Assessment of Agro-Morphologic Performance, Genetic Parameters and Clustering Pattern of Newly Developed Blast Resistant Rice Lines Tested in Four Environments
Abstract
:1. Introduction
2. Materials and Methods
2.1. Planting Materials
2.2. Experimental Environments
2.3. Experimental Scheme and Cultural Practices
2.4. Data Collection
2.5. Data Analysis
| Where: = phenotypic variance; X= mean of the trait | |
| Where; = genotypic variance; X = mean of the trait | |
| Where: = genotypic variance; = phenotypic variance | |
| Where: K = constant that represents the selection intensity (when k is 5% the value is 2.06); = standard deviation of phenotypic variance; = heritability in a broad sense |
3. Results
3.1. Agro-Morphologic Traits, Genotype and G×E Interactions
3.2. Genotypic and Phenotypic Coefficient of Variability (GCV and PCV)
3.3. Heritability and Genetic Advance
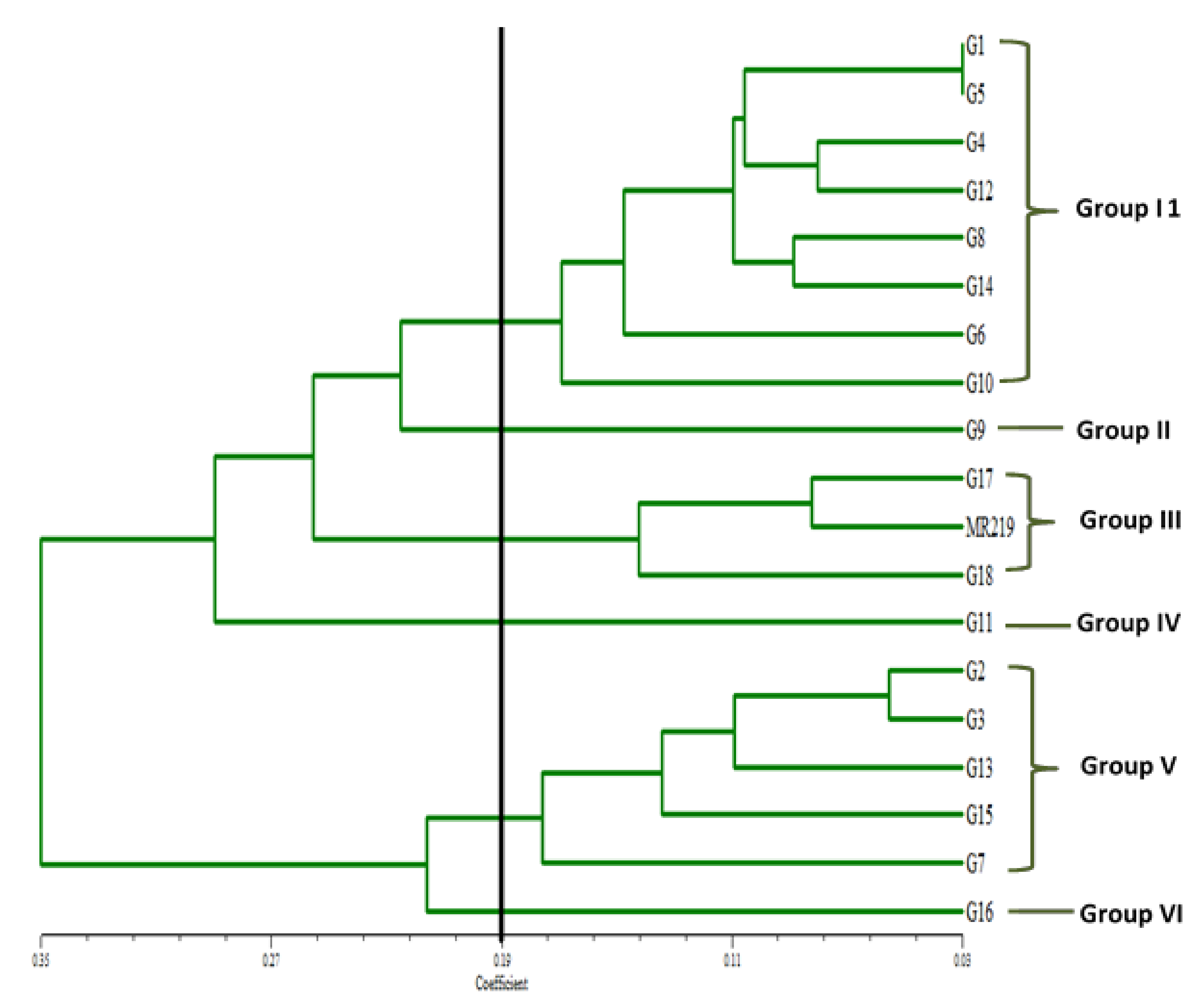
3.4. Correlation and Cluster Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chukwu, S.C.; Rafii, M.Y.; Ramlee, S.I.; Ismail, S.I.; Hasan, M.M.; Oladosu, Y.A.; Magaji, U.G.; Akos, I.; Olalekan, K.K. Bacterial leaf blight resistance in rice: A review of conventional breeding to molecular approach. Mol. Biol. Rep. 2019, 46, 1519–1532. [Google Scholar] [CrossRef]
- Prasad, R.; Shivay, Y.S.; Kumar, D. Current Status, Challenges, and Opportunities in Rice Production. In Rice Production Worldwide; Chauhan, B.S., Jabran, K., Mahajan, G., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–32. [Google Scholar]
- Conforti, P. Looking ahead in World Food and Agriculture: Perspectives to 2050; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2011. [Google Scholar]
- Maclean, J.; Hardy, B.; Hettel, G. Rice Almanac: Source Book for One of the Most Important Economic Activities on Earth; IRRI: Los Baños, Philippines, 2013. [Google Scholar]
- Vengedasalam, D.; Harris, M.; MacAulay, G. Malaysian rice trade and government interventions. In Proceedings of the 55th Annual Conference of the Australian Agricultural and Resource Economics Society, Melbourne, VI, Australia, 8–11 February 2011. [Google Scholar]
- Ramli, N.N.; Shamsudin, M.N.; Mohamed, Z.; Radam, A. Impact of price support policy on Malaysian rice industry. In Proceedings of the International Annual Symposium on Sustainability Science and Management, University Malaysia Terengganu (UMT), Terengganu, Malaysia, 9–11 July 2012. [Google Scholar]
- USDA. World Agricultural Production; Foreign Agricultural Service Circular Series WAP 08-17; USDA: Washington, DC, USA, 2017. [Google Scholar]
- Department of Statistics Malaysia. Supply and Utilization Accounts Selected Agricultural Commodities, Malaysia 2012–2016; Department of Statistics Malaysia: Sandakan, Malaysia, 2017. [Google Scholar]
- Arshad, F.M.; Alias, E.F.; Noh, K.M.; Tasrif, M. Food security: Self-sufficiency of rice in Malaysia. Int. J. Manag. Stud. 2011, 18, 83–100. [Google Scholar]
- Chukwu, S.C.; Rafii, M.Y.; Ramlee, S.I.; Ismail, S.I.; Oladosu, Y.; Kolapo, K.; Ahmed, M. Marker-assisted introgression of multiple resistance genes confers broad spectrum resistance against bacterial leaf blight and blast diseases in Putra-1 rice variety. Agronomy 2020, 10, 42. [Google Scholar] [CrossRef] [Green Version]
