1. Introduction
The formation of ice on pavement diminishes the surface’s traction [
1,
2], significantly compromising pavement safety and transportation efficiency [
3,
4]. Addressing methods to reduce winter pavement icing has become a focal point of interest among numerous scholars [
5,
6]. Currently, pavement snow removal methods primarily involve manual snow removal [
7], mechanical snow removal [
8,
9], and the application of deicing salt [
10]. However, these methods all, to varying degrees, consume traffic resources and suffer from delayed deicing effects [
11], thereby failing to address icy road surfaces promptly [
12,
13]. In this context, proactive snow melting and deicing technology for pavement has emerged as a crucial research subject.
Presently, proactive snow melting and deicing technologies primarily include self-stress elastic pavement technology [
14,
15,
16], self-melting ice asphalt pavement [
17], phase change material pavement [
18,
19,
20], and energy conversion pavement [
21,
22,
23]. Compared to the other three methods, self-melting ice asphalt pavement technology involves substituting deicing agents for aggregates or mineral powders during asphalt mixtures, which are mixed and laid down on the road surface. Under the influence of capillary force, osmotic pressure, and wheel “pumping,” solutes effectively diffuse from the confined spaces with higher internal concentrations in the mixture to the lower salt concentration surface of the road. As a result, this process disrupts the bond between ice and the road surface, lowering the freezing point of ice and snow on the road surface [
24,
25], thus restoring the pavement’s anti-skid functionality. Therefore, self-melting ice asphalt mixtures have sparked significant interest among researchers. This study focuses on the long-term salt release characteristics of self-melting ice asphalt mixtures and their impact on pavement performance.
To investigate the long-term salt release patterns of self-melting ice asphalt pavements, Zhong et al. [
26] utilized a conductivity meter to measure the salt leaching amount of high-elasticity storage asphalt mixtures at various temperatures, studying the influence of temperature on the salt release patterns. Zheng et al. [
27] analyzed the variation in salt release rate with temperature via the natural soaking method, obtaining the dissolution patterns of salt at different temperatures. Li et al. [
28] researchers utilized the solution conductivity analysis method based on the principle of similarity to study the precipitation variation in low-freezing-point fillers in low-freezing-point asphalt mixtures, proposing a long-term prediction model. Yang Xiong [
29] obtained the critical bonding force and critical concentration by analyzing the relationship between bond strength and effective components. Yu Yang [
30] analyzed the deicing and snow-melting performance of low-freezing-point pavements by continuously measuring the release of chloride ions from soaked specimens. Wu et al. [
31], using a Hamburg wheel-track device, simulated the effects of factors such as rainfall and load on salt dissolution. They established a salt precipitation prediction model with variables including anti-icing filler dosage, void ratio, rainfall intensity, temperature, and time. The addition of anti-freezing materials can have negative effects on the low-temperature cracking resistance and water stability of asphalt mixtures. Additionally, salt release can weaken the pavement performance of self-melting ice asphalt mixtures. Han et al. [
32] utilized Mafilon (MFL) as a substitute for mineral powder to prepare self-melting ice asphalt mixtures. They found that pavement performance decreased with increasing MFL content. Using high-elasticity and high-viscosity asphalt binders [
33,
34,
35] and fibers [
36] can mitigate the negative impact of deicing agents on the pavement performance of asphalt mixtures. Zhang et al. [
37] discovered via freeze–thaw tests that after 14 freeze–thaw cycles, the water stability, high-temperature stability, and low-temperature cracking resistance of self-melting ice asphalt mixtures decreased by 14.19%, 18.79%, and 11.96%, respectively. Zhang et al. [
38] pointed out that selecting granular anti-freezing agents can alleviate the adverse effects of salt release on pavement performance. The impact of anti-freezing materials on the pavement performance of asphalt mixtures is mainly related to factors such as the dosage [
39,
40,
41], type [
42,
43], application method [
44], and gradation type [
45] of deicing agents.
Currently, existing research on self-melting ice asphalt mixtures has several gaps:
There is a lack of test methods to determine salt release patterns quickly.
There is a lack of multifactor analysis, with the analysis of factors influencing salt release being relatively limited in scope.
There is insufficient research on the pavement performance of self-melting ice asphalt mixtures after complete salt release.
