3.1.1. Plant Growth and Metal Accumulation
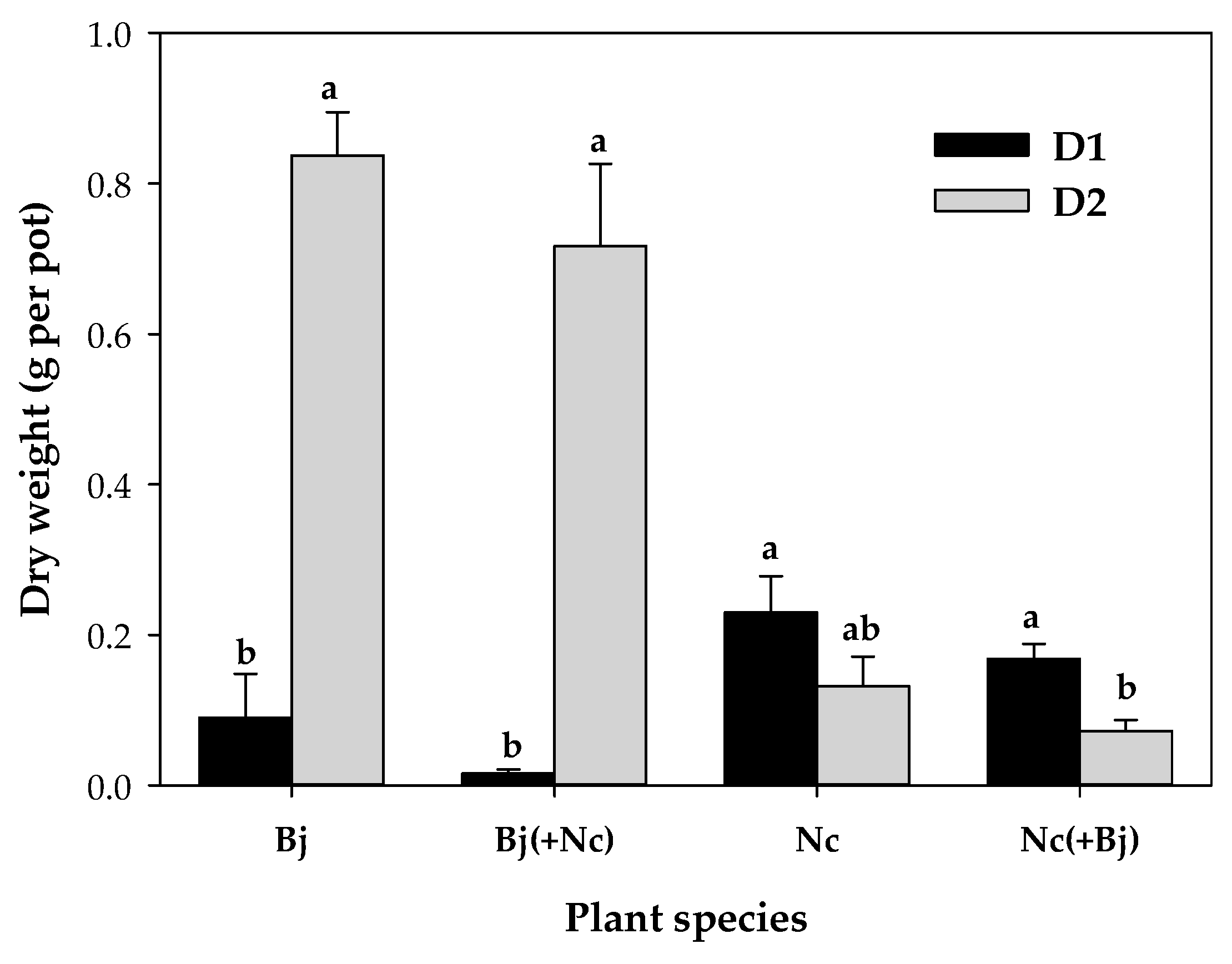
The dry weight of
B. juncea was significantly higher in soil D2, with the highest pH value, than in D1 (
Figure 1). Contrastingly,
N. caerulescens plants had higher dry weight in the soil of lower pH, D1. For this species, the growth conditions have been reported to be optimal at a pH value of about 5.1 [
36]. However, according to Broadhurst et al. [
37],
N. caerulescens plants do not exhibit phytotoxicity symptoms over a soil pH range of 5.5–7.0. In addition, in soil D1, the yield of
B. juncea plants was very low, and the determination of their elemental composition was not possible when cocultivated with
N. caerulescens. In fact,
B. juncea plants have low survival in soils with pH <4 [
20].
