Comparative Study on Protein Quality and Rheological Behavior of Different Wheat Species
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Trial and Sampling
2.2. Baking Quality Characterization
2.2.1. Wheat Flour Samples
2.2.2. Protein Fractions
2.2.3. High Molecular Weight Glutenin Subunits (HMW-GS) Analysis
2.3. Mixolab Analysis
2.4. Statistical Analysis
3. Results
3.1. Thousand Grain Weight and Grain Yield of Wheat Varieties
3.2. Basic Baking Quality
Baking Quality Characterization
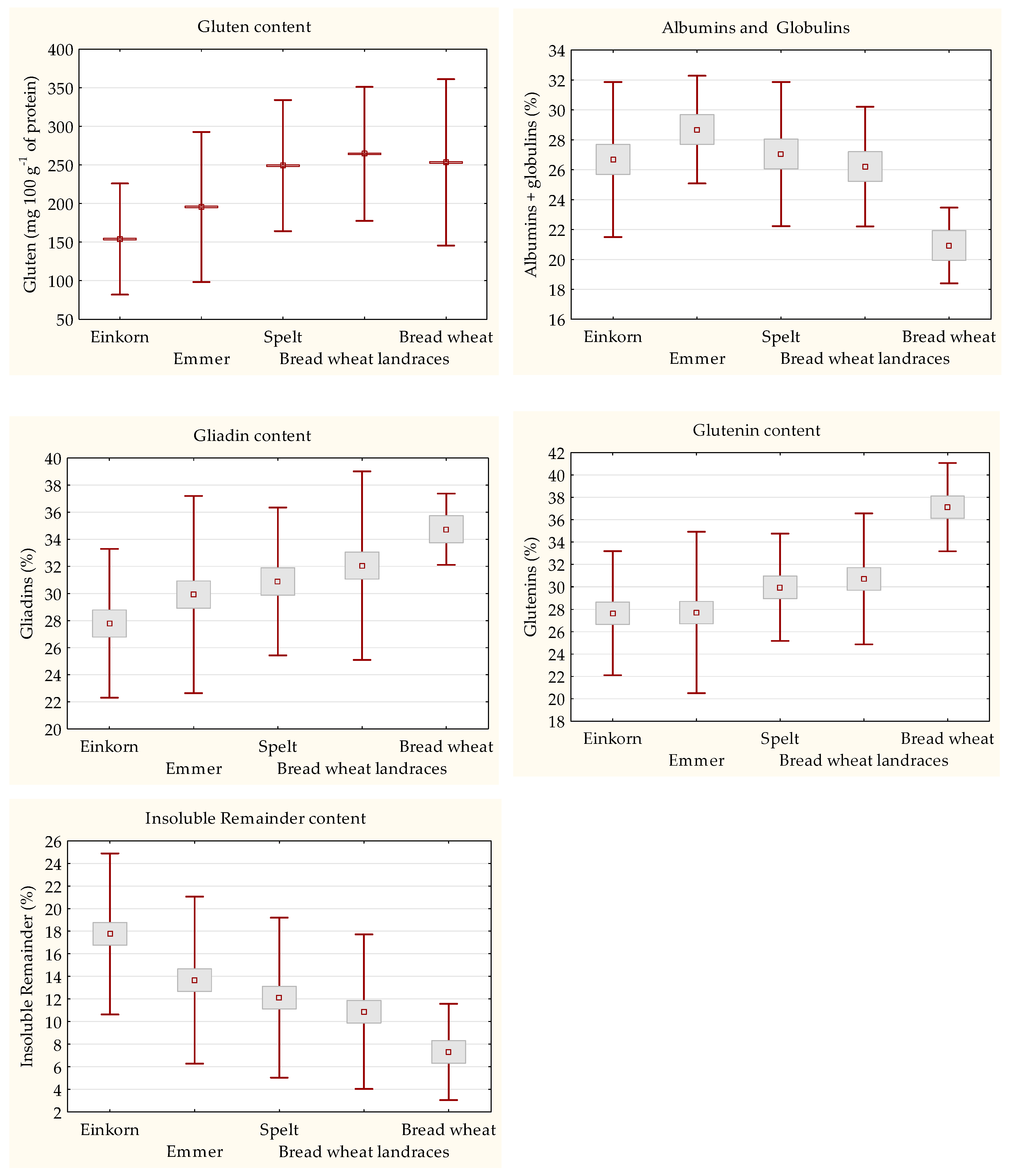
3.3. Quantitaion of Gluten Content and Protein Fractions Composition
3.4. Characterization of HMW-GSs from the Technological Viewpoint
3.5. Rheological Properties of Einkorn, Emmer, Spelt, and Bread Wheat
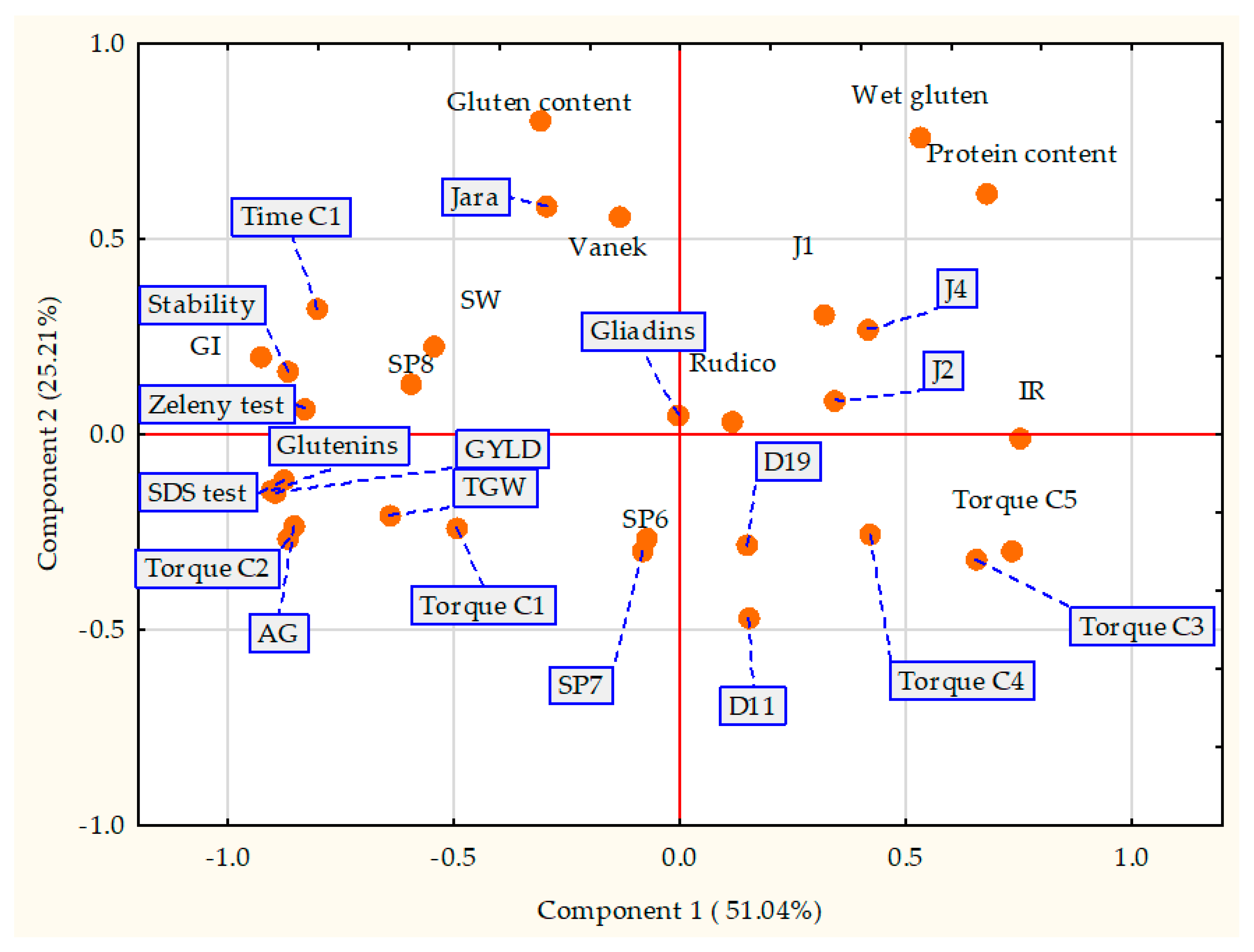
3.6. Principal Component Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- The World Wheat Book a History of Wheat Breeding, 1st ed.; William, J.; Angus, A.P.B. (Eds.) Lavoisier: Cachan, France, 2001; ISBN 1-898298-72-6. [Google Scholar]
- Zrcková, M.; Capouchová, I.; Paznocht, L.; Eliášová, M.; Dvořák, P.; Konvalina, P.; Janovská, D.; Orsák, M.; Bečková, L. Variation of the total content of polyphenols and phenolic acids in einkorn, emmer, spelt and common wheat grain as a function of genotype, wheat species and crop year. Plant Soil Environ. 2019, 65, 260–266. [Google Scholar] [CrossRef] [Green Version]
- Van Zeist, W.; Bakker-Heeres, J.A.H. Archaeobotanical studies in the Levant: I. Neolithic sites in the Damascus basin: Aswad, Ghoraife, Ramad. Palaeohistoria 2015, 24, 165–256. [Google Scholar]
- Crawford, D.J. Food: Tradition and change in Hellenistic Egypt. World Archaeol. 1979, 11, 136–146. [Google Scholar] [CrossRef]
- Arzani, A.; Ashraf, M. Cultivated Ancient Wheats (Triticum spp.): A Potential Source of Health-Beneficial Food Products. Compr. Rev. Food Sci. Food Saf. 2017, 16, 477–488. [Google Scholar] [CrossRef] [Green Version]
