Laboratory and Field Performance of Metarhizium anisopliae Isolate ICIPE 41 for Sustainable Control of the Invasive Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Insects
2.3. Fungal Isolate Culture, Viability and Suspension Preparation
2.4. Formulation of the Fungal Isolate Metarhizium anisopliae ICIPE 41
2.5. Pathogenicity of the Formulations of Metarhizium anisopliae ICIPE 41 against Second Instar of FAW in the Laboratory
2.6. Field Experiments
2.6.1. Mazao Achieve® Biopesticide
2.6.2. Field Site
2.6.3. Treatments and Experimental Layout
2.7. Assessment of FAW Adults before Treatment Application
2.7.1. Assessment of FAW Damage and Larval Mortality Induced by Corn Oil Formulation of Metarhizium anisopliae ICIPE 41
2.7.2. Conidial Persistence Assessment under Field Conditions
2.7.3. Effect of Different Treatments against FAW-Associated Parasitoids in Field
2.8. Maize Grain Yield
2.9. Data Analysis
3. Results
3.1. Pathogenicity of the various Metarhizium Anisopliae ICIPE 41 Formulations against Second-Instar FAW in the Laboratory
3.2. Assessment of the Effect of FAW Sex Pheromone Traps on the Population of FAW Moths
3.3. Fall Armyworm Larval Incidence and Damage in the Field
3.4. Cumulated Mortality of Collected Second-Instar FAW in the various Field Treatments after Applications
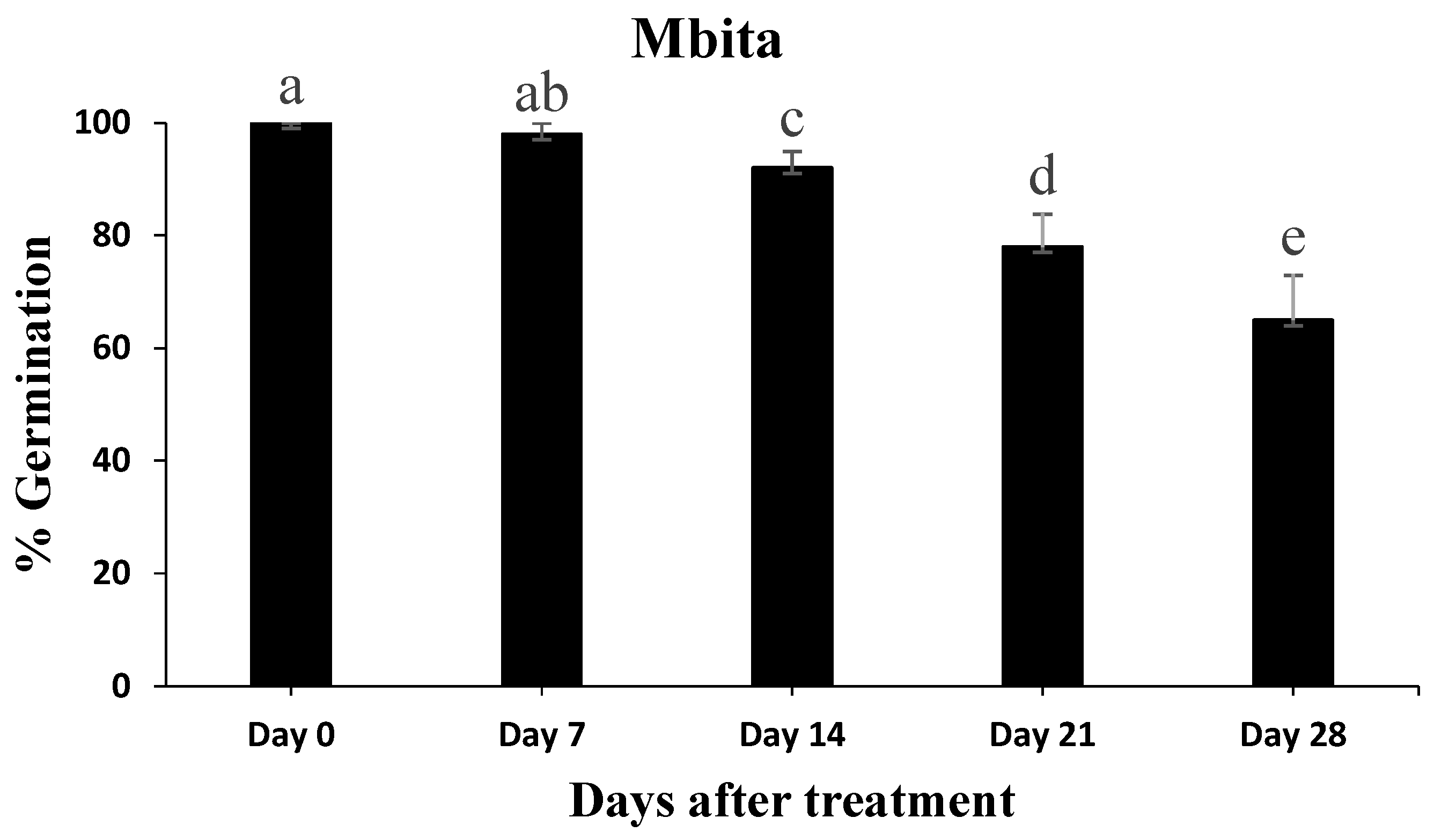
3.5. Conidial Persistence Assessment after Application
3.6. Effect of Entomopathogenic Fungi on Parasitoids Emergence
3.7. Maize Grain Yield
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Akutse, K.S.; Khamis, F.M.; Ambele, F.C.; Kimemia, J.W.; Ekesi, S.; Subramanian, S. Combining Insect Pathogenic Fungi and a Pheromone Trap for Sustainable Management of the Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae). J. Invertebr. Pathol. 2020, 177, 107477. [Google Scholar] [CrossRef] [PubMed]
- Ali-Olubandwa, A.M.; Kathuri, N.J.; Odero-Wanga, D.; Shivoga, W.A. Challenges Facing Small Scale Maize Farmers in Western Province of Kenya in the Agricultural Reform Era. Am. J. Exp. Agric. 2011, 1, 466–476. [Google Scholar] [CrossRef]
- Kfir, R.; Overholt, W.A.; Khan, Z.R.; Polaszek, A. Biology and Management of Economically Important Lepidopteran Cereal Stem Borers in Africa. Annu. Rev. Entomol. 2002, 47, 701–731. [Google Scholar] [CrossRef] [PubMed]
- Kumari, D.A.; Lakshmi, B.K.M.; Reddy, G.S.; Reddy, M.L. Influence of Abiotic Factors on the Incidence of Hopper and Chemical Control Strategies in Mango. Karnataka J. Agric. Sci. 2009, 22, 601–602. [Google Scholar]
- Sokame, B.M.; Subramanian, S.; Kilalo, D.C.; Juma, G.; Calatayud, P.A. Larval Dispersal of the Invasive Fall Armyworm, Spodoptera frugiperda, the Exotic Stemborer Chilo partellus, and Indigenous Maize Stemborers in Africa. Entomol. Exp. Appl. 2020, 168, 322–331. [Google Scholar] [CrossRef]