Therefore, this study designs indoor experiments to accelerate rapid salt release, analyzing the long-term salt release characteristics of self-melting ice asphalt mixtures. It investigates the effects of different salt release methods, deicing agent dosages, asphalt types, and mixture gradation types on salt release. Furthermore, it analyzes the pavement performance of asphalt mixtures after complete salt release. The research findings hold important guiding significance for long-term evaluation methods of snow melting and deicing, as well as sustainable research on self-melting ice asphalt pavements.
3. Results and Discussions
3.1. Salt Release Test Method Analysis
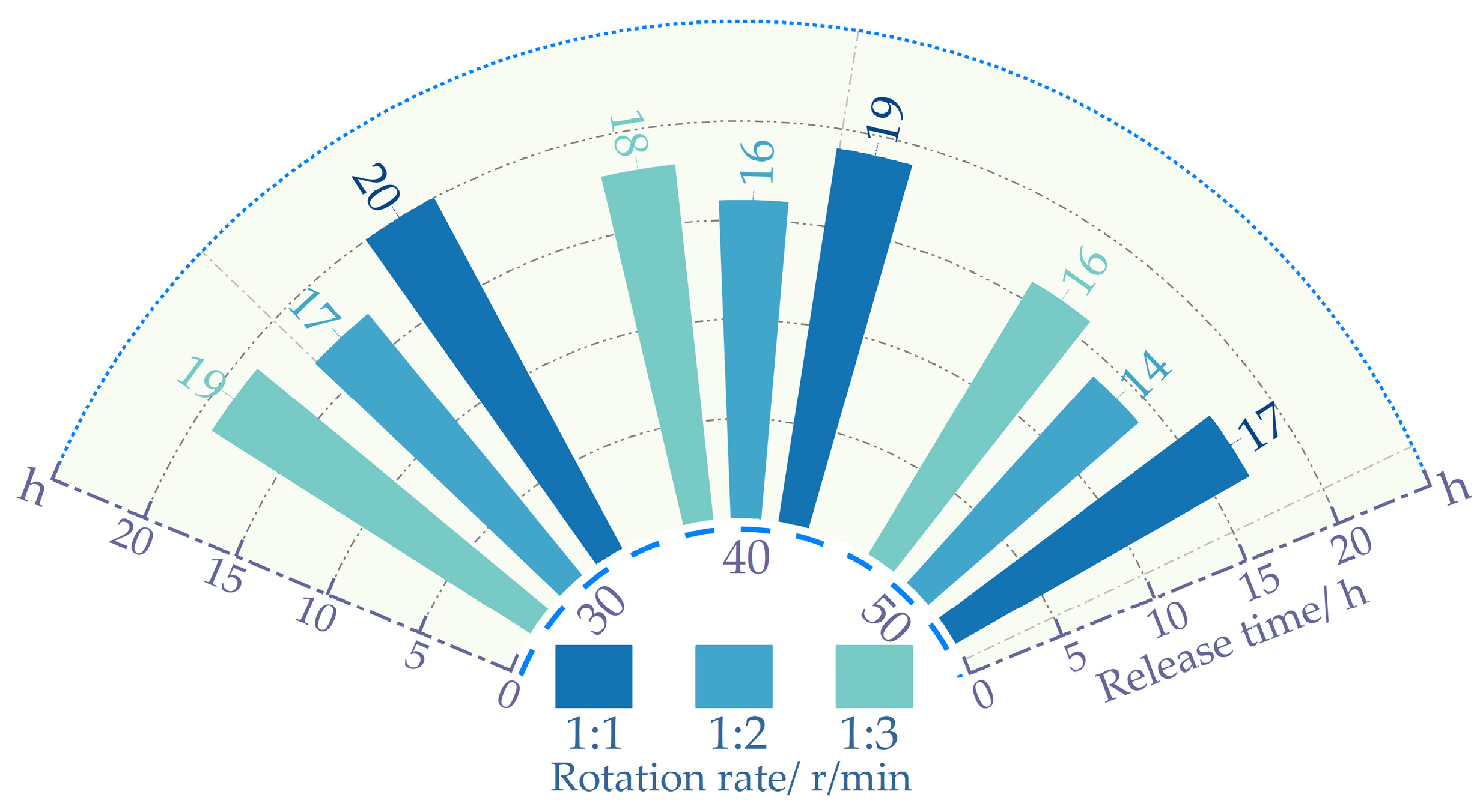
Figure 5 shows that at the same proportion, the salt release time decreases as the rotation speed increases. With the amplification of the rotational velocity, the frequency of water impacting the mixture intensifies. Accelerating the release of salt content within the asphalt. This results in a reduction in the release time for salt content in self-melting ice asphalt mixtures. Excessive rotational speed may lead to premature detachment of asphalt from the aggregate surface. The asphalt, once detached, sinks into the water, with salt release occurring at an extremely sluggish pace. Resulting in diminished uniformity in salt release. From
Figure 6, it is evident that when the rotation speed is 40 r/min, the process of mixing, separation, and re-mixing of the mixture with water is fully achieved, resulting in the best uniformity. At the same rotational speed, Excessive water content can also lead to premature detachment of asphalt from the surface of the mixture. Insufficient water content impedes the complete realization of the mixing–separation–mixing process between the mixture and water. In summary, a series of experiments have determined that the optimal process parameters for both the Los Angeles abrasion tester and the magnetic stirrer involve a rotational speed of 40 revolutions per minute (rpm). The water-to-mixture mass ratio for all three experimental methods is 1:2.
