Solvent-Evaporation-Induced Synthesis of Graphene Oxide/Peptide Nanofiber (GO/PNF) Hybrid Membranes Doped with Silver Nanoparticles for Antibacterial Application
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
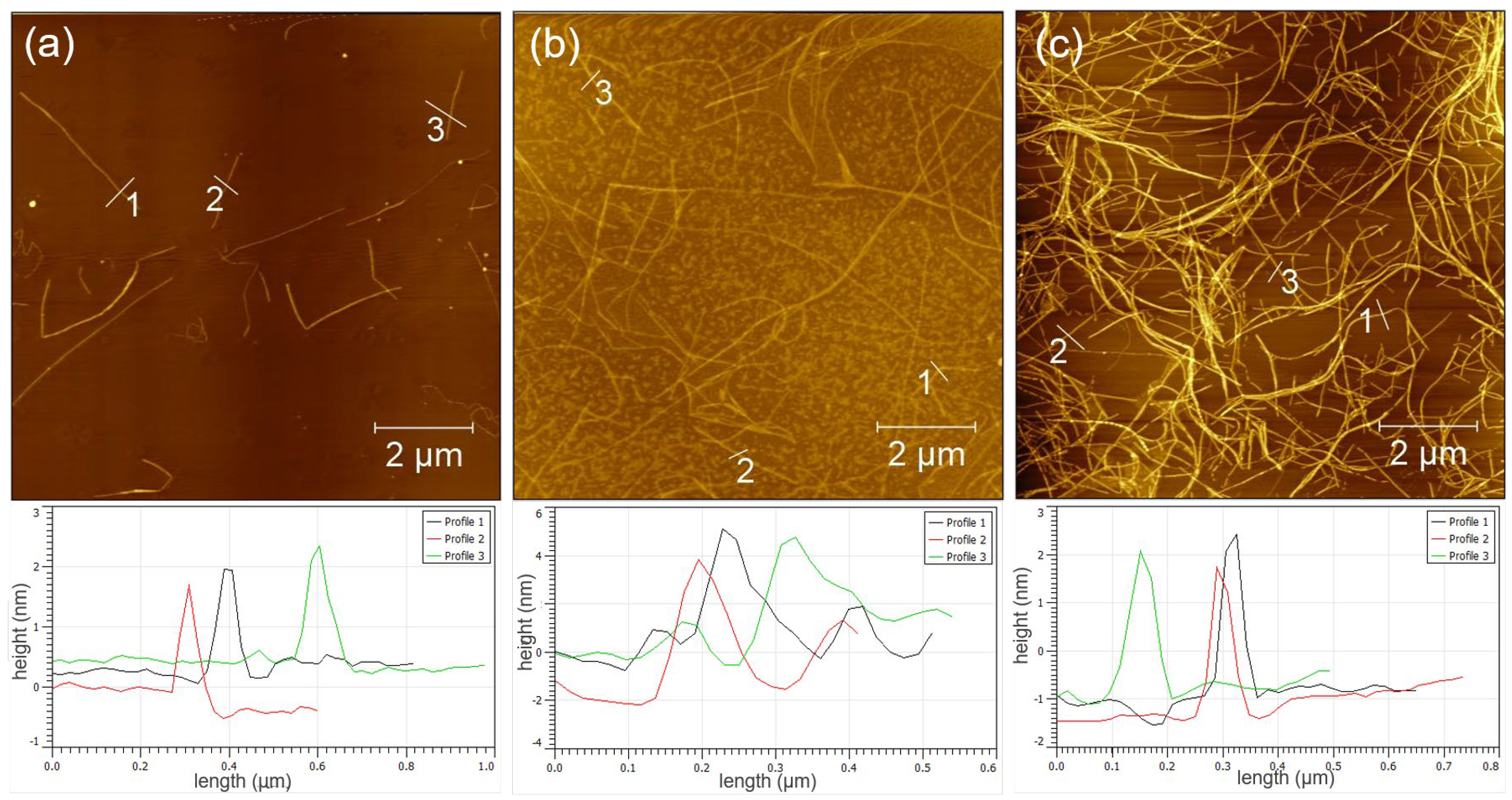
2.2. Preparation of PNFs via Self-Assembly
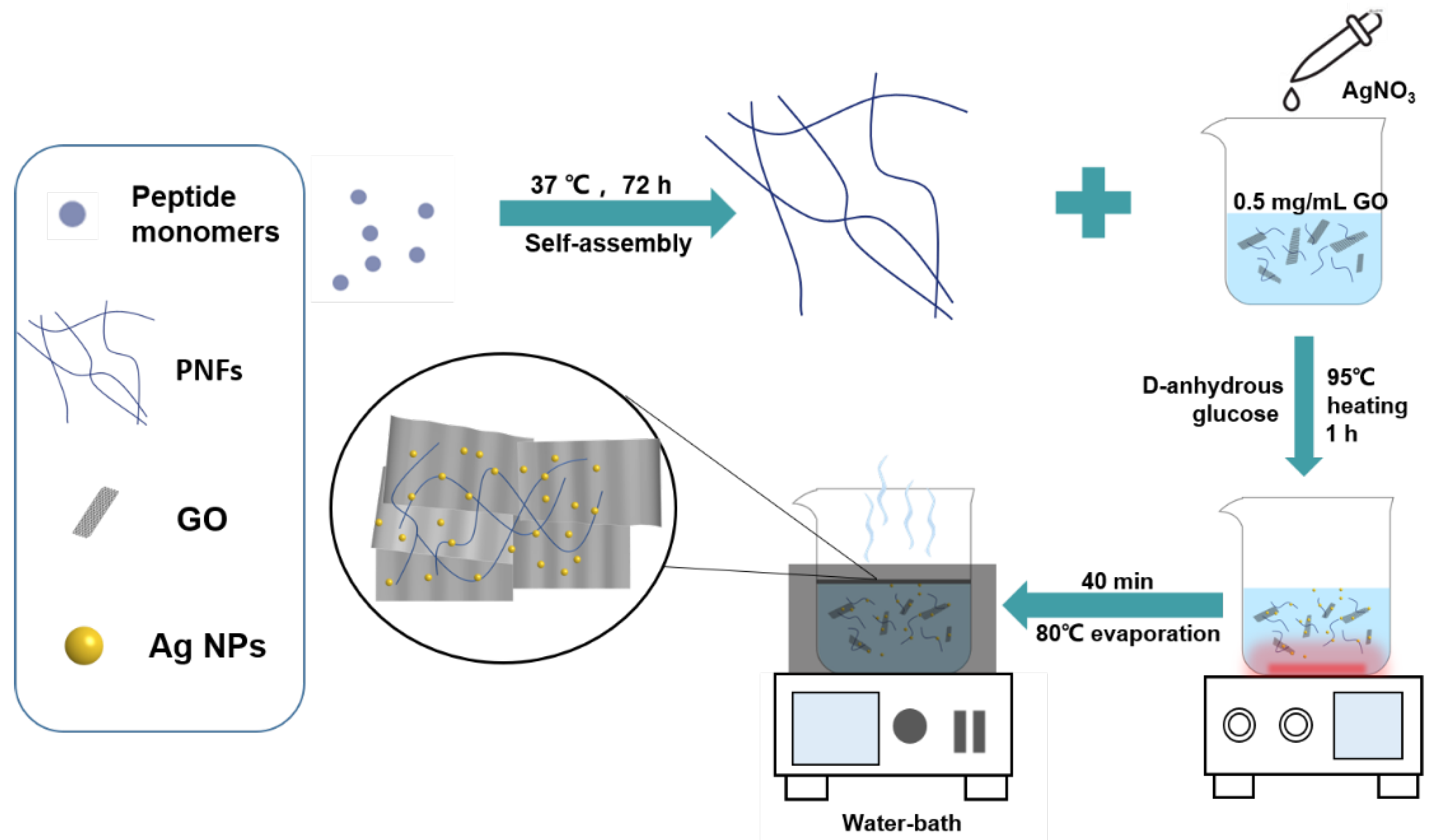
2.3. Fabrication of GO/PNF/AgNP Hybrid Membrane by Solvent Evaporation
2.4. Characterization Techniques
2.5. Antibacterial Tests
3. Results and Discussion
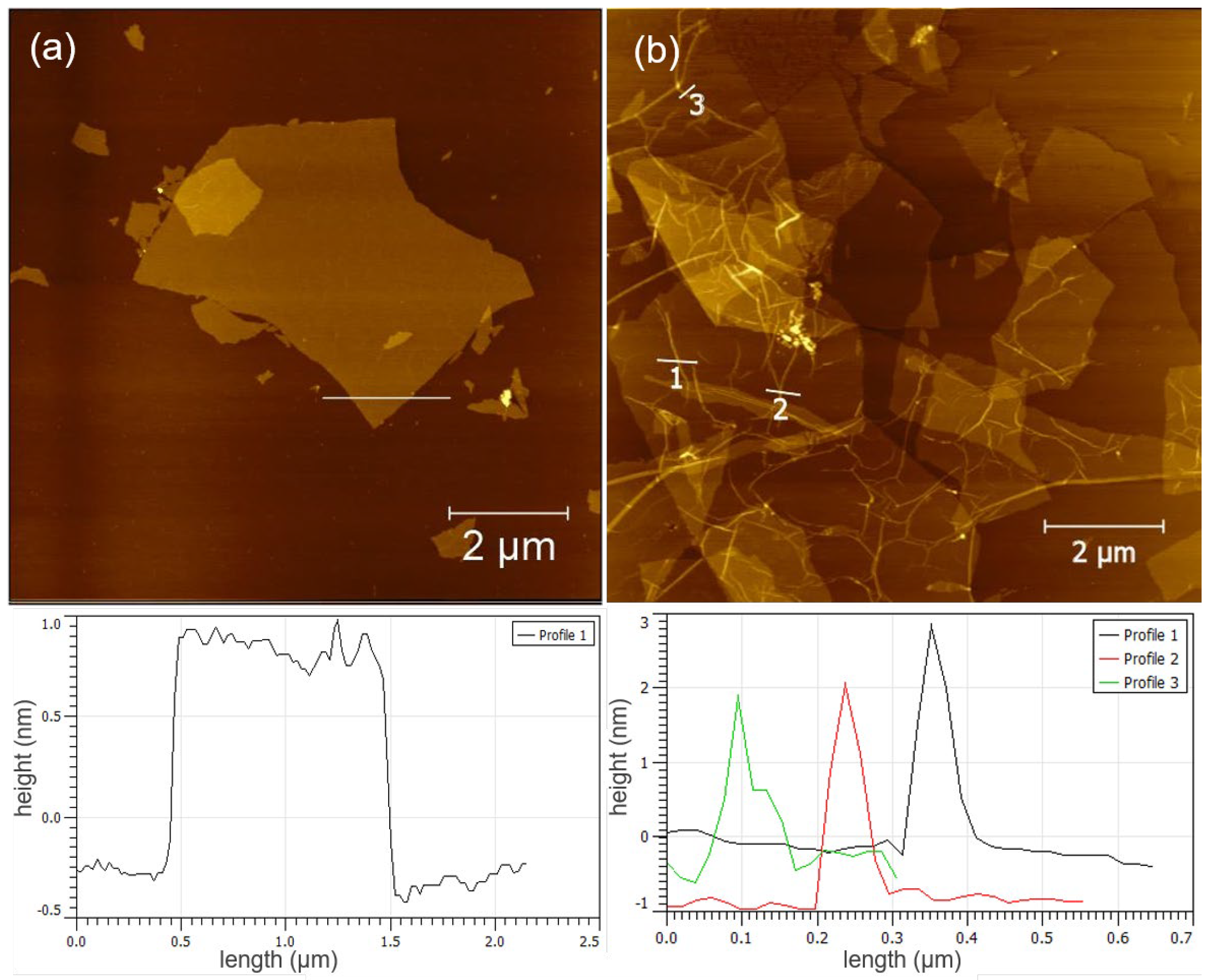
3.1. Synthesis and Characterization of GO/PNF Nanohybrids
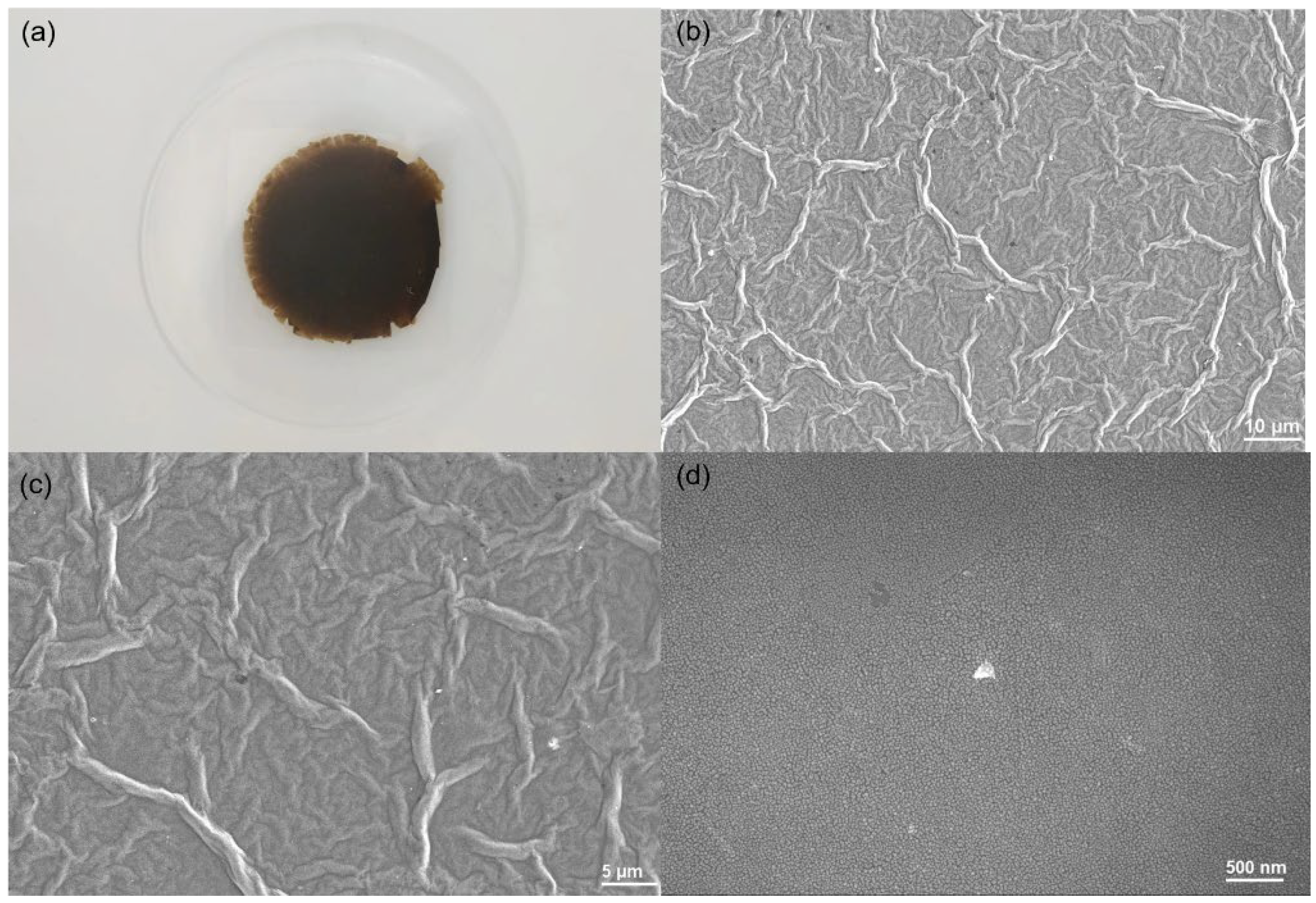
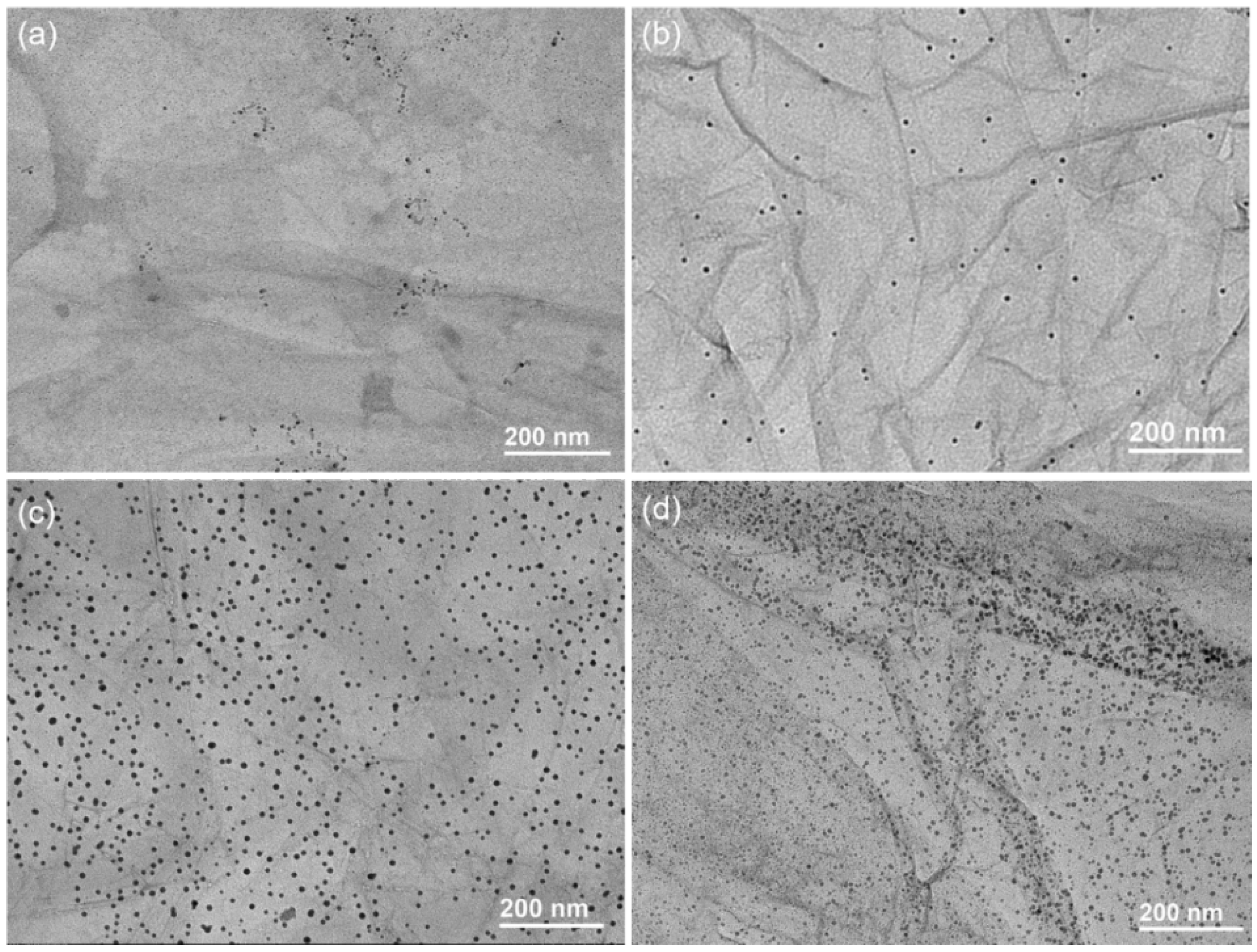
3.2. Synthesis and Characterization of GO/PNF/AgNP Hybrid Membranes
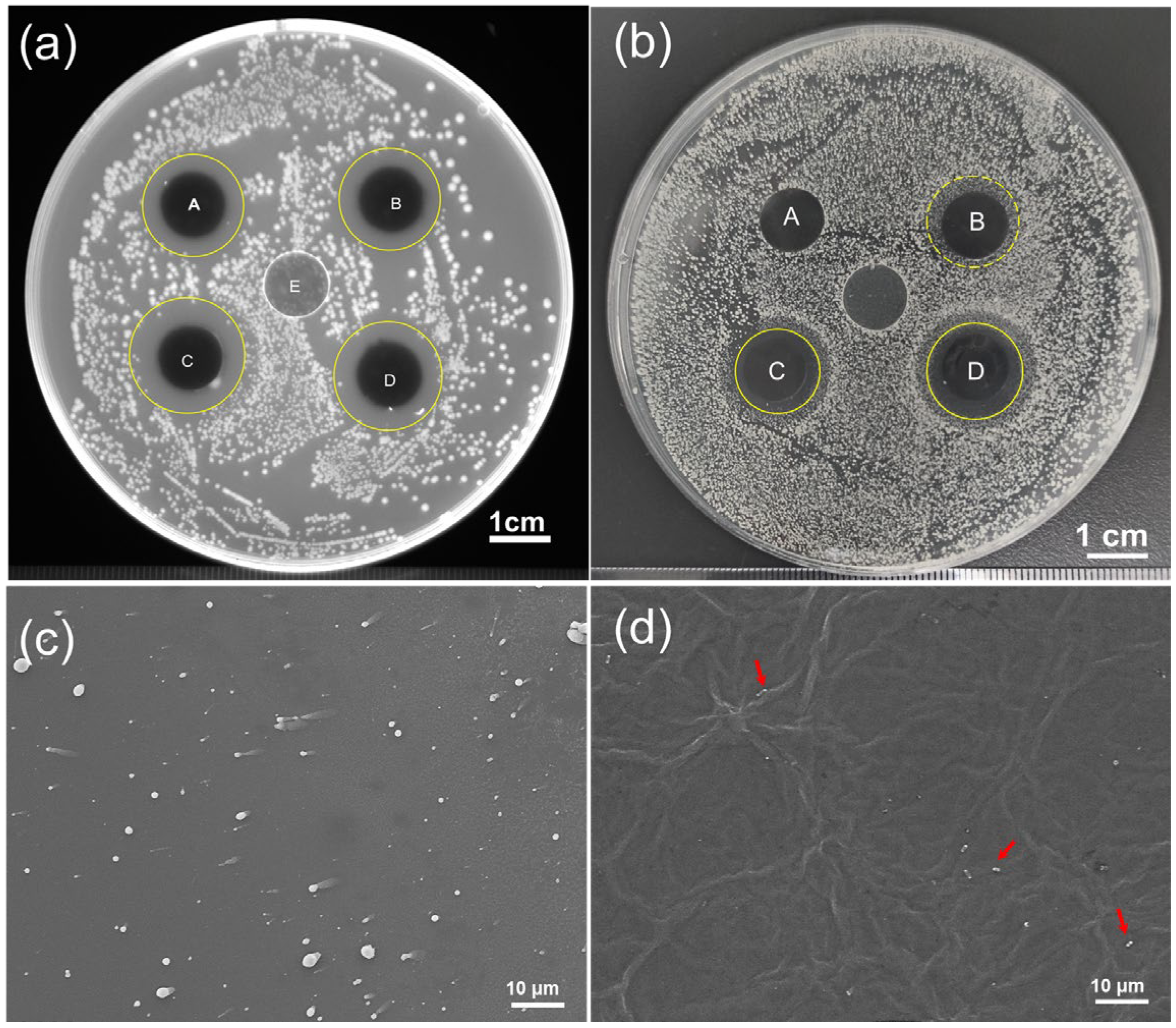
3.3. Antibacterial Performacne of the Fabricated GO/PNFs/AgNPs Hybrid Membranes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saraswathi, M.S.S.A.; Nagendran, A.; Rana, D. Tailored polymer nanocomposite membranes based on carbon, metal oxide and silicon nanomaterials: A review. J. Mater. Chem. A 2019, 7, 8723–8745. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Wei, S.; Hu, Y.H.; Sun, S.C. Membrane technology in wastewater treatment enhanced by functional nanomaterials. J. Clean. Prod. 2018, 197, 339–348. [Google Scholar] [CrossRef]
- Liu, G.P.; Jin, W.Q.; Xu, N.P. Two-Dimensional-Material Membranes: A New Family of High-Performance Separation Membranes. Angew. Chem. Int. Ed. 2016, 55, 13384–13397. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.G.; Nair, S. Membranes from nanoporous 1D and 2D materials: A review of opportunities, developments, and challenges. Chem. Eng. Sci. 2013, 104, 908–924. [Google Scholar] [CrossRef]