- Kumela, T.; Simiyu, J.; Sisay, B.; Likhayo, P.; Mendesil, E.; Gohole, L.; Tefera, T. Farmers’ Knowledge, Perceptions, and Management Practices of the New Invasive Pest, Fall Armyworm (Spodoptera frugiperda) in Ethiopia and Kenya. Int. J. Pest Manag. 2019, 65, 1–9. [Google Scholar] [CrossRef]
- Day, R.; Abrahams, P.; Bateman, M.; Beale, T.; Clottey, V.; Cock, M.; Colmenarez, Y.; Corniani, N.; Early, R.; Godwin, J.; et al. Fall Armyworm: Impacts and Implications for Africa. Outlooks on Pest Management. Outlooks Pest Manag. 2017, 28, 196–201. [Google Scholar] [CrossRef] [Green Version]
- Mweke, A.; Akutse, K.S.; Ulrichs, C.; Fiaboe, K.K.M.; Maniania, N.K.; Ekesi, S. Efficacy of Aqueous and Oil Formulations of a Specific Metarhizium anisopliae Isolate against Aphis craccivora Koch, 1854 (Hemiptera: Aphididae) under Field Conditions. J. Appl. Entomol. 2019, 143, 1182–1192. [Google Scholar] [CrossRef]
- Wokabi, S.M. Sustainability of Maize Production in Kenya. Kenya Agric. Res. Inst. Nairobi Kenya 2013, 5, 2. [Google Scholar]
- Cook, S.M.; Khan, Z.R.; Pickett, J.A. The Use of Push-Pull Strategies in Integrated Pest Management. Annu. Rev. Entomol. 2007, 52, 375–400. [Google Scholar] [CrossRef] [Green Version]
- Akutse, K.S.; Kimemia, J.W.; Ekesi, S.; Khamis, F.M.; Ombura, O.L.; Subramanian, S. Ovicidal Effects of Entomopathogenic Fungal Isolates on the Invasive Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). J. Appl. Entomol. 2019, 143, 626–634. [Google Scholar] [CrossRef]
- Sisay, B.; Simiyu, J.; Malusi, P.; Likhayo, P.; Mendesil, E.; Elibariki, N.; Wakgari, M.; Ayalew, G.; Tefera, T. First Report of the Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), Natural Enemies from Africa. J. Appl. Entomol. 2018, 142, 800–804. [Google Scholar] [CrossRef]
- Idrees, A.; Qadir, Z.A.; Akutse, K.S.; Afzal, A.; Hussain, M.; Islam, W.; Waqas, M.S.; Bamisile, B.S.; Li, J. Effectiveness of Entomopathogenic Fungi on Immature Stages and Feeding Performance of Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae) Larvae. Insects 2021, 12, 1044. [Google Scholar] [CrossRef]
- Shylesha, A.; Jalali, S.; Gupta, A.; Varshney, R.; Venkatesan, T.; Shetty, P.; Ojha, R.; Ganiger, P.; Navik, O.; Subaharan, K.; et al. Studies on New Invasive Pest Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae) and Its Natural Enemies. J. Biol. Control 2018, 32, 145–151. [Google Scholar] [CrossRef] [Green Version]
- Varshney, R.; Poornesha, B.; Raghavendra, A.; Lalitha, Y.; Apoorva, V.; Ramanujam, B.; Rangeshwaran, R.; Subaharan, K.; Shylesha, A.N.; Bakthavatsalam, N.; et al. Biocontrol-Based Management of Fall Armyworm, Spodoptera frugiperda (J E Smith) (Lepidoptera: Noctuidae) on Indian Maize. J. Plant Dis. Prot. 2021, 128, 87–95. [Google Scholar] [CrossRef]