- Shewry, P.R.; Halford, N.G. Cereal seed storage proteins: Structures, properties and role in grain utilization. J. Exp. Bot. 2002, 53, 947–958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Branlard, G.; Dardevet, M.; Saccomano, R.; Lagoutte, F.; Gourdon, J. Genetic diversity of wheat storage proteins and bread wheat quality. Euphytica 2001, 119, 59–67. [Google Scholar] [CrossRef]
- Bradová, J.; Šašek, A. Diversity of gliadins and HMW glutenin subunits in Czech registered wheat varieties. Czech J. Genet. Plant Breed. 2012, 41, 160–163. [Google Scholar] [CrossRef] [Green Version]
- Švec, I.; Hrušková, M. The Mixolab parameters of composite wheat/hemp flour and their relation to quality features. LWT Food Sci. Technol. 2015, 60, 623–629. [Google Scholar] [CrossRef]
- Harati, H.; Bekes, F.; Howell, K.; Noonan, S.; Florides, C.; Beasley, J.; Diepeveen, D.; Appels, R. Signatures for torque variation in wheat dough structure are affected by enzymatic treatments and heating. Food Chem. 2020, 316, 126357. [Google Scholar] [CrossRef]
- Axford, D.; Mcdermott, E.; Redman, D. Small-Scale Tests of Bread-Making Quality. Milling Feed Fertil. 1978, 161, 18–20. [Google Scholar]
- Capouchová, I.; Petr, J.; Tlaskalová-Hogenová, H.; Michalík, I.; Faměra, O.; Urminská, D.; Tučková, L.; Knoblochová, H.; Borovská, D. Protein fractions of oats and possibilities of oat utilisation for patients with coeliac disease. Czech J. Food Sci. 2011, 22, 151–162. [Google Scholar] [CrossRef] [Green Version]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Payne, P.I.; Lawrence, G.J. Catalogue of alleles for the complex gene loci, Glu-A1, Glu-B1, and Glu-D1 which code for high-molecular-weight subunits of glutenin in hexaploid wheat. Cereal Res. Commun. 1983, 11, 29–35. [Google Scholar]
- Bradová, J.; Matějová, E. Comparison of the Results of SDS PAGE and Chip Electrophoresis of Wheat Storage Proteins. Chroma 2008, 67, 83–88. [Google Scholar] [CrossRef]