- Divya, B.; Biswas, A.; Robin, S.; Rabindran, R.; Joel, A.J. Gene interactions and genetics of blast resistance and yield attributes in rice (Oryza sativa L.). J. Genet. 2014, 93, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Tanweer, F.A.; Rafii, M.Y.; Sijam, K.; Rahim, H.A.; Ahmed, F.; Latif, M.A. Current advance methods for the identification of blast resistance genes in rice. Comptesrendusbiologies 2015, 338, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Koutroubas, S.D.; Katsantonis, D.; Ntanos, D.A.; Lupotto, E. Blast disease influence on agronomic and quality traits of rice varieties under Mediterranean conditions. Turk. J. Agricult. For. 2009, 33, 487–494. [Google Scholar]
- Nasruddin, A.; Amin, N. Effects of Cultivar, Planting Period, and Fungicide Usage on Rice Blast Infection Levels and Crop Yield. J. Agricult. Sci. 2012, 5, 160. [Google Scholar] [CrossRef] [Green Version]
- Miah, G.; Rafii, M.Y.; Ismail, M.R.; Puteh, A.B.; Rahim, H.A.; Asfaliza, R.; Latif, M.A. Blast resistance in rice: A review of conventional breeding to molecular approaches. Mol. Biol. Rep. 2013, 40, 2369–2388. [Google Scholar] [CrossRef]
- Chukwu, S.C.; Rafii, M.Y.; Ramlee, S.I.; Ismail, S.I.; Oladosu, Y.; Okporie, E.; Jalloh, M. Marker-assisted selection and gene pyramiding for resistance to bacterial leaf blight disease of rice (Oryza sativa L.). Biotechnol. Biotechnol. Equip. 2019, 33, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Miah, G.; Rafii, M.Y.; Ismail, M.R.; Puteh, A.B.; Rahim, H.A.; Latif, M.A. Recurrent parent genome recovery analysis in a marker-assisted backcrossing program of rice (Oryza sativa L.). Comptesrendusbiologies 2015, 338, 83–94. [Google Scholar] [CrossRef]
- Tanweer, F.A.; Rafii, M.Y.; Sijam, K.; Rahim, H.A.; Ahmed, F.; Ashkani, S.; Latif, M.A. Introgression of blast resistance genes (putative Pi-b and Pi-kh) into elite rice cultivar MR219 through marker-assisted selection. Front. Plant Sci. 2015, 6, 1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jayawardana, W.A.D.; Jayasekera, G.A.U.; Wijesundera, R.L.C.; Dissanayake, D.M.; Sooriyapathirana, S.D.S.S.; Weebadde, C.K.; Hettige, P. Evaluation of DNA markers linked to blast resistant genes, Pikh, Pit (p), and Pita, for parental selection in Sri Lankan rice breeding. Trop. Agricult. Res. 2015, 26, 82–93. [Google Scholar] [CrossRef] [Green Version]
- Mackill, D.J.; Bonman, J.M. Inheritance of blast resistance in near-isogenic lines of rice. Phytopathology 1992, 82, 746–749. [Google Scholar] [CrossRef]
- Falconer, D.S. Introduction to Quantative Genetics; Longman: Harlow, UK, 1975. [Google Scholar]
- Azizi, P.; Rafii, M.Y.; Maziah, M.; Abdullah, S.N.; Hanafi, M.M.; Latif, M.A.; Rashid, A.A.; Sahebi, M. Understanding the shoot apical meristem regulation: A study of the phytohormones, auxin and cytokinin, in rice. Mech. Dev. 2015, 135, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.S. Breeding: Genotype-by-Environment Interaction. In Encyclopedia of Plant and Crop Science; Goodman, R.M., Ed.; Marcel-Dekker: New York, NY, USA; Taylor & Francis: Park Drive, UK, 2004; pp. 218–221. [Google Scholar]
