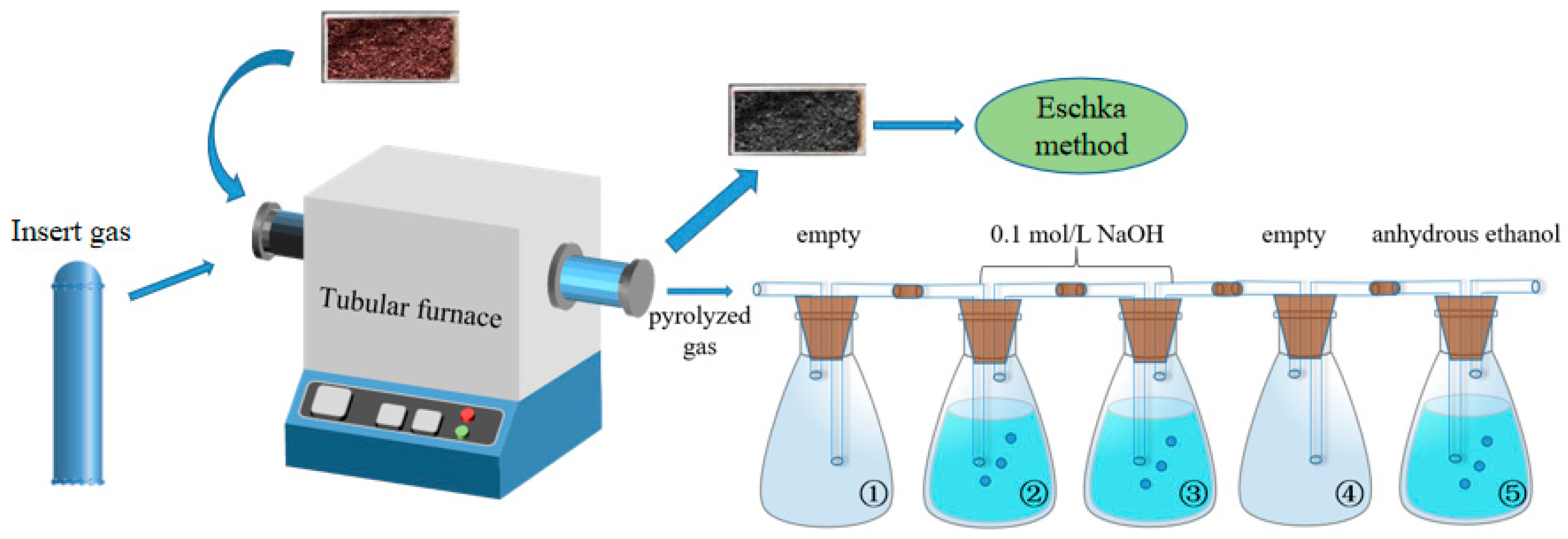
3.1. TG and DTG Results
The effect of CuO on the thermal degradation behavior of EPET was studied by TG, as shown in
Figure 3a,b. The characteristic temperatures in the TG and DTG curves are summarized in
Table 2. The pyrolysis process of 3EPET-1CuO had one stage starting from 292 °C to 482 °C. In addition, the peak thermal degradation rate was 6.2%/min and occurred at 409 °C. These characteristics are quite similar to that of EPET [
3]. EPET had a single pyrolysis stage ranging from 289 °C to 480 °C with a maximum weight loss rate occurring at 405 °C. The difference between these characteristic temperatures was only within
4 °C, which indicates that the introduction of CuO to the pyrolysis of EPET did not heavily influence the thermal degradation behavior of EPET.
PVC-3CuO decomposition during pyrolysis characteristically has four stages, as shown in
Figure 3c. During the first two stages (238–313 °C, 313–401 °C), it lost 5.5% and 3.5% (totaling 9.0%) of its weight, the third stage was 401–540 °C with a weight loss of 13.2%, and in the fourth stage (540–692 °C) it lost 29.0%. For PVC, as shown in
Figure 3d, there were two pyrolysis stages (203–371 °C, 371–536 °C), and the weight loss percentages were 65.3% and 28.9%, respectively. Mathematically, if CuO does not influence the weight loss of PVC, for the PVC-3CuO sample (weight ratio 1:3), its weight loss should be 25% of that of pure PVC. However, when pyrolysis was conducted together with CuO, the sum of the weight losses in the first two stages (238–401 °C) was only 13.8% of the weight loss in the first stage (203–371 °C) of PVC pyrolysis. However, the weight loss in the third stage (401–540 °C) was 45.7% of that in the second stage (371–536 °C) of PVC pyrolysis, which indicates the later increased thermal degradation of PVC. Meanwhile, an additional, fourth stage at a higher temperature range (540–692 °C) appeared in the thermal degradation of PVC-3CuO.