3.2. Analysis of Regulatory Patterns in Salt Release Affected by Various Factors
Utilizing the methods of the Los Angeles abrasion tester, magnetic stirrer, and natural soaking, this study investigates the impact of varying deicing agent concentrations, asphalt types, and asphalt mixture structures on salt release.
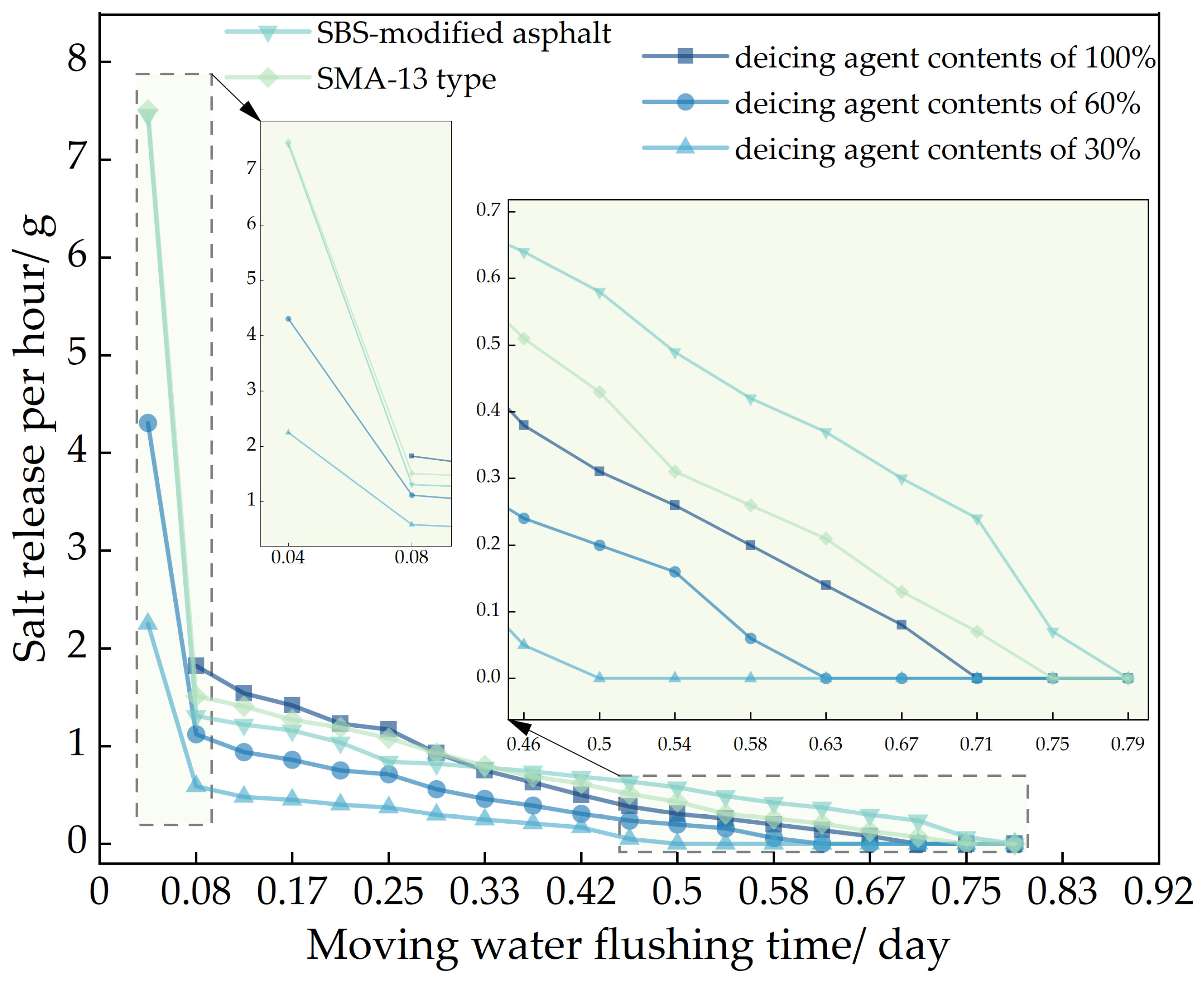