- Inthapanya, P.S.; Sihavong, P.; Sihathep, V.; Chanphengsay, M.; Fukai, S.; Basnayake, J. Genotypic performance under fertilized and non-fertilised conditions in rainfed lowland rice. Field Crops Res. 2000, 65, 1–14. [Google Scholar] [CrossRef]
- Wade, L.J.; McLaren, C.G.; Quintana, L.; Harnpichitvitaya, D.; Rajatasereekul, S.; Sarawgi, A.K.; Siopongco, J. Genotype by environment interactions across diverse rainfed lowland rice environments. Field Crops Res. 1999, 64, 35–50. [Google Scholar] [CrossRef]
- Cooper, M.; Byth, D.E.; Hammer, G.L. Understanding plant adaptation to achieve systematic applied crop improvement—A fundamental challenge. In Plant Adaptation and Crop Improvement; Centre for Agriculture and Bioscience International: Wallingford, UK, 1996; pp. 5–23. [Google Scholar]
- Oladosu, Y.; Rafii, M.Y.; Abdullah, N.; Magaji, U.; Miah, G.; Hussin, G.; Ramli, A. Genotype × Environment Interaction and Stability Analyses of Yield and Yield Components of Established and Mutant Rice Genotypes Tested in Multiple Locations in Malaysia. Acta Agricult. Scand. Sect. B Soil Plant Sci. 2017, 67, 590–606. [Google Scholar] [CrossRef]
- Xavier, A.; Jarquin, D.; Howard, R.; Ramasubramanian, V.; Specht, J.E.; Graef, G.L.; Nelson, R. Genome-Wide Analysis of Grain Yield Stability and Environmental Interactions in a Multiparental Soybean Population. G3 Genes Genomes Genet. 2017, 8, 519–529. [Google Scholar] [CrossRef] [Green Version]
- Hashemi, F.G.; Rafii, M.Y.; Ismail, M.R.; Mahmud, T.M.; Rahim, H.A.; Asfaliza, R.; Malek, M.A.; Latif, M.A. Biochemical, genetic and molecular advances of fragrance character in rice. Crit. Rev. Plant Sci. 2013, 32, 445–457. [Google Scholar] [CrossRef] [Green Version]
- Tariku, S.; Lakew, T.; Bitew, M.; Asfaw, M. Genotype by Environment Interaction and Grain Yield Stability Analysis of Rice (Oryza sativa L.) Genotypes Evaluated in North Western Ethiopia. Net J. Agricult. Sci. 2013, 1, 10–16. [Google Scholar]
- IRRI. Standard Evaluation System for Rice, 5th ed.; International Rice Research Institute: Manila, Philippines, 2013; p. 55. [Google Scholar]
- Sivasubramanian, S.; Menon, P.M. Genotypic and phenotypic variability in rice. Madras Agric. J. 1973, 60, 1093–1096. [Google Scholar]
- Johnson, H.W.; Robinson, H.F.; Comstock, R.E. Genotypic and Phenotypic Correlations in Soybeans and Their Implications in Selection 1. Agron. J. 1955, 47, 477–483. [Google Scholar] [CrossRef]
- Shakeel, A.; Abid, H.; Hakoomat, A.; Ashfaq, A. Transplanted fine rice (Oryza sativa L.) productivity as affected by plant density and irrigation regimes. Int. J. Agricult. Biol. 2005, 7, 445–447. [Google Scholar]
- Peng, S.; Khush, G.S.; Cassman, K.G. Evolution of the new plant ideotype for increased yield potential. In Breaking the Yield Barrier Proceedings of a Workshop on Rice Yield Potential in Favorable Environments; International Rice Research Institute: Los Banos, Philippines, 1994; pp. 5–20. [Google Scholar]
- Ranawake, A.L.; Amarasinghe, U.G.S. Relationship of yield and yield related traits of some traditional rice cultivars in Sri Lanka as described by correlation analysis. J. Sci. Res. Rep. 2014, 3, 2395–2403. [Google Scholar] [CrossRef]
