Abstract
The aim of the study was to determine the effect of herbicides and herbicides used with biostimulants on the content of total and true protein in potato tubers. The three-year field experiment was carried out using the random two-way split-plot arrangement in three repetitions. The examined factors were: I—three cultivars of potato: Bartek, Gawin, Honorata; II—five method applications of herbicides and herbicides with biostimulants: 1. Control—mechanical weeding, 2. Harrier 295 ZC (linuron + clomazone), 3. Harrier 295 ZC + Kelpak SL (linuron + clomazone and extract from algae Ecklonia maxima), 4. Sencor 70 WG (metribuzin), 5. Sencor 70 WG + Asahi SL (metribuzin and sodium p-nitrophenolate, sodium o-nitrophenolate, sodium 5-nitroguolacolate). On the plots sprayed with herbicides and herbicides mixed with biostimulants, a significant increase in the content of total and true proteins and the share of true protein in total protein in relation to tubers harvested from the control object was found. Genetic features of cultivars determined the content of total and true protein in potato tubers. The highest total and true protein was accumulated by tubers of the Bartek cultivar, and the least by the Honorata cultivar.
1. Introduction
Mechanical and chemical treatments using herbicides, herbicide mixtures or herbicides with other preparations used in potato plantations protect the crop against weeds [1,2,3,4], but can be phytotoxic, affect the yield and change the chemical composition of tubers [5,6,7]. One of the most important components of potato is protein, which in the fresh matter of the tuber is present in the amount of 1.7%–2.3%, and in the dry matter it ranges from 4.5% to 13.6% [8,9]. Although it occurs in a small amount, it has a high biological and nutritional value. Potato protein is rich in exogenous amino acids, such as: lysine, leucine, phenylalanine, threonine, valine and arginine, which the human body does not synthesize [10,11,12]. According to Pęksa et al. [8] and Bartová et al. [13], the biological value of potato protein is greater than that of most vegetable proteins, e.g., wheat, rice, peas, soy. According to Oser’s index, the Essential Amino Acid Index (EAA), characterizing potato protein in terms of amino acid composition indicates that it is comparable to the FAO egg standard of essential amino acid.
Due to the relatively high potato consumption in Poland of 92 kg per person, the protein contained in it plays an important role in human nutrition. The importance and popularity of potato in the world is also evidenced by the fact that out of 195 countries in the world, it is grown as a vegetable in 166 and provides the human diet with valuable nutrients [14].
The interest and use of biostimulants in plant production has increased in recent years. According to many authors [15,16,17,18,19], agricultural biostimulants include a variety of substances and microorganisms that stimulate natural physiological processes, promote nutrient uptake, improve crop yield, quality and plant resistance to stress. Calvo et al. [20] and Lea et al. [21] showed that the amino acids contained in biostimulants play an important role in the storage and transport of nitrogen in plants and increase plant tolerance to abiotic and biotic stress. Arafa et al. [22], Farouk [23] and Mystkowska [24] noted an increase in protein content in potato tubers, Matysiak et al. [25] in wheat grain and Szczepanek et al. [26] in carrot root after using biostimulants. Hence, the working hypothesis was adopted that herbicides and biostimulants may not only reduce weed infestation, but also have a positive effect on the content of protein compounds in the potato. The aim of the research was to determine the effect of herbicides and herbicides used with biostimulants on the content of total and true protein in Solanum tuberosum tubers.
2. Materials and Methods
2.1. Field Experiment and Agronomic Management
The research material, which was potato tubers, came from a three-year field experiment carried out in years 2012–2014, in Wojnów (52°12′59″ N, 22°34′37″ E) in Poland. The experiment was established as a two-way split-plot arrangement in three repetitions. The impact of two factors was analyzed:
- − Factor I—three cultivars of potato: Bartek, Gawin, Honorata [27] (Table 1);
 Table 1. Description of three potato cultivars grown in the experiment—factor I [27].
Table 1. Description of three potato cultivars grown in the experiment—factor I [27]. - − Factor II—five methods application of herbicides and herbicides with biostimulants (Table 2).
 Table 2. Description of herbicides and herbicide with biostimulants used in the experiment—factor II [28,29].