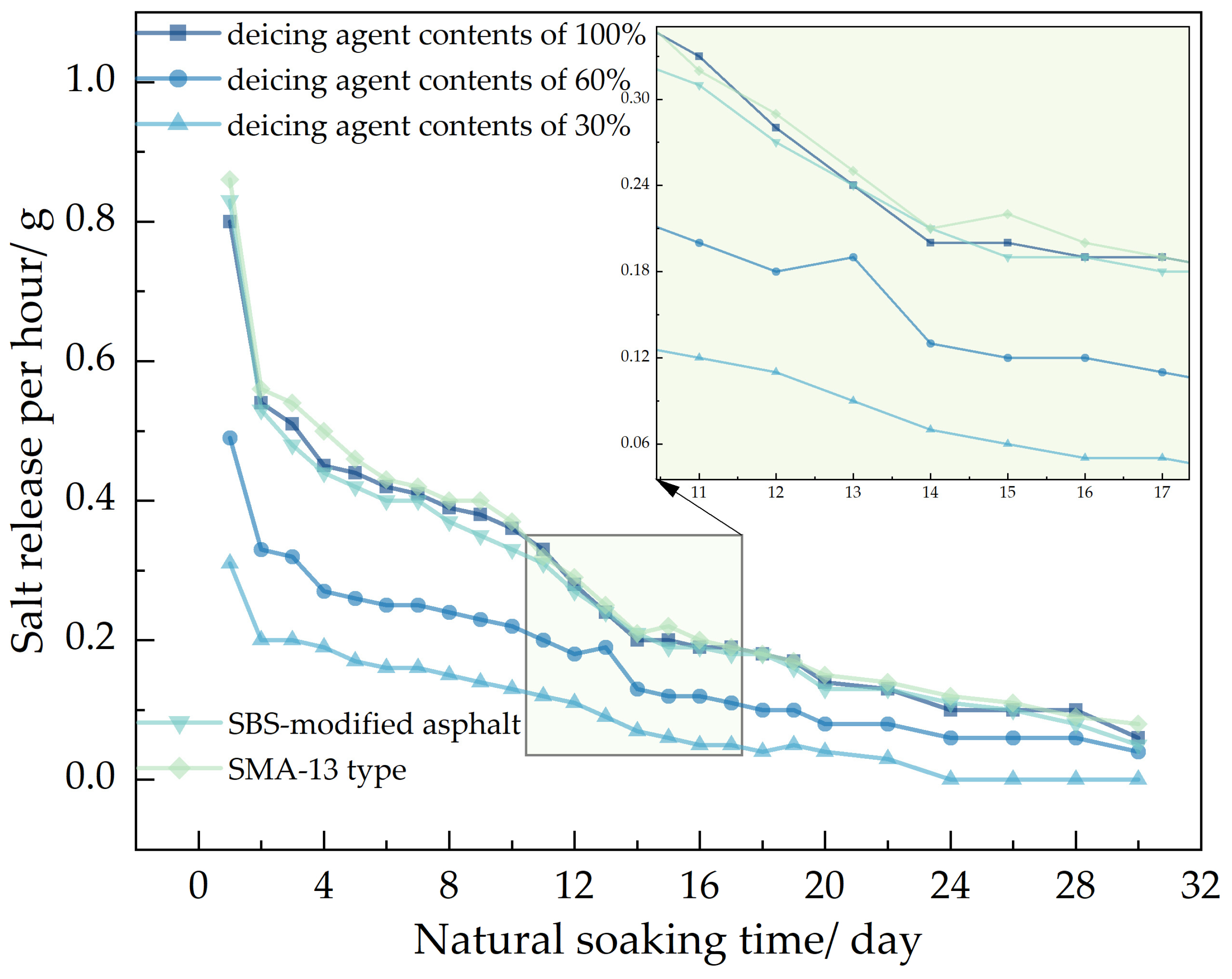
In the figure, showed that 70# base asphalt, AC-13 type asphalt mixture, with deicing agent contents of 100%, 60%, and 30%; SBS-modified asphalt, AC-13 type asphalt mixture, with a deicing agent content of 100% and 70# base asphalt, SMA-13 type asphalt mixture, with a deicing agent content of 100%.