- Oladosu, Y.; Rafii, M.Y.; Abdullah, N.; Abdul Malek, M.; Rahim, H.A.; Hussin, G.; Kareem, I. Genetic variability and selection criteria in rice mutant lines as revealed by quantitative traits. Sci. World J. 2014, 2014, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Zhang, J. Grain-filling problem in ‘super’ rice. J. Exp. Bot. 2009, 61, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Oladosu, Y.; Rafii, M.Y.; Magaji, U.; Abdullah, N.; Miah, G.; Chukwu, S.C.; Kareem, I. Genotypic and phenotypic relationship among yield components in rice under tropical conditions. Biomed. Res. Int. 2018, 2018, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Konate, A.K.; Zongo, A.; Kam, H.; Sanni, A.; Audebert, A. Genetic variability and correlation analysis of rice (Oryza sativa L.) inbred lines based on agro-morphological traits. Afr. J. Agricult. Res. 2016, 11, 3340–3346. [Google Scholar]
- Kumar, P.; Sao, A.; Salam, J.L.; Kanwar, R.R.; Poonam, K.; Gupta, A.K.; Kar, S. Formulating phenological equations for rainfed upland rice in Bastar plateau and assessment of genotype x environment interaction. Pak. J. Bot 2016, 48, 1609–1618. [Google Scholar]
- Begum, M.; Hossain, M.A.; Hossain, F.M.M.; Hasan, A.K. Genetic variability for grain yield and yield associated traits in transplant Aman rice (Oryza sativa L.). Res. Agricult. Livestock Fish. 2015, 2, 207–213. [Google Scholar] [CrossRef]
- Chouhan, S.K.; Singh, A.K.; Singh, A.; Singh, S.P.; Singh, N.K.; Singh, P.K. Agro-morphological diversity in wild rice accessions of Eastern Indo-Gangetic region of India. Bangladesh J. Bot. 2015, 43, 337–344. [Google Scholar] [CrossRef]
- Sajid, M.; Khan, S.A.; Khurshid, H.; Iqbal, J.; Muhammad, A.; Saleem, N.; Shah, S.M.A. Characterization of rice (Oryza Sativa L.) germplasm through various agro-morphological traits. Sci. Agricult. 2015, 9, 83–88. [Google Scholar]
- Islam, M.Z.; Khalequzzaman, M.; Bashar, M.K.; Ivy, N.A.; Haque, M.M.; Mian, M.A.K. Variability assessment of aromatic and fine rice germplasm in Bangladesh based on quantitative traits. Sci. World J. 2016, 2016, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosagoudar, G.N.; Kovi, B.S.; Sheela, H.K. Genetic Variability, Correlation and Leaf Blast Resistance Studies in Early Maturing Rice Genotypes under Hilly Conditions. Int. J. Curr. Microbiol. App. Sci. 2017, 6, 3451–3459. [Google Scholar] [CrossRef]
- Tuhina-Khatun, M.; Hanafi, M.M.; Rafii Yusop, M.; Wong, M.Y.; Salleh, F.M.; Ferdous, J. Genetic variation, heritability, and diversity analysis of upland rice (Oryza sativa L.) genotypes based on quantitative traits. BioMed Res. Int. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Sohrabi, M.; Rafii, M.Y.; Hanafi, M.M.; Siti Nor Akmar, A.; Latif, M.A. Genetic diversity of upland rice germplasm in Malaysia based on quantitative traits. Sci. World J. 2012, 2012, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Osman, K.A.; Mustafa, A.M.; Ali, F.; Yonglain, Z.; Fazhan, Q. Genetic variability for yield and related attributes of upland rice genotypes in semi arid zone (Sudan). Afr. J. Agricult. Res. 2012, 7, 4613–4619. [Google Scholar] [CrossRef]