- Luan, X.; Kong, H.; He, P.; Yang, G.; Zhu, D.; Guo, L.; Wei, G. Self-Assembled Peptide-Based Nanodrugs: Molecular Design, Synthesis, Functionalization, and Targeted Tumor Bioimaging and Biotherapy. Small 2023, 19, 2205787. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.N.; Zhang, P.P.; Chen, Y.T.; Su, Z.Q.; Wei, G. Recent advances in the fabrication and structure-specific applications of graphene-based inorganic hybrid membranes. Nanoscale 2015, 7, 5080–5093. [Google Scholar] [CrossRef]
- Zhang, X.T.; Kong, H.; Yang, G.Z.; Zhu, D.Z.; Luan, X.; He, P.; Wei, G. Graphene-Based Functional Hybrid Membranes for Antimicrobial Applications: A Review. Appl. Sci. 2022, 12, 4834. [Google Scholar] [CrossRef]
- Gao, L.F.; Li, C.; Huang, W.C.; Mei, S.; Lin, H.; Ou, Q.; Zhang, Y.; Guo, J.; Zhang, F.; Xu, S.X.; et al. MXene/Polymer Membranes: Synthesis, Properties, and Emerging Applications. Chem. Mater. 2020, 32, 1703–1747. [Google Scholar] [CrossRef]
- Zhu, Z.G.; Wang, W.; Qi, D.P.; Luo, Y.F.; Liu, Y.R.; Xu, Y.; Cui, F.Y.; Wang, C.; Chen, X.D. Calcinable Polymer Membrane with Revivability for Efficient Oily-Water Remediation. Adv. Mater. 2018, 30, 1801870. [Google Scholar] [CrossRef]
- Zhang, K.; Li, Z.J.; Kang, W.M.; Deng, N.P.; Yan, J.; Ju, J.G.; Liu, Y.; Cheng, B.W. Preparation and characterization of tree-like cellulose nanofiber membranes via the electrospinning method. Carbohyd. Polym. 2018, 183, 62–69. [Google Scholar] [CrossRef]
