Experimental Investigation on the Development of Environmentally Friendly Chitosan Quaternary Shale Inhibitor
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Experimental Apparatus
2.3. Experimental Methods
2.3.1. Preparation of QASC Inhibitor
2.3.2. Elemental Analysis
2.3.3. FTIR Analysis
2.3.4. Thermogravimetric Analysis
2.3.5. Solubility Test of QASC
2.3.6. Linear Expansion Test
2.3.7. Shale Rolling Recovery Test
2.3.8. Bentonite Slurry Inhibition Test
2.3.9. Preparation of Base Slurry
2.3.10. Zeta Potential Measurement
2.3.11. Particle Size Distribution Analysis
2.3.12. Compatibility Evaluation
3. Results and Discussion
3.1. Structural Characterization of QASC
3.1.1. Elemental Analysis Result
3.1.2. Thermogravimetric Analysis Results
3.1.3. FTIR Analysis Results
3.1.4. Solubility of QASC
3.2. Evaluation of Inhibitory Properties of QASC
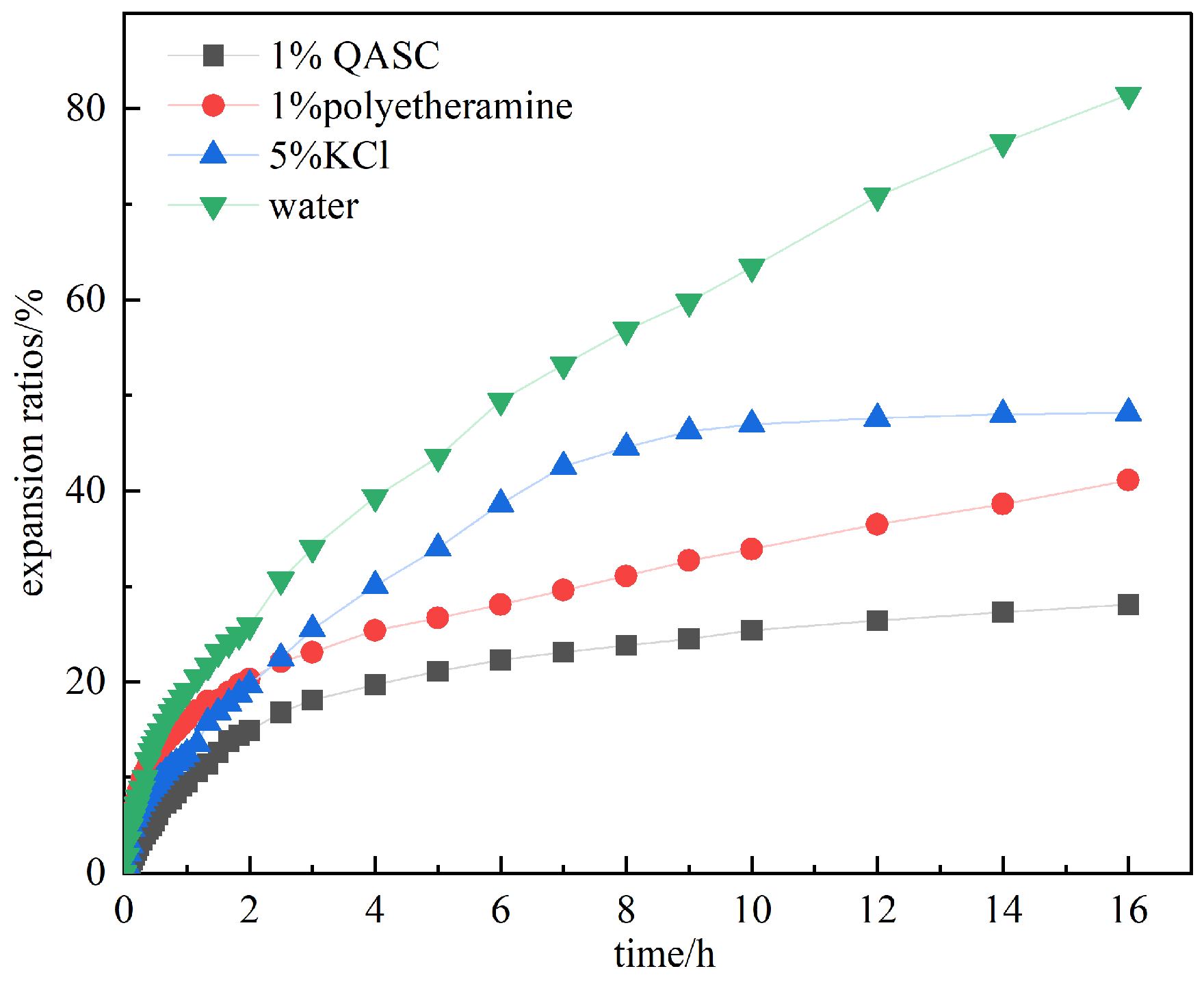
3.2.1. Results of the Shale Linear Expansion Experiment
3.2.2. Results of the Shale Rolling Recovery Experiment
3.2.3. Results of the Inhibiting Bentonite Slurry Formation Experiment
3.3. Microstructural Analysis
3.3.1. Results of Zeta Potential Measurement
3.3.2. Results of Particle Size Distribution Analysis
3.4. Analysis of the Inhibition Mechanism of QASC
3.5. The Effects of QASC on Drilling Fluids
4. Conclusions
- A novel environmentally friendly inhibitor, quaternary ammonium salt of chitosan (QASC), was successfully synthesized and characterized. In comparison to conventional inhibitors, QASC demonstrated significantly enhanced performance in terms of suppressing bentonite hydration swelling and improving shale cuttings recovery efficiency.
- Based on comprehensive microstructural analysis, the inhibition mechanism of QASC was systematically proposed. QASC can strongly adsorb onto negatively charged clay surfaces via synergistic electrostatic attraction and hydrogen bonding interactions, effectively reducing the zeta potential of sodium bentonite particles and suppressing diffuse double-layer formation. Furthermore, QASC molecules intercalate into bentonite interlayer galleries while simultaneously adsorbing onto basal surfaces, thereby creating a dual protective barrier against water invasion within the clay structure.
- Although the synthesis of QASC shale inhibitor is costly (due to complex purification and quaternization processes for chitosan) and its inhibition efficacy tends to diminish in high-salinity environments, with long-term thermal stability requiring further improvement, it still demonstrates promising application prospects as a high-performance, environmentally friendly shale inhibitor for water-based drilling fluids.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ahmad, H.M.; Kamal, M.S.; Hussain, S.M.S.; Al-Harthi, M. Synthesis of novel polymer nanocomposite for water-based drilling fluids. In Proceedings of the 35th International Conference of the Polymer Processing Society, Cesme-Izmir, Turkey, 26–30 May 2019. [Google Scholar] [CrossRef]