The concentrations of Fe found in
B. juncea plants, grown in both soils, were similar to those found in other studies (e.g., 91.0 µg g
−1) [
38] and generally within the range considered deficient in plants (50–150 µg g
−1) [
39]. This was also the case for
N. caerulescens plants grown in D1, while plants from D2 had slightly higher Fe concentrations (
Table 3). Interestingly, the Fe concentration found by Walker and Bernal [
13] in the same line of
N. caerulescens grown in a nutrient solution (103 µg g
−1) was similar to that found in the present experiment.
The concentrations of Cu in
B. juncea plants grown in D1 were near the threshold considered for plant toxicity of 20 μg g
−1 [
40]. The Cu concentrations were lower in D2, but not significantly so (
Table 3). The highest Cu concentration in
N. caerulescens was 15 µg g
−1 for plants grown alone in D2. This value is of the same magnitude as that obtained by Walker and Bernal [
13] in contaminated soil (7 µg g
−1) and is in the interval considered normal for plants (5–30 µg g
−1) [
40]; no significant differences were observed between soils D1 and D2
The
B. juncea plants had their highest concentration of Mn when grown in D1 (
Table 3), this value being similar to the results of Feigl et al. [
38] (61.4 µg g
−1). The concentrations of Mn in cocultivated
N. caerulescens plants grown in D1 were significantly higher than in soil D2. The values generally were lower than in previous reports for this species in a nutrient solution (e.g., 156 μg g
−1 [
13] and 178 μg g
−1 [
41]), but were similar to the results of McGrath et al. [
42] (35.5 µg g
−1) in contaminated soils.
The concentration of Zn in
B. juncea differed significantly between the soils, with the highest concentrations in soil D1, of low pH. In a field experiment conducted in the area of Aznalcóllar from which our soil samples were taken, high concentrations of Zn in the aerial part of
B. juncea (2029 µg g
−1) were also observed [
20]. Del Rio et al. [
15] also found increasing Zn concentrations in
B. juncea plants, from 37.5 µg g
−1 in uncontaminated soil to 94 μg g
−1 in soil with 462 μg g
−1 of total Zn, demonstrating a clear accumulator behavior. BCF found in this species ranged from 0.3 to 6.0, the latter when
B. juncea grew alone in D1. This BCF is much higher than that (1.52) found by Ali and Chaudhury [
43] in plants grown for 3 months in a noncontaminated soil.
The levels of Zn in
N. caerulescens did not reach hyperaccumulation (10,000 µg g
−1), neither in soil nor in the growth conditions, perhaps due to the multielemental contamination of the soils (discussed below). The highest concentrations of Zn in
N. caerulescens occurred when grown alone (
Table 3), while the lowest values were found in cocultivation with
B. juncea in soil D2 (pH 6.4). Zn BCF in the hyperaccumulator was approximately 12 when grown in D1 or alone in D2 but fell to 4.2 when grown with
B. juncea in D2. All these BCF values were lower than those found by McGrath et al. [
42] in a pot experiment using different contaminated soils. In soil D1,
B. juncea plants did not grow well and produced little biomass (
Figure 1), a clear symptom of toxicity. Therefore, competition/interaction between
B. juncea and
N. caerulescens for Zn uptake was not found in this soil.
The results indicate that the presence of
N. caerulescens did not improve the accumulation of Zn in the accumulator species (
B. juncea). In contrast, the presence of the latter reduced the hyperaccumulating potential (yield and Zn concentration) of the former, which may indicate competition between the two species for the uptake of nutrients and the bioavailable Zn fraction in soil D2. The level of Zn in these soils is not extremely high (although it is greater than the limits for agricultural soils [
32]), and Zn is an essential micronutrient, which is taken up from the same soil fractions by the roots of the hyperaccumulator and other plant species [
44].
N. caerulescens is able to bioaccumulate Zn even in soils with low or moderate concentrations [
45] due to its highly efficient transport system [
46]. According to Whiting et al. [
47], the absence of a large-scale active mechanism of Zn mobilization in the roots of
N. caerulescens can severely limit the rate of uptake from forms poorly available in the soil and the subsequent accumulation of Zn in the plant. This may have occurred in the high-pH soil D2, with low 0.1 M CaCl
2-extractable concentrations of Zn (discussed later;
Figure 2). Thus, in soil D2, the presence of high-biomass
B. juncea plants could have limited the development of the roots of the hyperaccumulator species, preventing them from exploring areas of the soil rich in Zn that were not dominated by the roots of
B. juncea. This factor is critical for
N. caerulescens to maintain its high accumulation of Zn [
47].
In fact, in soil D2, B. juncea showed a mean Zn phytoextraction value of 121 µg pot−1, similar to N. caerulescens (174 µg pot−1) when grown together. However, N. caerulescens reached the greatest Zn phytoextraction value when growing alone (1844 ± 964 µg pot−1).