- Yin, X.M.; Li, H.J.; Han, L.Y.; Meng, J.C.; Lu, J.H.; Song, Q. All Si3N4 Nanowires Membrane Based High-Performance Flexible Solid-State Asymmetric Supercapacitor. Small 2021, 17, 2008056. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.H.; Dong, K.Q.; Zhang, Y.Y.; Wang, X.; Aboalhassan, A.A.; Yu, J.Y.; Ding, B. Multifunctional flexible membranes from sponge-like porous carbon nanofibers with high conductivity. Nat. Commun. 2019, 10, 5584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, S.; Liu, X.H.; Liao, J.B.; Lin, H.; Liu, F. PEI modified multiwalled carbon nanotube as a novel additive in PAN nanofiber membrane for enhanced removal of heavy metal ions. Chem. Eng. J. 2019, 375, 122086. [Google Scholar] [CrossRef]
- Kong, H.; Chen, Y.; Yang, G.; Liu, B.; Guo, L.; Wang, Y.; Zhou, X.; Wei, G. Two-dimensional material-based functional aerogels for treating hazards in the environment: Synthesis, functional tailoring, applications, and sustainability analysis. Nanoscale Horiz. 2022, 7, 112–140. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, G.Z.; Liu, B.; Kong, H.; Xiong, Z.; Guo, L.; Wei, G. Biomineralization of ZrO2 nanoparticles on graphene oxide-supported peptide/cellulose binary nanofibrous membranes for high-performance removal of fluoride ions. Chem. Eng. J. 2022, 430, 132721. [Google Scholar] [CrossRef]
- Ding, L.; Wei, Y.Y.; Wang, Y.J.; Chen, H.B.; Caro, J.; Wang, H.H. A Two-Dimensional Lamellar Membrane: MXene Nanosheet Stacks. Angew. Chem. Int. Ed. 2017, 56, 1825–1829. [Google Scholar] [CrossRef]
