1. Introduction
Suspension stabilizers find widespread applications in various industrial processes, ranging from the formulation of consumable foods, and pharmaceuticals, to water treatment. Maintaining the suspension of solids in an aqueous medium not only enhances the visual quality of foods and drugs [
1,
2] but, in water treatment, it also improves the efficiency of removing both suspended and dissolved materials. Achieving these objectives relies on the utilization of stabilizing agents, which can be of chemical origin or derived from biological origin [
3,
4]. Nevertheless, the use or production of these stabilizers may generate waste, posing potential environmental challenges. Consequently, there is a growing interest in developing stabilizers from biological origins that are environmentally friendly and contribute to the principles of the circular economy.
A diverse array of stabilizing agents, including guar gum, xanthan gum, sodium alginate, pectins, carrageenans, gelatins, and locust bean gum, are extensively employed due to their exceptional functionality. These ingredients have significantly transformed the landscapes of the food and pharmaceutical and water treatment industries, with their availability in the market reflecting their synthetic, semi-synthetic, or natural origins [
3,
5,
6,
7]. With a heightened consumer awareness of environmental conservation and sustainability, there is a discernible preference for natural hydrocolloids, with algae emerging as prominent contenders in this category [
8,
9].
On the other hand, the extraction processes of commercial gums or hydrocolloids in many cases require the use of organic solvents and strong acidic or basic media [
10,
11], generating waste that negatively impacts the environment. Given this, the proposal of extraction with environmentally friendly methods is recurrent, so an alternative is extraction by atomization, and nostoc, being an algae with high moisture content (around 98%), is ideal to be subjected to this process.
While the majority of marine algae have undergone extensive study and exploitation in the food industry, the scientific knowledge and utilization of freshwater algae, particularly their extracted derivatives like the hydrocolloids of
Nostoc sphaericum, remain limited.
Nostoc sphaericum, an algae thriving as a renewable natural resource in the lagoons and wetlands of the Peruvian Andes, represents an area of untapped potential [
12,
13,
14]; it has a protein content of 24.01%, fat 1.88%, ash 6.19%, fiber 8.84%, carbohydrates 57.32%, humidity 10.57%, and pH 6.91 in dehydrated samples on a wet basis [
14].
Introducing novel stabilizers with broad applications poses a formidable challenge for the current industry, and the exploration of high Andean algae, such as
Nostoc spaericum, holds promise in meeting these expectations. However, understanding the key parameters of the stabilizers or hydrocolloids, including ζ potential, particle size, and rheological behavior, is imperative [
5,
15,
16,
17]. Additionally, comprehending their behavior during storage is essential. The stability of stabilizing agents in an aqueous medium over time is critical during application. This aspect becomes particularly significant in preventing the undesired sedimentation of suspended solids, a concern in various liquid products such as nectars, juices, and dairy-derived concentrates, and especially in the context of suspended drugs. Conversely, in applications where agglomeration is desired, such as certain processes [
18,
19,
20,
21,
22], sedimentation becomes a favorable outcome.
The stability of hydrocolloids in aqueous solution hinges on various factors, including hydrocolloid concentration, pH, temperature, and mixing speed. This stability can be characterized by understanding key parameters such as ζ potential, particle size, molecular weight, shear stress, strain rate, and activation energy [
18,
19,
20,
21,
22]. While these control parameters are well-established for commercial hydrocolloids, determining them for a new hydrocolloid is essential to define its potential.
In the high-altitude lagoons of Andahuaylas, a province of the Peruvian Andes, algae of the genus Nostoc flourishes at elevations above 4000 m. Remarkably, these algae serve as a folkloric food source for the residents of the local communities.
Despite its content of hydrocolloids with favorable techno-functional properties [
14,
23], this algae currently lacks commercial significance, potentially serving as a valuable alternative to commonly used industry hydrocolloids. The research is directed towards evaluating the aqueous stability of a newly developed spray-dried hydrocolloid derived from high Andean algae, specifically
Nostoc sphaericum.