- Habib, S.H.; Bashar, M.K.; Khalequzzaman, M.; Ahmed, M.S.; Rashid, E.S.M.H. Genetic analysis and morpho-physiological selection criteria for traditional Biroin Bangladesh rice germplasms. J. Biol. Sci. 2005, 5, 315–318. [Google Scholar]
- Anis, G.; El-Sabagh, A.; Ghareb, A.; El-Rewainy, I. Evaluation of Promising Lines in Rice (Oryza sativa L.) to Agronomic and Genetic Performance under Egyptian Conditions. Int. J. Agron. Agricult. Res. 2016, 8, 52–57. [Google Scholar]
- Nishanth, G.K.; Dushyanthakumar, B.M.; Gangaprasad, S.; Gowda, T.H.; Nataraju, S.P.; Shashidhar, H.E. Screening and Genetic Variability Studies in Submergence Tolerance Rice Germplasm Lines Under Flood Prone Lowlands of Hill Zone of Karnataka, India. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 1254–1260. [Google Scholar] [CrossRef] [Green Version]
- Hosagoudar, G.N.; Kovi, B.S. Evaluation of Advanced Varietal Rice Genotypes for Leaf Blast Reaction, Genotypic Performance and Correlation Studies. Int. J. Curr. Microbiol. Appl. Sci. 2018, 7, 75–81. [Google Scholar] [CrossRef] [Green Version]
- Hasan, M.M.; Yusop, M.R.; Ismail, M.R.; Mahmood, M.; Rahim, H.A.; Latif, M.A. Performance of yield and yield contributing characteristics of BC2F3 population with addition of blast resistant gene. Ciênc. Agrotecnol. 2015, 39, 463–476. [Google Scholar] [CrossRef]
- Srujana, G.; Suresh, B.G.; Lavanya, G.R.; Ram, B.J.; Sumanth, V. Studies on Genetic Variability, Heritability and Genetic Advance for Yield and Quality Components in Rice (Oryza sativa L.). J. Pharmacogn. Phytochem. 2017, 6, 564–566. [Google Scholar]
- Burton, G.W.; Devane, E.H. Estimating heritability in tall fescue (Festuca arundinacea) from replicated clonal material 1. Agron. J. 1953, 45, 478–481. [Google Scholar] [CrossRef]
- Kole, P.C.; Chakraborty, N.R.; Bhat, J.S. Analysis of variability, correlation and path coefficients in induced mutants of aromatic non-basmati rice. Trop. Agricult. Res. Ext. 2010, 11, 60. [Google Scholar] [CrossRef] [Green Version]
- Akinwale, M.G.; Gregorio, G.; Nwilene, F.; Akinyele, B.O.; Ogunbayo, S.A.; Odiyi, A.C. Heritability and Correlation Coefficient Analysis for Yield and Its Components in Rice (Oryza sativa L.). Afr. J. Plant Sci. 2011, 5, 207–212. [Google Scholar]
- Immanuel, S.C.; Pothiraj, N.; Thiyagarajan, K.; Bharathi, M.; Rabindran, R. Genetic parameters of variability, correlation and pathcoefficient studies for grain yield and other yield attributes among rice blast disease resistant genotypes of rice (Oryza sativa L.). Afr. J. Biotechnol. 2011, 10, 3322–3334. [Google Scholar] [CrossRef] [Green Version]
- Govintharaj, P.; Shalini, T.; Manonmani, S.; Robin, S. Estimates of genetic variability, heritability and genetic advance for blast resistance gene introgressed segregating population in rice. Int. J. Curr. Microbiol. App. Sci. 2016, 5, 672–677. [Google Scholar] [CrossRef] [Green Version]
- Chukwu, S.C.; Ekwu, L.G.; Onyishi, G.C.; Okporie, E.O.; Obi, I.U. Correlation between agronomic and chemical characteristics of maize (Zea mays L.) genotypes after two years of mass selection. Int. J. Sci. Res. 2015, 4, 1708–1712. [Google Scholar]
- Kumar, V.; Singh, D. Association Studies for Yield and Its Component Traits in Basmati Genotypes of Himachal Pradesh, India. Int. J. Curr. Microbiol. App. Sci. 2018, 7, 1243–1249. [Google Scholar] [CrossRef] [Green Version]