- Ahmad, H.M.; Murtaza, M.; Kamal, M.S.; Hussain, S.M.S.; Mahmoud, M. Cationic gemini surfactants containing biphenyl spacer as shale swelling inhibitor. J. Mol. Liq. 2021, 325, 115164. [Google Scholar] [CrossRef]
- Zhuang, G.Z.; Zhang, Z.P.; Peng, S.M.; Gao, J.H.; Jaber, M. Enhancing the rheological properties and thermal stability of oil-based drilling fluids by synergetic use of organo-montmorillonite and organo-sepiolite. Appl. Clay Sci. 2018, 161, 505–512. [Google Scholar] [CrossRef]
- Zhuang, G.Z.; Zhang, Z.P.; Yang, H.; Tan, J.J. Structures and rheological properties of organo-sepiolite in oil-based drilling fluids. Appl. Clay Sci. 2018, 154, 43–51. [Google Scholar] [CrossRef]
- Boek, E.; Coveney, P.; Skipper, N. Monte Carlo molecular modeling studies of hydrated Li-, Na-, and K-smectites: Understanding the role of potassium as a clay swelling inhibitor. J. Am. Chem. Soc. 1995, 117, 12608–12617. [Google Scholar] [CrossRef]
- Li, H.; Huang, X.B.; Sun, J.S.; Lv, K.H.; Meng, X.; Zhang, Z. Improving the anti-collapse performance of water-based drilling fluids of Xinjiang Oilfield using hydrophobically modified silica nanoparticles with cationic surfactants. Pet. Sci. 2023, 20, 1768–1778. [Google Scholar] [CrossRef]
- Ma, F.; Pu, X.; Wang, B.; Li, J.; Cao, C. Preparation and evaluation of polyampholyte inhibitor DAM. RSC Adv. 2017, 7, 49320–49328. [Google Scholar] [CrossRef]
- Zhong, H.; Qiu, Z.; Tang, Z.; Zhang, X.; Xu, J.; Huang, W. Study of 4, 4′-methylenebiscyclohexanamine as a high temperature-resistant shale inhibitor. J. Mater. Sci. 2016, 51, 7585–7597. [Google Scholar] [CrossRef]
- Peng, B.; Luo, P.Y.; Guo, W.Y.; Yuan, Q.Y. Structure–property relationship of polyetheramines as clay-swelling inhibitors in water-based drilling fluids. J. Appl. Polym. Sci. 2013, 129, 1074–1079. [Google Scholar] [CrossRef]
- Jiang, G.C.; Qi, Y.R.; An, Y.X.; Huang, X.B.; Ren, Y.J. Polyethyleneimine as shale inhibitor in drilling fluid. Appl. Clay Sci. 2016, 127, 70–77. [Google Scholar] [CrossRef]
- Zhong, H.Y.; Qiu, Z.S.; Sun, D.; Zhang, D.M.; Huang, W.A. Inhibitive properties comparison of different polyetheramines in water-based drilling fluid. J. Nat. Gas Sci. Eng. 2015, 26, 99–107. [Google Scholar] [CrossRef]