- Li, T.; Liu, G.; Kong, H.; Yang, G.; Wei, G.; Zhou, X. Recent advances in photonic crystal-based sensors. Coord. Chem. Rev. 2023, 475, 214909. [Google Scholar] [CrossRef]
- Zhu, D.; Liu, B.; Wei, G. Two-Dimensional Material-Based Colorimetric Biosensors: A Review. Biosensors 2021, 11, 259. [Google Scholar] [CrossRef]
- Jia, F.C.; Xiao, X.; Nashalian, A.; Shen, S.; Yang, L.; Han, Z.Y.; Qu, H.J.; Wang, T.M.; Ye, Z.; Zhu, Z.J.; et al. Advances in graphene oxide membranes for water treatment. Nano Res. 2022, 15, 6636–6654. [Google Scholar] [CrossRef]
- He, G.W.; Huang, S.Q.; Villalobos, L.F.; Zhao, J.; Mensi, M.; Oveisi, E.; Rezaei, M.; Agrawal, K.V. High-permeance polymer-functionalized single-layer graphene membranes that surpass the postcombustion carbon capture target. Energy Environ. Sci. 2019, 12, 3305–3312. [Google Scholar] [CrossRef] [Green Version]
- Akhavan, O.; Ghaderi, E. Toxicity of Graphene and Graphene Oxide Nanowalls Against Bacteria. ACS Nano 2010, 4, 5731–5736. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, M.; Liu, D.-K.; Chiao, Y.-H.; Hung, W.-S. Synergistic effect of one-dimensional silk nanofiber and two-dimensional graphene oxide composite membrane for enhanced water purification. J. Membr. Sci. 2020, 606, 118142. [Google Scholar] [CrossRef]
- Wu, Y.D.; Xin, W.W.; Kong, X.-Y.; Chen, J.J.; Qian, Y.C.; Sun, Y.; Zhao, X.L.; Chen, W.P.; Jiang, L.; Wen, L.P. Enhanced ion transport by graphene oxide/cellulose nanofibers assembled membranes for high-performance osmotic energy harvesting. Mater. Horiz. 2020, 7, 2702–2709. [Google Scholar] [CrossRef]
