Design of High-Performance Molecular Imprinted Magnetic Nanoparticles-Loaded Hydrogels for Adsorption and Photodegradation of Antibiotics from Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Imprinted Hydro Sample (Imp.Hydro)
2.2. Functionalization of Magnetite (Fe3O4) with Silane Derivative
2.3. Synthesis of Magnetic Hydrogel (HydroMAG)
2.4. Synthesis of Molecularly Imprinted Magnetic Hydrogels (Imp.HydroMAG)
3. Characterization
3.1. Adsorption Tests
3.2. Photodegradation Tests
3.3. Toxicity Tests
3.4. Calculation of Toxicity Values
4. Results and Discussion
4.1. Adsorption Tests
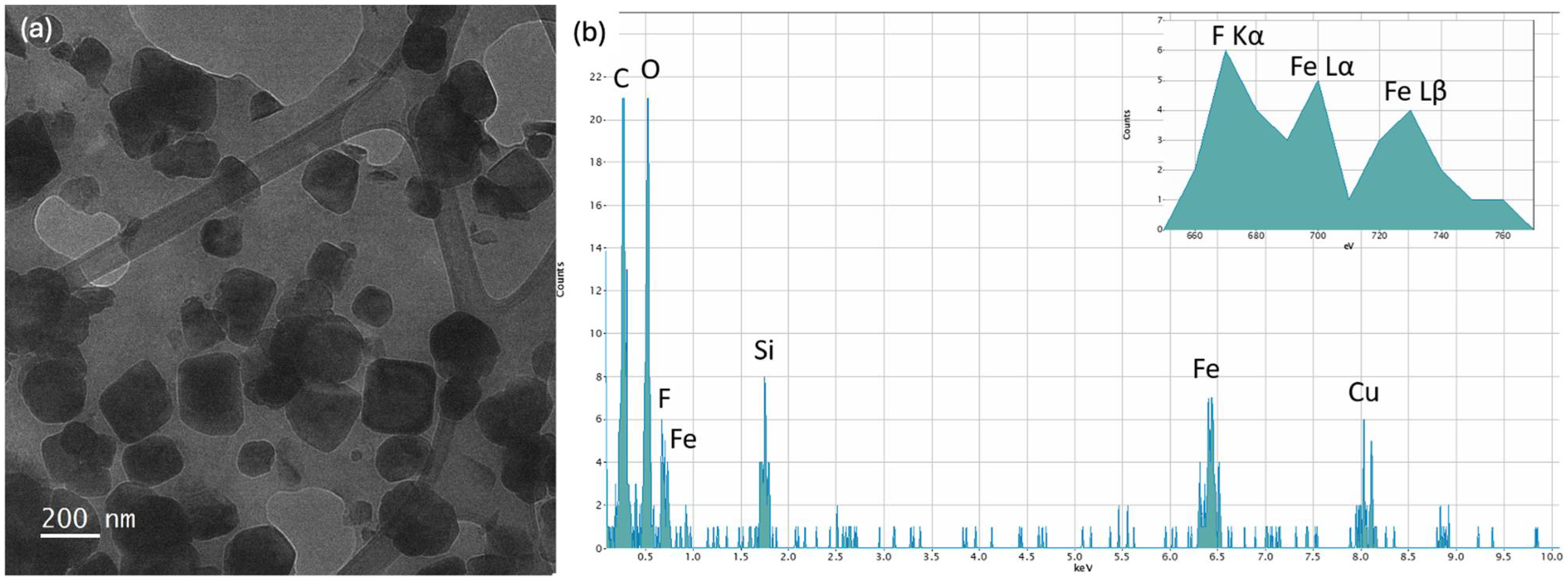
4.2. TEM Analysis
4.3. Photocatalytic Degradation Tests of Lome
4.4. Toxicity Evaluation of the Studied Materials
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Final Report to the Secretary-General of United Nations 2019. Available online: https://www.who.int/antimicrobial-resistance/interagency-coordination-group/final-report/en/ (accessed on 20 June 2024).
- Pei, M.; Zhang, B.; He, Y.; Su, J.; Gin, K.; Lev, O.; Shen, G.; Hu, S. State of the art of tertiary treatment technologies for controlling antibiotic resistance in wastewater treatment plants. Environ. Int. 2019, 131, 105026. [Google Scholar] [CrossRef] [PubMed]
- Zare, E.N.; Fallah, Z.; Le, V.T.; Doan, V.-D.; Mudhoo, A.; Joo, S.-W.; Vasseghian, Y.; Tajbakhsh, M.; Moradi, O.; Sillanpää, M.; et al. Remediation of pharmaceuticals from contaminated water by molecularly imprinted polymers: A review. Environ. Chem. Lett. 2022, 20, 2629–2664. [Google Scholar] [CrossRef] [PubMed]
- Von Sperling, M. Wastewater Characteristics, Treatment and Disposal; IWA Publishing: London, UK, 2007. [Google Scholar]
- Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), Establishing a European Chemicals Agency, Amending Directive 1999/45/EC and Repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as Well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC. Official Journal of the European Union, L 396, 30 December 2006. European Commission, Brussels, Belgium. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=OJ:L:2006:396:TOC (accessed on 20 June 2024).