- Konvalina, P.; Grausgruber, H.; Tran Dang, K.; Vlasek, O.; Capouchova, I.; Sterba, Z.; Suchy, K.; Stolickova, M.; Kyptova, M.; Bernas, J.; et al. Rheological and Technological Quality of Minor Wheat Species and Common Wheat. In Wheat Improvement, Management and Utilization; Wanyera, R., Owuoche, J., Eds.; BoD–Books on Demand: Norderstedt, Germany, 2017; ISBN 978-953-51-3151-9. [Google Scholar]
- Rosell, C.M.; Collar, C.; Haros, M. Assessment of hydrocolloid effects on the thermo-mechanical properties of wheat using the Mixolab. Food Hydrocoll. 2007, 21, 452–462. [Google Scholar] [CrossRef]
- Ferrer, A.; Rosell, C.M. Rheological Behaviour of Whole Wheat Flour. In Proceedings of the IUFOST—13th World Congress of Food Science & Technology: Food Is Life, Nantes, France, 17–21 September 2006. [Google Scholar] [CrossRef]
- Schmiele, M.; Ferrari Felisberto, M.H.; Pedrosa Silva Clerici, M.T.; Chang, Y.K. MixolabTM for rheological evaluation of wheat flour partially replaced by soy protein hydrolysate and fructooligosaccharides for bread production. LWT Food Sci. Technol. 2017, 76, 259–269. [Google Scholar] [CrossRef]
- Shewry, P.R.; Halford, N.G.; Tatham, A.S. High molecular weight subunits of wheat glutenin. J. Cereal Sci. 1992, 15, 105–120. [Google Scholar] [CrossRef]
- Shewry, P.R.; Tatham, A.S. The prolamin storage proteins of cereal seeds: Structure and evolution. Biochem. J. 1990, 267, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Gliadin and Glutenin: The Unique Balance of Wheat Quality; Wrigley, C.W.; Békés, F.; Bushuk, W. (Eds.) AACC International: St. Paul, MN, USA, 2006; ISBN 978-1-891127-51-9. [Google Scholar]
- Gianibelli, M.C.; Larroque, O.R.; MacRitchie, F.; Wrigley, C.W. Biochemical, Genetic, and Molecular Characterization of Wheat Glutenin and Its Component Subunits. Cereal Chem. 2001, 78, 635–646. [Google Scholar] [CrossRef]
- Wang, Y.G.; Khan, K.; Hareland, G.; Nygard, G. Quantitative Glutenin Composition from Gel Electrophoresis of Flour Mill Streams and Relationship to Breadmaking Quality. Cereal Chem. 2006, 83, 293–299. [Google Scholar] [CrossRef]