- Sowmiya, C.A.; Venkatesan, M. Studies on Correlation and Path Coefficient Analysis in Rice (Oryza sativa L.). Int. J. Curr. Microbiol. App. Sci. 2017, 6, 1757–1763. [Google Scholar] [CrossRef] [Green Version]
- Sarawagi, A.K.; Ojha, G.C.; Koshta, N.; Pachauri, A. Genetic Divergence and Association Study for Grain Yield in Rice (Oryza sativa L.) Germplasm Accessions. Ecoscan 2015, 9, 217–223. [Google Scholar]
- Ahmadikhah, A.; Nasrollanejad, S.; Alishah, O. Quantitative studies for investigating variation and its effect on heterosis of rice. Int. J. Plant Prod. 2008, 2, 297–308. [Google Scholar]
| Code | Planting Period | Location | Altitude (m) | Av. Temp. Min– Max (°C) | Av. Humidity (%) | Rainfall (Mean) |
|---|---|---|---|---|---|---|
| EN1 | September 2015–January 2016 | 3° 25’N 101° 10’E | 3 | 23–31 | 83 | 782.4 (195.6) |
| EN2 | February–June 2016 | 3° 25’N 101° 10’E | 3 | 25–37 | 65 | 482.7 (120.7) |
| EN3 | December 2016–March 2017 | 5° 59’N 100° 24’E | 18 | 25–38 | 63 | 486.9 (121.7) |
| EN4 | May–September 2017 | 3° 02’N 101° 42’E | 32 | 24–38 | 67 | 623.4 (115.9) |
| SOV | Blocks (Environment) | Genotypes (G) | Environments (S) | G×S | Error | |
|---|---|---|---|---|---|---|
| DF | 8 | 18 | 3 | 54 | 144 | |
| DTF | MS | 18.34** | 17.78* | 149.97** | 9.77** | 3.94 |
| TSS (%) | 9.18 | 8.9 | 75.06 | 4.89 | 1.97 | |
| DTM | MS | 133.93** | 19.96ns | 856.91** | 15.20** | 8.41 |
| TSS (%) | 12.95 | 1.93 | 82.84 | 1.47 | 0.81 | |
| PH | MS | 127.44** | 60.67** | 12,680.89** | 22.82** | 12.11 |
| TSS (%) | 0.99 | 0.47 | 98.27 | 0.18 | 0.09 | |
| NTH | MS | 95.95** | 105.02** | 726.42** | 11.81ns | 23.13 |
| TSS (%) | 9.97 | 10.91 | 75.49 | 1.23 | 2.4 | |
| NPH | MS | 68.43** | 78.49** | 478.86** | 12.40ns | 15.13 |
| TSS (%) | 10.47 | 12.01 | 73.3 | 1.9 | 2.32 | |
| PL | MS | 1.54** | 4.67** | 97.22** | 1.29** | 0.53 |
| TSS (%) | 1.46 | 4.44 | 92.37 | 1.23 | 0.5 | |
| FG | MS | 1791.21** | 2877.37ns | 38,887.19** | 1743.21** | 608.58 |
| TSS (%) | 3.9 | 6.27 | 84.71 | 3.8 | 1.32 | |
| UFG | MS | 1979.64** | 700.88* | 45,613.77** | 352.45** | 212.92 |
| TSS (%) | 4.05 | 1.43 | 93.36 | 0.72 | 0.44 | |
| TG | MS | 1384.19* | 1595.14ns | 5975.58** | 1327.15** | 686.72 |
| TSS (%) | 12.62 | 14.54 | 54.48 | 12.1 | 6.26 | |
| PFG | MS | 280.32** | 145.62* | 8234.20** | 78.53** | 34.06 |
| TSS (%) | 3.2 | 1.66 | 93.86 | 0.9 | 0.38 | |
| TGW | MS | 10.46** | 6.97* | 17.05** | 3.44** | 1.56 |
| TSS (%) | 26.49 | 17.66 | 43.19 | 8.71 | 3.95 | |
| TW | MS | 449.08** | 380.11** | 594.18** | 138.06** | 85.08 |
| TSS (%) | 27.27 | 23.09 | 36.09 | 8.38 | 5.17 | |
| YLD | MS | 11.49** | 9.73** | 15.19** | 3.53** | 2.18 |
| TSS (%) | 27.28 | 23.1 | 36.06 | 8.38 | 5.18 |
| Genotypes | DTF (Day) | DTM (Day) | PH (cm) | NTH (no) | NPH (no) | PL (cm) | FG (no) | UFG (no) | TG (no) | PFG (%) | TGW (g) | TW (g) | YLD(t/ha) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G1 | 72.25 ab | 104.25 | 98.52 ab | 19.92 de | 17.00 de | 24.30 abc | 192.33 | 47.91 | 240.25 | 79.35 | 23.21 ab | 38.60 ab | 6.17 ab |
| G2 | 72.00 ab | 103.17 | 100.72 ab | 19.83 e | 17.50 de | 24.81abc | 178.67 | 55.41 | 234.08 | 77.29 | 22.89 ab | 33.05b | 5.29 b |