Table 2. Description of herbicides and herbicide with biostimulants used in the experiment—factor II [28,29].
Mechanical procedures were performed on the control object and on objects with herbicides and biostimulants and were presented in another paper [5]. Other agrotechnical measures used in the experiment are presented in Table 3. Each year, potatoes were planted after winter wheat. The area of one plot was 18.73 m2 = 5.55 m × 3.375 m, i.e., 15 plants arranged in five rows and the experiment area was 843 m2 (18.73 m2 × 45 plots). The selection of herbicides was in line with the recommendations of the Institute of Plant Protection–National Research Institute Poznań in Poland and adapted to the occurring weeds [28]. Biostimulants Kelpak SL and Asahi SL are recommended in plant cultivation by the Ministry of Agriculture and Rural Development [29]. The experiment was carried out on soil belonging to Haplic Luvisol (LV-ha), with a sandy loam texture according to the World Reference Base for soil resources WRB FAO [30]. Soil parameters were determined before establishing the experiment: pH 5.60–6.35 in 1 M KCl, organic matter 15.0–18.7 g·kg−1, content of available nutrients in mg·kg–1 of soil: P ranging from 68.6 (high) to 110 (very high), K from 99.6 (low) to 149.4 (medium), Mg from 50.0 (high) to 56.0 (high), Fe (low) from 465.0 to 570.5, Mn (medium) from 80.8 to 83.4, Zn (low) from 7.5 to 7.9, Cu (low) from 2.7 to 2.9.

Table 3.
Agrotechnical treatments used in the field experiment.
2.2. Determination of Protein
During harvest, samples of tubers from 10 plants of potatoes per plot were collected for chemical analyses. Samples of potato tubers were dried at an initial temperature of 70 °C, and then dried at 105 °C to a constant weight using the SLW 115 SIMPLE dryer (Merazet, Poland) with forced air circulation. The total and true nitrogen was determined with Kjeldahl’s method on a 2300 Kjeltec Analyser Unit [31]. The content of total and true protein was calculated from the content of total and protein nitrogen with the use of the coefficient 6.25. True protein was separated from non-protein compounds by precipitation with 10% trichloroacetic acid and determined by the Bernstein method [32].
2.3. Statistical Analysis and Weather Conditions
The results of the study were analyzed statistically with an analysis of variance (ANOVA Cultivar × Methods × Years) for the two-way split-plot arrangement. The significance of differences between the compared averages was verified using Tukey’s test at the significance level p ≤ 0.05. Calculations were performed in Excel using the authors’ own algorithm based on the split-plot mathematical model. The above statistical procedures are presented in the work by Tretowski and Wójcik [33].
Thermal-humidity conditions in the three years of research were varied (Table 4). The average air temperatures in all years of the study were higher than the average from the long-term period. The sum of precipitation in 2012 was lower, and in 2013 and 2014 it was higher than the average from 1980–2009, although it was distributed unevenly during potato vegetation. According to the hydrothermal coefficient, 2012 was dry, 2013 optimal and 2014 quite dry. The accumulation of ingredients in tubers of medium-early varieties was determined mainly by hydrothermal conditions prevailing in July and August.

Table 4.
Weather conditions during potato growing period.
3. Results and Discussion
Potato is a valuable food product. Its nutritional value is determined by the nutrients contained in the tubers such as protein, starch, fat, vitamins, polyphenols, macro-elements and microelements. In addition, it is relatively low in calories, and anti-nutritional substances are found in tubers in very small amounts [8,11,35,36]. Our own research showed that the content of total protein in potato tubers depended significantly on cultivated cultivars, methods of using herbicides and biostimulants and atmospheric conditions in the years of the study and was in the range of 126.7–156.6 g·kg−1 (Table 5, Table 6 and Table 7). The cultivars grown in the experiment differed in the content of total protein in tubers. The Bartek cultivar gathered the highest, Gawin a lower and Honorata the lowest content of protein. Different authors also showed different protein contents depending on the cultivar [7,12,13,37].

Table 5.
Total and true protein content in tubers of potato depending on the treatments of herbicides and biostimulants, application and cultivars of dry matter.

Table 6.