- Payne, P.I.; Corfield, K.G. Subunit composition of wheat glutenin proteins, isolated by gel filtration in a dissociating medium. Planta 1979, 145, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Payne, P.I.; Nightingale, M.A.; Krattiger, A.F.; Holt, L.M. The relationship between HMW glutenin subunit composition and the bread-making quality of British-grown wheat varieties. J. Sci. Food Agric. 1987, 40, 51–56. [Google Scholar] [CrossRef]
- Banfalvi, A.; Nemeth, R.; Bagdi, A.; Gergely, S.; Rakszegi, M.; Bedo, Z.; Lang, L.; Vida, G.; Tomoskozi, S. A novel approach to the characterization of old wheat (Triticum aestivum L.) varieties by complex rheological analysis. J. Sci. Food Agric. 2020, 100, 4409–4417. [Google Scholar] [CrossRef] [PubMed]
- Koksel, H.; Kahraman, K.; Sanal, T.; Ozay, D.S.; Dubat, A. Potential Utilization of Mixolab for Quality Evaluation of Bread Wheat Genotypes. Cereal Chem. 2009, 86, 522–526. [Google Scholar] [CrossRef]
- Codina, G.G.; Mironeasa, S.; Bordei, D.; Leahu, A. Mixolab versus Alveograph and Falling Number. Czech J. Food Sci. 2010, 28, 185–191. [Google Scholar] [CrossRef] [Green Version]
- McKevith, B. Nutritional aspects of cereals. Nutr. Bull. 2004, 29, 111–142. [Google Scholar] [CrossRef]
- Grausgruber, H.; Sailer, C.; Ghambashidze, G.; Bolyos, L.; Ruckenbauer, P. Genetic variation in agronomic and qualitative traits of ancient wheats. In Proceedings of the 17th EUCARPIA General Congress, Tulln, Austria, 8–11 September 2004; pp. 19–22. [Google Scholar]
- Kiszonas, A.; Morris, C.F. Wheat Breeding for Quality: A Historical Review. Cereal Chem. J. 2017, 95, 17–34. [Google Scholar] [CrossRef] [Green Version]
- Konvalina, P.; Capouchova, I.; Stehno, Z.; Moudry, J., Jr.; Moudry, J. Fusarium Identification by PCR and DON Content in Grain of Ancient Wheat. J. Food Agric. Environ. 2009, 9, 321–325. Available online: http://orgprints.org/20762/ (accessed on 3 May 2012).