2. Materials and Methods
2.1. Raw Material
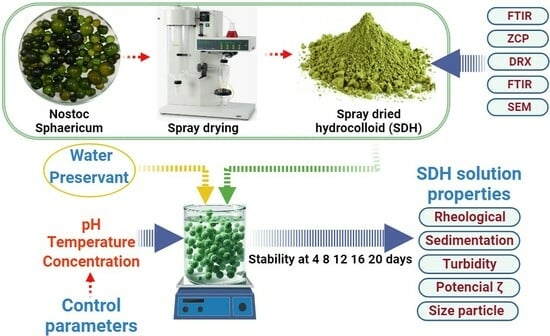
The samples of atomized hydrocolloid (SDH) of
Nostoc sphaericum were supplied by the Laboratory of Research in Advanced Materials for Water Treatment of the National University José María Arguedas, Peru. They were extracted by atomization according to the methodology proposed by Choque et al. [
14], which consists of liquefying the algae with distilled water in a 1/1 ratio, then being sieved at 45 microns, then atomized at 100 °C inlet temperature, air speed of 600 L/s, and suction speed of 38 m
3/h in a mini spray dryer model B-290, Buchi brand (Flawil, Switzerland).
2.2. Preparation of Hydrocolloid Suspensions
Suspensions were formulated in accordance with
Table 1. The process involved adjusting the pH of distilled water with 0.1 M citric acid. Subsequently, 100 mL of the solution was taken, and 1 mg of SDH along with potassium sorbate 1 mg was added (added to prevent microbial growth). The mixture was stirred at 1500 rpm for 1 min for homogenization and then left in agitation at 60 rpm for 24 h. To achieve uniform temperature, the suspensions were heated at 60 and 80 °C under continuous agitation at 60 rpm until a constant temperature was reached (refer to
Figure 1). Following this, the suspensions were cooled down to room temperature for subsequent evaluations.
2.3. SDH Characterization
The determination of the point of zero charge (PZC) involved preparing solutions with varying pH values (ranging from 2 to 12). In each case, 0.05 g of SDH was added to 50 mL of each solution and stirred at 150 rpm for 24 h at room temperature. The resulting solution’s pH was then measured, and the PZC was determined by identifying the intersection point of the initial pH and final pH curve [
24].
In a separate analysis, SDH was subject to examination using a Thermo Fisher (Waltham, MA, USA) FTIR spectrometer in ATR mode. This analysis focused on identifying influential functional groups within the range of 4000 to 400 cm−1, with a resolution of 4 cm−1.
Additionally, X-ray diffraction analysis was conducted using a Bruker diffractometer, model D8-Focus (Karlsruhe, Germany), (Cu Kα1 = 1.5406 A°) at 40 kV and 40 mA, with a PSD Lynxeye detector. The degree of crystallinity was determined by calculating the ratio between the area corresponding to the crystalline phase and the total area under the XRD curve using Equation (1) [
25].
Crystal size is not interchangeable with particle size, as crystals are contained within particles. Therefore, the average crystal size (
D) was calculated from the diffractogram using Scherrer’s formula (Equation (2)) [
26].
where k represents Scherrer’s constant (0.9),
λ is the wavelength of the X-ray source (0.15406 nm),
β denotes the peak width of the diffraction peak profile at half maximum height, a result of the small size of the crystallites (in radians), and
θ signifies the position of the peak (in radians). The data were analyzed using Origin Pro 2023 software.
The morphology of the SDH was examined using a scanning electron microscope (SEM), particularly the Prism E model by Thermo Fisher (Waltham, MA, USA), operating at an acceleration voltage of 25 kV and a magnification of 1000×.
2.4. Analysis of Rheological Behavior
The experimental samples underwent continuous testing using an Anton Paar rotational rheometer, specifically the MCR702e model (Graz, Austria). The rheometer featured a concentric cylinder arrangement, and the tests were conducted at controlled shear rates ranging from 1 to 300 s
−1 and at temperatures of 40, 60, and 80 °C. The acquired data were analyzed using shear stress models designed for non-Newtonian fluids, specifically the Power Law, Herschel–Bulkley, and Casson models, the details of which are presented in
Table 2.
The rheological models underwent adjustment via non-linear regression, employing the least squares difference as the convergence criterion and evaluated using the Quasi-Newton (QN), Simplex/Quasi-Newton (SQN), and Rosenbrock/Quasi-Newton (RQN) methods [
27,
28]. To assess the model’s quality, key metrics including the adjusted correlation coefficient (R
2), the residual mean square of the error (
MSE) calculated using Equation (6), and the mean absolute percentage of the error (
MAPE) determined through Equation (7) were considered.
In the given equations, represents the observed value; denotes the predicted value; signifies the number of observations, and represents the totalnumber of experimental observations.
Similarly, an assessment of the dispersion of residuals was conducted, employing criteria such as Random (R), Slightly Random (SR), and Tendentious (T). Models demonstrating the best fit exhibited a random distribution of residuals.
These analyses were conducted at a significance level of 5%, utilizing Excel sheets, the Solver utility, and Statistica V12 software (Statsoft, Tulsa, OK, USA).