Total and true protein content in dry matter of potato tubers depending on weather conditions.

Table 7.
Total and true protein content in tubers of potato tubers in study years of dry matter.
In the conducted studies, the content of total protein depended on the application of herbicides and herbicides with biostimulants. A significant increase in the content of this component was demonstrated after using the herbicides M (Sencor 70 WG) and LC (Harrier 295 ZC) and herbicides with biostimulants, i.e., M + S (Sencor 70 WG + Asahi SL), LC + E (Harrier 295 ZC + Kelpak SL) compared to the C** (control object). Mixtures of herbicides used in potato protection control a broader spectrum of weed species than a single herbicide [5]. L (linuron) inhibits the photosynthesis process in the plant in its initial stage, blocking the photolysis of water. In the light phase of photosynthesis, it acts as an inhibitor of electron transport, enabling the formation of active oxygen species. C (clomazone) inhibits the biosynthesis of photosynthetic pigments, especially chloroplast [2]. As a result of the action of M (metribuzin), the light energy is not converted into chemical energy, which is necessary for plants [4]. The active substances of biostimulators increase the biological potential without causing development defects, which may contribute to increasing the yield and improving the quality of crops and resistance to stress conditions [15,16]. S (sodium) works by increasing the accumulation of polyphenols (up to 30%), which protect plant cells and their enzymatic systems from damage, as well as support plants in reacting quickly to unfavorable habitat conditions. Previous studies have shown that phenols contained in S very easily penetrate into plant tissues and then into individual cells, where they stimulate faster cytoplasmic flow, which consequently affects more efficient biosynthesis of substances necessary for growth and development. There is a faster reaction to stress factors and activation of the natural defense mechanisms of plants [10]. E (Ecklonia maxima) extract from algae affects the increase in yield and tuber quality characteristics. Studies [15,24] show that the effect depends on the dose and frequency of treatment and the species of the crop plant.
Barbaś and Sawicka [10] also noted an increase in the amount of protein in tubers after spraying, at different doses, with the herbicides Sencor 70 WG (metribuzin) and Titus 25 WG (rimsulfuron), and Gugała et al. [38] after applying herbicides Command 480 EC (clomazone), Stomp 400 SC (pendimethalin) and herbicide mixtures Command 480 EC (clomazone) + Dispersive Afalon 450 SC (linuron), Stomp 400 SC (pendimethalin) + Dispersive Afalon 450 SC (linuron). In contrast, Miedzianka [39] showed a reduction in the protein content after the application of herbicides, which was most likely associated with a defense response to stress conditions, as a result of which the proportions of individual components may change in the direction of more intense storage of, e.g., carbohydrates. The beneficial effect of biostimulants on the content of total protein in potatoes was found by Mystkowska [24], Arafa et al. [22] and Farouk [23]. In turn, Haider et al. [40], after using the “Primo” seaweed extract as an organic biostimulant, in the Sante potato cultivar, observed a significant improvement in growth, yield as well as nitrogen and protein content in Solanum tuberosum tubers compared to control plants. Other authors noted an increase in total nitrogen accumulation in various plants after using biostimulants: Wierzbowska et al. [41] in potato tubers, Szczepanek et al. [26] in carrot roots, Kierzek et al. [42] in maize grain. Statistically proven cooperation of cultivars with the use of herbicides and herbicides with biostimulants proves that the reaction of cultivars was different (Table 5). The Bartek cultivar accumulated the highest content of total protein after the application of the LC + E (Harrier 295 ZC herbicide and the Kelpak SL biostimulant), and the Gawin and Honorata cultivars after spraying with M (Sencor 70 WG) or M + S (Sensor 70 WG + Asahi SL).
The content of total protein was significantly differentiated by weather conditions in individual vegetation periods (Table 6 and Table 7). The largest amount of this ingredient was accumulated by tubers in fairly dry and warm 2014, when July was warm and extremely dry, and significantly less in the other growing seasons. According to Bártová et al. [13], weather conditions during potato tuberization have a decisive impact on the value of this trait. Barbaś and Sawicka [10] and Gugała et al. [38] found that low rainfall in July and a temperature of around 19–20 °C favored the accumulation of protein in potato tubers. The interaction of years with the use of herbicides and herbicides with biostimulants confirms that the impact of chemical treatments on the content of total protein depended on meteorological conditions. The proven cooperation of cultivars with years indicates that the accumulation of the discussed ingredient in cultivated cultivars was shaped by weather conditions in the years of the experiment. The varied reaction of cultivars to climatic conditions in the case of total and true protein is confirmed by the studies conducted by Mystkowska [24].