- Menazea, A.A.; Ahmed, M.K. Synthesis and antibacterial activity of graphene oxide decorated by silver and copper oxide nanoparticles. J. Mol. Struct. 2020, 1218, 128536. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, W.S.; Zhang, L.; Li, J.F.; Su, Z.Q.; Wei, G. Sequence-Designed Peptide Nanofibers Bridged Conjugation of Graphene Quantum Dots with Graphene Oxide for High Performance Electrochemical Hydrogen Peroxide Biosensor. Adv. Mater. Interfaces 2017, 4, 1600895. [Google Scholar] [CrossRef]
- Yang, G.; He, P.; Zhu, D.; Wan, K.; Kong, H.; Luan, X.; Fang, L.; Wang, Y.; Wei, G. Functional regulation of polymer aerogels by graphene doping and peptide nanofiber-induced biomineralization as sustainable adsorbents of contaminants. Environ. Sci. Nano 2022, 9, 4497–4507. [Google Scholar] [CrossRef]
- Liu, G.P.; Jin, W.Q.; Xu, N.P. Graphene-based membranes. Chem. Soc. Rev. 2015, 44, 5016–5030. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.Y.; Yang, K.J.; Chen, B.L. Membranes prepared from graphene-based nanomaterials for sustainable applications: A review. Environ. Sci. Nano 2017, 4, 2267–2285. [Google Scholar] [CrossRef]
- Wei, L.; Chen, F.; Wang, H.; Zeng, T.H.; Wang, Q.; Chen, Y. Acetone-Induced Graphene Oxide Film Formation at the Water–Air Interface. Chem.-Asian J. 2013, 8, 437–443. [Google Scholar] [CrossRef]
- Ming, J.; Sun, D.; Wei, J.; Chen, X.; Zheng, N. Adhesion of Bacteria to a Graphene Oxide Film. ACS Appl. Bio Mater. 2020, 3, 704–712. [Google Scholar] [CrossRef] [Green Version]
