Evaluation of Electrospun PCL-PIBMD Meshes Modified with Plasmid Complexes in Vitro and in Vivo
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of MPs and MPs-pZNF580 Complexes Suspension
2.3. Electrospinning Process
2.4. Characterization of Electrospun Fibrous Meshes
2.4.1. Analysis of the Surface Morphology of Electrospun Fibrous Meshes
2.4.2. FT-IR Measurements
2.4.3. Mechanical Properties Tests
2.4.4. Static Water Contact Angle
2.5. Biological Evaluation
2.5.1. Culture of EA.hy926 Cells
2.5.2. Cell Proliferation Assay
2.5.3. Fluorescence Assay
2.5.4. Platelet Adhesion Assay
2.5.5. Subcutaneous Implantation
2.6. Statistical Analysis
3. Results and Discussion
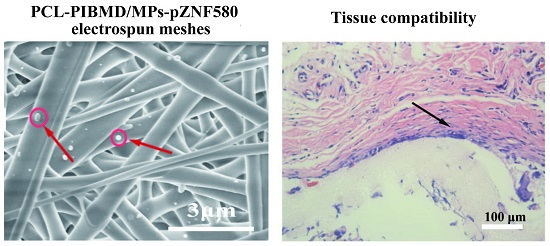
3.1. Morphology and Characterizations of Fibrous Meshes
3.2. ATR-FTIR Analysis
3.3. Mechanical Properties of Fibrous Meshes
3.4. WCA of Fibrous Meshes
3.5. Morphology of EA.hy926 Cells on PCL-PIBMD/MPs Meshes
3.6. Proliferation of EA.hy926 on PCL-PIBMD/MPs Meshes
3.7. Platelet Adhesion Examination
3.8. Tissue Response of Meshes in Subcutaneous Implantation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dzingina, M.D.; Higginson, I.J. Public health and palliative care in 2015. Clin. Geriatr. Med. 2015, 31, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Kurobe, H.; Maxfield, M.W.; Breuer, C.K.; Shinoka, T. Concise review: Tissue-engineered vascular grafts for cardiac surgery: Past, present, and future. Stem Cells Transl. Med. 2012, 1, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Sengupta, D.; Chien, S. Vascular tissue engineering: From in vitro to in situ. Wiley Interdiscip. Rev. Syst. Biol. Med. 2014, 6, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Bouten, C.V.; Dankers, P.Y.; Driessen-Mol, A.; Pedron, S.; Brizard, A.M.; Baaijens, F.P. Substrates for cardiovascular tissue engineering. Adv. Drug Deliver. Rev. 2011, 63, 221–241. [Google Scholar] [CrossRef] [PubMed]
- Hasan, A.; Memic, A.; Annabi, N.; Hossain, M.; Paul, A.; Dokmeci, M.R.; Dehghani, F.; Khademhosseini, A. Electrospun scaffolds for tissue engineering of vascular grafts. Acta Biomater. 2014, 10, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Ravi, S.; Chaikof, E.L. Biomaterials for vascular tissue engineering. Regen. Med. 2010, 5, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Wise, S.G.; Byrom, M.J.; Waterhouse, A.; Bannon, P.G.; Weiss, A.S.; Ng, M.K. A multilayered synthetic human elastin/polycaprolactone hybrid vascular graft with tailored mechanical properties. Acta Biomater. 2011, 7, 295–303. [Google Scholar] [CrossRef] [PubMed]
- McKenna, K.A.; Hinds, M.T.; Sarao, R.C.; Wu, P.C.; Maslen, C.L.; Glanville, R.W.; Babcock, D.; Gregory, K.W. Mechanical property characterization of electrospun recombinant human tropoelastin for vascular graft biomaterials. Acta Biomater. 2012, 8, 225–233. [Google Scholar] [CrossRef]
- Steinhoff, G.; Stock, U.; Karim, N.; Mertsching, H.; Timke, A.; Meliss, R.R.; Pethig, K.; Haverich, A.; Bader, A. Tissue engineering of pulmonary heart valves on allogenic acellular matrix conduits—In vivo restoration of valve tissue. Circulation 2000, 102, 50–55. [Google Scholar] [CrossRef]
- Smith, M.J.; McClure, M.J.; Sell, S.A.; Barnes, C.P.; Walpoth, B.H.; Simpson, D.G.; Bowlin, G.L. Suture-reinforced electrospun polydioxanone-elastin small-diameter tubes for use in vascular tissue engineering: A feasibility study. Acta Biomater. 2008, 4, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Feng, Y.; Zhang, L. Biodegradable carrier/gene complexes to mediate the transfection and proliferation of human vascular endothelia cells. Polym. Adv. Technol. 2015, 12, 1370–1377. [Google Scholar] [CrossRef]