It can be seen from
Figure 7,
Figure 8 and
Figure 9, using the Los Angeles abrasion tester to accelerate the salt release of self-melting ice asphalt mixtures, the salt release rate increased by 91 times compared to the magnetic stirrer immersion flushing method and by 114 times compared to the natural soaking method. Analyze the reasons for this phenomenon, due to the high rotational speed of the Los Angeles abrasion tester driving the water flow, resulting in a strong flushing force exerted by the water flow on the self-melting ice asphalt mixture. Salt is continuously released from the surface or interior of the asphalt emulsion.
In the analysis of salt release trends across the three testing methods, it was observed that self-melting ice asphalt mixtures using SBS-modified asphalt exhibited a longer salt release duration compared to those using 70# base asphalt. However, SBS-modified asphalt mixtures showed poorer uniformity in salt release. Analyze the reasons for this phenomenon. Deicing agents primarily exist within the asphalt emulsion, while SBS-modified asphalt is a three-dimensional structure formed by adding an SBS modifier to the base asphalt and then subjecting it to shear and mixing. Some deicing agents are placed within a mesh-like structure, constrained by the mesh-like structure, the release process of salt is more challenging, salt cannot be fully released, and the release time is prolonged, resulting in poor uniformity of salt release.
Regarding different deicing agent contents in self-melting ice asphalt mixtures, the salt release time was directly proportional to the deicing agent content, with minimal impact on the uniformity of salt release. When comparing asphalt types and deicing agent contents, it was found that, under similar conditions, SMA-13 asphalt mixtures exhibited poorer uniformity in salt release compared to AC-13 asphalt mixtures. Analyze the reasons for this phenomenon. SMA-type asphalt mixtures belong to a skeletal-dense structure, characterized by a high proportion of coarse aggregates, mineral filler, and asphalt, with fewer fine aggregates; compared to AC-type asphalt mixtures, SMA-type asphalt mixtures have a large flushing area, the excessive area causes the mixture to be initially affected by moisture, resulting in an excessive release of salt, resulting in poor uniformity of release.
3.3. Analysis of the Impact of Salt Seepage on Pavement Performance
3.3.1. Analysis of High-Temperature Stability
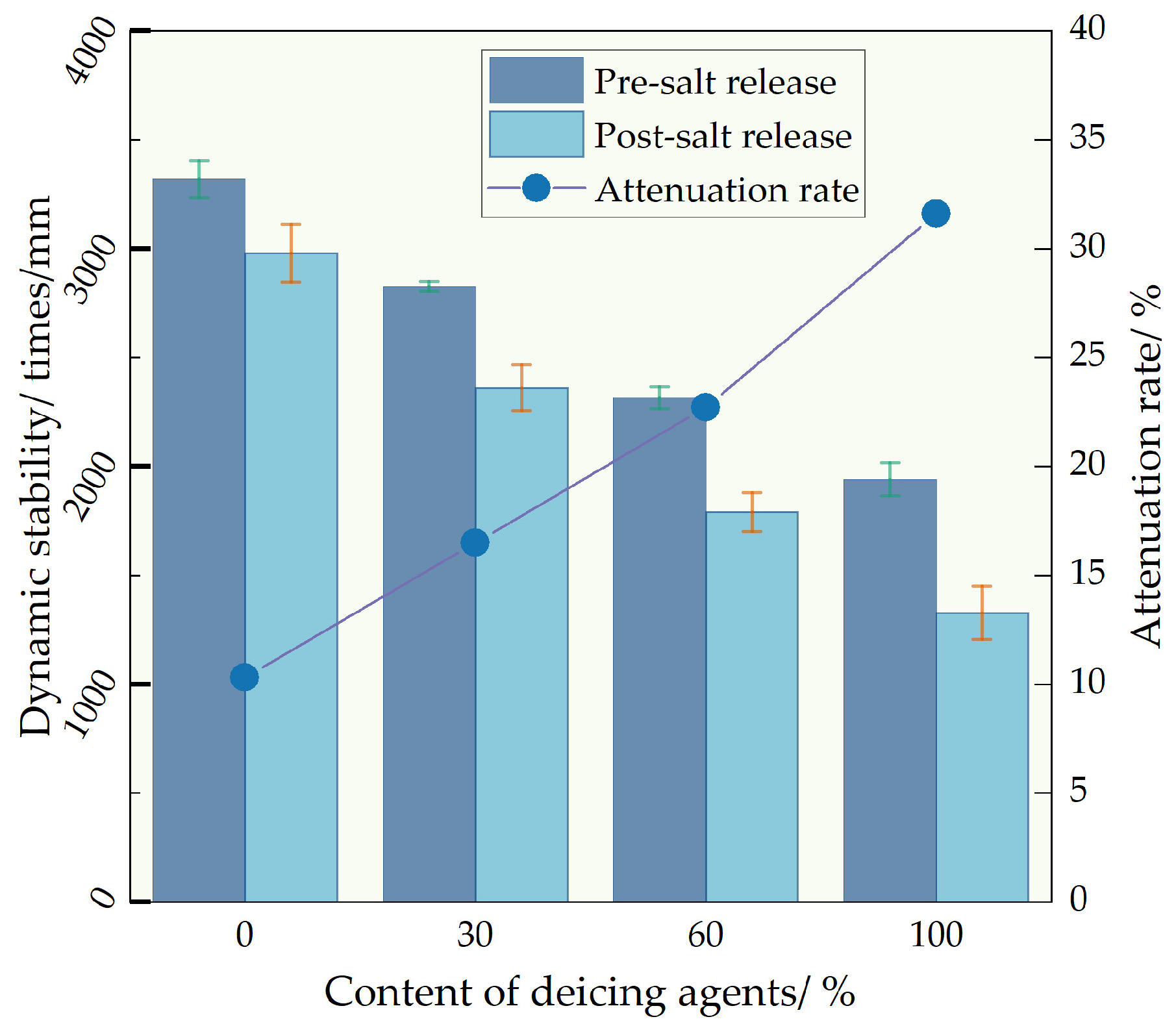
The impact of deicing agent concentrations on the high-temperature performance variation in self-melting ice asphalt mixture after complete salt release was investigated via trajectory testing. It can be seen from