The chemical analysis showed that the content of true protein in potato tubers significantly depended on the variety, methods of application of herbicides and biostimulants and hydrothermal conditions in the years of the study and ranged from 91.6 to 111.7 g·kg−1 of dry mass (Table 5, Table 6 and Table 7). The highest concentration of true protein was found in Bartek, was significantly lower in Gawin and the lowest in Honorata. Sawicka et al. [43] and Baranowska et al. [44] also noted differences in the accumulation of this component.
Herbicides and biostimulants used in the experiment increased the accumulation of true protein compared to the control object. The largest significant increase in tuber specific protein content was found after the application of the M (Sencor 70 WG) herbicide and M + S (Sencor 70 WG with Asahi SL) biostimulant. Similar results were obtained by Gugała et al. [38] using a single herbicide Stomp 400 SC (pendimethalin) and a mixture of herbicides Stomp 400 SC (pendimethalin) with Afalon Dispersive 450 SC (linuron). In contrast, Mystkowska [24] noted an increase in the content of the protein in question by spraying potato plants during vegetation with biostimulants: Kelpak SL, Titanit, GreenOk and BrunatneBio Złoto. In the conducted experiment, the interaction of potato cultivars with the use of herbicides and herbicides with biostimulants, and methods of application of preparations with weather conditions, was found.
During the three-year study period, the highest content of true protein in tubers was recorded in fairly dry 2014, and the least in dry 2012. The interaction of cultivars with conditions in the years of research has been demonstrated, which indicates a different reaction of cultivars to hydrothermal agents. The least true protein was in the Gawin cultivar in 2013, and in the Bartek and Honorata, no differences between years occurred in cultivars in 2012. According to Kalbarczyk and Kalbarczyk [45], optimal conditions for yielding and collecting ingredients in potato tubers are provided by weather conditions with a moderate average air temperature from May to September of 15.2 °C and a rainfall of 347.1 mm.
An important feature, from the point of view of the consumer, is the share of true protein in the total protein, which in the experiment was quite high and ranged from 69.6% to 75.6% (Figure 1).
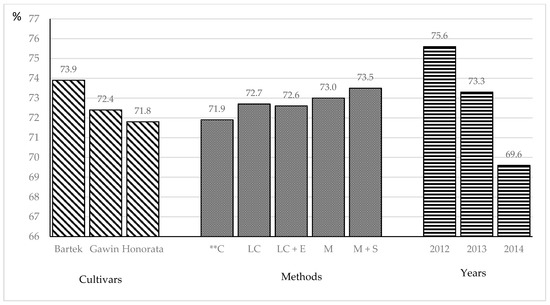
Figure 1.
Percentage of true protein in potato tubers total protein. ** C—Control object; LC - linuron + clomazone; LC + E - linuron + clomazone + Ecklonia maxima; M—metribuzin; M + S—metribuzin + sodium.
In the studies by Bárta and Bártová [46], this share was within the range of 44.5%–62.1%; in the experiment by Barbaś and Sawicka [10], 64.0%–65.1%; and in Mystkowska [24], 47.9%–49.9% depending on the cultivar, weather conditions and application of preparations. The genetic properties of the varieties studied in the experiment differentiated this trait—the Bartek cultivar had the largest share, Gawin had a smaller one and Honorata had the smallest, which was confirmed by the studies of other authors [10,24]. Under the influence of the use of herbicides and biostimulants, an increase in the proportion of true protein in total protein was observed. The control compared to the share of true protein was noted after the application of the M (Sencor 70 WG) herbicide and M + S (Sencor 70 WG with the Asahi SL) biostimulant. Barbaś and Sawicka [10] found the largest share of true protein in the total protein after spraying the plots with herbicides Sencor 70 WG and Titus 25 WG before potato plant rise. Meteorological conditions also determined the share of true protein overall. The highest value of this trait was obtained in dry 2012, and the lowest in 2014. Our results confirm the data presented by Barbaś and Sawicka [10] and Gugała et al. [38], who proved that in the dry years, the share of specific protein in the total protein was the highest. However, Bárta and Bártová [46] found the highest proportion of true protein in the season with the highest humidity.