2.5. Determination of Temperature Dependence
The influence of temperature on the rheological behavior was examined by assessing the activation energy (
Ea), providing insights into the behavior of colloidal solutions, interpenetrating networks, and nanofluid flow. This parameter is linked to the energy necessary for the interchain displacement of polymers, with higher
Ea values indicating elevated crosslinking [
29,
30]. The calculation of
Ea was carried out using the Arrhenius equation (Equation (8)) based on the consistency index values.
where
k is the consistency index of the fitted model;
k0 is the pre-exponential factor;
R is the universal gas constant (8.314 kJ/kmol.K), and T is absolute temperature, K.
2.6. SDH Suspension Stability Evaluation
The stability of the suspensions was evaluated for 20 days, employing indicators such as turbidity, sedimentation, color, and ζ potential.
For each SDH suspension formulation, 20 mL was dispensed into flat-bottomed tubes with a diameter of 1.6 cm. Subsequently, 10 mL of suspension was extracted from the top of each treatment at rest, and turbidity was measured at intervals of 4 days for 20 days. The measurements were conducted by recording the transmittance at 560 nm using a UV-Vis spectrophotometer, specifically the Thermo Fisher Genesys 150 UV model (Waltham, MA, USA). Distilled water with potassium sorbate at the study pH served as a control. It is noteworthy that samples were discarded after each measurement.
To assess sedimentation, 8 mL of the tube’s remaining volume was extracted and discarded, leaving 2 mL. The residual content was vigorously vortexed at 3000 rpm for 2 min to achieve sediment homogenization. Subsequently, the homogenized sample was subject to spectrophotometric analysis, measuring transmittance at 560 nm. This process was repeated over a span of days, with readings taken at 4-day intervals.
The color stability of the suspensions was evaluated in the CIE L* a* b* color space, employing specific criteria. Luminosity (L*) was gauged on a scale from 0 = black and 100 = white, while chroma values a* and b* were utilized to determine color characteristics (+a = red, −a = green, +b = yellow and –b = blue) [
31]. For this analysis, treatment samples were examined using a Konica Minolta colorimeter, model CR-5 (Japan), and readings were recorded in the reflectance module. Additionally, the color index (CI*) was calculated using Equation (9), providing a singular numerical representation of the color index as follows [
32]:
If CI* −40 to −20, colors range from blue-violet to deep green.
If CI* −20 to −2, the colors range from deep green to yellowish green.
If CI* −2 to +2, represents greenish yellow.
If CI* +2 to +20, colors range from pale yellow to deep orange.
If CI* +20 to +40, colors range from deep orange to deep red.
For the ζ potential measurements, a 2 mL aliquot was extracted from each treatment and transferred to a polystyrene cell. Then, the samples were subject to analysis using dynamic light scattering equipment (DLS, Zetasizer ZSU3100, Malvern Instruments, Worcestershire, UK). The instrument operated at 632.8 nm, a scattering angle of 90°, and an electric field strength of 5 V/cm. To ensure accuracy and reproducibility, readings were performed in triplicate.
2.7. Statistical Analysis
The data on the stability properties of the SDH solutions were collected in Excel sheets and were evaluated by measuring the main effects and interactions of the input variables using the Statistica V12 software. The PCA analysis was carried out by standardizing the data of the response variables to integer values through the Origin Pro 2022b Software.
4. Conclusions
Nostoc sphaericum, a freshwater algae native to the Peruvian Andes, serves as a valuable source for extracting spray-drying hydrocolloid (SDH). This hydrocolloid exhibits favorable characteristics, rendering it highly stable in aqueous medium, with zero charge point at approximately pH 8.1, a crystallinity degree of 71.98%, and an average particle size of 4.12 nm. In the aqueous medium within the pH range of 4.5 to 6.5, SDH demonstrates dilatant behavior conforming to the Power Law (R2 > 0.99). Notably, its viscosity ranges from 8.5 to 10 cP at temperatures spanning 60 to 90 °C, displaying an activation energy fluctuating between 8.19 to 37.74 kJ/mol. Extended storage stability tests conducted up to day 20 reveal consistent turbidity, minimal sedimentation, ζ potential ranging between −31 to −17 mV, and particle size maintaining a steady range of 300 to 500 nm with low variability over time. Given these attributes, SDH emerges as a potential alternative stabilizer for application in aqueous media due to its exceptional stability. Although there is still the challenge of overcoming the depigmentation of the hydrocolloid, as well as the sensory study applied in food systems.