- Nboyine, J.A.; Asamani, E.; Agboyi, L.K.; Yahaya, I.; Kusi, F.; Adazebra, G.; Badii, B.K. Assessment of the Optimal Frequency of Insecticide Sprays Required to Manage Fall Armyworm (Spodoptera frugiperda J.E Smith) in Maize (Zea mays L.) in Northern Ghana. CABI Agric. Biosci. 2022, 3, 3. [Google Scholar] [CrossRef]
- Sombra, K.E.S.; de Aguiar, C.V.S.; de Oliveira, S.J.; Barbosa, M.G.; Zocolo, G.J.; Pastori, P.L. Potential Pesticide of Three Essential Oils against Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae). Chil. J. Agric. Res. 2020, 80, 617–628. [Google Scholar] [CrossRef]
- Akutse, K.S.; Subramanian, S.; Maniania, K.N.; Dubois, T.; Ekesi, S. Biopesticide Research and Product Development in Africa for Sustainable Agriculture and Food Security – Experiences from the International Centre of Insect Physiology and Ecology (icipe). Front. Sustain. Food Syst. 2020, 4, 563016. [Google Scholar] [CrossRef]
- Dimbi, S.; Maniania, N.K.; Lux, S.A.; Ekesi, S.; Mueke, J.K. Pathogenicity of Metarhizium anisopliae (Metsch.) Sorokin and Beauveria bassiana (Balsamo) Vuillemin, to Three Adult Fruit Fly Species: Ceratitis capitata (Weidemann), C. Rosa var. fasciventris Karsch and C. Cosyra (Walker) (Diptera: Tephritidae). Mycopathologia 2003, 156, 375–382. [Google Scholar] [CrossRef]
- Prasanna, B.; Huesing, J.E.; Eddy, R.; Peschke, V.M. Fall Armyworm in Africa: A Guide for Integrated Pest Management, 1st ed.; Feed the Future—The US Government’s Global Hunger &, Food Security Initiative, USAID: Washington, DC, Eds.; CIMMYT: Mexico City, Mexico, 2018; pp. 1–120. [Google Scholar]
- Akutse, K.S.; Maniania, N.K.; Fiaboe, K.K.M.; Van den Berg, J.; Ekesi, S. Endophytic Colonization of Vicia faba and Phaseolus vulgaris (Fabaceae) by Fungal Pathogens and Their Effects on the Life-History Parameters of Liriomyza huidobrensis (Diptera: Agromyzidae). Fungal Ecol. 2013, 6, 293–301. [Google Scholar] [CrossRef]
- Goettel, M.S.; Douglas Inglis, G. Fungi: Hyphomycetes. Manual of Techniques in Insect Pathology; Academic Press: Cambridge, MA, USA, 1997; ISBN 9780124325555. [Google Scholar]
- Paradza, V.M.; Khamis, F.M.; Yusuf, A.A.; Subramanian, S.; Akutse, K.S. Heliyon Virulence and Horizontal Transmission of Metarhizium anisopliae by the Adults of the Greenhouse Whitefly Trialeurodes vaporariorum (Hemiptera : Aleyrodidae) and the efficacy of Oil Formulations against Its Nymphs. Heliyon 2021, 7, e08277. [Google Scholar] [CrossRef] [PubMed]
- Opisa, S.; du Plessis, H.; Akutse, K.S.; Fiaboe, K.K.M.; Ekesi, S. Effects of Entomopathogenic Fungi and Bacillus thuringiensis-Based Biopesticides on Spoladea recurvalis (Lepidoptera: Crambidae). J. Appl. Entomol. 2018, 142, 617–626. [Google Scholar] [CrossRef]