- Wang, H.; Feng, Y.; Yuan, W.; Zhao, H.; Fang, Z.; Khan, M.; Guo, J. Fabrication and characterization of electrospun biocompatible PU/PEGMA hybrid nanofibers by in-situ UV photopolymerization. Sci. China Phys. Mech. 2012, 55, 1189–1193. [Google Scholar] [CrossRef]
- Kannan, R.Y.; Salacinski, H.J.; Butler, P.E.; Hamilton, G.; Seifalian, A.M. Current status of prosthetic bypass grafts: A review. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 74, 570–581. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Yang, F.; Seyednejad, H.; Chen, Z.; Hennink, W.E.; Anderson, J.M.; van den Beucken, J.J.; Jansen, J.A. Biocompatibility and degradation characteristics of PLGA-based electrospun nanofibrous scaffolds with nanoapatite incorporation. Biomaterials 2012, 33, 6604–6614. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.; Juran, C.; McClendon, M.; Eyadiel, C.; McFetridge, P.S. Development of a mechanically tuneable 3D scaffold for vascular reconstruction. J. Biomed. Mater. Res. A 2012, 100, 3480–3489. [Google Scholar] [CrossRef] [PubMed]
- Tugulu, S.; Silacci, P.; Stergiopulos, N.; Klok, H.A. RGD-functionalized polymer brushes as substrates for the integrin specific adhesion of human umbilical vein endothelial cells. Biomaterials 2007, 28, 2536–2546. [Google Scholar] [CrossRef] [PubMed]
- Avci-Adali, M.; Ziemer, G.; Wendel, H.P. Induction of EPC homing on biofunctionalized vascular grafts for rapid in vivo self-endothelialization-a review of current strategies. Biotechnol. Adv. 2010, 28, 119–129. [Google Scholar] [CrossRef] [PubMed]
- De-Mel, A.; Jell, G.; Stevens, M.M.; Seifalian, A.M. Biofunctionalization of biomaterials for accelerated in situ endothelialization: A review. Biomacromolecules 2008, 9, 2969–2979. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Jia, X.; Han, F.; Zhao, J.; Zhao, Y.; Fan, Y.; Yuan, X. Dual-delivery of VEGF and PDGF by double-layered electrospun membranes for blood vessel regeneration. Biomaterials 2013, 34, 2202–2212. [Google Scholar] [CrossRef] [PubMed]
- Naito, Y.; Shinoka, T.; Duncan, D.; Hibino, N.; Solomon, D.; Cleary, M.; Rathore, A.; Fein, C.; Church, S.; Breuer, C. Vascular tissue engineering: Towards the next generation vascular grafts. Adv. Drug Deliver. Rev. 2011, 63, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Savoji, H.; Hadjizadeh, A.; Maire, M.; Ajji, A.; Wertheimer, M.R.; Lerouge, S. Electrospun nanofiber scaffolds and plasma polymerization: A promising combination towards complete, stable endothelial lining for vascular grafts. Macromol. Biosci. 2014, 14, 1084–1095. [Google Scholar] [CrossRef] [PubMed]
- Del Gaudio, C.; Baiguera, S.; Boieri, M.; Mazzanti, B.; Ribatti, D.; Bianco, A.; Macchiarini, P. Induction of angiogenesis using VEGF releasing genipin-crosslinked electrospun gelatin mats. Biomaterials 2013, 34, 7754–7765. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, H.; Zheng, W.; Zhang, J.; Zhao, Q.; Wang, S.; Yang, Z.; Kong, D. Highly stable surface modifications of poly(ε-caprolactone) (PCL) films by molecular self-assembly to promote cells adhesion and proliferation. Chem. Commun. 2011, 47, 8901–8903. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wang, Z.; Song, L.; Zhao, Q.; Zhang, J.; Li, D.; Wang, S.; Han, J.; Zheng, X.L.; Yang, Z.; Kong, D. Endothelialization and patency of RGD-functionalized vascular grafts in a rabbit carotid artery model. Biomaterials 2012, 33, 2880–2891. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Wan, H.; Xia, T.; Guo, X.; Wang, H.; Liu, Y.; Li, X. Promoted regeneration of mature blood vessels by electrospun fibers with loaded multiple pDNA-calcium phosphate nanoparticles. Eur. J. Pharm. Biopharm. 2013, 85, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Yao, F.; Huang, J.; Han, G.; Li, Q.; Khan, M.; Feng, Y.; Zhang, W. Proliferation and migration of human vascular endothelial cells mediated by ZNF580 gene complexed with MPEG-b-p(MMD-co-GA)-g-PEI microparticles. J. Mater. Chem. B 2014, 2, 1825–1837. [Google Scholar] [CrossRef]
- Shi, C.; Li, Q.; Zhang, W.; Feng, Y.; Ren, X. REDV peptide conjugated nanoparticles/pZNF580 complexes for actively targeting human vascular endothelial cells. ACS Appl. Mater. Interfaces 2015, 7, 20389–20399. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Yao, F.; Li, Q.; Khan, M.; Ren, X.; Feng, Y.; Huang, J.; Zhang, W. Regulation of the endothelialization by human vascular endothelial cells by ZNF580 gene complexed with biodegradable microparticles. Biomaterials 2014, 35, 7133–7145. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Cuddihy, M.J.; Kotov, N.A. Three-dimensional cell culture matrices: State of the art. Tissue Eng. B Rev. 2008, 14, 61–86. [Google Scholar] [CrossRef]
- Yang, J.; Chen, H.; Yuan, Y.; Sarkar, D.B.; Zheng, J. Synthesis and characterization of biocompatible polyurethanes for controlled release of hydrophobic and hydrophilic drugs. Front. Chem. Sci. Eng. 2014, 8, 498–510. [Google Scholar] [CrossRef]
- Hakkarainen, M.; Höglund, A.; Odelius, K.; Albertsson, A.C. Tuning the release rate of acidic degradation products through macromolecular design of caprolactone-based copolymers. J. Am. Chem. Soc. 2007, 129, 6308–6312. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, J.; Zhang, Y.Z.; Ramakrishna, S. Fabrication of modified and functionalized polycaprolactone nanofibre scaffolds for vascular tissue engineering. Nanotechnology 2005, 16, 2138–2142. [Google Scholar] [CrossRef] [PubMed]
- Battig, A.; Hiebl, B.; Feng, Y.; Lendlein, A.; Behl, M. Biological evaluation of degradable, stimuli-sensitive multiblock copolymers having polydepsipeptide- and poly(ε-caprolactone) segments in vitro. Clin. Hemorheol. Microcirc. 2011, 48, 161–172. [Google Scholar] [PubMed]
- McBane, J.E.; Sharifpoor, S.; Cai, K.; Labow, R.S.; Santerre, J.P. Biodegradation and in vivo biocompatibility of a degradable, polar/hydrophobic/ionic polyurethane for tissue engineering applications. Biomaterials 2011, 32, 6034–6044. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Feng, Y.; Yang, J.; Guo, J.; Zhang, W. Targeting REDV peptide functionalized polycationic gene carrier for enhancing the transfection and migration capability of human endothelial cells. J. Mater. Chem. B 2015, 3, 3379–3391. [Google Scholar] [CrossRef]
- Feng, Y.; Behl, M.; Kelch, S.; Lendlein, A. Biodegradable multiblock copolymers based on oligodepsipeptides with shape-memory properties. Macromol. Biosci. 2009, 9, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.; Li, Q.; Lv, J.; Yu, L.; Ren, X.; Zhang, L.; Feng, Y.; Zhang, W. CREDVW-linked polymeric micelles as a targeting gene transfer vector for selective transfection and proliferation of endothelial cells. ACS Appl. Mater. Interfaces 2015, 7, 12128–12140. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Shi, C.; Zhang, W.; Behl, M.; Lendlein, A.; Feng, Y. Nanoparticles complexed with gene vectors to promote proliferation of human vascular endothelial cells. Adv. Healthc. Mater. 2015, 4, 1225–1235. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Feng, Y.; Li, Q.; Hao, X.; Liu, W.; Zhou, W.; Shi, C.; Ren, X.; Zhang, W. PLGA/SF blend scaffolds modified with plasmid complexes for enhancing proliferation of endothelial cells. React. Funct. Polym. 2015, 91–92, 19–27. [Google Scholar] [CrossRef]