- Konvalina, P.; Stehno, Z.; Capouchová, I.; Zechner, E.; Berger, S.; Grausgruber, H.; Janovská, D.; Moudrý, J. Differences in grain/straw ratio, protein content and yield in landraces and modern varieties of different wheat species under organic farming. Euphytica 2014, 199, 31–40. [Google Scholar] [CrossRef]
- Goesaert, H.; Brijs, K.; Veraverbeke, W.S.; Courtin, C.M.; Gebruers, K.; Delcour, J.A. Wheat flour constituents: How they impact bread quality, and how to impact their functionality. Trends Food Sci. Technol. 2005, 16, 12–30. [Google Scholar] [CrossRef]
- Šramková, Z.; Gregová, E.; Šturdík, E. Genetic improvement of wheat—A review. Nova Biotechnol. 2009, 9, 27–51. [Google Scholar]
- Jiang, P.; Xue, J.; Duan, L.; Gu, Y.; Mu, J.; Han, S.; Chen, L.; Li, Y.; Ma, W.; Yan, Y.; et al. Effects of high-molecular-weight glutenin subunit combination in common wheat on the quality of crumb structure. J. Sci. Food Agric. 2019, 99, 1501–1508. [Google Scholar] [CrossRef] [PubMed]
- Vasil, I.K.; Anderson, O.D. Genetic engineering of wheat gluten. Trends Plant Sci. 1997, 2, 292–297. [Google Scholar] [CrossRef]
- Stehno, Z.; Bradová, J.; Dotlačil, L.; Konvalina, P. Landraces and obsolete cultivars of minor wheat species in the Czech collection of wheat genetic resources. Czech J. Genet. Plant Breed. 2010, 46, 100–105. [Google Scholar] [CrossRef] [Green Version]
- Dhaka, V.; Gulia, N. Application of Mixolab to Assess the Bread Making Quality of Wheat Varieties. J. Food Process. Technol. 2012, 1. [Google Scholar] [CrossRef]
- Khatkar, B. Effects of high Mr glutenin subunits on dynamic rheological properties and bread making qualities of wheat gluten. J. Food Sci. Technol. 2006, 43, 382–387. [Google Scholar]





| Species | Variety | HMW-GSs | ||
|---|---|---|---|---|
| Glu-A1 | Glu-B1 | Glu-1D | ||
| Einkorn | J1 | 0 | ||
| J2 | 0 | |||
| J4 | 0 | |||
| J6 | 0 | |||
| Emmer | D11 | 1 | (7 + 8) | |
| D12 | 1 | (7 + 8) | ||
| D13 | 2 * | 6 (21) | ||
| D14 | 0 | 0 | ||
| D17 | 1 | 6 (21) | ||
| D18 | 1 | (7 + unk) | ||
| D19 | 1 | (7+8) | ||
| RU | 1 | (7+8) | ||
| Spelt | SP1 | 1 | 7+8 | 2+12 |
| SP2 | 1 | 6 + 8 | 2 + 12 | |
| SP4 | 1 | 6 + 8 | 2 + 12 | |
| SP6 | 1 | 7 + 8 | 2 + 12 | |
| SP7 | 1 | 7 + 8 | 2 + 12 | |
| SP8 | 1 | 7 + 8 | 2 + 12 | |
| SP9 | 1 | 6 + 8 | 2 + 12 | |
| Landraces of bread wheat | P1 | 2 * | 7 | 2 + 12 |
| P2 | 0 | 7 + 9 | 5 + 10 | |
| P3 | 0 | 7 + 9 | 5 + 10 | |
| P4 | 0 | 7 + 9 | 5 + 10 | |
| Bread wheat | JR | 2 * | 7 + 9 | 5 + 10 |
| VK | 1 | 7 + 9 | 5 + 10 | |
| SWK | 1 | 14 + 15 | 5 + 10 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, K.D.; Konvalina, P.; Capouchova, I.; Janovska, D.; Lacko-Bartosova, M.; Kopecky, M.; Tran, P.X.T. Comparative Study on Protein Quality and Rheological Behavior of Different Wheat Species. Agronomy 2020, 10, 1763. https://doi.org/10.3390/agronomy10111763
Tran KD, Konvalina P, Capouchova I, Janovska D, Lacko-Bartosova M, Kopecky M, Tran PXT. Comparative Study on Protein Quality and Rheological Behavior of Different Wheat Species. Agronomy. 2020; 10(11):1763. https://doi.org/10.3390/agronomy10111763
Chicago/Turabian StyleTran, Khoa Dang, Petr Konvalina, Ivana Capouchova, Dagmar Janovska, Magdalena Lacko-Bartosova, Marek Kopecky, and Phuong Xuan Thi Tran. 2020. "Comparative Study on Protein Quality and Rheological Behavior of Different Wheat Species" Agronomy 10, no. 11: 1763. https://doi.org/10.3390/agronomy10111763
APA StyleTran, K. D., Konvalina, P., Capouchova, I., Janovska, D., Lacko-Bartosova, M., Kopecky, M., & Tran, P. X. T. (2020). Comparative Study on Protein Quality and Rheological Behavior of Different Wheat Species. Agronomy, 10(11), 1763. https://doi.org/10.3390/agronomy10111763