- Tang, S.; Zheng, J. Antibacterial Activity of Silver Nanoparticles: Structural Effects. Adv. Healthc. Mater. 2018, 7, 1701503. [Google Scholar] [CrossRef] [PubMed]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramírez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Badawy, A.M.; Silva, R.G.; Morris, B.; Scheckel, K.G.; Suidan, M.T.; Tolaymat, T.M. Surface Charge-Dependent Toxicity of Silver Nanoparticles. Environ. Sci. Technol. 2011, 45, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.F.; Liu, Y.Q.; Liu, M.L.; Wang, Y.; He, W.F.; Shi, G.Q.; Hu, X.H.; Zhan, R.X.; Luo, G.X.; Xing, M.; et al. Synthesis of graphene oxide-quaternary ammonium nanocomposite with synergistic antibacterial activity to promote infected wound healing. Burn. Trauma 2018, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Jian, Z.W.; Wang, H.; Liu, M.L.; Chen, S.Y.; Wang, Z.H.; Qian, W.; Luo, G.X.; Xia, H.H. Polyurethane-modified graphene oxide composite bilayer wound dressing with long-lasting antibacterial effect. Mat. Sci. Eng. C 2020, 111, 110833. [Google Scholar] [CrossRef]
- Xiao, S.F.; Lu, X.M.; Gou, L.; Li, J.L.; Ma, Y.Q.; Liu, J.J.; Yang, K.; Yuan, B. Graphene oxide as antibacterial sensitizer: Mechanically disturbed cell membrane for enhanced poration efficiency of melittin. Carbon 2019, 149, 248–256. [Google Scholar] [CrossRef]
- Li, J.C.; Liu, X.Y.; Lu, J.Q.; Wang, Y.D.; Li, G.L.; Zhao, F.B. Anti-bacterial properties of ultrafiltration membrane modified by graphene oxide with nano-silver particles. J. Colloid Interf. Sci. 2016, 484, 107–115. