1. Introduction
Polylactic acid (PLA) and polybutylene succinate (PBS) are versatile biopolymers with many environmental benefits [
1,
2]. PLA has outstanding mechanical stiffness and strength [
3] and also breaks down under industrial conditions (58 °C) [
4]. It is therefore suitable for biomedical, agricultural, textile, and food packaging applications [
1,
2]. Conversely, PBS breaks down at lower temperatures (28 °C) [
4], making it a sustainable alternative to polyethylene terephthalate and polypropylene (which have similar properties), and potentially suitable for home composting [
5]. Whereas conventional plastics derived from fossil resources persist in the environment [
6], PLA and PBS are derived from biobased raw materials, so biodegradation is a sustainable end-of-life scenario, particularly if disposal occurs close to the site of use, thus reducing the impact of logistics [
7,
8]. The demand for environmentally friendly products and packaging solutions has driven the development and applications of PLA and PBS [
9]. However, the market share of both polymers is influenced by the cost of raw materials [
10]. The PLA market share was 18.9% in 2021, and the PBS market share is expected to reach 16% by 2026 [
11]. One interesting application is the melt binding of fibers in teabags, which would reduce the 9000 tons of persistent waste generated by the disposal of 30 billion used teabags per year in Germany [
4].
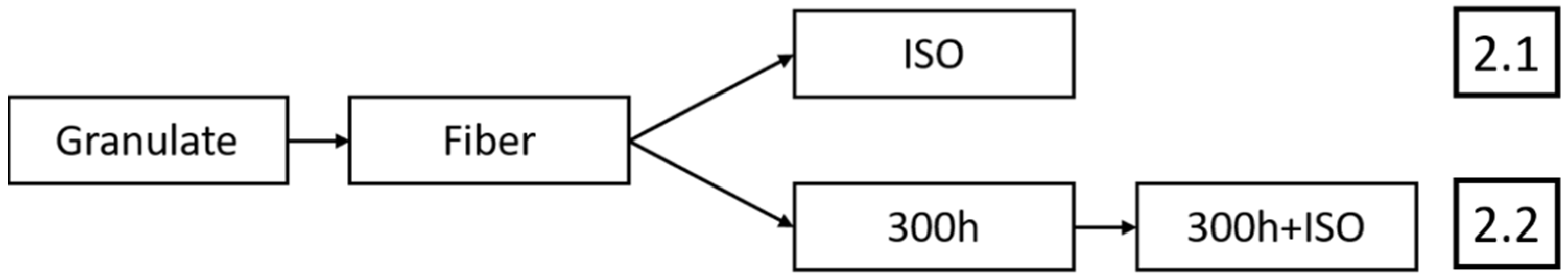
To support the transition to biobased and biodegradable plastics, it is necessary to assess the environmental performance of polymers throughout their life cycle (
Figure 1). This is often achieved by life cycle assessment (LCA), but such studies require the availability of high-quality data [
12,
13]. This is difficult to achieve for new and emerging technologies because, even if data are available, the technological readiness level (TRL) tends to be too low for comparison to established technologies [
14]. There is also a lack of primary data modeling end-of-life scenarios, such as home composting [
15]. It is therefore necessary to build reliable datasets that identify the real environmental benefits of biopolymers such as PLA and PBS.
Like their petrochemical counterparts, biopolymers processed by melt spinning experience thermomechanical stress that causes their degradation, the severity of which depends on the temperature, shear stress, and dwell time [
16,
17,
18]. The temperature can be maintained when scaling up, but shear stress can only by modeled. The dwell time of the polymer in the extruder can be measured precisely at different process scales, therefore offering an excellent parameter for comparison.