Figure 10. With an increase in deicing agent concentration, the high-temperature performance of the self-melting ice asphalt mixture experiences a decline. Before salt release, the dynamic stability of deicing agent concentrations D30, D60, and D100 decreased by 14.9%, 30.2%, and 41.5%, respectively, compared to the D0 concentration. This decline can be attributed to the less effective interaction between the deicing agent and asphalt compared to the interaction between alkaline limestone filler and asphalt. Consequently, the adhesion between asphalt mortar and aggregate is compromised, leading to a reduction in the dynamic stability of the mixture.
Salt release adversely affects the high-temperature performance of self-melting ice asphalt mixture, and an increase in the blending ratio exacerbates the degradation of high-temperature performance. After salt release, the self-dynamic stability of D0, D30, D60, and D100 concentrations decreases by 10.3%, 16.5%, 22.7%, and 31.6%, respectively. The emergence of porous silica pores after salt release increases the specimen’s porosity. Simultaneously, salt precipitation alters the properties of the asphalt binder, resulting in a decline in adhesion between asphalt and aggregate, thereby reducing the high-temperature resistance to rutting of the asphalt pavement.
3.3.2. Analysis of Low-Temperature Stability
The variations in the low-temperature performance of self-melting ice asphalt mixtures before and after salt release were investigated via beam flexural testing and can be seen in
Figure 11. The addition of deicing agents adversely affected the low-temperature performance of self-melting ice asphalt mixtures, with a more significant decrease observed as the anti-freezing agent dosage increased. Prior to salt release, the maximum bending strain of D30, D60, and D100, relative to D0, decreased by 7.1%, 21.1%, and 26.9%, respectively. This decline is primarily attributed to the weakening of the interaction between aggregates and asphalt due to the addition of deicing agents, reducing low-temperature performance. Additionally, deicing agents are primarily distributed in the asphalt, and their dispersal characteristics may reduce the continuity of asphalt on the fracture surface, diminishing its toughness and consequently affecting the low-temperature performance of the mixture. After complete salt release, the maximum bending strain of self-melting ice asphalt mixtures is somewhat reduced, with a significantly greater decrease than conventional asphalt mixtures. Following salt release, the maximum bending strain of D0, D30, D60, and D100 concentrations decreases by 5.2%, 9.3%, 11.2%, and 15.4%, respectively. This reduction may be attributed to the release of deicing agents after immersion, causing partial damage to the internal structure of the asphalt mixture. Furthermore, the porosity increases with the release of chloride ions in the mixture, further disrupting the adhesive relationship between asphalt and aggregate and weakening crack resistance performance.