- Sisay, B.; Simiyu, J.; Mendesil, E.; Likhayo, P.; Ayalew, G.; Mohamed, S.; Subramanian, S.; Tefera, T. Fall Armyworm, Spodoptera frugiperda Infestations in East Africa: Assessment of Damage and Parasitism. Insects 2019, 10, 195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mweke, A.; Ulrichs, C.; Nana, P.; Akutse, K.S.; Fiaboe, K.K.M.; Maniania, N.K.; Ekesi, S. Evaluation of the Entomopathogenic Fungi Metarhizium anisopliae, Beauveria bassiana and Isaria sp. for the Management of Aphis craccivora (Hemiptera: Aphididdae). J. Econ. Entomol. 2018, 111, 1587–1594. [Google Scholar] [CrossRef] [PubMed]
- Abbott, W.S. A Method of Computing the Effectiveness of an Insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. An Analysis of Variance Test for Normality (Complete Samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; R Development Core Team: Vienna, Austria, 2019; Available online: https://www.R-Project.Org/ (accessed on 12 June 2018).
- Hun Lee, T.; Hau Lee, C.; Alia Azmi, N.; Kavita, S.; Wong, S.; Znati, M.; Ben Jannet, H. Characterization of Polar and Non-Polar Compounds of House Edible Bird’s Nest (EBN) from Johor, Malaysia. Chem. Biodivers. 2020, 17, 1900419. [Google Scholar] [CrossRef] [Green Version]
- Rongai, D.; Cerato, C.; Lazzeri, L.; Palmieri, S.; Patalano, G. Vegetable Oil Formulation as Biopesticide to Control California Red Scale (Aonidiella aurantii Maskell). J. Pest Sci. 2008, 81, 179–185. [Google Scholar] [CrossRef]
- Alavo, T.B.C. Biological Control Agents and Environmentally-Friendly Compounds for the Integrated Management of Helicoverpa armigera Hübner (Lepidoptera: Noctuidae) on Cotton: Perspectives for Pyrethroid Resistance Management in West Africa? Arch. Phytopathol. Plant Prot. 2006, 39, 105–111. [Google Scholar] [CrossRef]
- Bugeme, D.M.; Knapp, M.; Boga, H.I.; Wanjoya, A.K.; Maniania, N.K. Influence of Temperature on Virulence of Fungal Isolates of Metarhizium anisopliae and Beauveria bassiana to the Two-Spotted Spider Mite Tetranychus urticae. Mycopathologia 2009, 167, 221–227. [Google Scholar] [CrossRef]
- Wang, X.; Wu, Y. High Levels of Resistance to Chlorantraniliprole Evolved in Field Populations of Plutella xylostella. J. Econ. Entomol. 2012, 105, 1019–1023. [Google Scholar] [CrossRef]
- Caballero, R.; Cyman, S.; Schuster, D.J.; Portillo, H.E.; Slater, R. Baseline Susceptibility of Bemisia tabaci (Genn.) Biotype B in Southern Florida to Cyantraniliprole. Crop Prot. 2013, 44, 104–108. [Google Scholar] [CrossRef]
- Mohapatra, S.; Ahuja, A.K.; Deepa, M.; Sharma, D.; Jagadish, G.K.; Rashmi, N. Persistence and Dissipation of Flubendiamide and Des-Iodo Flubendiamide in Cabbage (Brassica oleracea Linne) and Soil. Bull. Environ. Contam. Toxicol. 2010, 85, 352–356. [Google Scholar] [CrossRef]
- Mweke, A.; Akutse, K.S.; Ulrichs, C.; Fiaboe, K.K.M.; Maniania, N.K.; Ekesi, S. Integrated Management of Aphis craccivora in Cowpea Using Intercropping and Entomopathogenic Fungi under Field Conditions. J. Fungi 2020, 6, 60. [Google Scholar] [CrossRef]