- Dirion, B.; Cobb, Z.; Schillinger, E.; Andersson, L.I.; Sellergren, B. Water-Compatible Molecularly Imprinted Polymers Obtained via High-Throughput Synthesis and Experimental Design. J. Am. Chem. Soc. 2003, 125, 15101–15109. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Peng, K.; Su, Y.; Song, X.; Qiu, J.; Xiong, R.; He, L. Preparation of surface molecularly imprinted polymer and its application for the selective extraction of teicoplanin from water. RSC Adv. 2021, 11, 13615–13623. [Google Scholar] [CrossRef] [PubMed]
- Anvari, M.; Shadjou, N.; Mahmoudian, M. Synthesis of Innovative Series of MIPs using Acrylic Acid and Diallyl Dimethylammonium Chloride for Adsorption and Environmental Assessment of Chlordiazepoxide. J. Polym. Environ. 2023, 31, 4533–4545. [Google Scholar] [CrossRef]
- Roland, R.M.; Bhawani, S.A.; Ibrahim MN, M. Synthesis of molecularly imprinted polymer for the removal of cyanazine from aqueous samples. Chem. Biol. Technol. Agric. 2023, 10, 92. [Google Scholar] [CrossRef]
- Cantarella, M.; Carroccio, S.C.; Dattilo, S.; Avolio, R.; Castaldo, R.; Puglisi, C.; Privitera, V. Molecularly imprinted polymer for selective adsorption of diclofenac from contaminated water. Chem. Eng. J. 2019, 367, 180–188. [Google Scholar] [CrossRef]
- Refaat, D.; Aggour, M.G.; Farghali, A.A.; Mahajan, R.; Wiklander, J.G.; Nicholls, I.A.; Piletsky, S.A. Strategies for molecular imprinting and the evolution of MIP nanoparticles as plastic antibodies—Synthesis and applications. Int. J. Mol. Sci. 2019, 20, 6304. [Google Scholar] [CrossRef] [PubMed]
- Tasfaout, A.; Ibrahim, F.; Morrin, A.; Brisset, H.; Sorrentino, I.; Nanteuil, C.; Laffite, G.; Nicholls, I.A.; Regan, F.; Branger, C. Molecularly imprinted polymers for per- and polyfluoroalkyl substances enrichment and detection. Talanta 2023, 258, 124434. [Google Scholar] [CrossRef]
- Munoz, M.; Nieto-Sandoval, J.; Serrano, E.; De Pedro, Z.M.; Casas, J.A. CWPO intensification by induction heating using magnetite as catalyst. J. Environ. Chem. Eng. 2020, 8, 104085. [Google Scholar] [CrossRef]
- Huang, D.-L.; Wang, R.-Z.; Liu, Y.-G.; Zeng, G.-M.; Lai, C.; Xu, P.; Lu, B.-A.; Xu, J.-J.; Wang, C.; Huang, C. Application of molecularly imprinted polymers in wastewater treatment: A review. Environ. Sci. Pollut. Res. 2015, 22, 963–977. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Torrellas, S.; Munoz, M.; Mondejar, V.; de Pedro, Z.M.; Casas, J.A. Boosting the catalytic activity of natural magnetite for wet peroxide oxidation. Environ. Sci. Pollut. Res. 2020, 27, 1176–1185. [Google Scholar] [CrossRef]
- Santana, R.M.d.R.; Napoleão, D.C.; Rodriguez-Diaz, J.M.; Gomes, R.K.d.M.; Silva, M.G.; de Lima, V.M.E.; Neto, A.A.d.M.; Vinhas, G.M.; Duarte, M.M.M.B. Efficient microbial cellulose/Fe3O4 nanocomposite for photocatalytic degradation by advanced oxidation process of textile dyes. Chemosphere 2023, 326, 138453. [Google Scholar] [CrossRef]
- Brillas, E. A review on the photoelectro-Fenton process as efficient electrochemical advanced oxidation for wastewater remediation. Treatment with UV light, sunlight, and coupling with conventional and other photo-assisted advanced technologies. Chemosphere 2020, 250, 126198. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Huang, X.; Ma, J.; Wei, S.; Zhang, H. A novel electrochemical sensor based on molecularly imprinted polymer with binary functional monomers at Fe-doped porous carbon decorated Au electrode for the sensitive detection of Lomefloxacin. Ionics 2020, 26, 4183–4192. [Google Scholar] [CrossRef]