PLA has been subjected to accelerated aging in extrusion and injection molding studies [
19], and different molecular weights of PLA have been tested by hydrolysis [
20]. PLA fibers have also been compared to cellulose fibers, such as hemp, jute and sisal, for their degradation in farmland soil for up to 11 days under controlled conditions [
21]. Poly(butylene succinate-co-butylene adipate) samples melt blended with TiO
2 have been tested by UV degradation [
22], and PBS prepared by injection molding has been compared to low-density polyethylene by accelerated weathering [
23]. PBS mixed with Mg–Al was prepared by melt compounding and tested in a marine degradation setting [
24]. PLA blended with various polymers has been tested by accelerated aging [
25], and PBS in a composite with jute has been tested in a biodegradation setting [
26]. Blends of PLA and PBS have also been investigated in disintegration studies with inorganic fillers such as talc and chalk [
27]. Poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) in medical devices has been tested for hydrolysis [
28]. Polymers are also prone to degrade during processing [
16,
29]. Even broader studies of biodegradable man-made fibers look only at burial trials in natural soil or compost for PLA, polyhydroxyalkanoate-PLA blends, or bi-component fibers prepared from PBS and PLA [
30].
The studies above examined individual polymers and their degradability in detail. Multiple grades, polymer blends, or different polymers are often evaluated using a single degradation mechanism. The possibility of pre-exposure causing a second degradation mechanism is disregarded in studies focusing on one degradation mechanism alone. In addition, the influence of different production scales (and therefore differences in dwell time) has been overlooked. The relationship between dwell time and degradation in two consecutively tested degradation mechanisms can