The thermal degradation of 9EPET-1PVC with and without CuO was further studied. According to
Figure 3e,f and
Table 2, 9EPET-1PVC-3CuO had three pyrolysis stages of 242–328 °C, 328–439 °C, and 439–636 °C with weight loss percentages of 4.3%, 22.5%, and 18.3%, respectively, while 9EPET-1PVC had two stages of 214–325 °C and 325–528 °C with weight loss percentages of 8.5% and 39.5%, respectively. Similar to the analysis outlined in the previous paragraph, if ignoring the effect of CuO and according to the mass ratios, the mathematically calculated weight loss of 9EPET-1PVC-3CuO should be 76.9% of that of 9EPET-1PVC. However, comparing the first pyrolysis stages of the two samples, the proportion was only 50.6%. Meanwhile, although the proportion of weight loss in stage II of EPET-PVC-CuO pyrolysis accounted for less than the mathematically calculated one, it should be noted that the upper limit of this temperature range (328–439 °C) is 89 °C lower than that (325–528 °C) of EPET-PVC. Additionally, EPET-PVC-CuO also has an additional pyrolysis stage at a higher temperature range (439–636 °C). We think this might be attributed to the delayed decomposition of polymers, especially PVC, in the presence of CuO; additionally, according to the literature [
16], in the latter pyrolysis process, carbon atoms in char react with chloride salts and generate volatile products.
There are several similar changing trends between the two above-discussed pairs of samples, 1PVC-3CuO/PVC and 9EPET-1PVC-3CuO/9EPET-1PVC, when comparing the characteristic parameters of the two samples of each pair. The presence of CuO led to much lower weight losses in the early pyrolysis stages, and the beginning temperatures of weight losses had 35 °C and 28 °C delays, respectively. These demonstrated that CuO decreases and adversely affects the emission of the volatile pyrolysis products produced. However, all of the peak DTG temperatures in the first pyrolysis stage of the 1PVC-3CuO, 9EPET-1PVC-3CuO, and 9EPET-1PVC samples, which were 281, 279, and 284 °C, were unaffected by EPET and were very comparable to that of pure PVC (288 °C). Additionally, CuO rarely influences EPET pyrolysis; additionally, as demonstrated in the literature, most of the weight loss of 9EPET-1PVC in early pyrolysis results from PVC decomposition [
28]. The first stage of PVC pyrolysis is very important for studying the emissions of chlorine-containing pollutants, as it is well known that most of the chlorine is released in the form of HCl in the first pyrolysis stage and contributes the most to weight loss [
5,
7,
16]. Since the temperature ranges of the first and second pyrolysis stages of 9EPET-1PVC-3CuO overlapped with that of the first stage of PVC pyrolysis, it can be deduced that the changing trends in these two stages are closely related to the interactions between CuO and PVC, and it is reasonable to estimate that CuO inhibits HCl emission and/or stabilizs HCl into a solid, resulting in decreased weight loss in the early co-pyrolysis stages of EPET, PVC, and CuO.
3.2. Py-GC/MS Results
In order to further study the co-pyrolysis of EPET and PVC in the presence of CuO, Py-GC/MS was performed on the 9EPET-1PVC-1CuO sample to analyze the main organic pyrolysis products. The first detection temperature was set at an integer value of 440 °C which is approximately equal to the termination temperature of the second pyrolysis stage (439 °C). This setting was chosen because, as discussed previously, in the first two pyrolysis stages, the interactions between CuO and PVC are significant, and the pyrolysis products can reflect these interactions. The second detection temperature was set at 640 °C, which is after the end of the whole pyrolysis process, to detect the final pyrolysis products.
Table 3 summarizes the main pyrolysis products with contents larger than 1wt.%. At 440 °C, the pyrolysis products mainly consisted of benzoic acid, 1,2-ethyleneglycol dibenzoate, propylene ketone, and vinyl formate, and the total content accounted for 73.85 wt.% of the products. These products were demonstrated to be mostly derived from EPET [
3,
8,
29]. Benzene was also detected in the products, though in a small amount (2.24 wt.%). Benzene is one of our main concerns since it is a main product of PVC in early pyrolysis stages and is strongly correlated with other accompanying products such as HCl, etc. In our previous study [
3] where CuO was absent, the 9EPET-1PVC pyrolysis produced 13.21wt.% of benzene at a temperature of 325 °C, and until this temperature, the weight loss of this sample was much lower than that of 9EPET-1PVC-1CuO at 439 °C (8.5% vs. 26.8%). This result illustrates that the presence of CuO decreases the yield of benzene, which is also in accordance with the literature [
16,
18,
21,
22]. Meanwhile, the reduction in benzene emission indicates the reduced formation of polyenes, as it is the decomposition of polyenes and the following aromatization of produced cyclohexene and cyclohexadiene rings that generate benzene [
7]. Several studies have also reported that CuO promotes the crosslinking of PVC and thereafter inhibits the generation of polyenes. Therefore, it can be deduced that in the co-pyrolysis of EPET, PVC, and CuO, the crosslinking of PVC is prominent.