- Mfuti, D.K.; Subramanian, S.; van Tol, R.W.; Wiegers, G.L.; de Kogel, W.J.; Niassy, S.; du Plessis, H.; Ekesi, S.; Maniania, N.K. Spatial Separation of Semiochemical Lurem-TR and Entomopathogenic Fungi to Enhance Their Compatibility and Infectivity in an Autoinoculation System for Thrips Management. Pest Manag. Sci. 2016, 72, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Hardke, J.T.; Temple, J.H.; Leonard, B.R.; Jackson, R.E. Laboratory Toxicity and Field Efficacy of Selected Insecticides against Fall Armyworm (Lepidoptera : Noctuidae). Florida Entomol. 2011, 94, 272–278. [Google Scholar] [CrossRef]
- Curl, C.L.; Spivak, M.; Phinney, R.; Montrose, L. Synthetic Pesticides and Health in Vulnerable Populations: Agricultural Workers. Curr. Environ. Health Rep. 2020, 7, 13–29. [Google Scholar] [CrossRef]
- Alves, R.T.; Bateman, R.P.; Gunn, J.; Prior, C.; Leather, S.R. Effects of Different Formulations on Viability and Medium-Term Storage of Metarhizium anisopliae Conidia. Neotrop. Entomol. 2002, 31, 91–99. [Google Scholar] [CrossRef] [Green Version]
- Bayissa, W.; Ekesi, S.; Mohamed, S.A.; Kaaya, G.P.; Wagacha, J.M.; Hanna, R.; Maniania, N.K. Selection of Fungal Isolates for Virulence against Three Aphid Pest Species of Crucifers and Okra. J. Pest Sci. (2004) 2017, 90, 355–368. [Google Scholar] [CrossRef]
- Alves, R.T.; Bateman, R.P.; Prior, C.; Leather, S.R. Effects of Simulated Solar Radiation on Conidial Germination of Metarhizium anisopliae in Different Formulations. Crop Prot. 1998, 17, 675–679. [Google Scholar] [CrossRef]
- Morley-Davies, J.; Moore, D.; Prior, C. Screening of Metarhizium and Beauveria spp. Conidia with Exposure to Simulated Sunlight and a Range of Temperatures. Mycol. Res. 1996, 100, 31–38. [Google Scholar] [CrossRef]
- Goettel, M.; Hajek, A. Evaluation of Non-Target Effects of Pathogens Used for Management of Arthropods. In Evaluating Indirect Ecological Effects of Biological Control; CABI: Wallingford, UK, 2001; pp. 81–97. [Google Scholar]
- Ogendo, J.O.; Deng, A.L.; Omolo, E.O.; Matasyoh, J.C.; Tuey, R.K.; Khan, Z.R. Management of Stem Borers Using Selected Botanical Pesticides in a Maize-Bean Cropping System. Egert. J. Sci. Technol. 2013, 13, 21–38. [Google Scholar]


| Fungal Formulation | Larval Cumulated Mortality (%) | Lethal Time 50% (LT50) (Days) ± SE |
|---|---|---|
| Canola oil-formulated M. anisopliae ICIPE 41 | 76.0 ± 6.4 b | 2.0 (1.9–2.2) a |
| Corn oil-formulated M. anisopliae ICIPE 41 | 72.5 ± 5.5 b | 2.2 (2.0–2.5) a |
| Olive oil-formulated M. anisopliae ICIPE 41 | 70.4 ± 6.6 b | 2.5 (2.0–2.9) a |
| Aqueous-formulated M. anisopliae ICIPE 41 | 15.2 ± 2.8 a | 8.1 (6.5–0.6) a |
| Treatments | Percentage of FAW Moths | |
|---|---|---|
| Migori | Mbita | |
| Spinetoram pesticide | 0.00 d | 0.00 d |
| Corn oil formulation of M. anisopliae ICIPE 41 | 79.25 ± 11.85 b | 92.76 ± 7.83 b |
| Mazao Achieve® | 32.10 ± 8.49 c | 37.50 ± 9.15 c |
| Control | 88.00 ± 12.21 a | 95.50 ± 5.45 a |
| Treatments | FAW Damage per Plant (%) | |