be clearly defined as a knowledge gap. We therefore investigated the influence of dwell time during the processing, accelerated weathering, and disintegration of PLA and PBS. Three process scales were identified with different dwell times: laboratory, pilot, and industrial scales. We also examined the influence of accelerated weathering on disintegration to demonstrate the realistic degradation of biopolymer products that end up in the environment due to accidental or deliberate improper disposal. We focused on the relationship between the laboratory and industrial fibers in virgin and pre-exposed settings, thus contributing useful data for future LCA studies.
5. Conclusions
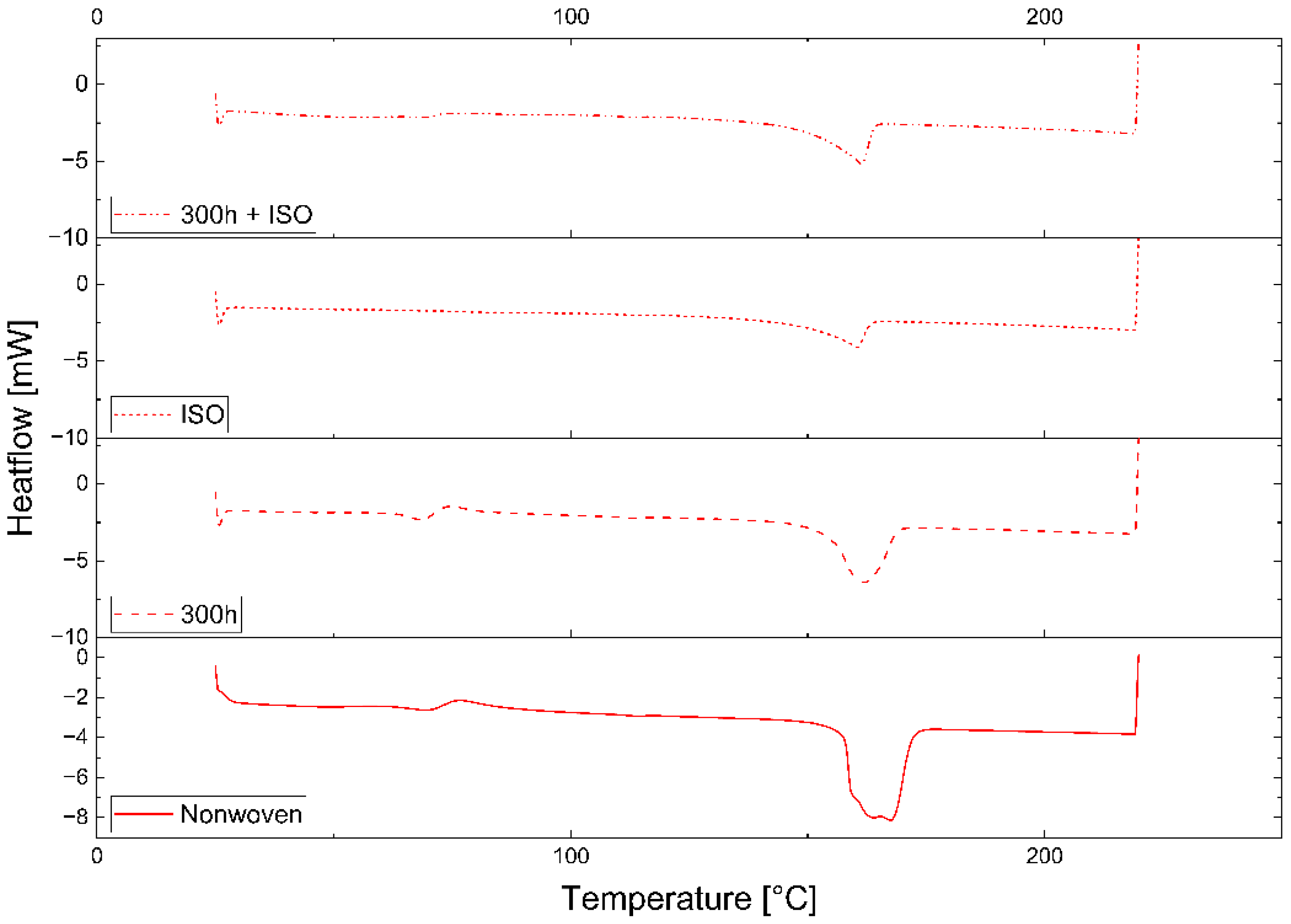
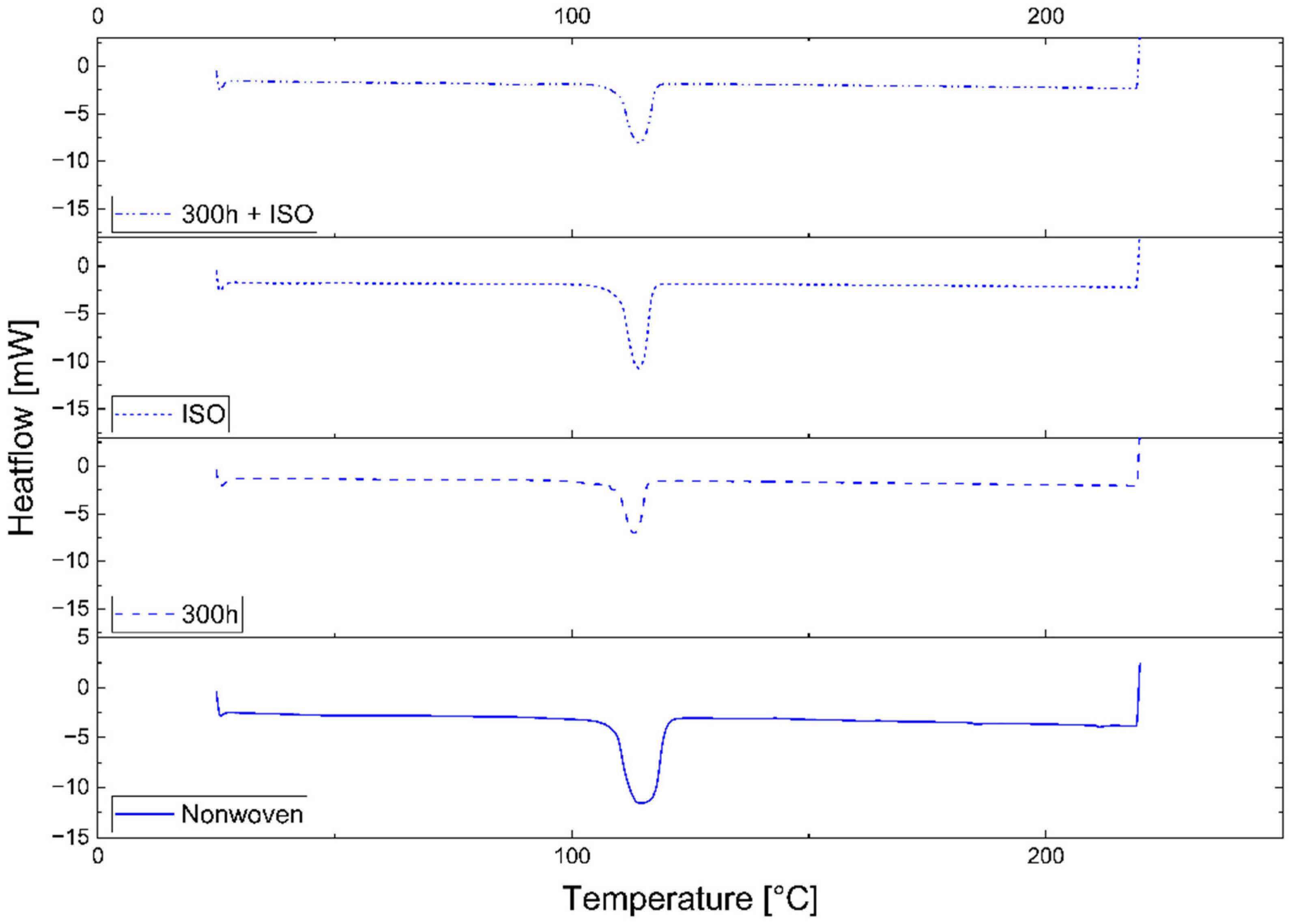
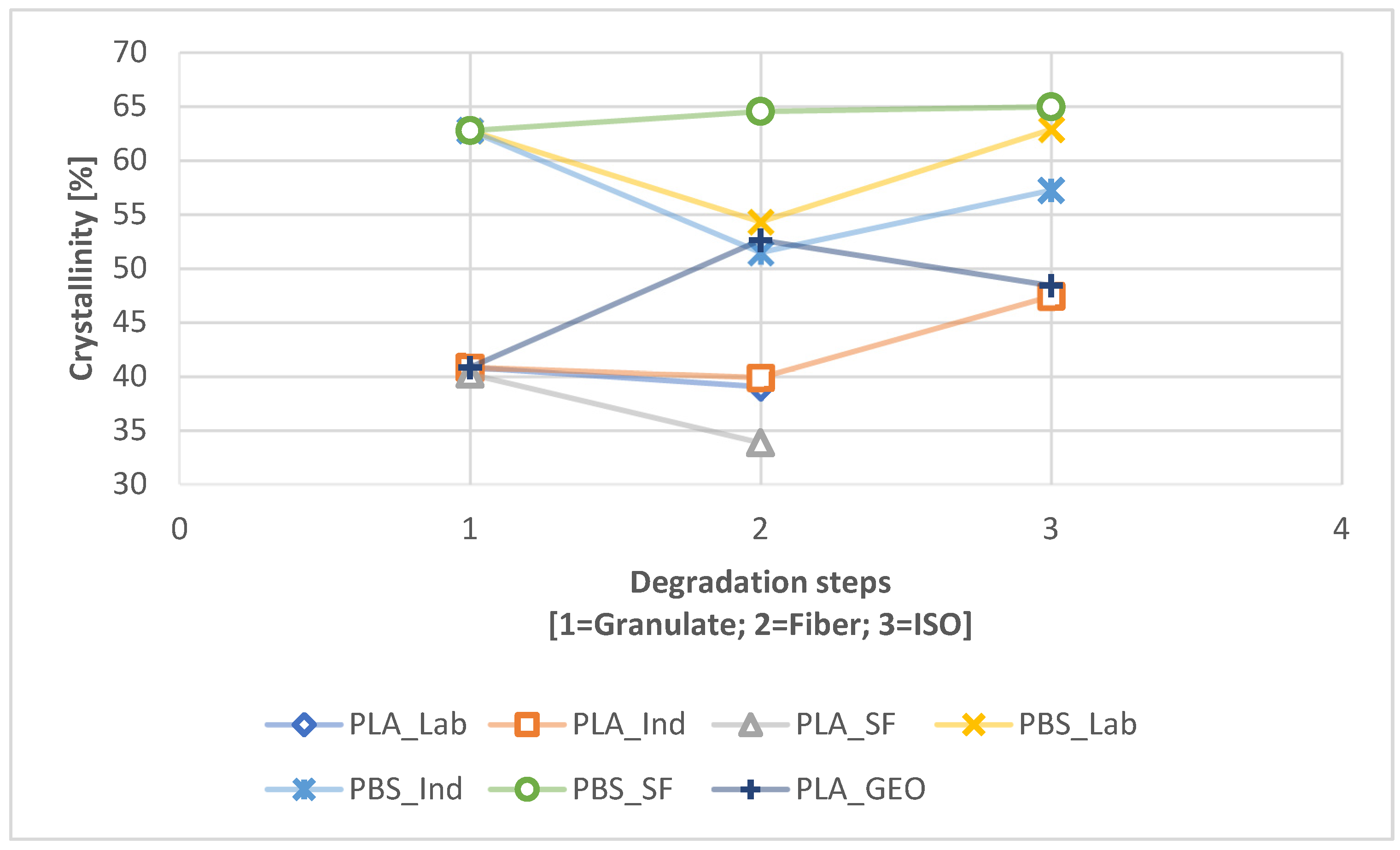
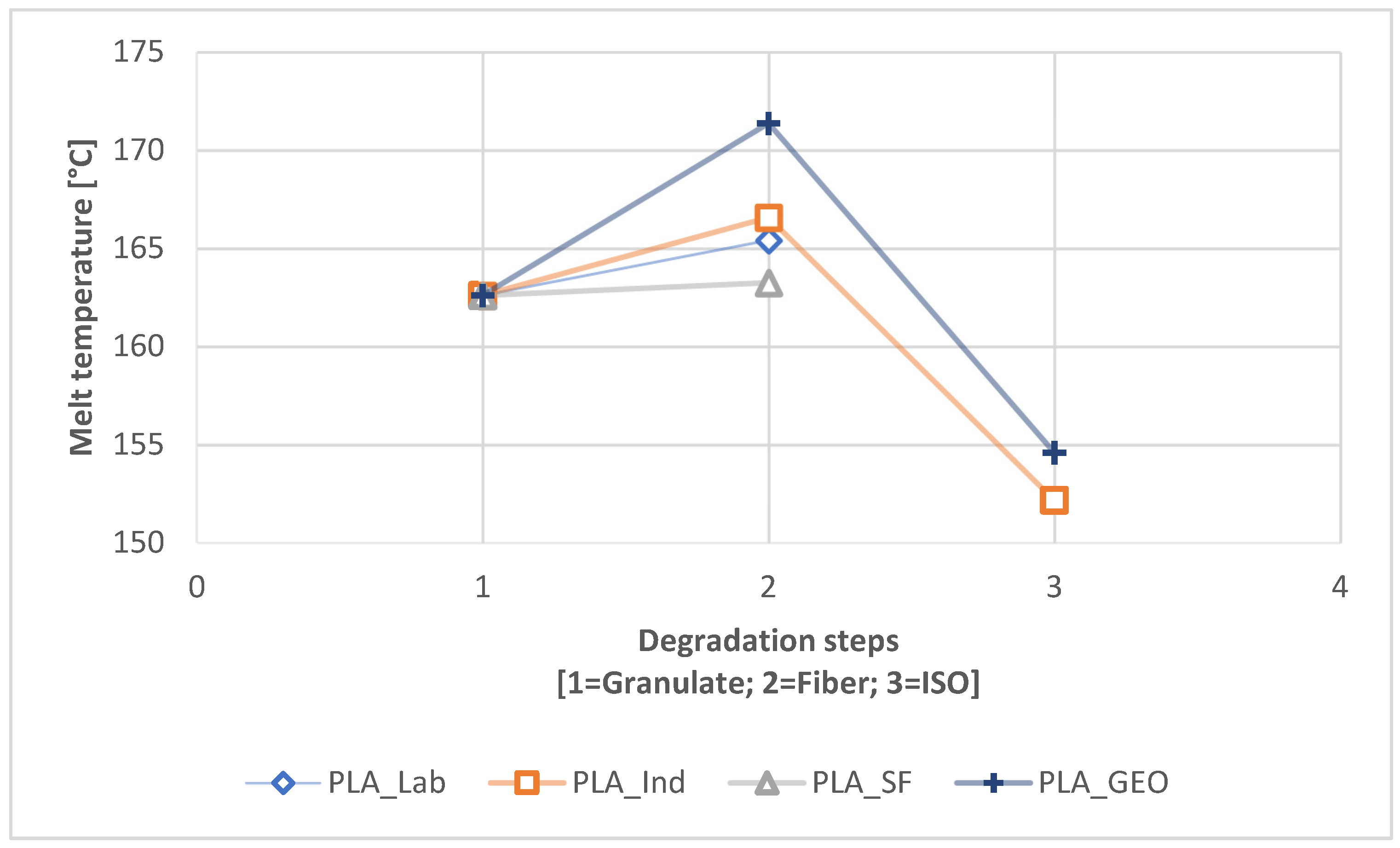
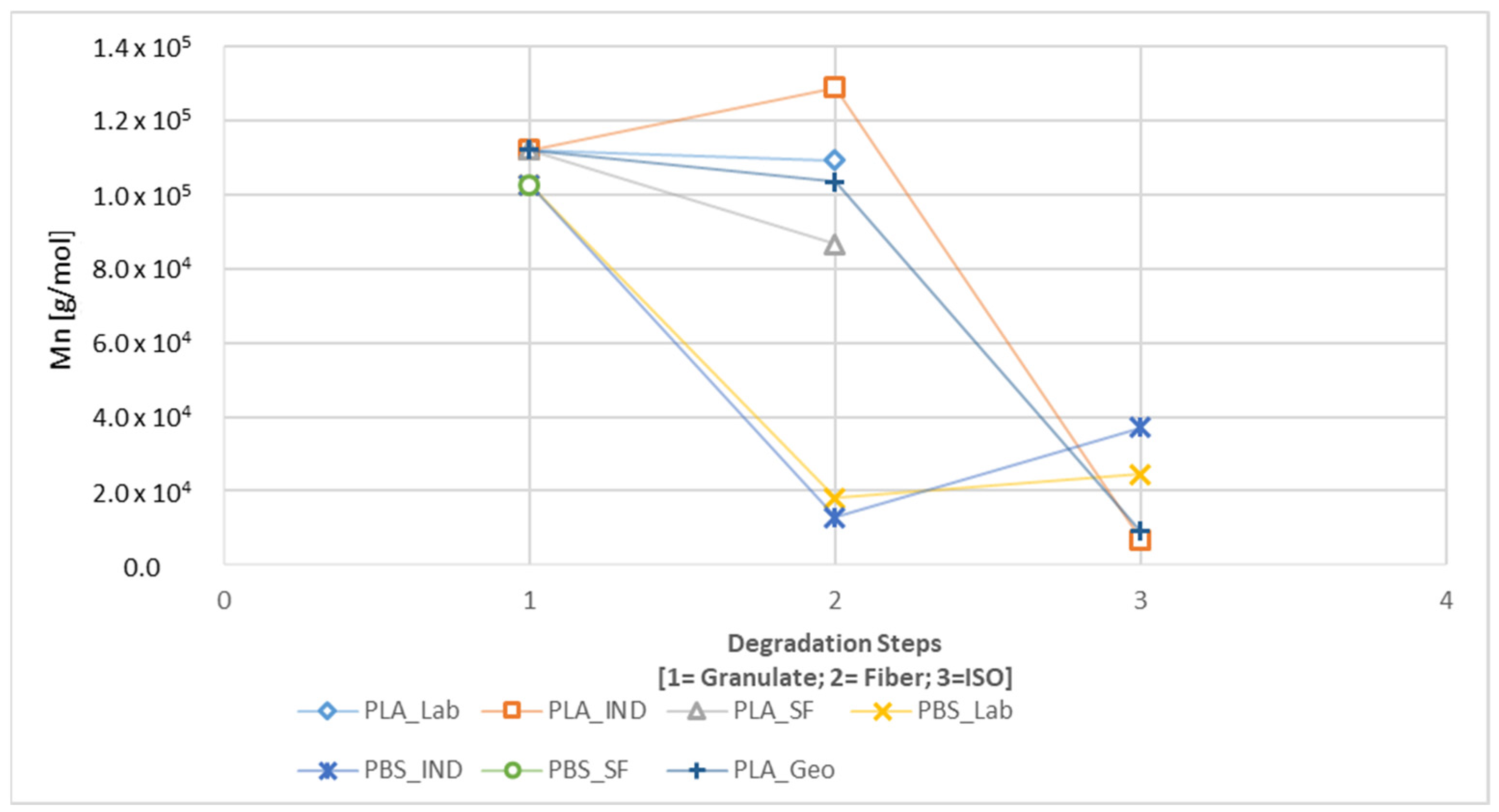