4. Conclusions
The conducted study showed that the genetic characteristics of the cultivars determined the content of total and true protein in potato tubers. The Bartek cultivar was characterized by the highest content of these components and the largest share of the true protein in the total protein. Herbicides and mixtures of herbicides with biostimulants used in potato cultivation contributed to an increase in the content of total and true protein and the share of true protein in total protein in relation to control tubers. The obtained results indicate that the selection of cultivar, herbicides and biostimulants is important in relation to the accumulation capacity of total and true protein in potato tubers. In the future, in order to use the potential of potato protein in food or processing, it will be necessary to choose the right cultivars and preparations not only depending on the starch content, but also on the content of total and true protein.
Author Contributions
Conceptualization, K.Z.; methodology, K.Z. and M.G.; software, M.G.; writing—original draft preparation, K.Z.; conducting chemical analysis, I.M.; resources, A.S.; writing—review and editing, K.Z. and I.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research within the research theme No. 31/20/B was financed from the science grant by the Ministry of Science and Higher Education in Poland.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Boydston, R.A. Managing weeds in potato rotations with herbicides. Am. J. Pot. Res. 2010, 87, 420–427. [Google Scholar] [CrossRef]
- Correia, N.M.; Carvalho, A.D.F. Herbicide selectivity for potato crop. Hortic. Bras. 2019, 37, 302–308. [Google Scholar] [CrossRef]
- Hashim, S. Chemical weed control efficiency in potato (Solanum tuberosum L.) under agro-climatic conditions of Peshawar, Pakistan. Pak. J. Weed Sci. Res. 2003, 9, 105–110. [Google Scholar]
- Hutchinson, P.J.S.; Beutler, B.R.; Hancock, D.M. Weed control and potato (Solanum tuberosum) crop response with low rates of sulfentrazone applied postemergence with metribuzin. Weed Technol. 2006, 20, 1023–1029. [Google Scholar] [CrossRef]
- Gugała, M.; Zarzecka, K.; Dołęga, H.; Sikorska, A. Weed Infestation and Yielding of Potato Under Conditions of Varied Use of Herbicides and Bio-Stimulants. J. Ecol. Eng. 2018, 19, 191–196. [Google Scholar] [CrossRef]
- Ilić, O.; Nikolić, L.; Ilin, Ž.; Mišković, A.; Vujasinović, V.; Kukić, B. Effect of cultural practices on weeds community in function of potato yield. Acta Sci. Pol. Hortorum Cultus 2016, 15, 31–43. [Google Scholar]
- Järvan, M.; Edesi, L. The effect of cultivation methods on the yield and biological quality of potato. Agron. Res. 2009, 7, 289–299. [Google Scholar]
- Pęksa, A.; Rytel, E.; Kita, A.; Lisińska, G.; Tajner-Czopek, A. The Properties of Potato Protein. Food 2009, 3, 79–87. [Google Scholar]
- Jansen, G.; Flamme, W.; Schüller, K.; Wandrey, M. Tuber and starch quality of wild and cultivated potato species and cultivars. Potato Res. 2001, 44, 137–146. [Google Scholar] [CrossRef]
- Barbaś, P.; Sawicka, B. Total and true protein content in two varieties of potato tubers depending on methods of weed control. Fragm. Agron. 2017, 34, 7–18. (In Polish) [Google Scholar]
- Camire, M.; Kubow, S.; Donelly, D.J. Potatoes and Human Health. Crit. Rev. Food Sci. Nutr. 2009, 49, 823–840. [Google Scholar] [CrossRef]
- Öztürk, E.; Polat, T. The effect of long term storage on physical and chemical properties of potato. Field Crops 2016, 21, 218–223. [Google Scholar] [CrossRef]