For
B. juncea, the concentration of Cd was below the detection limit (0.01 µg g
−1) when grown combined with the hyperaccumulator species in soil D2, while its maximum Cd concentration occurred in D1 when grown alone (6.22 µg g
−1;
Table 3). Contrastingly, the Cd concentration in
N. caerulescens was highest (23.1 µg g
−1) when grown alone in D2, which resulted in a Cd BCF of 11.6, similar to that reported by Martínez-Alcalá et al. [
48] for
N. caerulescens plants grown in TE-contaminated soil (around 13).
As with Zn, the low biomass of
B. juncea in D1 precluded any effect on the Cd uptake by the hyperaccumulator, but its presence in soil D2 greatly reduced the Cd concentration in
N. caerulescens. All the Cd concentrations found were below the minimum value (100 µg g
−1) considered to represent hyperaccumulation, confirming the report of Walker and Bernal [
13] that this line of
N. caerulescens is not a Cd hyperaccumulator.
3.1.2. Soil Properties
The soil pH values at the end of the experiment were slightly acidic in soil D1, and there was no significant effect of the plant species grown in the soil or of the cocultivation of the two species (
Figure 2). Soil D2 showed, as expected, higher pH values that were, in addition, higher with cocultivation. Kim et al. [
49] found that the pH of the soil solution extracted from the rhizosphere of
B. juncea plants was higher than that of the bulk soil solution (<6.5). This effect was not observed in our experiment, as the pH values did not change compared to the initial values of soils D1 and D2. Although the rhizosphere had not been separated from the bulk soil, the abundance of visually detected roots, which completely covered the soil, could imply a relevant contribution of the rhizosphere in the soil samples. Martínez-Alcalá et al. [
48] found no significant changes in the pH of the rhizosphere of
N. caerulescens plants, with respect to the bulk soil, in soils with different pH. In fact, the mechanism of metal mobilization by a hyperaccumulator species does not necessarily involve a pH reduction in the rhizosphere [
42,
50].
In general, soil D1 had higher concentrations of extractable metals (0.1 M CaCl
2-extractable) than soil D2 as a consequence of its lower pH (
Figure 2). No significant differences were found between the extractable Cd concentrations in the soils where
B. juncea or
N. caerulescens plants were grown, whether in monoculture or together (
Figure 2). This was also true for Cu, Fe, and Mn in both soils.
The extractable Zn concentrations in soil D1 were highest when
B. juncea was grown individually (
Figure 2), likely because of the scarce plant biomass of
B. juncea (
Figure 1) and the strong Zn uptake by
N. caerulescens (
Table 2) in this soil. The uptake of Zn by the hyperaccumulator species likely first involves soluble and exchangeable forms (extractable in 0.1 M CaCl
2). In fact, active soil mobilization by hyperaccumulator species appears to be less important than their highly active and efficient metal uptake systems [
48,
51], which results in a depletion of the concentration of highly soluble forms of the metals in the soil. Moreover, the rate of replenishment of soluble forms of Zn in the soil solution from less labile forms has been found to be slower than the rate of metal uptake by
N. caerulescens [
52]. Contrastingly, extractable Zn concentrations in soil D2 were much lower than in D1 and were not altered in the soils with
N. caerulescens with respect to the other species. Hammer and Keller [
53] found that EDTA extraction was more suitable to assess the uptake of Zn by
N. caerulescens in acidic soils than extraction with a neutral salt (0.1 M NaNO
3), which can only extract the easily soluble Zn fraction, quickly replaced from less-soluble forms at acidic pH.
3.1.3. Correlations
Simple linear correlations between soil and plant parameters for
B. juncea and
N. caerulescens were calculated. Highly significant negative correlations were found between soil pH and the extractable concentrations of Cd, Cu, Mn, and Zn in the soil, which confirms that pH is the key factor influencing the solubility of these metals in the soil (
Table 4). Soil pH was correlated positively with the dry weight of
B. juncea plants and negatively with the Cd and Zn concentrations in this species. Several authors have observed the difficulty of this species to grow in acidic soils [
20,
54]. Negative correlations between the extractable concentrations of Cu, Mn, and Zn and the plant dry weight indicate that high concentrations of these elements in easily available forms may be toxic for
B. juncea.
In agreement with this, the Cd and Zn concentrations in
B. juncea were negatively correlated with plant dry weight. The positive correlation between the concentrations of Zn in the soil (0.1 M CaCl
2-extractable) and in the plants reflects the Zn accumulator character of
B. juncea and its potential to serve as an indicator of Zn availability in soil [
20].
In
N. caerulescens, a negative correlation between soil pH and plant biomass was found (
Table 4), which indicates that this species grows better in the slightly acidic soils. As a consequence, plant dry weight was positively correlated with extractable metal concentrations in the soil. The extractable concentrations of Cu correlated positively with the corresponding concentrations in
N. caerulescens, which may indicate that the Cu concentration in the soils was not high enough to cause toxicity in this species [
12], with Cu acting only as an essential micronutrient.