A connection between longer dwell times and a change in mechanical properties was established for PLA but was only indicated for PBS because the PBS sample with the longest dwell time was insoluble and could not be characterized by GPC. The connection between a longer dwell time and lower T
m was established for PLA. A longer dwell time was also connected to a lower M
n for all samples, in agreement with previous reports [
29,
43]. The samples increased in crystallinity, and in the case of PBS_SF (with the longest dwell time), this was almost linear from the granular sample along the entire degradation path. All other (PBS and PLA) samples decreased in crystallinity after spinning. Increasing crystallinity is linked to the degradation of amorphous structures first. For PLA, the samples with the longest dwell times showed the lowest crystallinity and M
n values.
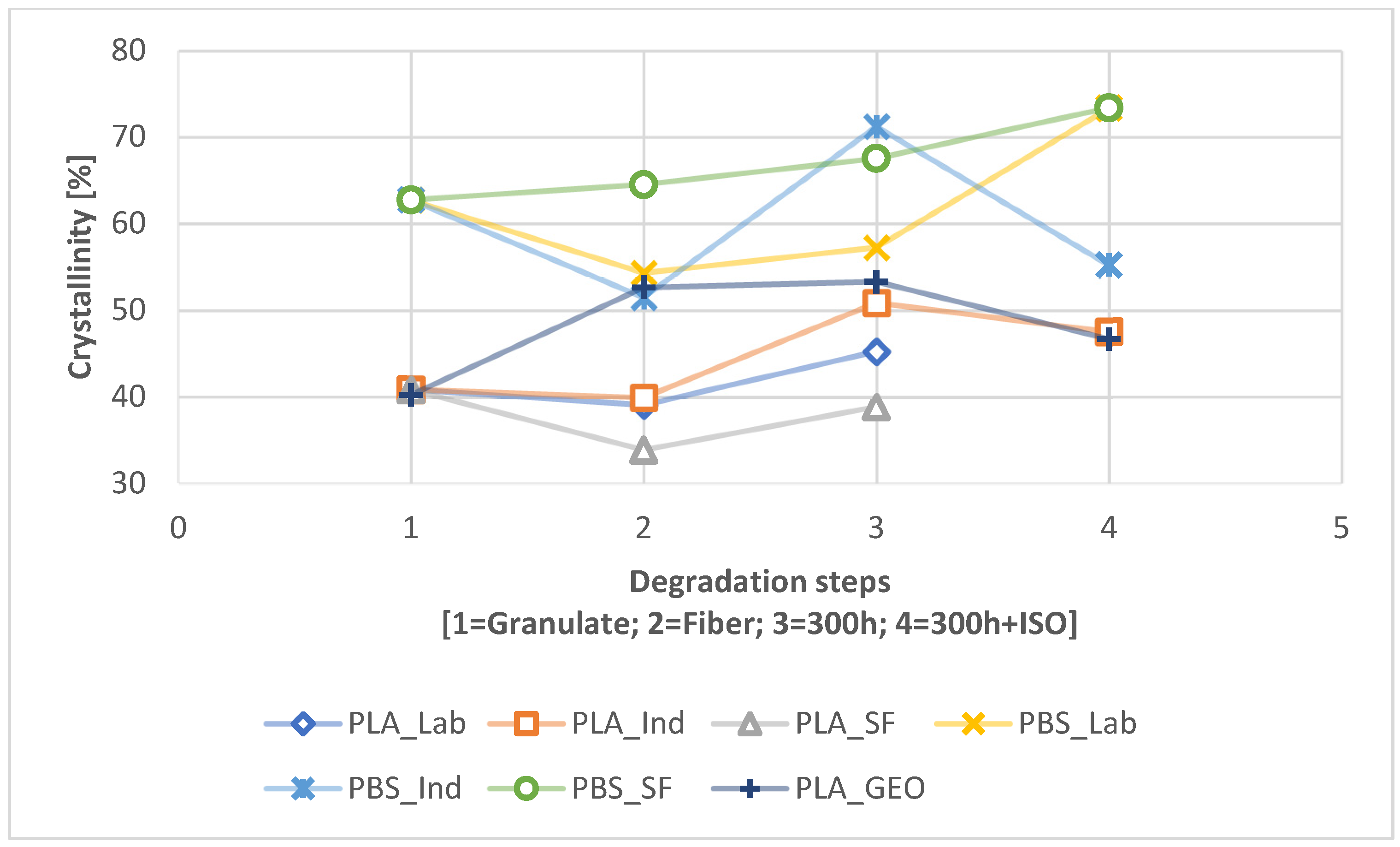
UV pre-exposure had a strong effect on the disintegration of the PLA and PBS samples, significantly increasing the CI, VI, and crystallinity while slightly reducing the M