- Bártová, V.; Bárta, J.; Diviš, J.; Švajner, J.; Peterka, J. Crude protein content in tubers of starch processing potato cultivars in dependence on different agroecological conditions. J. Central Eur. Agric. 2009, 10, 57–66. [Google Scholar]
- Dzwonkowski, W. Potato Market. State and Perspectives; Institute of Agricultural and Food Economics−National Research Institute, Ed.; Minister of Agriculture and Rural Development: Warsaw, Poland, 2019; Volume 46, pp. 1–35. (In Polish)
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 1–12. [Google Scholar] [CrossRef]
- Piotrowski, K.; Romanowska-Duda, Z. Positive impact of biostimulators on growth and physiological activity of willow in climate change conditions. Int. Agrophys. 2018, 32, 279–286. [Google Scholar] [CrossRef]
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants application in horticultural crops under abiotic stress conditions. Agronomy 2019, 9, 306. [Google Scholar] [CrossRef]
- Niewiadomska, A.; Sulewska, H.; Wolna-Maruwka, A.; Ratajczak, K.; Waraczewska, Z.; Budka, A. The Influence of Bio-Stimulants and Foliar Fertilizerson Yield, Plant Features, and the Level of Soil Biochemical Activity in White Lupine (Lupinus albus L.) Cultivation. Agronomy 2020, 10, 150. [Google Scholar] [CrossRef]
- Wadas, W.; Dziugieł, T. Changes in Assimilation Area and Chlorophyll Content of Very Early Potato (Solanum tuberosum L.) Cultivars as Influenced by Biostimulants. Agronomy 2020, 10, 387. [Google Scholar] [CrossRef]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostimulants. Plant Soil. 2014. [Google Scholar] [CrossRef]
- Lea, P.J.; Sodek, L.; Parry, M.A.J.; Shewry, P.R.; Halford, N.G. Asparagine in plants. Ann. Appl. Biol. 2006, 150, 1–26. [Google Scholar] [CrossRef]
- Arafa, A.A.; Hussien, S.F.M.; Mohamed, H.S.G. Response of tuber yield quantity and quality of potato plants and its economic consideration to certain bioregulators or effective microorganisms under potassium fertilization. J. Plant Prod. Mansoura Univ. 2012, 3, 131–150. [Google Scholar] [CrossRef]
- Farouk, S. Imroving growth and productivity of potato (Solanum tuberosum L.) by some biostimulants and lithovit with or without boron. J. Plant Prod. Mansoura Univ. 2015, 6, 2187–2206. [Google Scholar] [CrossRef]
- Mystkowska, I.T. Content of total and true protein in potato tubers in changing weather conditions under the influence of biostimulators. Acta Agroph. 2018, 25, 475–483. [Google Scholar] [CrossRef]
- Matysiak, K.; Miziniak, W.; Kaczmarek, S.; Kierzek, R. Herbicides with natural and synthetic biostimulants in spring wheat. Ciência Rural 2018, 48, 1–10. [Google Scholar] [CrossRef]
- Szczepanek, M.; Wilczewski, E.; Pobereżny, J.; Wszelaczyńska, E.; Keutgen, A.; Ochmian, I. Effect of biostimulants and storage on macroelement content in storage roots of carrot. J. Elem. 2015, 20, 1021–1031. [Google Scholar] [CrossRef]
- Nowacki, W. Characteristic of Native Potato Cultivars Register, 22nd ed.; Plant Breeding Acclimatization Institute—National Research Institute: Jadwisin, Poland, 2019; pp. 1–43. (In Polish) [Google Scholar]
- Institute of Plant Protection—National Research Institute. Plant Protection Recommendations for 2014/15; Institute of Plant Protection—National Research Institute: Poznań, Poland, 2014. (In Polish) [Google Scholar]
- List of Plant Conditioners. List of Plant Growth Stimulants. Register of Plant Protection Products. 2020. Available online: https://www.gov.pl/web/rolnictwo/rejestr-rodkow-ochrony-roslin (accessed on 20 April 2020). (In Polish)