- Wang, Z.L.; Liu, J.P.; Lv, K.H.; Shao, Z.H.; Zhang, X.F.; Xu, Z.; Zhang, F.; Dong, X.; Sun, J.S. Hydrophobically modified low molecular weight polymers as high temperature resistant shale inhibitor. J. Mol. Liq. 2023, 382, 121856. [Google Scholar] [CrossRef]
- Bai, X.D.; Wang, H.; Luo, Y.M.; Zheng, X.X.; Zhang, X.Y.; Zhou, S.; Pu, X.L. The structure and application of amine-terminated hyperbranched polymer shale inhibitor for waterbased drilling fluid. J. Appl. Polym. Sci. 2017, 134, 45466. [Google Scholar] [CrossRef]
- Liu, X.J.; Liu, K.; Gou, S.H.; Liang, L.X.; Luo, C.; Guo, Q.P. Water-soluble acrylamide sulfonate copolymer for inhibiting shale hydration. Ind. Eng. Chem. Res. 2014, 53, 2903–2910. [Google Scholar] [CrossRef]
- Huang, X.B.; Shen, H.K.; Sun, J.S.; Lv, K.H.; Liu, J.P.; Dong, X.D.; Luo, S. Nanoscale laponite as a potential shale inhibitor in water-based drilling fluid for stabilization of wellbore stability and mechanism study. ACS Appl. Mater. Interfaces 2018, 10, 33252–33259. [Google Scholar] [CrossRef]
- Huang, X.B.; Sun, J.S.; Lv, K.H.; Liu, J.P.; Shen, H.K.; Zhang, F. Application of core-shell structural acrylic resin/nano-SiO2 composite in water based drilling fluid to plug shale pores. J. Nat. Gas Sci. Eng. 2018, 55, 418–425. [Google Scholar] [CrossRef]
- Rana, A.; Arfaj, M.K.; Saleh, T.A. Advanced developments in shale inhibitors for oil production with low environmental footprints—A review. Fuel 2019, 247, 237–249. [Google Scholar] [CrossRef]
- Xu, Y.; Xu, G.L.; Jiang, G.C. Synthesis and Application of Environmentally Friendly Modified Biopeptide Water-Based Drilling Fluid Inhibitor. Drill. Fluid Complet. Fluid 2022, 39, 301–306. [Google Scholar]
- Cescon, L.; Dos, S.; Quartarone, P.; Ribeiro, S.P.; Da, S.; Nascimento, R.S.V. Cationic starch derivatives as reactive shale inhibitors for water-based drilling fluids. J. Appl. Polym. Sci. 2018, 135, 46621. [Google Scholar] [CrossRef]
- Ghasemi, A.; Jalalifar, H.; Norouzi Apourvari, S.; Sakebi, M.R. Mechanistic study of improvement of wellbore stability in shale formations using a natural inhibitor. J. Pet. Sci. Eng. 2019, 181, 106222. [Google Scholar] [CrossRef]
- Jiang, G.C.; Li, X.L.; Zhu, H.H.; Yang, L.L.; Li, Y.Y.; Wang, T.D.; Wu, X.J. Improved shale hydration inhibition with combination of gelatin and KCl or EPTAC, an environmentally friendly inhibitor for water-based drilling fluids. J. Appl. Polym. Sci. 2019, 136, 47585. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Suter, J.L.; Coveney, P.V.; Anderson, R.L.; Greenwell, H.C.; Cliffe, S. Rule based design of clay-swelling inhibitors. Energy Environ. Sci. 2011, 4, 4572. [Google Scholar] [CrossRef]