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, D.; Cho, M.J.; Lee, S.S.; Zhang, F.Z.; Biswas, P.; Fortner, J.D. In Situ Photocatalytic Synthesis of Ag Nanoparticles (nAg) by Crumpled Graphene Oxide Composite Membranes for Filtration and Disinfection Applications. Environ. Sci. Technol. 2016, 50, 2514–2521. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.B.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.R.; Kong, J.; Chen, Y. Antibacterial Activity of Graphite, Graphite Oxide, Graphene Oxide, and Reduced Graphene Oxide: Membrane and Oxidative Stress. Acs Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef]
- Wang, M.; Zhu, H.; Shen, J. Synthesis and molecular dynamics simulation of CuS@GO–CS hydrogel for enhanced photothermal antibacterial effect. New J. Chem. 2021, 45, 6895–6903. [Google Scholar] [CrossRef]
- Liang, Y.; Wang, M.; Zhang, Z.; Ren, G.; Liu, Y.; Wu, S.; Shen, J. Facile synthesis of ZnO QDs@GO-CS hydrogel for synergetic antibacterial applications and enhanced wound healing. Chem. Eng. J. 2019, 378, 122043. [Google Scholar] [CrossRef]
- Pender, M.J.; Sowards, L.A.; Hartgerink, J.D.; Stone, M.O.; Naik, R.R. Peptide-mediated formation of single-wall carbon nanotube composites. Nano Lett. 2006, 6, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Salleh, A.; Naomi, R.; Utami, N.D.; Mohammad, A.W.; Mahmoudi, E.; Mustafa, N.; Fauzi, M.B. The Potential of Silver Nanoparticles for Antiviral and Antibacterial Applications: A Mechanism of Action. Nanomaterials 2020, 10, 1566. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, L.; Pompa, P.P. Nanosilver-based antibacterial drugs and devices: Mechanisms, methodological drawbacks, and guidelines. Chem. Soc. Rev. 2014, 43, 1501–1518. [Google Scholar] [CrossRef]