At 640 °C, the main products turned out to be benzoic acid, 1,2-ethyleneglycol dibenzoate, benzene, ethylamine, propylene ketone, vinyl benzoate, vinyl formate, etc. Comparing the pyrolysis products at 440 °C and 640 °C, the contents of benzoic acid, benzene, biphenyl, and vinyl benzoate increased and the contents of other substances decreased. These trends might be because the molecules of pyrolysis products tend to be smaller and the structures become simpler with the increase in the pyrolysis temperature. Additionally, ethylamine appeared in the products, and we believe it derived from EPET since the elemental analysis showed that the EPET contained 0.46 wt.% nitrogen [
3]. Meanwhile, the content of benzene increased to 7.39 wt.%, which might not only be attributed to PVC pyrolysis, since at high temperatures the main product, benzoic acid, from EPET can decompose and generate benzene [
8,
11,
30]. Therefore, at 640 °C, the benzene cannot be regarded as a standard to evaluate PVC pyrolysis alone.
It is particularly noteworthy that no chlorine-containing compound was found in products with contents over 1 wt.% at 440 and 640 °C. Nevertheless, to better understand the pyrolysis mechanism, the main organic chlorinated compounds (COCs) and their contents are summarized in
Table 4. The products are characteristic of four main types, including chloroaromatics, chloroaromatic derivatives, chloroesters, and acyl chlorides. At 440 °C, the total COC content was 2.54 wt.%, while at 640 °C it increased to 3.35 wt.%, which indicates that a higher temperature may benefit the formation of COCs. A similar tendency was also observed for the 9EPET-1PVC sample, as reported in our previous publication [
3]; namely, at 325 °C, which was the end temperature of the first pyrolysis stage, there was only 0.35 wt.% of COCs, and at end the pyrolysis at 528 °C the content reached 11.6 wt.%. Comparing the two samples, it can be clearly seen that CuO dramatically reduced the emission of COCs in the co-pyrolysis of EPET and PVC. Additionally, the most abundant products changed from chloroesters (9.28 wt.%) to chlorinated aromatics and their derivatives, and the contained chloroesters were only 0.34 wt.%. As demonstrated previously, chloroesters formed from an addition reaction between the HCl and C=C bond of the vinyl esters that were generated in EPET pyrolysis [
3,
8]. Here, in 9EPET-1PVC-3CuO pyrolysis, this addition reaction was greatly hindered. Since CuO does not affect EPET pyrolysis, it can be deduced that it influences HCl or this addition reaction.
It was also found that in the chlorine-containing aromatics and their derivatives, chlorine atoms were often directly attached to benzene rings. For example, there was 0.91 wt.% chlorobenzene in the total 1.23 wt.% of the chloroaromatics. It was demonstrated that chlorobenzene is the main chlorinated hydrocarbon from PVC pyrolysis [
5,
21,
31,
32]. Additionally, the formation of chlorobenzenes was intended to rechlorinate the polyenes, which was achieved via the recombination of chlorine with double bonds, and Cu (II) was suggested to be able to promote this rechlorination [
33]. However, due to decreased polyenes in the co-pyrolysis, the amount of chlorobenzenes was very low. Additionally, another possible mechanism of chlorobenzene formation was proposed to be the Friedel–Crafts chlorination of benzene through Lewis acid, and the literature reports that FeCl
3 was the Lewis acid [
34]. Cu (II) is a weak Lewis acid, and therefore this Friedel–Crafts chlorination may have also occurred in the present pyrolysis system.
Other typical chorine-containing products with relatively high proportions are chlorobenzoic acids and their derivatives, including 2-chlorobenzoic acid, 2,5-dichlorobenzoic acid, and 2-chlorobenzoic acid-2-oxo-2-phenylethyl ester. Compared with the pyrolysis products of 9EPET-1PVC, among which there was only a very small amount of 3-chlorobenzoic acid-2-chloroethyl ester, herein, the amounts of chlorobenzoic acids and their derivatives increased and their types varied. As benzoic acid was the dominant pyrolysis product in the co-pyrolysis (see
Table 3) and was generated from EPET, it is reasonable to assume a chlorination of benzoic acid that produces chlorobenzoic acids. A study on learning the effect of CuCl
2 on the thermal degradation of PET found that there was also chorobenzoic acid in the products; however, no further explanation was given [
29]. According to the abovementioned results, we assume that the generated CuCl
2 from the reaction of CuO and HCl promotes the formation of chlorobenzoic acids and their derivatives. The reaction mechanism is discussed in
Section 3.4.
