, there are still some problems to be solved in covalent cross-linking, self-healing rubber materials, such as low self-healing efficiency, high cost and complex preparation processes. Therefore, it is important to develop composite materials with excellent properties by simple, economic and efficient preparation methods.
Eucommia ulmoides rubber (EUR) is mainly composed of trans-1, 4-polyisoprene, which can be used as a shape memory material with a low cross-linking density [
33,
34]. EUR exhibits excellent shape memory properties, which enables the material to recover its original shape when subjected to external forces such as stretching, compression, etc. There are relatively few research papers on the shape memory properties and self-healing properties of Eucommia rubber. Yue et al. modified Eucommia ulmoides rubber by the epoxidation method and prepared a composite with both shape memory and self-healing properties with a tensile strength of ~12.1 MPa and elongation of ~876% [
35].
Here, we manufactured EUR- and SR-based blends with a moderately cross-linking approach. Surlyn resin (SR) is an ethylene–methacrylic acid copolymer thermoplastic resin whose internal ionic bonds give it unique properties and potential applications for self-healing [
36,
37,
38,
39,
40,
41]. Semi-crystalline and lightly cross-linked EUR and SR enable blends to complete shape memory processes with proper programming procedures while electrostatic interactions of ionomers provide the prerequisite for self-healing. The blends with a EUR/SR blending ratio of 10/90 showed an R
f of 96.46% and an R
r of 82.47%, respectively, and EUR/SR blends with a blending ratio of 30/70 achieved the highest self-healing efficiency of 87.41%, which was much higher than the self-healing efficiency of ~40% at the same blending ratio (SR/PCO = 70/30) in similar Surlyn 9520 ionomer and polycyclooctene systems [
42]. Therefore, this work provides a convenient and facile approach for manufacturing rubber-based composites with high shape memory and high self-healing properties. EUR/SR blends have potential applications in medical devices and various sensor fields.
2. Experimental Procedure
Herein, we demonstrate, for the first time, the fabrication of a composite with excellent mechanical shape memory and self-healing properties by using EUR and SR as the matrix via a simple blending method. The double cross-linking network constructed by the covalent cross-linking of the EUR component and the ion cross-linking of the SR component gave the composite the dual effect of heat-induced shape memory and self-healing. Significantly, the self-healing efficiency of the composites reached 87.41%, which is difficult for other covalent cross-linking materials to achieve.
2.1. Materials
The bark of
Eucommia ulmoides was crushed and dissolved in the organic solvent chloroform, the dissolved EUR was precipitated using freezing ice water and then the gum was filtered to obtain solid EUR. The lyophilized resin 8920 Na-ionomer contained methacrylic acid groups (5.4 mol%) and MA groups, sixty percent of which were neutralized by sodium (DuPont Co., Ltd., Wilmington, DE, USA). The chemical structure of EUR and SR is shown in
Figure 1. Dicumyl peroxide (DCP, purity 96%) was purchased from Aladdin Co., Ltd. (Shanghai, China). Other reagents were obtained from commercial sources and used without further purification.
2.2. Preparation of the Blends
EUR and SR blends were prepared by a high-temperature, open mill with a temperature of 90 °C for approximately 10 min using a standard mixing sequence. After the melting and mixing process, these composites were pressed into plates at 160 °C with a thickness of 2 ± 0.1 mm. The compound formulations are shown in
Table 1.
2.3. Curing Characteristics
The curing characteristics of the EUR/SR blends were monitored by a Monsanto oscillating disc rheometer (MDR-2000, GOTECH TESTING MACHINES Inc., Taichung, Taiwan) at 160 °C, according to ASTM D-2084-11, and torque was recorded over time. The scorch time T10, optimum cure time T90, minimum torque and maximum torque were determined from curing graphs.
2.4. Mechanical Characterization
Vulcanized slabs were dumbbell-shaped by die-cutting according to ASTM Die C. The mechanical tests were conducted following the ASTM D-412-16 and ASTM D-624-00 standard procedures using a tensile testing machine (AI-7000S, High-Speed Rail Co., Ltd., Taiwan). For each set of samples, five splines were tested and the average value was used as the experimental result. In addition, according to ASTM D2240-2015, the hardness of the composites was determined using a hardness tester (LX-A, Shanghai Liuling Instrument Factory, Shanghai, China).