- Ren, G.; Wan, K.; Kong, H.; Guo, L.; Wang, Y.; Liu, X.; Wei, G. Recent advance in biomass membranes: Fabrication, functional regulation, and antimicrobial applications. Carbohydr. Polym. 2023, 305, 120537. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, P.; Yang, M.; Lei, Y.; Guo, L.; Wang, Y.; Wei, G. Solvent-Evaporation-Induced Synthesis of Graphene Oxide/Peptide Nanofiber (GO/PNF) Hybrid Membranes Doped with Silver Nanoparticles for Antibacterial Application. Polymers 2023, 15, 1321. https://doi.org/10.3390/polym15051321
He P, Yang M, Lei Y, Guo L, Wang Y, Wei G. Solvent-Evaporation-Induced Synthesis of Graphene Oxide/Peptide Nanofiber (GO/PNF) Hybrid Membranes Doped with Silver Nanoparticles for Antibacterial Application. Polymers. 2023; 15(5):1321. https://doi.org/10.3390/polym15051321
Chicago/Turabian StyleHe, Peng, Minghao Yang, Yu Lei, Lei Guo, Yan Wang, and Gang Wei. 2023. "Solvent-Evaporation-Induced Synthesis of Graphene Oxide/Peptide Nanofiber (GO/PNF) Hybrid Membranes Doped with Silver Nanoparticles for Antibacterial Application" Polymers 15, no. 5: 1321. https://doi.org/10.3390/polym15051321
APA StyleHe, P., Yang, M., Lei, Y., Guo, L., Wang, Y., & Wei, G. (2023). Solvent-Evaporation-Induced Synthesis of Graphene Oxide/Peptide Nanofiber (GO/PNF) Hybrid Membranes Doped with Silver Nanoparticles for Antibacterial Application. Polymers, 15(5), 1321. https://doi.org/10.3390/polym15051321