To clarify the chlorine distribution in liquid pyrolysis products,
Table 5 summarizes the fractions of chlorinated organic compounds for the 9EPET-1PVC-3CuO sample pyrolyzed at 640 °C. According to the carbon atom number, the chlorinated liquid products were separated into three categories (C5–C9, C10–C13, and >C13). It can be seen that the chloroaromatics and their derivatives were distributed in all the categories, but the light liquids (C5–C9) were the main fractions. Meanwhile, chloroesters were only found in medium liquids (C10–C13).
3.4. Proposed Reaction Mechanism of Co-Pyrolysis
Based on the abovementioned results and discussion, the variation of the main chlorine-containing pollutants from the pyrolysis of PVC, 9EPET-1PVC, and 9EPET-1PVC-3CuO is summarized and shown in
Scheme 1. The emission of HCl dramatically decreased in the co-pyrolysis of EPET, PVC, and CuO. Its variation mechanisms are as follows. (i) In PVC pyrolysis, a large amount of HCl was produced, and HCl also acted as a catalyst for the further dehydrochlorination of PVC, as shown on the left of
Scheme 1; (ii) in 9EPET-1PVC pyrolysis, the HCl formed from PVC was mainly consumed by an addition reaction with vinyl esters formed from EPET, as shown in the middle of
Scheme 1; and (iii) in 9EPET-1PVC-3CuO pyrolysis, besides the addition reaction described in (ii), HCl could also be fixed by CuO in the solid phase by forming CuCl
2.
Besides this research, there exist several studies that have investigated the effects of Cu (II) on PVC pyrolysis; therefore, there exist plenty of foundations upon which we can systemically summarize the reaction mechanisms. Herein, according to the literature and the results of this research, possible reactions between Cu (II) and PVC as well as chlorinated organic formation reactions closely related to Cu (II) in the co-pyrolysis process are proposed, as shown in
Scheme 2 and
Scheme 3. Reactions (1), (2), and (4) in
Scheme 2 show the crosslinking of PVC itself and its product, polyene, respectively, in the presence of Cu (II) [
7,
16,
38]. Additionally, Reactions (1) and (4) are reductive coupling mechanisms that benefit HCl fixation, while Reaction (2) is the Lewis acid mechanism and produces HCl. However, the produced HCl in Reactions (2) and (3) can be absorbed by CuO, as indicated in Reaction (5). These mechanisms are not only supported by the fact that HCl emissions are reduced, but the reduced benzenes in the pyrolysis products also indicate an enhanced crosslinking of PVC and polyene, as the crosslinking was demonstrated to greatly inhibit the generation of benzenes [
16,
21,
22,
33,
38].
For chlorinated organics, the addition of CuO causes a dramatic decrease in chloroesters in the product, and this is attributed to decreased HCl formation that leads to a weakened addition of HCl to vinyl esters, as indicated in the middle of
Scheme 1. Moreover, the main product type turns out to be chlorinated aromatics and their derivatives, for example, chlorobenzene and chlorobenzoic acid. The possible reactions that generate these products are listed in
Scheme 3. Reactions (6) and (7) show a mechanism of Friedel–Crafts chlorination of benzene and benzoic acid through the Lewis acid CuCl
2, respectively [
34,
39,
40]. Reactions (8–10) represent the recombination of chlorine with double bonds of polyene followed by cyclization reactions (10), resulting in chlorobenzene [
5,
34,
41]. Particularly, it was reported that Lewis acids such as CuCl
2 accelerate the recombination and are beneficial to chlorobenzene generation (Reactions 9–10) [
34,
41]. It is noteworthy that although the representative reactions shown in
Scheme 3 are feasible, they are not dominant secondary reactions during the pyrolysis process because the reaction products (e.g., chlorinated aromatics) only account for a small amount of the pyrolysis products (
Section 3.2). It also should be mentioned that although
Scheme 1,
Scheme 2 and
Scheme 3 comprehensively list the main and most likely reaction mechanisms that affect the generation of chlorine-containing pollutants, and the reaction mechanisms were carefully deduced from the results of this research and those in the literature, parts of them still require further study and proof.