- Stearns-Kurosawa, D.J.; Kurosawa, S.; Mollica, J.S.; Ferrell, G.L.; Esmon, C.T. The endothelial cell protein C receptor augments protein C activation by the thrombin-thrombomodulin complex. Proc. Natl. Acad. Sci. USA 1996, 93, 10212–10216. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.W.; Kim, H.W.; Kim, Y.B.; Rhee, Y.H. Poly(ethylene glycol)-grafted poly(3-hydroxyundecenoate) networks for enhanced blood compatibility. Int. J. Biol. Macromol. 2003, 32, 17–22. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Sill, T.J.; Von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef] [PubMed]
- Gaumer, J.; Prasad, A.; Lee, D.; Lannutti, J. Structure-function relationships and source-to-ground distance in electrospun polycaprolactone. Acta Biomater. 2009, 5, 1552–1561. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Krikorian, V.; Yu, J.H.; Thomas, E.L.; Rutledge, G.C. Electrospun polymer nanofibers with internal periodic structure obtained by microphase separation of cylindrically confined block copolymers. Nano Lett. 2006, 6, 2969–2972. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, P.; Tang, L.; Sun, P.; Liu, W.; Sun, P.; Zuo, A.; Liang, D. Temperature-tuned DNA condensation and gene transfection by PEI-g-(PMEO2MA-b-PHEMA) copolymer-based nonviral vectors. Biomaterials 2010, 31, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Barth, A. The infrared absorption of amino acid side chains. Prog. Biophys. Mol. Biol. 2000, 74, 141–173. [Google Scholar] [CrossRef]
- Zhou, W.; Feng, Y.; Yang, J.; Fan, J.; Lv, J.; Zhang, L.; Guo, J.; Ren, X.; Zhang, W. Electrospun scaffolds of silk fibroin and poly(lactide-co-glycolide) for endothelial cell growth. J. Mater. Sci. Mater. Med. 2015, 26, 5386. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Kim, H.Y.; Khil, M.S.; Ra, Y.M.; Lee, D.R. Characterization of nano-structured poly(ε-caprolactone) nonwoven mats via electrospinning. Polymers 2003, 44, 1287–1294. [Google Scholar] [CrossRef]
- Kurane, A.; Simionescu, D.T.; Vyavahare, N.R. In vivo cellular repopulation of tubular elastin scaffolds mediated by basic fibroblast growth factor. Biomaterials 2007, 28, 2830–2838. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.H.; Lee, J.H. Hydrophilization of synthetic biodegradable polymer scaffolds for improved cell/tissue compatibility. Biomed. Mater. 2013, 8, 014101. [Google Scholar] [CrossRef] [PubMed]
- Soria, J.M.; Ramos, C.M.; Bahamonde, O.; Cruz, D.M.; Sanchez, M.S.; Esparza, M.A.; Casas, C.; Guzman, M.; Navarro, X.; Ribelles, J.L.; et al. Influence of the substrate’s hydrophilicity on the in vitro schwann cells viability. J. Biomed. Mater. Res. A 2007, 83A, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.-H.; Wang, M.-J.; Chang, C.-W.; Wei, T.-C.; Lai, J.-Y.; Tsai, W.-B.; Lee, C. Improvement of hemocompatibility on materials by photoimmobilization of poly(ethylene glycol). J. Mater. Chem. 2012, 22, 9991–9999. [Google Scholar] [CrossRef]