- Zagni, C.; Dattilo, S.; Mecca, T.; Gugliuzzo, C.; Scamporrino, A.A.; Privitera, V.; Puglisi, R.; Carroccio, S.C. Single and dual polymeric sponges for emerging pollutants removal. Eur. Polym. J. 2022, 179, 111556. [Google Scholar] [CrossRef]
- Thach, U.D.; Thi HH, N.; Pham, T.D.; Mai, H.D.; Nhu-Trang, T.T. Synergetic effect of dual functional monomers in molecularly imprinted polymer preparation for selective solid phase extraction of Ciprofloxacin. Polymers 2021, 13, 2788. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.M.; Foguel, M.V.; Sotomayor, M.d.P.T. Use of two functional monomers for a new approach to the synthesis of a magnetic molecularly imprinted polymer for Ciprofloxacin. J. Mater. Res. Technol. 2021, 15, 511–523. [Google Scholar] [CrossRef]
- Hamad, B. The antibiotics market. Nat. Rev. Drug Discov. 2010, 9, 675–676. [Google Scholar] [CrossRef]
- Ussia, M.; Di Mauro, A.; Mecca, T.; Cunsolo, F.; Nicotra, G.; Spinella, C.; Cerruti, P.; Impellizzeri, G.; Privitera, V.; Carroccio, S.C. ZnO-pHEMA Nanocomposites: An Ecofriendly and Reusable Material for Water Remediation. ACS Appl. Mater. Interfaces 2018, 10, 40100–40110. [Google Scholar] [CrossRef]
- Yaseen, M.; Khan, A.; Humayun, M.; Farooq, S.; Shah, N.; Bibi, S.; Khattak, Z.A.K.; Rehman, A.U.; Ahmad, S.; Ahmad, S.M.; et al. Facile Synthesis of Fe3O4−SiO2 Nanocomposites for Wastewater Treatment. Macromol. Mater. Eng. 2023, 308, 2200695. [Google Scholar] [CrossRef]
- OECD Guideline 202 for Testing of Chemicals, Daphnia sp., Acute Immobilisaiton Test; Organisation for Economic Cooperation and Development (OECD): Paris, France, 2004; ISBN 9789264069947. [CrossRef]
- OECD Guidelines for the Testing of Chemicals. Freshwater Alga and Cyanobacteria, Growth Inhibition Test; Organisation for Economic Cooperation and Development (OECD): Paris, France, 2011; ISBN 9789264069923. [CrossRef]
- Aruoja, V.; Dubourguier, H.C.; Kasemets, K.; Kahru, A. Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci. Total Environ. 2009, 407, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Joonas, E.; Aruoja, V.; Olli, K.; Syvertsen-Wiig, G.; Vija, H.; Kahru, A. Potency of (doped) rare earth oxide particles and their constituent metals to inhibit algal growth and induce direct toxic effects. Sci. Total Environ. 2017, 593–594, 478–486. [Google Scholar] [CrossRef] [PubMed]
- ISO 21338:2010; Water Quality—Kinetic Determination of the Inhibitory Effects of Sediment, Other Solids and Coloured Samples on the Light Emission of Vibrio fischeri. Organization for Standardization (ISO): Geneva, Switzerland, 2010.
- Sihtmäe, M.; Silm, E.; Kriis, K.; Kahru, A.; Kanger, T. Aminocatalysts are More Environmentally Friendly than Hydrogen-Bonding Catalysts. ChemSusChem 2022, 15, e202201045. [Google Scholar] [CrossRef] [PubMed]
- Vindimian, E. REGTOX Macro Excel ver. 2020: Redmond, WA, USA. Available online: https://www.normalesup.org/~vindimian/en_index.html (accessed on 6 May 2024).