|---|---|---|
| Migori | Mbita | |
| Spinetoram pesticide | 2.2 ± 0.7 a | 2.4 ± 0.6 a |
| Corn oil formulation of M. anisopliae ICIPE 41 | 13.3 ± 2.0 b | 12.0 ± 2.8 b |
| Mazao Achieve® | 36.1 ± 5.5 c | 35.1 ± 5.0 c |
| Control | 75.7 ± 8.3 d | 71.7 ± 7.3 d |
| Treatments | % FAW Larvae Mortality | % Mycosis | Lethal Time 50% ± SE |
|---|---|---|---|
| Migori | |||
| Spinetoram-based pesticide | 96.1 ± 2.5 a | 0.0 ± 0.0 c | 3.8 (3.80–3.90) b |
| Mazao Achieve® | 73.0 ± 1.3 c | 30.0 ± 0.7 b | 5.1 (5.12–5.26) a |
| Corn oil formulation of M. anisopliae ICIPE 41 | 81.3 ± 2.6 b | 70.0 ± 0.5 a | 5.2 (5.22–5.32) a |
| Control | - | 0.0 ± 0.0 c | - |
| Mbita | |||
| Spinetoram-based pesticide | 98.7 ± 1.3 a | 0.0 ± 0.0 c | 3.8 (3.80–3.88) b |
| Mazao Achive® | 74.3 ± 2.5 c | 25.0 ± 0.4 b | 4.7 (4.70–4.82) a |
| Corn oil formulation of M. anisopliae ICIPE 41 | 83.6 ± 1.5 b | 66.3 ± 0.7 a | 4.6 (4.56–4.68) a |
| Control | - | 0.0 ± 0.0 c | - |
| Treatments | Percentage of Cotesia icipe Emerged | |
|---|---|---|
| Migori | Mbita | |
| Spinetoram pesticide | 0.00 | 0.00 |
| Corn oil formulation of M. anisopliae ICIPE 41 | 9.25 ± 0.85 b | 11.00 ± 1.83 b |
| Mazao Achive® | 2.00 ± 0.49 c | 2.50 ± 0.15 c |
| Control | 10.00 ± 0.71 a | 14.75 ± 1.25 a |
| χ2 | 1.71 | 3.13 |
| Df | 3 | 3 |
| p-value | <0.0001 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munywoki, J.; Omosa, L.K.; Subramanian, S.; Mfuti, D.K.; Njeru, E.M.; Nchiozem-Ngnitedem, V.-A.; Akutse, K.S. Laboratory and Field Performance of Metarhizium anisopliae Isolate ICIPE 41 for Sustainable Control of the Invasive Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). Agronomy 2022, 12, 2636. https://doi.org/10.3390/agronomy12112636
Munywoki J, Omosa LK, Subramanian S, Mfuti DK, Njeru EM, Nchiozem-Ngnitedem V-A, Akutse KS. Laboratory and Field Performance of Metarhizium anisopliae Isolate ICIPE 41 for Sustainable Control of the Invasive Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). Agronomy. 2022; 12(11):2636. https://doi.org/10.3390/agronomy12112636
Chicago/Turabian StyleMunywoki, Joseph, Leonidah Kerubo Omosa, Sevgan Subramanian, David Kupesa Mfuti, Ezekiel Mugendi Njeru, Vaderament-A. Nchiozem-Ngnitedem, and Komivi Senyo Akutse. 2022. "Laboratory and Field Performance of Metarhizium anisopliae Isolate ICIPE 41 for Sustainable Control of the Invasive Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae)" Agronomy 12, no. 11: 2636. https://doi.org/10.3390/agronomy12112636
APA StyleMunywoki, J., Omosa, L. K., Subramanian, S., Mfuti, D. K., Njeru, E. M., Nchiozem-Ngnitedem, V.-A., & Akutse, K. S. (2022). Laboratory and Field Performance of Metarhizium anisopliae Isolate ICIPE 41 for Sustainable Control of the Invasive Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). Agronomy, 12(11), 2636. https://doi.org/10.3390/agronomy12112636