3.3.3. Analysis of Water Stability
Based on the variation in the freeze–thaw splitting strength of the self-melting ice asphalt mixture with the release of salt, an analysis was conducted on the influence of deicing agent dosage and salt precipitation on the water stability performance of the asphalt mixture. The experimental results are depicted in
Figure 12. It is evident that with the increase in deicing agent dosage, the splitting strength significantly decreases, indicating an adverse impact of the deicing agent on the water stability of the mixture. Before salt release, the freeze–thaw splitting strength ratios of D30, D60, and D100 to D0 decreased by 4.9%, 6.4%, and 8.4%, respectively. This is attributed to the deicing agent being unfavorable for moisture resistance, as its increased dosage leads to a reduction in structural asphalt within the asphalt, consequently compromising the integrity of the asphalt mixture and causing a decline in moisture resistance performance.
Furthermore, after the deicing agent dissolves in water, the micro-pores of the asphalt mixture enlarge, making it easier for water to penetrate the interior of the asphalt mixture. This could be one of the reasons for the decrease in the residual stability ratio. With the complete release of salt, the freeze–thaw splitting strength of all specimens significantly decreases, with a more pronounced decrease in the freeze–thaw splitting strength of asphalt mixture specimens with an added deicing agent. After salt release, the maximum bending strain of D0, D30, D60, and D100 concentrations decreases by 8%, 15.1%, 21.6%, and 26.7%, respectively. A plausible explanation is that the deicing agent continues to release underwater immersion conditions, and chloride salt solution erodes the interface between asphalt and aggregate. Simultaneously, the permeation of chloride ions leads to the dissolution of the light components of asphalt, accelerating the aging of asphalt binder and further reducing the adhesive properties between asphalt and aggregate. This results in asphalt being more prone to detach from the aggregate surface, leading to a deterioration in the water stability performance of the mixture.
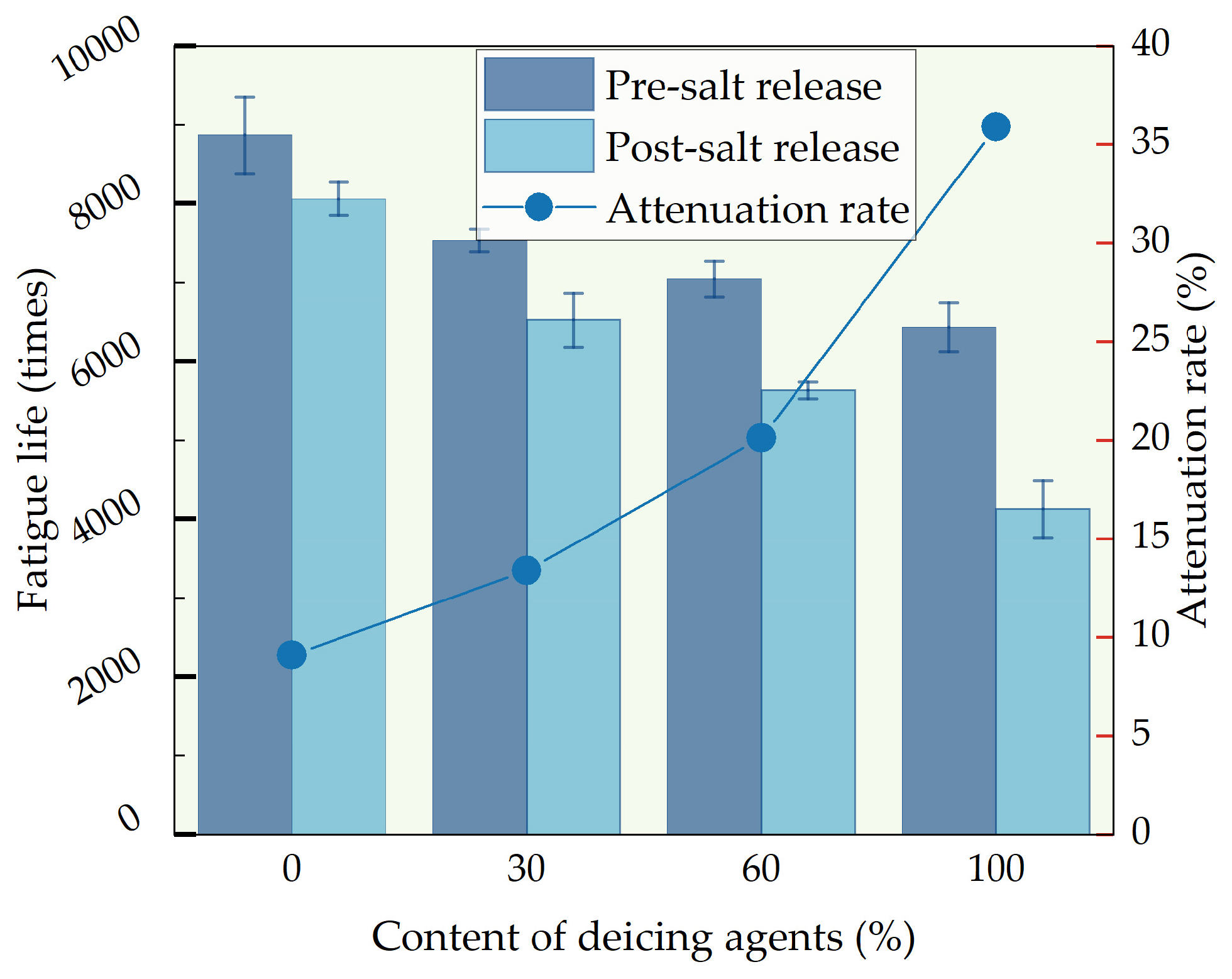
3.3.4. Analysis of Four-Point Bending Fatigue Life Test
Using a four-point bending fatigue testing machine to determine the fatigue life of compacted asphalt mixtures under repeated bending loads. From