- Elsabee, M.Z.; Naguib, H.F.; Morsi, R.E. Chitosan based nanofibers, review. Mater. Sci. Eng. C 2012, 32, 1711–1726. [Google Scholar] [CrossRef]
- Xu, Y.X.; Ren, X.; Hanna, M.A. Chitosan/clay nanocomposite film preparation and characterization. J. Appl. Polym. Sci. 2006, 99, 1684–1691. [Google Scholar] [CrossRef]
- Xuan, Y.; Jiang, G.C.; Song, R.R.; Wang, Z.F.; Lin, Y.X. Study on Hyper-Branched Polyethyleneimine as Shale Inhibitor for Drilling Fluid. J. China Univ. Pet. (Nat. Sci. Ed.) 2017, 41, 178–186. [Google Scholar]
- Lv, K.H.; Huang, X.B.; Li, H.; Sun, J.; Du, W.; Li, M. Modified biosurfactant cationic alkyl polyglycoside as an effective additive for inhibition of highly reactive shale. Energy Fuels 2020, 34, 1680–1687. [Google Scholar] [CrossRef]









| No. | Bentonite | FA367 | BTM-2 | AP-1 | DSP-1 | NP-1 | Emulsified Asphalt | Barite |
|---|---|---|---|---|---|---|---|---|
| Content (wt%) | 4 | 0.15 | 0.5% | 0.5 | 1.5% | 10.4 | 3 | 85 |
| Sample | Mass Fraction of Different Elements | |
|---|---|---|
| C (%) | N (%) | |
| CS | 41.52 | 7.80 |
| QASC | 36.42 | 7.16 |
| Experimental Conditions | Formulation | AV (mPa·s) | PV (mPa·s) | YP (Pa) | YP/PV (Pa) | G10s/10min (Pa/Pa) | FLAPI (mL) |
|---|---|---|---|---|---|---|---|
| indoor temperature | Original formulation | 80 | 59 | 19 | 0.3 | 1.5/5.0 | 2.6 |
| Original formulation +1% QASC | 88 | 67 | 21 | 0.2 | 2.5/6.0 | 3.4 | |
| hot-rolled at 150 °C for 16 h | Original formulation | 64 | 48 | 16 | 0.3 | 1.5/4.5 | 3.8 |
| Original formulation +1% QASC | 80 | 63 | 17 | 0.3 | 2.0/5.0 | 4.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Duan, Z.; Ouyang, Y.; Si, D.; Huang, Z.; Zhou, Y.; Hui, C. Experimental Investigation on the Development of Environmentally Friendly Chitosan Quaternary Shale Inhibitor. Polymers 2026, 18, 561. https://doi.org/10.3390/polym18050561
Duan Z, Ouyang Y, Si D, Huang Z, Zhou Y, Hui C. Experimental Investigation on the Development of Environmentally Friendly Chitosan Quaternary Shale Inhibitor. Polymers. 2026; 18(5):561. https://doi.org/10.3390/polym18050561
Chicago/Turabian StyleDuan, Zhifeng, Yong Ouyang, Daichun Si, Zhanying Huang, Yu Zhou, and Cheng Hui. 2026. "Experimental Investigation on the Development of Environmentally Friendly Chitosan Quaternary Shale Inhibitor" Polymers 18, no. 5: 561. https://doi.org/10.3390/polym18050561
APA StyleDuan, Z., Ouyang, Y., Si, D., Huang, Z., Zhou, Y., & Hui, C. (2026). Experimental Investigation on the Development of Environmentally Friendly Chitosan Quaternary Shale Inhibitor. Polymers, 18(5), 561. https://doi.org/10.3390/polym18050561