2.5. Differential Scanning Calorimetry (DSC)
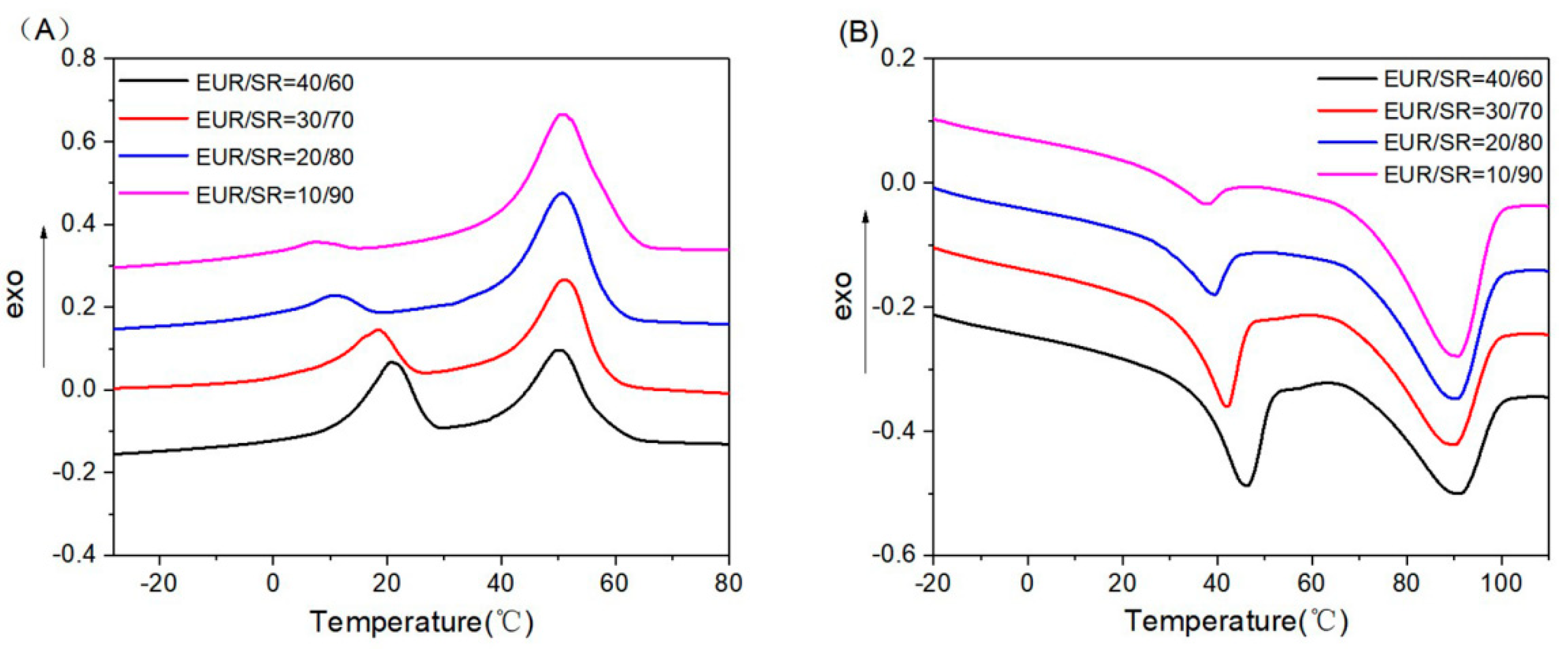
DSC measurements were carried out on a DSC-Q20 (TA Instruments, New Castle, DE, USA) under a nitrogen atmosphere over the temperature range from −50 to 120 °C at a heating speed of 10 °C min
−1. Samples were maintained at 120 °C for 3 min to eliminate their thermal histories before cooling. The exothermic curves of heat flow as a function of time were recorded to calculate the degree of crystallinity (X
c) for each portion of the composites using Equation (1):
where ΔH
m and ΔH
m* are the melting enthalpy of the polymer and its theoretical melting enthalpy (ca. 186.8 J∙g
−1 for EUR), respectively. ΔH
m in Surlyn resin is calculated by the mass fraction of the target component.