| G3 | 73.50 ab | 105.33 | 100.93 ab | 22.17 bcde | 18.58 cde | 24.85abc | 176.83 | 55.75 | 232.59 | 76.06 | 23.93 ab | 34.30 b | 5.49 b |
| G4 | 72.50 ab | 104.75 | 100.12 ab | 22.25 bcde | 18.25 cde | 24.86abc | 186.58 | 48.25 | 234.83 | 78.39 | 24.19 ab | 33.83 b | 5.41 b |
| G5 | 73.00 ab | 105 | 99.32 ab | 20.75 cde | 18.67 cde | 24.38abc | 194.25 | 49.33 | 243.58 | 79.97 | 23.85 ab | 39.95 ab | 6.39 ab |
| G6 | 71.67 ab | 104.17 | 101.73 a | 19.75 e | 16.58 e | 25.15 a | 193.42 | 40.75 | 234.17 | 81.91 | 23.22 ab | 37.80 ab | 6.05 ab |
| G7 | 70.92 b | 101 | 99.55 ab | 22.75bcde | 19.83bcde | 24.53abc | 165.92 | 64.33 | 230.25 | 72.49 | 23.65 ab | 33.27 b | 5.32 b |
| G8 | 72.33 ab | 103.83 | 98.93 ab | 23.17bcde | 20.25bcde | 24.67abc | 185.08 | 44.75 | 229.83 | 80.13 | 23.07 ab | 38.12 ab | 6.10 ab |
| G9 | 74.25 ab | 106.17 | 97.25 ab | 25.17 ab | 20.75abcde | 23.36abc | 169.42 | 48.17 | 217.59 | 79.14 | 22.54 ab | 35.16 b | 5.63 b |
| G10 | 71.17 b | 102.75 | 94.35 b | 22.08bcde | 19.83bcde | 23.53abc | 195.33 | 51.92 | 247.25 | 79.71 | 22.75 ab | 36.82 ab | 5.89 ab |
| G11 | 73.92 ab | 105.58 | 98.61 ab | 22.83bcde | 19.92bcde | 25.15 a | 226.58 | 43.33 | 269.92 | 82.92 | 22.49 ab | 41.00 ab | 6.56 ab |
| G12 | 75.92 a | 106.25 | 98.05 ab | 21.25 cde | 18.25 cde | 24.36abc | 191.92 | 46.5 | 238.42 | 80.47 | 23.00 ab | 34.21b | 5.47 b |
| G13 | 73.25 ab | 103.83 | 99.04 ab | 22.83bcde | 19.00 cde | 23.37bc | 175.17 | 60.5 | 235.67 | 74.42 | 23.06 ab | 33.44 b | 5.35 b |
| G14 | 72.00 ab | 103.58 | 94.57 ab | 24.67abcde | 21.33abcde | 23.48abc | 178.67 | 47.08 | 225.75 | 79.23 | 24.04 ab | 40.91 ab | 6.55 ab |
| G15 | 71.58 ab | 103.58 | 94.28 b | 26.92 ab | 23.25 abc | 23.17 c | 170.58 | 56.42 | 227 | 74.75 | 21.63 b | 33.76 b | 5.40 b |
| G16 | 73.67 ab | 105.17 | 97.12 ab | 21.17 cde | 17.67 de | 24.48abc | 156.92 | 58.59 | 215.5 | 72.97 | 23.01 ab | 31.01 b | 4.96 b |
| G17 | 73.83 ab | 105.33 | 98.79 ab | 25.08 abcd | 22.08abcd | 24.62abc | 194.42 | 39.25 | 233.66 | 83.61 | 23.08 ab | 43.95 ab | 7.03 ab |
| G18 | 73.25 ab | 104.75 | 100.48 ab | 29.58 a | 25.00 ab | 25.06 ab | 199.92 | 38.33 | 238.25 | 83.89 | 24.78 a | 54.23 a | 8.68 a |
| MR219 | 72.83 ab | 105.5 | 101.10 ab | 29.67 a | 25.67 a | 24.44abc | 193.17 | 38.83 | 232 | 82.83 | 24.42 ab | 45.79 ab | 7.33 ab |
| Mean | 72.83 | 104.42 | 98.6 | 23.25 | 19.97 | 24.36 | 185.53 | 49.23 | 234.77 | 78.92 | 23.31 | 37.85 | 6.06 |
| HSD(p = 0.05) | 4.31 | 5.88 | 7.2 | 5.18 | 5.31 | 1.71 | 62.97 | 28.31 | 54.94 | 13.37 | 2.8 | 17.72 | 2.83 |
| CV | 4.03 | 3.9 | 5 | 26.93 | 23.76 | 4.34 | 20.54 | 43.32 | 13.94 | 11.98 | 7.73 | 32.64 | 32.64 |
| Max | 77.33 | 112.67 | 111.73 | 49 | 30.33 | 26.7 | 275 | 132.33 | 315.33 | 90.97 | 28.36 | 70.17 | 11.23 |
| Min | 65.33 | 95.67 | 88.13 | 12.67 | 10.33 | 22.07 | 108 | 25.33 | 169.33 | 46.92 | 18.32 | 13.78 | 2.2 |
| SOV | DTF (Day) | DTM (Day) | PH (cm) | NTH (No) | NPH (No) | PL (cm) | FG (No) | UFG (No) | TG (No) | PFG (%) | TGW (g) | TW (g) | YLD (t/ha) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| σ2 g | 0.67 | 0.40 | 3.15 | 7.08 | 5.34 | 0.28 | 94.51 | 29.04 | 22.33 | 5.59 | 0.29 | 20.17 | 0.52 |