- FAO. World Reference Base for Soil Resources: International Soil Classification System for Naming Soils and Creating Legends for Soil; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2014. [Google Scholar]
- Ostrowska, A.; Gawliński, S.; Szczubiałka, Z. Methods of Analysis and Assessment of Soil and Plant Properties; Institute of Environment Protection: Warszawa, Poland, 1991; pp. 1–334. (In Polish) [Google Scholar]
- Horwitz, W.; Latimer, G.W. Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MA, USA, 2006. [Google Scholar]
- Trętowski, J.; Wójcik, R. Methodology of Agricultural Experiments; Wyższa Szkoła Rolniczo-Pedagogiczna: Siedlce, Poland, 1991; pp. 331–334. (In Polish) [Google Scholar]
- Skowera, B.; Jędrszczyk, E.S.; Kopcińska, J.; Ambroszczyk, A.M.; Kołton, A. The Effects of Hydrothermal Conditions during Vegetation Period on Fruit Quality of Processing Tomatoes. Pol. J. Environ. Stud. 2014, 23, 195–202. [Google Scholar]
- Leszczyński, W. Nutrition value of potato and potato products (Review of literature). Biuletyn Instytutu Hodowli Aklimatyzacji Roślin 2012, 266, 5–20. (In Polish) [Google Scholar]
- Beals, K.A. Potatoes, Nutrition and Health. Am. J. Potato Res. 2019, 96, 102–110. [Google Scholar] [CrossRef]
- Gumul, D.; Ziobro, R.; Noga, M.; Sabat, R. Characterisation of five potato cultivars according to their nutritional and pro-health components. Acta Sci. Pol. Technol. Aliment. 2011, 10, 73–81. [Google Scholar]
- Gugała, M.; Zarzecka, K.; Sikorska, A. The effect of herbicides on the protein content in tubers of edible potato cultivars. Acta Sci. Pol. Agric. 2017, 16, 17–23. [Google Scholar]
- Miedzianka, J. Functional Properties of Potato Protein Subjected to Chemical Modification. Ph.D. Thesis, Uniwersytet Przyrodniczy Wrocław, Wrocław, Poland, 2011; pp. 1–102. (In Polish). [Google Scholar]
- Haider, M.W.; Ayyub, C.M.; Pervez, M.A.; Asad, H.U.; Manan, A.; Raza, S.A.; Ashraf, I. Impact of foliar application of seaweed extract on growth, yield and quality of potato (Solanum tuberosum L.). Soil Environ. 2012, 31, 157–162. [Google Scholar]
- Wierzbowska, J.; Cwalina-Ambroziak, B.; Głosek-Sobieraj, M.; Sienkiewicz, S. Effect of biostimulators on yield and selected chemical properties of potato tubers. J. Elem. 2015, 20, 757–768. [Google Scholar] [CrossRef][Green Version]
- Kierzek, R.; Dubas, M.; Matysiak, K. Effect of biostimulator Aminoplant mixtures with terbuthylazine and bromoxynil (Zeagran 340 SE) on herbicidal effect and yield of maize. Prog. Plant Prot. 2015, 55, 164–169. (In Polish) [Google Scholar] [CrossRef][Green Version]
- Sawicka, B.; Danilcenko, H.; Jariene, E.; Krochmal-Marczak, B. Phenotypic changeability of features of foreign cultivars of potato in Poland. Zeszyty Problemowe Postępów Nauk Rolniczych 2009, 542, 447–463. (In Polish) [Google Scholar]
- Baranowska, A.; Zarzecka, K.; Mystkowska, I.; Gugała, M.; Zarzecka, M. Crude and true protein content in potato tubers (Solanum tuberosum L.) depending on the ways of application of the microbiological preparation UGmax. Fresenius Environ. Bull. 2018, 27, 7967–7972. [Google Scholar]
- Kalbarczyk, R.; Kalbarczyk, E. The needs and deficiency in atmospheric precipitation in cultivated mid-late and late potato in Poland. Infrastruktura Ekologia Terenów Wiejskich 2009, 3, 129–140. (In Polish) [Google Scholar]
- Bárta, J.; Bártová, V. Patatin, the major protein of potato (Solanum tuberosum L.) tubers, and its occurrence as genotype effect: Processing versus table potatoes. Czech J. Food Sci. 2008, 26, 347–359. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).