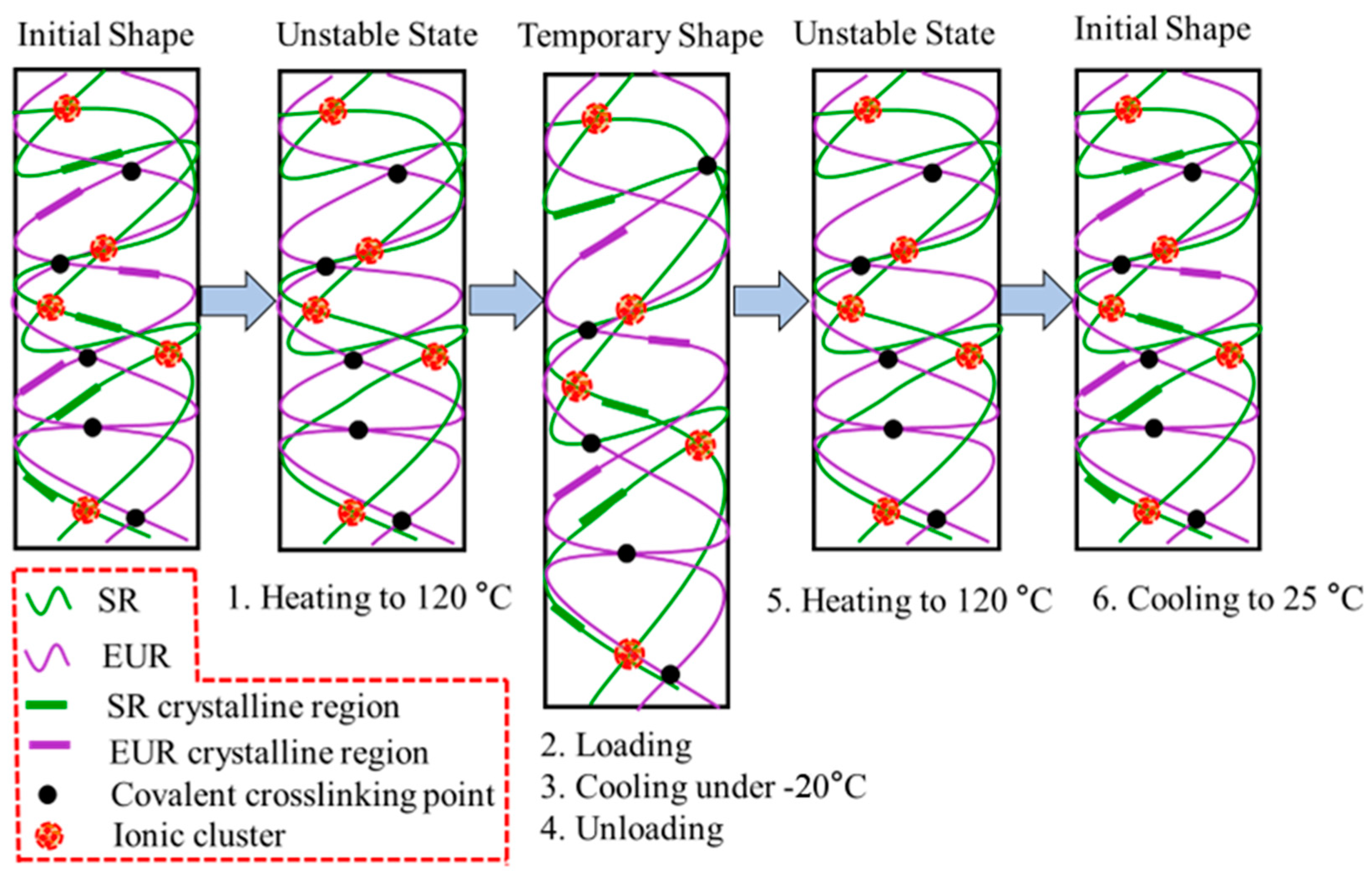
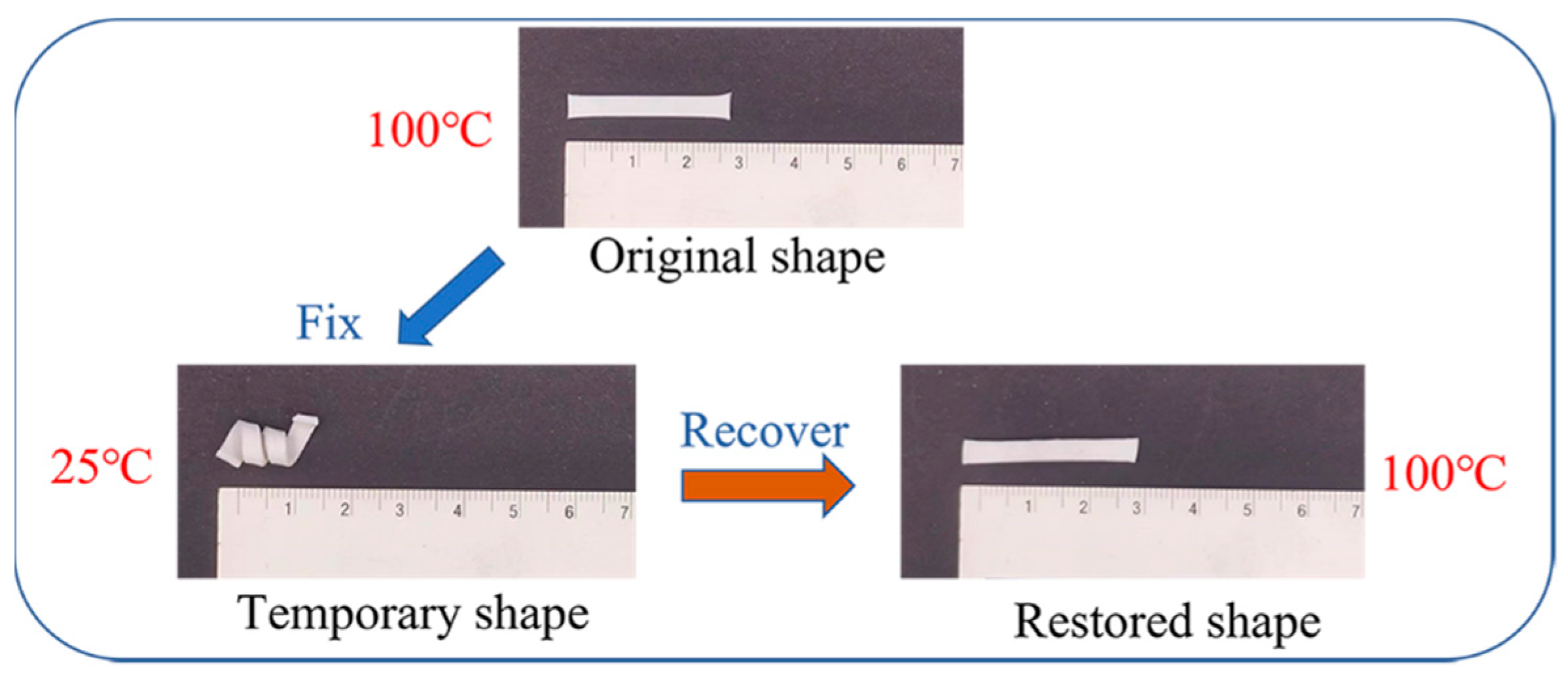
2.6. Shape Memory Effect
A TA Instruments DMA Q800 with the initial clamp gap set to 5.0~10.0 mm was used to analyze the shape memory properties of the composites. The specifications of the test sample and the test procedure for the shape memory effect were the same as the works we reported earlier [
43,
44]. First, the sample was maintained isothermally at 120 °C to melt the crystalline regions completely. Then, a load of 0.02 MPa was applied, after which the sample was cooled to −20 °C to freeze the crystalline domain completely. Then, the load was removed, and the sample was reheated to 120 °C and isothermally maintained for 15 min. The critical parameters for shape memory effect (SME) characterization, shape fixity ratio (R
f) and shape recovery ratio (R
r) can be quantified as follows:
where ε
0 is the initial strain, ε
1,load represents the maximum strain under the load, ε
1 is the strain after cooling and load removal, and ε
0,rec is the recovered strain.