Figure 13, it is evident that prior to salt release, the fatigue life of D30, D60, and D100 decreased by 15.1%, 20.5%, and 20.75%, respectively, compared to D0. The addition of de-icing agents is demonstrated to diminish the fatigue resistance of asphalt mixtures. This is due to the infiltration of chloride ions, leading to a decrease in the content of lightweight components in asphalt, resulting in the hardening and brittleness of the asphalt mixture. Therefore, under repeated loading, fatigue failure is more likely to occur, resulting in a lower fatigue life being manifested.
After complete salt release, the fatigue life of D0, D30, D60, and D100, respectively, decreased by 9.1%, 13.4%, 20.1%, and 35.9%. This phenomenon arises from the ease with which cations such as sodium and magnesium in the salt solution interact with the polar components in asphalt, forming organic metal salts with high solubility. The organic metal salts erode the adhesive interface between asphalt and aggregates, significantly weakening the adhesive strength formed between the aggregates and asphalt. This results in the asphalt on the surface of the mixture detaching from the aggregate surface, further reducing the fatigue life of the self-melting ice asphalt mixture.
3.4. Analysis of the Influence of Salt Precipitation Extract on Steel
The investigation focuses on the corrosion of standard steel hangers caused by leachates with varying salt concentrations. Based on the results of the corrosion test depicted in
Figure 14. The saline solution used for precipitation accelerates the corrosion of the standard steel hangers. Under equivalent immersion durations, when the concentration of the deicing agent is at 100%, the corrosion of standard steel hangers is most severe. By the eighth day, the corrosion rate can reach 0.06%. Through the analysis of the reasons, As the concentration of salt precipitates increases, the rate of electrochemical reaction of standard steel hangers is accelerated. As a result, the corrosion of standard steel hangers has deepened. The experimental design considers the most adverse conditions. The actual concentration of salt in the runoff from self-melting ice asphalt pavement is significantly lower than the concentration of precipitated solution in the experiment. Thus, the corrosion rate of steel caused by the salt analysis runoff from self-melting ice asphalt pavement should be less than 0.06%. Hence, in actual environmental conditions, the impact of self-melting ice asphalt mixture on the corrosion rate of steel is minor.