| σ2gs | 1.94 | 2.26 | 3.57 | 0.00 | 0.00 | 0.25 | 378.21 | 46.51 | 213.48 | 14.82 | 0.63 | 17.66 | 0.45 |
| σ2e | 3.94 | 8.41 | 12.11 | 20.04 | 14.38 | 0.53 | 608.58 | 212.92 | 686.72 | 34.06 | 1.56 | 85.08 | 2.18 |
| σ2p | 6.55 | 11.07 | 18.83 | 27.12 | 19.72 | 1.06 | 1081.30 | 288.47 | 922.53 | 54.47 | 2.48 | 122.91 | 3.15 |
| Mean | 72.83 | 104.42 | 98.60 | 23.25 | 19.97 | 24.36 | 185.54 | 49.23 | 234.77 | 78.92 | 23.31 | 37.85 | 6.06 |
| h2B (%) | 10.23 | 3.61 | 16.72 | 26.11 | 27.08 | 26.42 | 8.74 | 10.07 | 2.42 | 10.26 | 11.70 | 16.41 | 16.51 |
| GCV (%) | 1.12 | 0.61 | 1.80 | 11.44 | 11.57 | 2.17 | 5.24 | 10.95 | 2.01 | 3.00 | 2.31 | 11.87 | 11.91 |
| PCV (%) | 3.51 | 3.19 | 4.40 | 22.40 | 22.24 | 4.23 | 17.72 | 34.50 | 12.94 | 9.35 | 6.76 | 29.29 | 29.30 |
| GA (%) | 0.74 | 0.24 | 1.52 | 12.05 | 12.40 | 2.30 | 3.19 | 7.15 | 0.65 | 1.98 | 1.63 | 9.90 | 9.97 |
| DTF | DTM | PH | NTH | NPH | PL | FG | UFG | TG | PFG | TGW | TW | YLD | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DTF | 0.43** | -0.30** | 0.183** | -0.02 | -0.14* | 0.20** | -0.19** | 0.06 | 0.22** | -0.12 | -0.07 | -0.07 | |
| DTM | -0.30** | 0.28** | 0.42** | -0.06 | 0.30** | -0.27** | 0.12 | 0.31** | 0.13* | 0.122 | 0.128 | ||
| PH | -0.49** | -0.27** | 0.72** | -0.51** | 0.71** | 0.05 | -0.73** | 0.25** | 0.02 | 0.02 | |||
| NTH | 0.80** | -0.31** | 0.36** | -0.41** | 0.05 | 0.45** | -0.03 | 0.38** | 0.38** | ||||
| NPH | -0.14* | 0.32** | -0.28** | 0.14* | 0.33** | 0.11 | 0.45** | 0.45** | |||||
| PL | -0.16** | 0.55** | 0.33** | -0.49** | 0.26** | 0.22** | 0.22** | ||||||
| FG | -0.63** | 0.65** | 0.79** | -0.14* | 0.46** | 0.46** | |||||||
| UFG | 0.18** | -0.96** | 0 | -0.20** | -0.20** | ||||||||
| TG | 0.06 | -0.17** | 0.39** | 0.38** | |||||||||
| PFG | -0.04 | 0.31** | 0.31** | ||||||||||
| THW | 0.22** | 0.22** | |||||||||||
| TW | 1** | ||||||||||||
| YLD |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabri, R.S.; Rafii, M.Y.; Ismail, M.R.; Yusuff, O.; Chukwu, S.C.; Hasan, N. Assessment of Agro-Morphologic Performance, Genetic Parameters and Clustering Pattern of Newly Developed Blast Resistant Rice Lines Tested in Four Environments. Agronomy 2020, 10, 1098. https://doi.org/10.3390/agronomy10081098
Sabri RS, Rafii MY, Ismail MR, Yusuff O, Chukwu SC, Hasan N. Assessment of Agro-Morphologic Performance, Genetic Parameters and Clustering Pattern of Newly Developed Blast Resistant Rice Lines Tested in Four Environments. Agronomy. 2020; 10(8):1098. https://doi.org/10.3390/agronomy10081098
Chicago/Turabian StyleSabri, Raieah Saiyedah, Mohd Y. Rafii, Mohd Razi Ismail, Oladosu Yusuff, Samuel C. Chukwu, and Nor’Aishah Hasan. 2020. "Assessment of Agro-Morphologic Performance, Genetic Parameters and Clustering Pattern of Newly Developed Blast Resistant Rice Lines Tested in Four Environments" Agronomy 10, no. 8: 1098. https://doi.org/10.3390/agronomy10081098
APA StyleSabri, R. S., Rafii, M. Y., Ismail, M. R., Yusuff, O., Chukwu, S. C., & Hasan, N. (2020). Assessment of Agro-Morphologic Performance, Genetic Parameters and Clustering Pattern of Newly Developed Blast Resistant Rice Lines Tested in Four Environments. Agronomy, 10(8), 1098. https://doi.org/10.3390/agronomy10081098