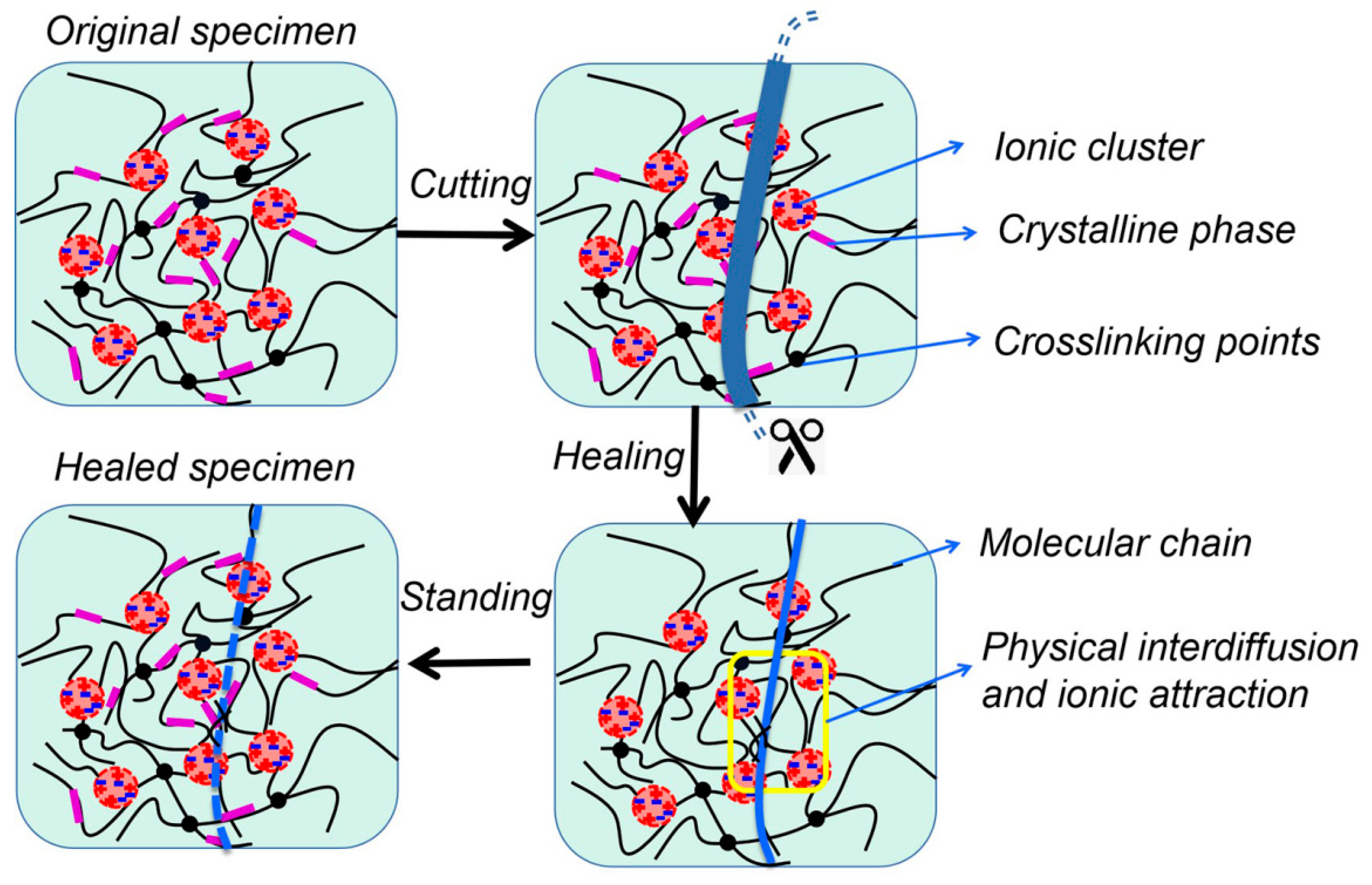
2.7. Self-Healing Effect
To perform the self-healing test, the standard dumbbell-type tensile splines were first cut into two independent parts from the middle with a blade, the fracture surfaces were spliced and the healing process started at 120 °C and 10 MPa pressure. Finally, the specimens were left to stand at room temperature for 5 days. The healing efficiency was quantified by comparing the tensile strengths of the healed sample and the original sample by stretching to fracture at a cross-head velocity of 500 mm·min
−1 at 25 °C, as shown in Equation (4). For each set of samples, three splines were tested and the average value was used as the tensile strength. In addition, the self-healing cracks were sprayed with gold and placed on the aluminum base with conductive tape to observe the surface morphology under a scanning electron microscope (SEM) (JEOL JSM-6700F, Tokyo, Japan).