- Khan, M.; Yang, J.; Shi, C.; Lv, J.; Feng, Y.; Zhang, W. Surface tailoring for selective endothelialization and platelet inhibition via a combination of SI-ATRP and click chemistry using Cys-Ala-Gly-peptide. Acta Biomater. 2015, 20, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Feng, Y.; Guo, J.; Wang, H.; Li, Q.; Yang, J.; Hao, X.; Lv, J.; Ma, N.; Li, W. Surface modification and endothelialization of biomaterials as potential scaffolds for vascular tissue engineering applications. Chem. Soc. Rev. 2015, 44, 5745–5745. [Google Scholar] [CrossRef] [PubMed]
- Hubbell, J.A.; Massia, S.P.; Desai, N.P.; Drumheller, P.D. Endothelial cell-selective materials for tissue engineering in the vascular graft via a new receptor. Nat. Biotechnol. 1991, 9, 568–572. [Google Scholar] [CrossRef]
- Ninan, L.; Monahan, J.; Stroshine, R.L.; Wilker, J.J.; Shi, R.Y. Adhesive strength of marine mussel extracts on porcine skin. Biomaterials 2003, 24, 4091–4099. [Google Scholar] [CrossRef]
- Lin, Q.-K.; Hou, Y.; Ren, K.-F.; Ji, J. Selective endothelial cells adhesion to Arg-Glu-Asp-Val peptide functionalized polysaccharide multilayer. Thin Solid Films 2012, 520, 4971–4978. [Google Scholar] [CrossRef]
- Sun, H.; Wei, S.; Xu, R.; Xu, P.; Zhang, W. Sphingosine-1-phosphate induces human endothelial VEGF and MMP-2 production via transcription factor ZNF580: Novel insights into angiogenesis. Biochem. Biophys. Res. Commun. 2010, 395, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Cheng, B.W.H.; Che, C.T.; Hsieh, D.P.H. Cytotoxicity assessment of Ma-huang (Ephedra) under different conditions of preparation. Toxicol. Sci. 2000, 56, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Yang, P.; Qin, W.; Maitz, M.F.; Zhou, S.; Huang, N. The effect of coimmobilizing heparin and fibronectin on titanium on hemocompatibility and endothelialization. Biomaterials 2011, 32, 4691–4703. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Lv, J.; Gao, B.; Zhang, L.; Yang, D.; Shi, C.; Guo, J.; Li, W.; Feng, Y. Modification of polycarbonateurethane surface with poly(ethylene glycol) monoacrylate and phosphorylcholine glyceraldehyde for anti-platelet adhesion. Front. Chem. Sci. Eng. 2014, 8, 188–196. [Google Scholar]
- Zhang, M.; Wang, K.; Wang, Z.; Xing, B.; Zhao, Q.; Kong, D. Small-diameter tissue engineered vascular graft made of electrospun PCL/lecithin blend. J. Mater. Sci. Mater. Med. 2012, 23, 2639–2648. [Google Scholar] [CrossRef] [PubMed]
- Badylak, S.F.; Gilbert, T.W. Immune response to biologic scaffold materials. Semin. Immunol. 2008, 20, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Wang, H.; Liu, J.; Zhang, M.; Zhang, W. ROS-induced ZNF580 expression: A key role for H2O2/NF-κB signaling pathway in vascular endothelial inflammation. Mol. Cell. Biochem. 2012, 359, 183–191. [Google Scholar]








© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, Y.; Liu, W.; Ren, X.; Lu, W.; Guo, M.; Behl, M.; Lendlein, A.; Zhang, W. Evaluation of Electrospun PCL-PIBMD Meshes Modified with Plasmid Complexes in Vitro and in Vivo. Polymers 2016, 8, 58. https://doi.org/10.3390/polym8030058
Feng Y, Liu W, Ren X, Lu W, Guo M, Behl M, Lendlein A, Zhang W. Evaluation of Electrospun PCL-PIBMD Meshes Modified with Plasmid Complexes in Vitro and in Vivo. Polymers. 2016; 8(3):58. https://doi.org/10.3390/polym8030058
Chicago/Turabian StyleFeng, Yakai, Wen Liu, Xiangkui Ren, Wei Lu, Mengyang Guo, Marc Behl, Andreas Lendlein, and Wencheng Zhang. 2016. "Evaluation of Electrospun PCL-PIBMD Meshes Modified with Plasmid Complexes in Vitro and in Vivo" Polymers 8, no. 3: 58. https://doi.org/10.3390/polym8030058