- Song, Y.; Rong, C.; Shang, J.; Wang, Y.; Zhang, Y.; Yu, K. Synthesis of an inorganic-framework molecularly imprinted Fe-doped TiO2 composite and its selective photo-Fenton-like degradation of acid orange II. J. Chem. Technol. Biotechnol. 2017, 92, 2038–2049. [Google Scholar] [CrossRef]
- Zhang, X.; Gao, Y.; Li, Y.; Zhou, Y.; Ma, H.; Shang, J.; Cheng, X. Synthesis of magnetic NiFe2O4/CuS activator for degradation of Lomefloxacin via the activation of peroxymonosulfate under simulated sunlight illumination. Sep. Purif. Technol. 2022, 288, 120664. [Google Scholar] [CrossRef]
- De Vries, H.; Beijersbergen Van Henegouwen, G.M.J. Photochemical decomposition of Lomefloxacin in vitro and in vivo. J. Photochem. Photobiol. B Biol. 2000, 58, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Bilinska, L.; Gmurek, M.; Ledakowicz, S. Application of Advanced Oxidation Technologies for Decolorization and Mineralization of Textile Wastewaters. J. Adv. Oxid. Technol. 2015, 18, 185–194. [Google Scholar] [CrossRef]
- Sanderson, H.; Johnson, D.J.; Wilson, C.J.; Brain, R.A.; Solomon, K.R. Probabilistic hazard assessment of environmentally occurring pharmaceuticals toxicity to fish, daphnids and algae by ECOSAR screening. Toxicol. Lett. 2003, 144, 383–395. [Google Scholar] [CrossRef]
- Blaise, C.; Gagné, F.; Férard, J.F.; Eullaffroy, P. Ecotoxicity of selected nano-materials to aquatic organisms. Environ. Toxicol. 2008, 23, 591–598. [Google Scholar] [CrossRef]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef] [PubMed]






| Test Organism | Bacteria Vibrio fischeri | Crustaceans Daphnia magna | Algae Raphidocelis subcapitata | |
| Toxicity endpoint, contact time: | Inhibition of luminescence, 30 min | Immobilisation, 48 h | Inhibition of growth, 72 h | |
| 1 | Magnetite (Fe3O4) | Not toxic (EC50 > 100 mg/L) | Not toxic (LC50 > 100 mg/L) | Toxic (EC50 2.2 mg/L) |
| 2 | HEMA Hydro | Not toxic (EC50 > 100 mg/L) | Not toxic (LC50 > 100 mg/L) | Not toxic (EC50 > 100 mg/L) |
| 3 | Imp.Hydro + Lome | Not toxic (EC50 > 100 mg/L) | Not toxic (LC50 > 100 mg/L) | Not toxic (EC50 > 100 mg/L) |
| 4 | Imp.Hydro | Not toxic (EC50 > 100 mg/L) | Not toxic (LC50 > 100 mg/L) | Not toxic (EC50 > 100 mg/L) |
| 5 | HydroMAG | Not toxic (EC50 > 100 mg/L) Inhibition at 100 mg/L less than 5% | Not toxic (EC50 > 100 mg/L) Immobilisation at 100 mg/L–0 | Harmful (EC50 13.8 mg/L) |
| 6 | Imp.HydroMAG + Lome | Not toxic (EC50 > 100 mg/L) Inhibition at 100 mg/L less than 5% | Not toxic (EC50 > 100 mg/L) Immobilisation at 100 mg/L–0 | Harmful (EC50 30.7 mg/L) |
| 8 | ZnSO4x7H2O, Alfa Aesar (positive control) | Toxic EC50 = 5.9 mg Zn/L | Toxic LC50 = 1.4 mg Zn/L3 | Extremely toxic EC50 = 0.08 mg Zn/L |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Curcuruto, G.; Scamporrino, A.A.; Puglisi, R.; Nicotra, G.; Sfuncia, G.; Impellizzeri, G.; Dattilo, S.; Kahru, A.; Sihtmäe, M.; Aruoja, V.; et al. Design of High-Performance Molecular Imprinted Magnetic Nanoparticles-Loaded Hydrogels for Adsorption and Photodegradation of Antibiotics from Wastewater. Polymers 2024, 16, 2096. https://doi.org/10.3390/polym16152096
Curcuruto G, Scamporrino AA, Puglisi R, Nicotra G, Sfuncia G, Impellizzeri G, Dattilo S, Kahru A, Sihtmäe M, Aruoja V, et al. Design of High-Performance Molecular Imprinted Magnetic Nanoparticles-Loaded Hydrogels for Adsorption and Photodegradation of Antibiotics from Wastewater. Polymers. 2024; 16(15):2096. https://doi.org/10.3390/polym16152096
Chicago/Turabian StyleCurcuruto, Giusy, Andrea A. Scamporrino, Roberta Puglisi, Giuseppe Nicotra, Gianfranco Sfuncia, Giuliana Impellizzeri, Sandro Dattilo, Anne Kahru, Mariliis Sihtmäe, Villem Aruoja, and et al. 2024. "Design of High-Performance Molecular Imprinted Magnetic Nanoparticles-Loaded Hydrogels for Adsorption and Photodegradation of Antibiotics from Wastewater" Polymers 16, no. 15: 2096. https://doi.org/10.3390/polym16152096
APA StyleCurcuruto, G., Scamporrino, A. A., Puglisi, R., Nicotra, G., Sfuncia, G., Impellizzeri, G., Dattilo, S., Kahru, A., Sihtmäe, M., Aruoja, V., Blinova, I., & Carroccio, S. C. (2024). Design of High-Performance Molecular Imprinted Magnetic Nanoparticles-Loaded Hydrogels for Adsorption and Photodegradation of Antibiotics from Wastewater. Polymers, 16(15), 2096. https://doi.org/10.3390/polym16152096