n compared to samples that were not exposed. This was also found in previous studies, where UV pre-exposure influenced the degradation rate of various polymers [
45]. Disintegration is a degradation method that proceeds from the surface inwards. Faster degradation results from the UV radiation changing the surface topography [
46].
Two of three PLA samples disintegrated completely when maintained at 38 °C for 45 days. Degradation is usually connected to the glass transition temperature (T
g) of a polymer, meaning that PLA should be degraded at temperatures exceeding 60 °C [
4]. We observed the complete disintegration of PLA_LAB and PLA_SF, which was not solely linked to the dwell time of the spinning trial. Indeed, the longer PLA was processed in the extruder (longer dwell time), the lower the crystallinity, the lower the M
n, and the lower the T
m, leading to a combinatorial effect that accelerated the degradation. For PBS, a longer dwell time was connected to the highest crystallinity overall in this study. Furthermore, PLA was more stable during processing but was more prone to degradation in the ISO disintegration trial, whereas the opposite was true for PBS.
Interestingly, we found that laboratory-scale fibers (the focus of state-of-the-art studies thus far) behave differently to pre-loaded industrial-scale fibers, representing products placed on the market that end up in the environment. The scale of the spinning process increases the dwell time, which results in shorter polymer chains and faster degradation. If a product is manufactured at the laboratory scale, the findings of any tests may differ significantly from those applied to products from industrial-scale processes. Tests for degradability should therefore accommodate pre-loading that reflects the real-world situation. In our study, quick weathering as a pre-loading method paints a more accurate picture than the testing of pristine fibers, and we recommend this approach for product testing in the future.
In addition to the dwell time, the textile structure, temperature, and many other parameters affect the degradation process. In this study, the dwell time was isolated as far as possible at three different production scales to show its impact on degradation. Our results suggest that the degradability of biopolymers could be tailored by manipulating the dwell time during production, and this should be validated in further studies with parallel spinning trials involving a wider range of dwell times. Our findings could revolutionize the approach to biopolymer production, leading to more sustainable and environmentally friendly products. The assessment of changes in crystallinity, FTIR-based indices, and the K value determined from GPC data, offer profound insight into the changes that take place in polymer samples as they are broken down.